Abstract
In this report, we summarize our experience of rescuing four children with severe type A H3N2 influenza from January to February 2017 in Weifang People’s Hospital, Shandong Province, China for reference in clinical treatment. Two boys and two girls, ranging in age from 3 months to 6 years, with fever, cough, and asthma, were admitted to the pediatric intensive care unit. All children had severe pulmonary infection with respiratory distress. Three children had myocardial damage, two had liver damage, and one had encephalitis. One child had a history of bronchial asthma and one had severe spinal muscular atrophy. After all four children were admitted to the pediatric intensive care unit, they were provided active and effective organ function support and ventilator-assisted respiration. They were treated with gamma globulin, methylprednisolone, and antibiotics. Three children were treated with anti-influenza drugs and recovered from influenza; one child died even before antiviral treatment intervention on the first day. Definite diagnosis of the cases was through clinical manifestations, supplemented by laboratory tests, such as influenza virus H3N2 rapid antigen detection and nucleic acid detection. Early antiviral therapy, high-dose glucocorticoids and immunoglobulins, and systemic comprehensive rescue might be important for rescuing children with severe influenza A (H3N2).
Introduction
Influenza is an acute respiratory disease caused by the influenza virus. Every year, 5% to 10% of adults and 20% to 30% of children worldwide suffer from influenza and approximately 3 to 5 million patients are critically ill, resulting in approximately 250,000 to 500,000 deaths. 1 Influenza viruses are classified into A, B, and C types according to differences in nuclear proteins and matrix protein antigens. Influenza A virus has high human pathogenicity and has caused many global pandemics. 2 Outbreak of influenza caused by the H3N2 subtype in influenza A virus has been gaining an increasing amount of attention.3,4 Children are at high risk for the flu and becoming critically ill. Critically ill cases of the flu are potentially dangerous and the prognosis is poor. A search of relevant international and domestic databases, such as PubMed, Wanfang, and Chinese National Knowledge Infrastructure, showed no reports on the rescue of severe influenza A cases in the most recent 10 years. From the end of 2016 to the beginning of 2017, an influenza pandemic broke out in Weifang. The main virus strain was type A H3N2. Most children could be cured by oral medication, with a few children requiring hospitalization. From January to February 2017, four pediatric patients with severe H3N2 influenza A were treated in the Pediatric Department of Weifang People’s Hospital, of whom three were successfully treated and one died. We summarize our clinical experience of these children.
Patients
Our patients were two boys and two girls aged from 3 months to 6 years (Table 1). All four patients showed symptoms of severe pulmonary infection and respiratory failure, such as high fever (39°C–40°C), respiratory distress, heart rate of 160 to 200 beats/minute, shortness of breath (45–64 breaths/minute), cyanosis of the lips, wheeze on auscultation of the lungs, and blisters. Chest computed tomography showed severe pneumonia. Three of the children had myocardial damage, two had liver damage, and one had encephalitis (Figure 1). Leukocytes or neutrophils were greatly elevated in all of the patients. C-reactive protein levels were greatly elevated, with high procalcitonin levels, which suggested a serious bacterial infection (Table 2). None of the four patients were treated with antiviral drugs before admission and they were not vaccinated. One patient had a history of bronchial asthma and one patient had severe spinal muscular atrophy. In all four patients, influenza virus H3N2 rapid antigen and nucleic acid antigen detection were positive, and one patient had mycoplasma infection.

Chest computed tomographic images of the four children. Case 1 was a 6-year-old boy. Case 2 was a 5-year-old girl. Case 3 was a 2-year-old boy. Case 4 was a 3-month-old girl.
Clinical data of the four cases.

IVIG: intravenous immunoglobulin.
Laboratory examination data of the four children at the beginning of admission.
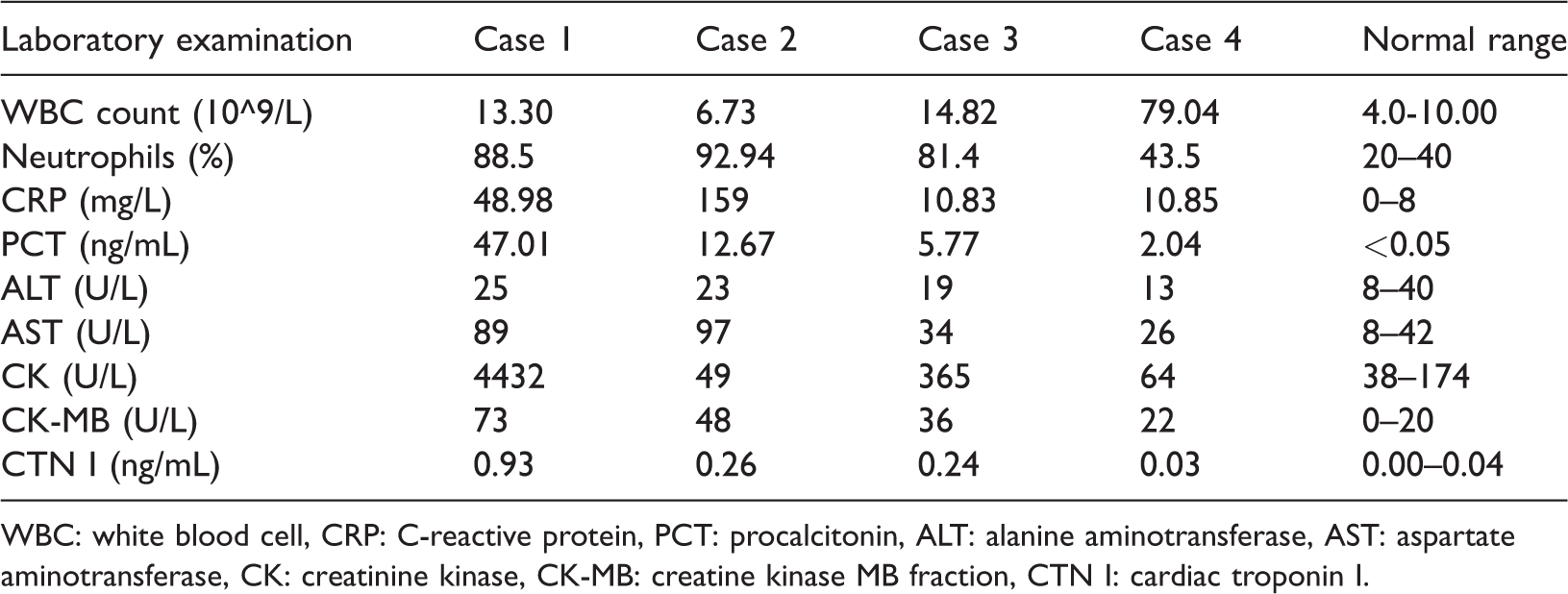
WBC: white blood cell, CRP: C-reactive protein, PCT: procalcitonin, ALT: alanine aminotransferase, AST: aspartate aminotransferase, CK: creatinine kinase, CK-MB: creatine kinase MB fraction, CTN I: cardiac troponin I.
Because the condition of the four patients was urgent, the study was approved verbally by the president of the ethics review committee of Weifang People’s Hospital at the beginning of the study. The study was then approved by the ethics committee of Weifang People’s Hospital to remedy the written protocol after rescue (No: P2017004). The four patients’ parents provided written informed consent.
Treatment and results
The four children were admitted to the pediatric intensive care unit. The children were provided effective organ function support and ventilator-assisted breathing, antibiotics, anti-inflammatory drugs, high-dose corticosteroids, anti-inflammatory treatment (methylprednisolone), gamma globulin, and other treatments. Three of the patients were treated with the anti-influenza drug peramivir, but one patient was not and died within 13 hours following loss of consciousness and respiratory failure. because of severity of illness, insufficient rescue time, and failure of prompt treatment with antiviral drugs. The three surviving patients were discharged from hospital. The average hospital stay was 13 days.
Analysis of the main causes of death for the non-surviving child revealed the following: her condition at time of admission was more severe than that of the other three patients, and this included symptoms of severe respiratory failure, a heart rate of 200 beats/minute or more, breathing rate of 64 beats/minute, and oxygen saturation of 66%; the disease progressed rapidly and the stay in hospital was insufficient; and the patient had previous underlying diseases, such as spinal muscular atrophy, and her physical and mental development were delayed for her age.
Treatment experience and discussion
All of our patients were admitted to hospital in time to carry out a full and systematic rescue. Influenza rapidly progresses in children and the prognosis is poor if not rapidly diagnosed. A correct diagnosis must be made in the first attempt and a comprehensive rescue must be performed. Most patients with severe influenza have symptoms of respiratory failure, and thus ventilator-assisted breathing should be started as early as possible. While conducting comprehensive treatment, organ system complications need to be prevented by providing supportive treatment for the circulatory system, renal system, and other organs. Anti-shock treatment, fluid resuscitation, correction of internal environment disorders, maintenance of water, electrolytes, and acid-base balance, should also be performed for these patients. These treatments may have been the basis for the successful resolution of reported cases.
All of our patients were treated with high-dose glucocorticoids. Glucocorticoids regulate body metabolism and the immune response, and they have anti-inflammatory and anti-shock effects. The use of glucocorticoids is currently controversial. 5 However, for rescuing severe infection, sepsis, and especially respiratory failure, respiratory distress, and other critical illnesses in children, glucocorticoids have positive effects if administered on time and with the full amount of application. The amount of methylprednisolone used in our four patients reached 6 to 8 mg/kg daily, with individual doses reaching 10 mg. This may have played an important role in saving the children's lives, but further observation and studies are required in the future to confirm this possibility.
All of our patients were treated with early anti-influenza virus and anti-infection treatment. Early detection of influenza, early diagnosis and early treatment, and early intervention of anti-influenza virus drugs can shorten the course of the disease, relieve symptoms, reduce complications, and reduce mortality. 6 Treatment should be started as soon as possible within 48 hours, but late intervention of treatment can still produce positive results. The World Health Organization recommends the anti-influenza drugs oseltamivir, peramivir, and zanamivir. 7 The recommended course of treatment for hospitalized pediatric patients is intravenous peramivir for 5 days. If the condition of the patient does not improve or there is still viral replication after 5 days of treatment, the treatment course can be extended.8,9 Gamma globulin has the effect of promoting phagocytosis of monocytes (opsonization), neutralizing the toxicity of bacterial toxins, and binding to viral antigens to neutralize the ability of the virus to infect host cells. Therefore, timely application of gamma globulin has a positive effect in reducing the risk of influenza virus infection. Some other infections, such as mycoplasma can complicate influenza A infection. 10 Therefore, active treatment of secondary infections is crucial in a positive outcome of influenza infection. The most common secondary infections in children are Staphylococcus aureus, Streptococcus pneumoniae, and other Streptococcus bacteria.11,12
Children are at high risk for flu and serious illness, especially infants and young children with underlying diseases.13,14 Underweight, neuromuscular system diseases, and gastrointestinal diseases are risk factors for children with influenza A (H3N2). Influenza in children is more likely to be secondary to bacterial infection, pneumonia, sepsis, and other symptoms compared with adults. Clinical symptoms of influenza are severe and this disease rapidly develops. If not treated in time, influenza can rapidly develop into acute respiratory distress syndrome, shock, multiple organ dysfunction or failure, and can become life-threatening. Among our patients, one patient died because of severe basic disease. This led to an ineffective rescue, and other factors were related to the cause of death in that child.11,12
Footnotes
Declaration of conflicting of interest
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
