Abstract
Pyoderma gangrenosum (PG) is a rare ulcerative inflammatory dermatosis easily confused with wound infection following surgery. There have been seven case reports of PG occurring after total knee arthroplasty (TKA), all of which used routine tissue culture for differential diagnosis. Notably, all previous cases involved delayed diagnosis. We report a case of PG after TKA where we used shotgun metagenomics for differential diagnosis. Metagenomic analysis is a new method that can be used for pathogen detection; it is fast and sensitive, compared with traditional culture. Early application of metagenomic analysis in cases of suspicious wound infection after surgery can detect the pathogen of the infection for target therapy; it can also exclude infection for differential diagnosis of non-infectious diseases, such as autoimmune disorders. This case is presented to support the use of metagenomic analysis by surgeons and physicians for early and rapid differential diagnosis in patients who exhibit postoperative wound infections.
Keywords
Introduction
Pyoderma gangrenosum (PG) is a rare autoinflammatory neutrophilic dermatosis, which results in ulceration and necrosis of the skin. It presents with a variety of clinical forms; thus, diagnosis can be challenging. Although rare, PG is known to occur following trauma or operative procedures. 1 Thus far, seven cases2–8 have been reported following total knee arthroplasty (TKA), all of which used routine tissue culture for differential diagnosis; notably, this culture method resulted in delayed diagnosis. Herein, we report a case of PG following knee arthroplasty, in which we used shotgun metagenomics for rapid differential diagnosis. Shotgun metagenomics, which involves the direct sequencing of DNA from a sample without culture or target-specific amplification or capture, has been widely used in virus discovery and bacterial pathogen detection.9–12
Case report
A 65-year-old Chinese woman, with a past history of type 2 diabetes mellitus, systemic hypertension and hypothyroidism, underwent cemented TKA in October 2017 for osteoarthritis. A standard postoperative regimen of intravenous cefuroxime was followed. On postoperative day 2, the patient demonstrated a fever of 39.3°C and erythema of the wound, with laboratory tests revealing a white blood cell count (WBC) of 25.5 × 109 cells/L, C-reactive protein levels of 201 mg/L (normal: 0–10) and procalcitonin levels of 0.16 ng/mL (normal: 0–0.05). The patient was treated with systemic antibiotics following diagnosis of surgical wound infection. The wounds failed to improve and became painful, after 8 days of antibiotics. On postoperative day 9 (hospital day 12), the patient underwent surgical irrigation and debridement with retention of the prosthesis. Because of superficial necrosis around the wound, negative pressure wound therapy was initiated during the surgery. Notably, the patient then exhibited a suboptimal response to insulin therapy. Although all blood cultures, Gram staining of direct smears from the discharge, and wound cultures of several specimens from debridement were negative, histology revealed neutrophil infiltration into the soft tissue of the knee, which was consistent with wound infection. The fever remained and the lesion rapidly worsened and grew in size. The wound with yellow white pus and black crusts showed a clear red border. Furthermore, a pus blister appeared on the ipsilateral lower leg after 2 days of debridement (Figure 1). Therefore, emergent debridement and resection of the suspected-infected prosthesis was arranged on hospital day 17. The patient was transferred to the intensive care unit (ICU) after operation, because the presence of 2 quickSOFA points (30–35 breaths per minute, somnolence) indicated complicated infection. Interestingly, the wound demonstrated minimal purulence and persistent superficial necrosis. All wound cultures remained negative; however, the pus swab culture obtained during monitoring in the ICU revealed extensively drug-resistant Acinetobacter baumannii. Despite two rounds of debridement and treatment with broad-spectrum antibiotics, the lesion continued to worsen. Determination of whether the wound was infectious confused both physicians and surgeons. Given the absence of an etiological diagnosis and the continued aggravated wound breakdown, an unbiased metagenomic approach was used to detect the causative pathogen. The results suggested that A. baumannii was detected from superficial tissue swabs of pus blister on the ipsilateral lower leg, and deep sample was still negative. The ANCA, ANA, and anti-CCP tests for vasculitis and autoimmune were negative.
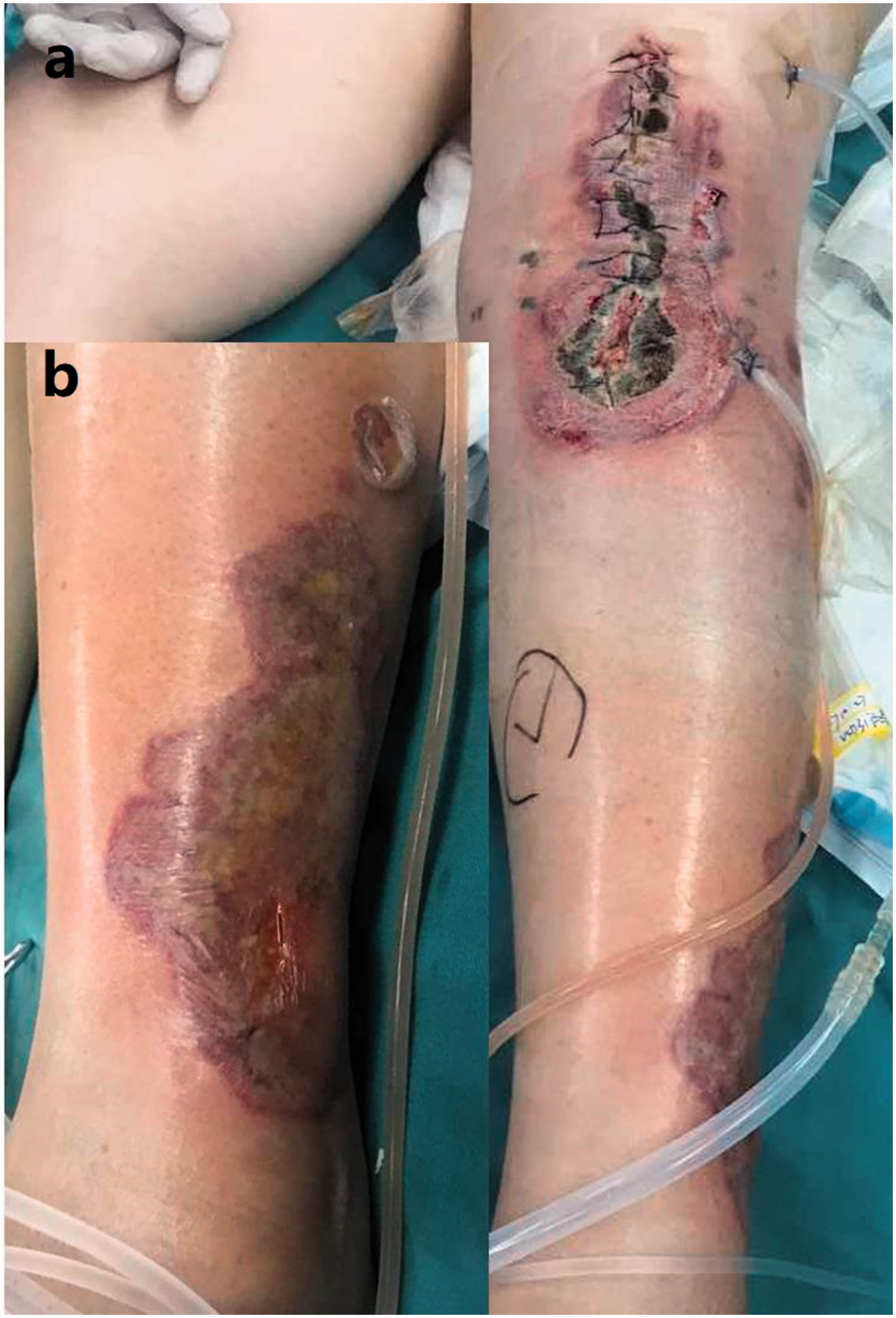
(a) Wound before the second debridement on hospital day 17. (b) A pus blister on the ipsilateral lower leg.
Because of the lack of response to debridement and antibiotics, combined with the unremarkable results of metagenomic sequencing, a dermatology consult was obtained. A new biopsy after the second debridement demonstrated abundant neutrophilic inflammation in the dermis and subcutaneous cellular tissue (Figure 2). A diagnosis of PG was made, and the patient was treated with intravenous prednisone 40 mg daily and intravenous immunoglobulin (10 g*5 days, 15 g*5 days, 25 g*5 days) on day 26. Proliferation of the ulcer ceased, and the wound improved dramatically within a few days. Laboratory tests showed a WBC of 9.4 × 109 cells/L, C-reactive protein levels of 8.9 mg/L (normal: 0–10) and procalcitonin levels of 0.21 ng/mL (normal: 0–0.05) on day 34. With immunosuppressive therapy, the wound healed within 2 months.
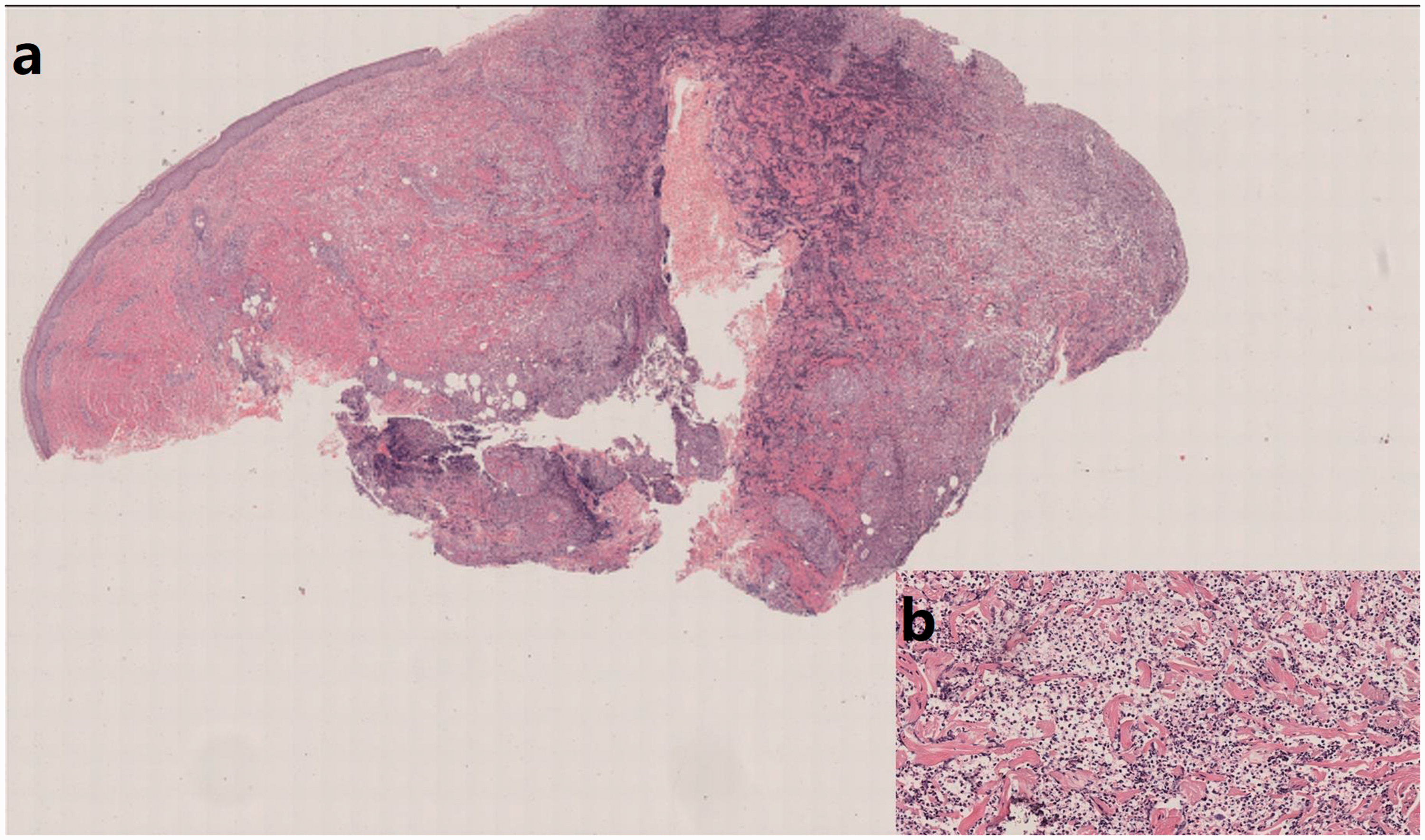
Histopathology findings. (a) A predominantly dermal and hypodermal neutrophilic infiltrate (hematoxylin and eosin (H&E) ×10). (b) The infiltrate in greater detail (H&E ×200).
The present study was approved by the ethics committee of The First Affiliated Hospital of ZheJiang Chinese Medical University (approval no. 2018-KL-011), and the patient provided written informed consent for publication of her information and images.
Discussion
Although skin biopsy aids in confirming the diagnosis of PG, PG is considered a “diagnosis of exclusion” due to the lack of definitive laboratory or histopathological diagnostic criteria. 13 Clinical presentation of PG following TKA mimics a surgical wound infection. Thus, PG is frequently misdiagnosed. There have been seven previous cases2–8 that followed TKA. All cases were initially misdiagnosed as wound infection. Delayed PG diagnosis results in repeated debridement, arthrotomy, and washout of the joint. This leads to a vicious cycle, as surgery aggravates the condition via the pathergy response. Therefore, it is important to identify clinical findings that suggest PG, including severe inflammatory syndrome despite aggressive antibiotic treatment, increased breakdown after debridement, and a lack of bacterial growth from wound swabs despite high WBC. 4 Cell culture methods have existed for many years, but are cumbersome, time-consuming, and lack both sensitivity and specificity. Procalcitonin is a commonly used marker for severe infection, used in diagnosis and assessment of therapeutic interventions. Biomarkers can support clinical decision-making, but cannot distinguish responses to infectious and non-infectious insults with 100% sensitivity and specificity, largely because of the complex, overlapping pathomechanism of pathogen-associated and damage-associated molecular patterns. 14 Thus, there is a need for improved methods to exclude a diagnosis of infection. Metagenomic analysis, using next generation sequencing, is a method that has recently been applied to pathogen detection in cases involving infectious diseases. Compared with traditional cell culture methods, metagenomic analysis is more sensitive and more rapid. Importantly, it also overcomes the limitations of targeted diagnostic methods, such as PCR, because it requires no prior knowledge or assumptions regarding the causative pathogen; therefore, it enables detection of novel and unexpected pathogens. 9 Earlier diagnosis of a sterile wound enables earlier consideration of PG.
Because of the high false negative rate, the negative result of cultures obtained before the patient was transferred to the ICU had not convinced us of the non-infectious nature of the wound; however, metagenomic analysis confirmed it. XDR-A. baumannii was considered to have colonized the wound after its initial establishment, as the lesions substantially compromised the skin barrier. To the best of our knowledge, this is the first report regarding detection of PG following TKA via metagenomic analysis, as well as the use of metagenomic analysis to exclude infection during differential diagnosis of PG. In our case, metagenomic analysis was performed when both diagnosis and treatment were unclear. In review, our efforts at debridement and prosthesis resection were not necessary, because they resulted in a worse short-term outcome. When there is suspicion of wound infection following TKA, we recommend the use of metagenomic analysis screening.
Conclusion
In conclusion, PG is a rare complication after TAK; however, it is an important entity to recognize, because prompt diagnosis is necessary to initiate appropriate treatment. When the wound is suspected of infection, metagenomic analysis should be performed as early as possible; if the metagenomic results are inconclusive, PG should be considered.
Footnotes
Declaration of conflicting interests
The authors declare that there is no conflict of interest.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
