Abstract
Objective
To study the effects of methyl jasmonate on multidrug resistance in a mouse model of hepatocellular carcinoma.
Methods
Multidrug resistant H22 (H22/FAP) hepatocellular carcinoma cells were produced in vitro by continuous exposure to increasing doses of doxorubicin, cisplatin and 5-fluorouracil (FAP regimen). Cell toxicity was measured using the 3 -(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolum bromide (MTT) assay. Survival time was calculated for BALB/c mice that received intraperitoneal injections of H22/FAP cells followed by treatment with methyl jasmonate or verapamil in combination with FAP for 7 days. Adenosine triphosphate (ATP) hydrolysis was used to measure the activity of permeability-glycoprotein (P-gp) ATPase activity in plasma membranes.
Results
The MTT assay showed that methyl jasmonate significantly enhanced the cytotoxicity of the FAP regimen in multidrug resistant H22/FAP cells. Methyl jasmonate (10 mg/kg and 5 mg/kg) combined with FAP significantly increased survival time in BALB/c mice by 44.25% and 48.01%, respectively, compared with FAP. Methyl jasmonate increased P-gp ATPase activity.
Conclusion
The combined use of methyl jasmonate and the FAP regimen might be a novel strategy for overcoming the multidrug resistance often observed in hepatocellular carcinoma.
Keywords
Introduction
Hepatocellular carcinoma (HCC) is the fifth most common cancer and is the third leading cause of cancer-related death in the world.1–3 In North America and Western Europe, the annual incidence of HCC is <10 cases per 100 000, but in parts of Africa and Asia it increases to 50–150 cases per 100 000.4,5 During the past 50 years, the treatment of HCC has greatly improved; however, therapeutic efficacy remains unsatisfactory and one of the reasons for this is the emergence of multidrug resistance (MDR) to some chemotherapeutic agents in HCC cells.6,7 MDR is defined as resistance of tumour cells to an antitumour drug, with the simultaneous cross-resistance to a variety of structurally and mechanistically diverse antitumour agents.8,9 Upregulation of permeability-glycoprotein (P-gp) is thought to be largely responsible for the development of MDR. 10 P-gp, which is located in the plasma membrane, is a member of the membrane transporter superfamily. 11 In eukaryotes, adenosine triphosphate (ATP)-binding cassette (ABC) transporters function as transporters, regulators and channels. 12 P-gp is the primary member of the ABC transporter superfamily that has been identified in eukaryotes, and the hydrolysis by P-gp is tightly coupled with its function, which is to transport a broad range of substrates across cell membranes.13,14
Effective reversal agents are needed to overcome MDR. Many reversal agents (such as verapamil and cyclosporin A) produce high in vivo toxicity, which limits their clinical application.15,16 Methyl jasmonate, the volatile methyl ester derivative of jasmonic acid, is a novel growth regulator that is widely found in plants. 17 The jasmonates were accepted as a novel class of phytohormones at the 16th International Conference on Plant Growth Substances, 18 and they mediate plant growth, development and the stress response via signal transduction pathways. 17 Methyl jasmonate inhibits the growth of multiple tumour cells by inducing apoptosis and promoting differentiation; consequently, the jasmonates are now being considered as a new family of potential anticancer drugs.19–21 Methyl jasmonate reverses MDR in drug-resistant human cancer cells, 22 but few in vivo studies report the reversal of MDR in HCC, in particular the reversal of MDR via inhibitory effects on P-gp ATPase activity. Furthermore, it has been proven that HCC displays high resistance to chemotherapeutic agents such as cisplatin, 5-fluorouracil and doxorubicin.23–25
In the current study, a clinically simulated FAP (doxorubicin + cisplatin + 5-fluorouracil) regimen was employed in a BALB/c mouse model of multidrug resistant HCC, which was previously established by our laboratory, to investigate the effect of methyl jasmonate (in combination with the FAP regimen) on P-gp ATPase activity and the reversal of MDR.
Materials and methods
Cell lines and animals
The HCC cell line H22 was prepared in the Department of General Surgery, Third Xiangya Hospital, Central South University, Changsha, Hunan Province, China. Multidrug resistant H22/FAP HCC cells were induced by step-by-step, continuous exposure to increasing doses of doxorubicin (Pfizer, New York, NY, USA), cisplatin (Merck, Darmstadt, Germany) and 5-fluorouracil (Qilu-Pharma, Jinan, Shandong Province, China), known as the FAP regimen, in cell culture, as previously described. 26 BALB/c mice (weight range 20–22 g; 5 weeks old) were purchased from Hunan Experimental Animal Centre (Changsha, Hunan Province, China). The mice were given food and water ad libitum. They were housed in a room maintained at 25 ± 1℃ with 55% relative humidity and a 12-h light/12-h dark cycle. The animals had been raised from birth in a specific pathogen-free environment and were handled according to the institutional guidelines complying with Chinese legislation. The study protocol was reviewed and approved by the Ethics Committee on the Use of Animals in Experimentation of Central South University. The study was conducted according to the Regulations for the Management of Experimental Animals that were formulated by the Ministry of Science and Technology of the People’s Republic of China.
Preparation of methyl jasmonate and verapamil
Methyl jasmonate was purchased from Sigma-Aldrich (CAS number: 39924–52-2; Sigma-Aldrich, St Louis, MO, USA). Methyl jasmonate was first dissolved in ethanol and appropriate volumes were made up with distilled water and filter-sterilized before administering to cell cultures, as previously described. 27 Verapamil was purchased from Central Pharmaceutical (lot number: 090101; Central Pharmaceutical, Tianjin, China). Verapamil was first dissolved in RPMI 1640 medium (Gibco, Gaithersburg, MD, USA) and filter-sterilized before being administered to cell cultures.
Determination of maximum nontoxic dose of methyl jasmonate and verapamil in H22/FAP HCC cells
The 3 -(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolum bromide (MTT) assay was used to determine the maximum nontoxic dose of methyl jasmonate and verapamil in H22/FAP HCC cells. H22/FAP HCC cells were cultured in RPMI 1640 medium with 10% fetal bovine serum (FBS; Gibco) and penicillin (100 IU/ml), streptomycin (100 µg/ml), and 25 mM 4-(2-hydroxyethyl)-1-piperazine-ethanesulphonic acid (HEPES) (pH 7.4) in a humidified incubator at 37℃. H22/FAP HCC cells were cultured as an ascetic tumour suspension because their biological characteristics more closely resemble human HCC in this suspension than when they are cultured in vitro. Ascitic tumour cell suspensions were collected from the ascitic fluid of BALB/c mice harbouring H22/FAP HCC cells, pelleted by centrifugation at 180
Determination of resistance index
The resistance index of doxorubicin, cisplatin and 5-fluorouracil in H22 HCC and H22/FAP HCC cells was determined using the MTT assay. Briefly, ascitic tumour cells were collected from the ascitic fluid of BALB/c mice harbouring H22 HCC cells and multidrug resistant H22/FAP HCC cells, as described previously. The suspensions of H22 HCC cells and H22/FAP HCC cells were prepared (as described previously) using the Ficoll-Histopaque discontinuous density-gradient centrifugation method, and were cultured in RPMI 1640 medium containing 10% FBS. The H22 HCC cells and H22/FAP HCC cells were then treated with different concentrations of doxorubicin, cisplatin and 5-fluorouracil for 48 h. For the H22 HCC cells, the final concentrations of doxorubicin and cisplatin were 0.025, 0.05, 0.1, 0.2, 0.4 and 0.8 mg/l; the final concentrations of 5-fluorouracil were 1.0, 2.0, 4.0, 8.0, 16.0 and 32.0 mg/l. For the H22/FAP HCC cells, the final concentrations of doxorubicin and cisplatin were 0.25, 0.5, 1.0, 2.0, 4.0 and 8.0 mg/l; the final concentrations of 5-fluorouracil were 4.0, 8.0, 16.0, 32.0, 64.0 and 128.0 mg/l. Control cultures contained no doxorubicin, cisplatin or 5-fluorouracil. Following incubation for 48 h, a 20 -µl aliquot of 5 mg/ml MTT solution was added to each well and incubated for 4 h, and the MTT assay was undertaken as described above. The inhibitory rate of cell growth was calculated using the following formula: inhibitory rate of cell growth (%) = (mean optical density of the control group‐mean optical density of the treatment group)/mean optical density of the control group × 100%. The half maximal inhibitory concentration (IC50) was calculated, and the resistance index was calculated using the following formula: resistance index = IC50 value of chemotherapeutic agents on H22/FAP HCC cells/IC50 value of chemotherapeutic agents on H22 HCC cells.
Assessment of survival
The H22/FAP HCC cells were cultured in RPMI 1640 medium with 10% FBS and penicillin (100 IU/ml), streptomycin (100 µg/ml), and 25 mM HEPES (pH 7.4) in a humidified incubator at 37℃. The H22/FAP HCC cells (2 × 106 cells) were suspended in 0.1 ml of 0.1 M PBS (pH 7.2). A total of 2 × 106 H22/FAP HCC cells were intraperitoneally injected into each BALB/c mouse (0.2 ml per mouse, 1 × 108 cells/l). The mice were then randomly divided into eight groups using a computer-generated randomization schedule, each group included 10 animals: (i) normal saline (NS) group; (ii) FAP regimen + 20 mg/kg methyl jasmonate group; (iii) FAP regimen + 10 mg/kg methyl jasmonate group; (iv) FAP regimen + 5 mg/kg methyl jasmonate group; (v) FAP regimen + 3.0 mg/kg verapamil group; (vi) FAP regimen + 1.5 mg/kg verapamil group; (vii) FAP regimen + 0.75 mg/kg verapamil group; and (viii) FAP regimen group. These doses of methyl jasmonate and verapamil were used because they were well tolerated by mice in preliminary experiments. The FAP regimen was given at a concentration of ½ IC50 (namely, 42.50 mg/kg 5-fluorouracil) on day 1, 2.10 mg/kg doxorubicin on day 1 and 1.40 mg/kg cisplatin on days 1–5; NS, methyl jasmonate and verapamil were administered on days 1–7. All regimens were administered by injection via the tail vein. The mice were observed over a period of 40 days and the duration of survival was calculated, based on the time to death from ascites.
Preparation of plasma membranes
The H22/FAP HCC cells were cultured as described above and 2 × 106 cells were suspended in 0.1 ml of 0.1 M PBS (pH 7.2). A total of 2 × 106 H22/FAP HCC cells were intraperitoneally injected into each BALB/c mouse (0.2 ml per mouse, 1 × 108 cells/l). The mice were then randomly divided into nine groups as described above, each containing 10 animals: (i) NS group; (ii) FAP regimen + 20 mg/kg methyl jasmonate group; (iii) FAP regimen + 10 mg/kg methyl jasmonate group; (iv) FAP regimen + 5 mg/kg methyl jasmonate group; (v) FAP regimen + 2.5 mg/kg methyl jasmonate group; (vi) FAP regimen + 3.0 mg/kg verapamil group; (vii) FAP regimen + 1.5 mg/kg verapamil group; (viii) FAP regimen + 0.75 mg/kg verapamil group; (ix) FAP regimen + 0.375 mg/kg verapamil group. All drugs were injected via the tail vein and the FAP regimen was given using the protocol described above. After 7 days, ascitic tumour cells were collected and prepared as for the MTT assay described above. 1 × 105 cells were then incubated in 6 ml of lysis solution (Qproteome Plasma Membrane Protein Kit; QIAGEN, Valencia, CA, USA) at 4℃ for 20 min and then centrifuged at 4000
Determination of P-gp ATPase activity
Approximately 20 µg of plasma membrane was sampled from each mouse, then mixed with 50 µl of Tris-HCl (pH 6.8; Sinopharm Chemical Reagent Co. Ltd, Shanghai, China), 2 µl of dithiothreitol (DTT; Sinopharm Chemical Reagent Co. Ltd), 5 µl of MgCl2 (Sinopharm Chemical Reagent Co. Ltd), 2 µl of strophanthin (for scavenging Na+, K+-ATPase activity; Sigma-Aldrich), 2 µl of egtazic acid (for scavenging Ca2+-ATPase activity; Sigma-Aldrich) and 5 µl of sodium azide (for scavenging mitochondrial ATPase activity; Sinopharm Chemical Reagent Co. Ltd). Next, 50 µl of 10 mmol/l Mg-ATP (pH 7.4; Sigma-Aldrich) was added. Finally, 1 ml of phosphorus reagent (item no: 467868; Beckman Coulter, Brea, CA, USA) was added at 37℃ for 30 min and the optical density was measured at 720 nm using a spectrophotometer (UniCel DxC 680i Synchron® Access® Clinical System; Beckman Coulter). Cornish-Bowden plots were constructed using the concentrations of study drug plotted on the negative portion of the abscissa (x-axis) and the P-gp ATPase activities plotted on the vertical co-ordinate (y-axis) for each drug. Cornish-Bowden plots provide a graphical method for determining the type of enzyme inhibition and the dissociation constant for an enzyme–inhibitor complex or for an enzyme–inhibitor–substrate complex. The effect on the enzymatic rate is determined at two or more substrate concentrations and over a range of inhibitor concentrations.
Statistical analyses
Data were presented as mean ± SD (all were <5%) of three experiments. All statistical analyses were performed using the SPSS® statistical software package, version 18.0 (SPSS Inc., Chicago, IL, USA) for Windows®. Statistical comparisons between the groups were undertaken using Student’s t-test. A P-value < 0.05 was considered statistically significant.
Results
The MTT assay, which was used to determine the maximum nontoxic dose of methyl jasmonate and verapamil in the multidrug resistant H22/FAP HCC cells, demonstrated that doses of 1.0 g/l methyl jasmonate and 0.15 g/l verapamil inhibited the growth of <10% of cells. If each mouse was administered an injection based on a volume of 0.02 ml/g, the maximum administration of methyl jasmonate and verapamil to each mouse was 20 and 3 mg/kg, respectively. These determinations of the maximum nontoxic doses of methyl jasmonate and verapamil provided the basis for dose selection for the in vivo treatment protocols described above, which ensured that the reversal of MDR by methyl jasmonate and verapamil was not caused by cytotoxicity.
The half maximal inhibitory concentration (IC50) and the resistance index for doxorubicin, cisplatin and 5-fluorouracil in H22 hepatocellular carcinoma cells and multidrug resistant H22 (H22/FAP) hepatocellular carcinoma cells cultured in vitro, as determined using the 3 -(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolum bromide assay.

Data presented as mean ± SD (all were <5%) of three experiments.
Following the intraperitoneal injection of H22/FAP HCC cells into BALB/c mice, treatment with FAP regimen + 10 mg/kg methyl jasmonate, FAP regimen + 5 mg/kg methyl jasmonate or FAP regimen + 1.5 mg/kg verapamil significantly prolonged the survival of mice compared with treatment with the FAP regimen alone (P < 0.05 for each comparison) (Figure 1). The mean ± SD survival times were 30.74 ± 2.61, 31.54 ± 2.49 and 32.58 ± 2.75 days following treatment with the FAP regimen + 10 mg/kg methyl jasmonate, FAP regimen + 5 mg/kg methyl jasmonate, and FAP regimen + 1.5 mg/kg verapamil, respectively: life prolongation rates of 44.25%, 48.01% and 52.89%, respectively, were achieved, in comparison with the survival time observed in mice treated with the FAP regimen alone (mean ± SD survival time 21.31 ± 1.98 days). There were no significant differences detected between the FAP regimen + 10 mg/kg methyl jasmonate, FAP regimen + 5 mg/kg methyl jasmonate and the FAP regimen + 1.5 mg/kg verapamil groups. The mean ± SD survival times were 23.62 ± 2.44, 20.57 ± 1.82 and 19.63 ± 2.15 days following treatment with the FAP regimen + 20 mg/kg methyl jasmonate, FAP regimen + 3.0 mg/kg verapamil, and FAP regimen + 0.75 mg/kg verapamil, respectively, which were not significantly different from the times observed in the mice treated with the FAP regimen alone. In addition, no significant difference was observed in the survival time between the NS group and the FAP regimen group, which further confirmed the development of resistance of the H22/FAP HCC cells to the FAP regimen.
The effect of methyl jasmonate (MJ) and verapamil (VER) in combination with doxorubicin, cisplatin and 5-fluorouracil (FAP) on the survival of BALB/c mice that had received an intraperitoneal injection of multidrug resistant H22 (H22/FAP) hepatocellular carcinoma cells. Survival time (days) was measured from the last day of drug administration (i.e. from day 8 after intraperitoneal injection of H22/FAP cells). Data presented as mean ± SD. *P < 0.05 compared with mice treated with FAP regimen alone; Student’s t-test. Columns: 1, normal saline; 2, FAP; 3, FAP + MJ (20 mg/kg); 4, FAP + MJ (10 mg/kg); 5, FAP + MJ (5 mg/kg); 6, FAP + VER (3 mg/kg); 7, FAP + VER (1.5 mg/kg); 8, FAP + VER (0.75 mg/kg).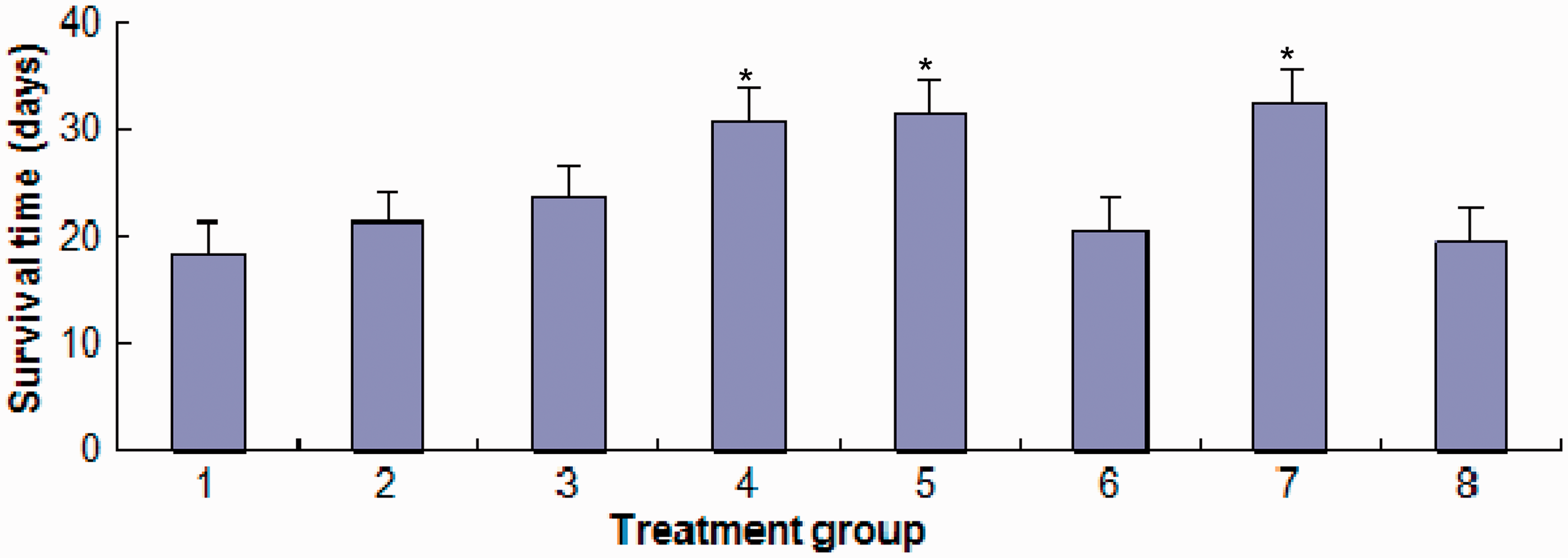
Both methyl jasmonate and verapamil increased the activity of P-gp ATPase in plasma membranes, which had been purified from multidrug resistant H22/FAP HCC cells injected intraperitoneally into BALB/c mice and treated with methyl jasmonate or verapamil in combination with the FAP regimen for 7 days, in a concentration-dependent manner (Figures 2 and 3).
The effect of methyl jasmonate on the activity of permeability glycoprotein ATPase in plasma membranes purified from multidrug resistant H22 (H22/FAP) hepatocellular carcinoma cells injected intraperitoneally into BALB/c mice and treated with methyl jasmonate in combination with doxorubicin, cisplatin and 5-fluorouracil for 7 days. ATPase activity is presented relative to the normal saline control group. The effect of verapamil on the activity of permeability glycoprotein ATPase in plasma membranes purified from multidrug resistant H22 (H22/FAP) hepatocellular carcinoma cells injected intraperitoneally into BALB/c mice and treated with verapamil in combination with doxorubicin, cisplatin and 5-fluorouracil for 7 days. ATPase activity is presented relative to the normal saline control group.

Cornish-Bowden plots for concentrations of methyl jasmonate and verapamil are presented in Figures 4 and 5, respectively. The Michaelis constant (Km) and maximal velocity (Vmax) of methyl jasmonate for P-gp ATPase were 1.11 mg/kg and 184 nmol/min per mg, respectively, while the Km and Vmax of verapamil for P-gp ATPase were 0.34 mg/kg and 190 nmol/min per mg, respectively.
Cornish-Bowden plot showing the effect of methyl jasmonate on the activity of permeability glycoprotein ATPase in plasma membranes purified from multidrug resistant H22 (H22/FAP) hepatocellular carcinoma cells injected intraperitoneally into BALB/c mice and treated with methyl jasmonate in combination with doxorubicin, cisplatin and 5-fluorouracil for 7 days. Cornish-Bowden plot showing the effect of verapamil on the activity of permeability glycoprotein ATPase in plasma membranes purified from multidrug resistant H22 (H22/FAP) hepatocellular carcinoma cells injected intraperitoneally into BALB/c mice and treated with verapamil in combination with doxorubicin, cisplatin and 5-fluorouracil for 7 days.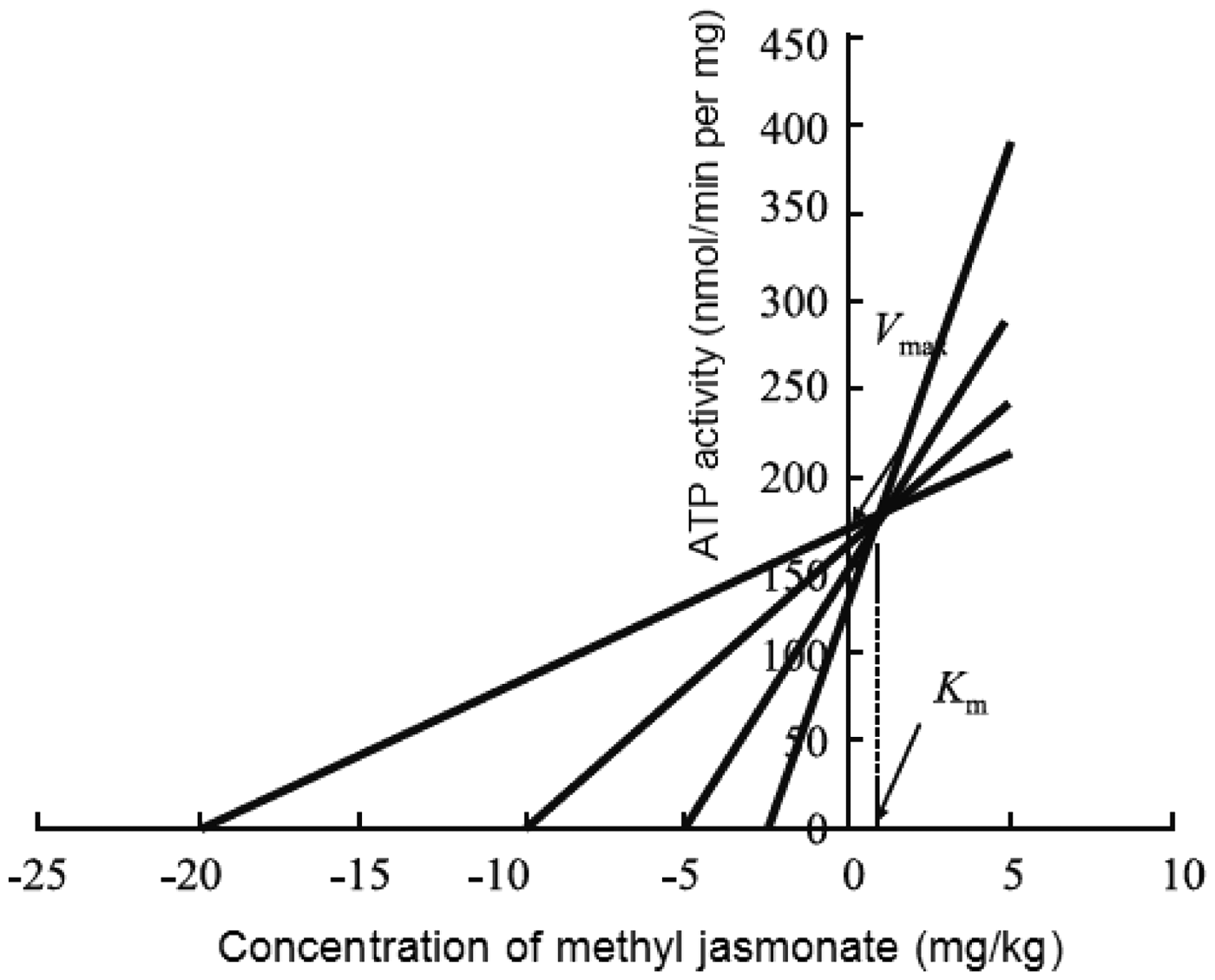

Discussion
Permeability-glycoprotein plays a critical role in the development of MDR by tumours; studying the mechanisms of action of reversal agents would provide valuable knowledge about how to overcome P-gp-mediated tumour MDR.28–30 The present study simulated resistance to the FAP regimen in a BALB/c mouse model of multidrug resistant HCC, which was previously established by our laboratory, in order to explore if MDR could be reversed by the addition of methyl jasmonate to the FAP regimen. If this could be demonstrated, the study also sought to investigate the underlying mechanism of action. In addition, the current study observed the effect of methyl jasmonate and verapamil, in combination with the FAP regimen, on survival time in BALB/c mice implanted with multidrug resistant H22/FAP HCC cells. The results showed that the combination of methyl jasmonate and the FAP regimen prolonged the survival time of mice that had received an intraperitoneal injection of multidrug resistant H22/FAP HCC cells, and could partially reverse the MDR of the H22/FAP HCC cells.
The ATPase activity located on the plasma membrane includes a mixture of Na+, K+-ATPase, Ca2+-ATPase, mitochondrial ATPase and P-gp ATPase, so in order to measure the activity of P-gp ATPase alone, the biochemical analysis undertaken in our current study used DTT, egtazic acid, strophanthin and sodium azide to scavenge all of the other ATPase activities.31–33 Enzyme kinetic analysis revealed that verapamil could increase P-gp ATPase activity, which was in agreement with previous studies.34–36 Methyl jasmonate also increased ATPase activity in a concentration-dependent manner, which would be expected to inhibit the efflux function of P-gp, thereby reversing the MDR of the H22/FAP HCC cells. Cornish-Bowden plots showed that the reversal of MDR by methyl jasmonate was similar to that observed for verapamil. The mechanism of action of MDR reversal by methyl jasmonate might be explained by an increase in P-gp ATPase activity and inhibition of P-gp efflux function. By detecting the accumulation and efflux of rhodamine 123, research has demonstrated that the inhibition of P-gp efflux was responsible for the reversal of MDR by isotetrandrine. 37 Methyl jasmonate is structurally similar to isotetrandrine, so it can be speculated that a direct effect on P-gp efflux function might be one of the mechanisms of action involved in the reversal of MDR, in multidrug-resistant H22/FAP HCC cells.
Methyl jasmonate is a promising MDR reversal agent that deserves further investigation. The present study demonstrated that the combined use of methyl jasmonate and the FAP regimen might be a novel strategy for overcoming the MDR that is often observed in HCC during chemotherapy. Such a strategy would increase the sensitivity of drug-resistant tumours to chemotherapeutic agents in patients with cancer, which could have considerable significance for the treatment of HCC tumours in future.
Footnotes
Declaration of conflicting interest
The authors declare that there are no conflicts of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
