Abstract
Objective
High-quality data indicating the advantages of preoperative Helicobacter pylori screening and eradication as well the clinical outcomes of patients with and without H. pylori after bariatric surgery are lacking.
Methods
In total, 96 morbidly obese patients with H. pylori preparing for bariatric surgery were retrospectively reviewed.
Results
Of 96 biopsy specimens, 73 (76%) were positive for H. pylori on initial Giemsa staining. These patients were treated with the standard 7-day antibiotic treatment protocol corrected by the individual patient’s creatinine clearance rate and body mass index and received a 30% higher dose because of their H. pylori positivity. A linear correlation was found between the effective antibiotic dose and the BMI with a recurrence rate of only 2.1% (2/96 patients). The preoperative percent estimated weight loss before surgery (17%) and in the first year of follow-up (68%–88%) was statistically equal between H. pylori-positive and -negative patients. Two early postoperative infectious complications and two postoperative surgical complications occurred in the preoperatively H. pylori-positive patients.
Conclusions
A patient-tailored H. pylori eradication protocol prior to bariatric surgery is mandatory to improve the eradication rate and reduce the incidence of postoperative complications in mostly asymptomatic H. pylori-positive bariatric candidates.
Introduction
Infection with Helicobacter pylori is one of the most common human infections and is found in approximately half of the global population. 1 Epidemiological studies have shown higher rates of H. pylori infection in developing countries and among socioeconomically deprived populations with a low degree of hygiene.2,3
Helicobacter pylori infection is clearly associated with peptic ulcers, gastric mucosa-associated lymphoid tissue, and gastric cancer. 4 The spectrum of H. pylori-related diseases has recently been addressed in the field of bariatric surgery, and some authors have examined the correlation between postoperative complications of sleeve gastrectomy and the presence of gastric mucosal inflammation and edema secondary to H. pylori infection.5,6
However, the link between H. pylori infection and morbid obesity remains unclear and controversial. Additionally, the eradication rate in the obese population is reportedly insufficient compared with that in the non-obese population. 7 Although it is recommended to routinely check for and eradicate the bacteria before planning a bariatric procedure,8,9 high-quality evidence of the advantages of preoperative H. pylori screening and eradication and data on the clinical outcomes of patients with and without H. pylori after bariatric surgery are lacking.
Therefore, the present study was performed to 1) evaluate the prevalence of H. pylori infection, 2) evaluate the H. pylori eradication rates, 3) evaluate clinical outcomes and postoperative complications regardless of the type of bariatric procedure performed, 4) identify any influence of bacterial eradication on body weight under the same circumstances among Slovenian candidates for bariatric surgery, and 5) present an individualized and tailored approach to H. pylori eradication and overall reduction of surgical and nonsurgical complications.
Materials and methods
Participants
Morbidly obese men and women preparing for bariatric surgery were reviewed. The inclusion criteria were an absence of any symptoms (i.e., no epigastric pain, reflux episodes, nausea, vomiting, bloated feeling, appetite change, or unexplained weight change) and a body mass index (BMI) of >35 kg/m2. The exclusion criteria were a BMI of <35 kg/m2, upper gastrointestinal symptoms (dyspepsia, gastroesophageal reflux, dysphagia, melena, or hematemesis), a history of H. pylori infection with eradication therapy, and an allergy to esomeprazole, amoxicillin, or metronidazole. We also excluded patients with recent (within 2 months before the examination) administration of drugs (nonsteroidal anti-inflammatory drugs, anti-thrombogenics, or proton pump inhibitors), a history of gastric surgery, hemorrhagic disease, liver cirrhosis, end-stage renal disease requiring dialysis, severe heart failure with any symptoms, and early or advanced gastric cancer because of the potentially harmful effects of these conditions on the gastric mucosa.10,11
Study design
Basic anthropometric and biochemical data; comorbidities; medication taken; endoscopic, histopathologic, and microbiologic findings of upper gastrointestinal endoscopy (UGE); perioperative and postoperative complications; and percentage of excess weight loss (% EWL) 1 year after the surgical procedure were obtained from the electronic medical records system at our clinical department of abdominal surgery. The prevalence of H. pylori infection in the adult Slovenian population according to national cohort data was used as a comparator against the bariatric patient group. Patients from the national registry were interviewed regarding their BMI and nutritional habits, and a control group of 75 participants with similar parameters (age, obesity, and type of nutrition) was selected.
Preoperatively, each patient’s general status, mental condition, and dietary habits were assessed by a bariatrician, psychologist, and dietitian. The study design was explained to each patient before the initial assessment, and each patient provided written informed consent before participation. The trial protocol was approved by The National Medical Ethics Committee of the Republic of Slovenia.
Anthropometry and biochemical measurements
The patients’ basic anthropometric characteristics were obtained using standardized techniques and equipment. The BMI was calculated in units of kg/m2, and normal weight was classified as a BMI of 18.5 to 24.9 kg/m2 according to the World Health Organization recommendations for adults. 12 Body composition was evaluated by bioelectrical impedance analysis (BIA) (InBody 720; InBody, Seoul, South Korea). Following an overnight fast, venipuncture was performed in the antecubital vein to obtain blood samples for analysis of blood glucose and lipoprotein levels using standard methods in a certified laboratory.
Diagnosis of H. pylori infection
The indication for UGE in preoperatively asymptomatic patients was a pre-bariatric surgery evaluation due to the association of H. pylori infection with the previously mentioned pathologies. Endoscopic assessment was performed by a well-trained endoscopist using a high-resolution gastrointestinal videoendoscope (Optera; Olympus, Tokyo, Japan). Each patient underwent three biopsies: two specimens were taken from the gastric antrum, and one was taken from the gastric body. One biopsy specimen was used to identify urease enzyme in or on the gastric mucosa with a commercially available bedside rapid urease test (RUT). Because of the low sensitivity of the RUT (65%), we used this test to obtain a quick result, begin the treatment protocol immediately, and avoid prolonging the preoperative time. The other two biopsy specimens were processed for hematoxylin and eosin and modified Giemsa staining (100% sensitivity) and histopathologically examined for the presence of H. pylori by an experienced pathologist. All endoscopically retrieved positive biopsy specimens were transported to the laboratory for culture and antibiotic susceptibility testing of specific H. pylori strains.
Dose adjustment based on BMI
In agreement with local antibiotic sensitivity to H. pylori, each patient received the standard 7-day antibiotic treatment protocol (esomeprazole at 40 mg every 12 hours, amoxicillin at 1300 mg every 12 hours, and metronidazole at 750 mg every 12 hours) corrected by the individual patient’s creatinine clearance rate (CrCl, mL/minute) and BMI. All patients histologically positive for H. pylori based on Giemsa staining received a 30% higher dose. Of these patients, those with an early positive RUT result (within 45 minutes) received the antibiotics within 24 hours, and those with a late positive RUT result (within 24 hours) received the antibiotics within 48 hours. Patients with a negative RUT result but histologically positive findings received antibiotics 2 weeks after UGE. UGE was routinely repeated in all H. pylori-positive patients after the 1-month drug-free interval. To calculate the individual dose of the antibiotics, the Cockcroft–Gault formula (CrCl = {((140 – age in years) × weight in kg) / (72 × SCr)} × 0.85 if female, where SCr is the serum creatinine concentration in mg/dl), body surface area, and volume distribution were used.
Statistical analysis
Data analyses were conducted using IBM SPSS Statistics, version 22.0 (IBM Corp., Armonk, NY, USA). Statistical significance was considered at p < 0.05. Student’s t-test was used to compare the differences between morbidly obese patients and the non-obese Slovenian population.
Results
In total, 96 morbidly obese patients (men, 12 [12.5%]; women, 84 [87.5%]) of middle social class with a mean age of 36 years (range, 28–57 years) and mean BMI of 46 ± 8.3 kg/m2 were enrolled. Helicobacter pylori colonization was significantly higher in morbidly obese patients than in the non-obese Slovenian population (20–49 years of age) (73.0% vs. 29.5%, respectively; p < 0.001), without significant differences in antimicrobial susceptibility of H. pylori isolates to the prescribed antibiotic scheme. The obese patients had a normal range of fat-free body mass (lean body mass) but a significantly higher fat mass and water retention than the non-obese population (p < 0.001). Compared with the non-obese population, the morbidly obese patients also had a significantly higher rate of dyslipidemia (19% vs. 10%, p < 0.001) and a significantly higher rate of hyperglycemia, ranging from higher basal glycemia to type 2 diabetes mellitus (35% vs. 6%, p < 0.04), than the non-obese patients. No patients were taking thiazide diuretics, corticosteroids, or beta-blockers or had any comorbidities affecting the lipoprotein level. Obstructive sleep apnea (OSA) was detected in 54.7% of H. pylori-positive patients, representing 41.6% of all morbidly obese patients (p < 0.001). The RUT turned positive within 10 minutes in 60 of 73 (63%) and within 24 hours in 63 of 73 (65%) of all histologically positive patients. In total, 73 of 96 (76%) biopsy specimens were positive for H. pylori on the initial Giemsa review. The average preoperative % EWL was 17% in both H. pylori-positive and -negative patients. Individual nutritional counseling and physical activity were introduced during the first outpatient visit. The patient-tailored protocol treatment, augmented to the BMI (Figures 1, 2), resulted in an eradication rate of 92.0% for the obese patients and 84.3% for the non-obese patients using the same antibiotic treatment schema. Recurrence was found in only 2 of 96 (2.1%) patients. The % EWL following the eradication therapy and prior to the operation was 17%, which was statistically equal to that in H. pylori-negative patients, while the % EWL in the first year of follow-up ranged from 68% to 88%, again statistically equal to that in H. pylori-negative patients.

Antibiotic dose adjustment based on body mass index.
Two early postoperative infectious complications were treated with a prolonged antibiotic scheme (first-generation cephalosporin, 3 g every 8 h): one patient had a small, ultrasound-proven abscess (1.5 cm in diameter) in the proximal resection line due to a hematoma infection, and the other patient had an elevated white blood cell count and C-reactive protein level with negative findings on abdominal ultrasound. During the postoperative course, two patients who were preoperatively positive for H. pylori developed postoperative complications. The first patient, a polymorbid woman who had undergone laparoscopic sleeve gastrectomy (LSG), developed resection line leakage on postoperative day 16 and was treated conservatively with clip placement (Ovesco, Cary, NC, USA) and percutaneous drain positioning. The second patient, a woman who had undergone laparoscopic Roux-en-Y gastric bypass (LRYGB), developed a marginal ulcer perforation 7 months after surgery and was treated with laparoscopic suturing and omentoplasty. Standard medical care was provided for both patients. At the time of these postoperative complications, both patients were negative for H. pylori based on the results of blood tests and H. pylori stool antigen tests. Despite iron supplementation, late moderate anemia was detected in seven patients who had undergone sleeve gastrectomy and one patient who had undergone single-anastomosis bypass (8/96 [8.3%]). Preoperative oral iron supplementation was not routinely prescribed; based on endocrinological testing, however, vitamin D supplementation was prescribed in all surgical patients.
Discussion
In this study of Slovenian candidates for bariatric surgery, the percentage of patients with H. pylori infection was 2.5 times higher among the morbidly obese patients than among the non-obese population (i.e., 29.5%; unpublished data). In contrast to these findings, other studies have reported a lower prevalence of H. pylori infection in obese patients than in the general population. 7 However, the prevalence of preoperative H. pylori in morbidly obese patients is highly variable, ranging from 8.7% in a German cohort 13 to 85.5% in a Saudi cohort. 14 We believe that the major reason for this discrepancy may be the variation in diagnostic testing. Several direct diagnostic tests (including histopathology and/or immunohistochemistry, RUT, and culture) and indirect diagnostic tests (serology and urine tests, urea breath test, and H. pylori stool antigen test) are currently available for H. pylori detection. To the best of our knowledge, a single noninvasive test that is considered the gold standard with 100% sensitivity and 100% specificity for H. pylori detection is not yet available. Therefore, the clinician should consider all of the advantages and disadvantages of several tests prior to testing.1,15–17 However, in contrast to the majority of studies that largely used indirect noninvasive tests such as serology, which have low diagnostic accuracy (80%–84%), 7 and in agreement with the European guidelines following the Maastricht IV/Florence Consensus Report, 1 we used an endoscopy-based strategy of direct invasive diagnostic testing (i.e., histopathology, RUT, and culture) to obtain quick results and start treatment immediately. Several factors support our decision. The RUT is an inexpensive, rapid, widely available, and highly specific test, 18 making it the most routinely used technique in clinical practice. In the present study, however, its sensitivity was lower than other commercially available RUTs (65% vs. 85%–95%, respectively). 18 Therefore, histopathological confirmation of H. pylori in biopsy samples stained with modified Giemsa (100% sensitivity) was mandatory to support our tailored eradication protocol. Preliminary results using this same approach were previously observed in a small group of morbidly obese patients who were not candidates for obesity surgery interventions (unpublished data). Although culturing is generally not considered as a routine diagnostic method and has limited sensitivity along with a time-consuming procedure, 19 it enabled us to perform antimicrobial susceptibility testing and subsequently implement an appropriate treatment regimen that was tailored to regional antibiotic resistance patterns and individual characteristics. From a clinical viewpoint, wide application of antibiotics in H. pylori eradication treatment increases the number of antibiotic-resistant H. pylori strains, 20 attributing to a global resistance rate of 49.3%, with 40.0% of strains resistant to metronidazole and 19.5% to clarithromycin. 18 The most recently reported resistance rates among adults in the Slovenian general population are comparable, albeit lower (27% and 16% to metronidazole and clarithromycin, respectively 21 ); in our opinion, however, the main reasons for treatment failure are the use of subtherapeutic doses and poor patient compliance.
Although the antimicrobial susceptibility of H. pylori isolates to the prescribed antibiotics did not differ between the two groups in the present study, the eradication rates differed and were significantly higher in the morbidly obese patients. We believe that this supports patient-tailored therapy as a viable alternative to empiric therapy with respect to the correlation of the CrCl, BMI, and volume of distribution calculated according to Cockcroft–Gault formula. Additionally, the body surface area and excessive total body water as measured with BIA were used, and the results suggested a 30% antibiotic dose adjustment; hence, a higher dose than that suggested in the Maastricht guidelines was used. 1 We propose three possible explanations for why dose augmentation based on the relationship between the BMI and CrCl may be crucial for successful treatment of H. pylori infection in morbidly obese patients. The first is delayed gastric emptying, partly caused by consumption of a high-caloric, high-fat diet prior to nutritional counseling and a resultant decrease in the drug absorption rate regardless of the drug characteristics, as well as a sedentary lifestyle and increased gastric distension. 22 The second reason involves pharmacokinetic variables based on body composition, such as the drug distribution to tissue, volume of distribution, and clearance, all of which might be altered in obese patients because of the excess body fat, increased organ mass, reduced lean tissue, and increased water retention. The third reason may have been especially prevalent in our cohort of morbidly obese patients and involves the use of hydrophilic antibiotics such as metronidazole and amoxicillin, thus explaining the linear correlation between the effective dose and the BMI. Analysis of our cohort of morbidly obese patients revealed altered physiological characteristics and adaptations including an increased glomerular filtration rate and increased renal plasma flow; renal hyperfiltration as a consequence of renal vasodilation represents a compensatory response to increased tubular reabsorption of sodium. Increased hydrostatic pressure in afferent arterioles leads to hypertrophy and renal disease, which is strongly affected by hyperlipidemia as well as leptin and hormones originating from adipose tissue. The observed population had significantly increased fat free mass as measured by BIA (average of 66.8%), thus also supporting a negative impact of obesity on a reduced filtering capacity over time. To support our tailored approach, we re-assessed the patients’ renal function and performed dosage adjustments of metronidazole and amoxicillin, which are excreted by the kidneys. We also considered age, muscle mass, diet, and proximal tubule secretion of creatinine. The Cockcroft–Gault equation is a standard method for estimating the CrCl to allow for dosing adjustments. The CrCl increases in a linear manner with lean body weight and may improve the accuracy of the estimated CrCl by the Cockcroft–Gault method. 23 In our patient cohort, we used the adjusted body weight (adjustment factor of 0.3 or 0.4), which represents a gain in lean body weight between the total body weight and ideal body weight as presented in BIA measurements. We used the Cockcroft–Gault formula to determine the drug dosing. Our calculations showed that an average 30% upward dose adjustment was needed. In support of this idea, the literature strongly supports dosing based on body weight or body surface area; drug pharmacokinetic parameters increase in proportion with increasing body size. 24 The BIA measurements regarding excessive body water (overhydration) showed an average 21% EBW, thus also supporting the different volume of distribution in our patient cohort. These data were used to format Figure 1 and Figure 2, which show strong correlations.
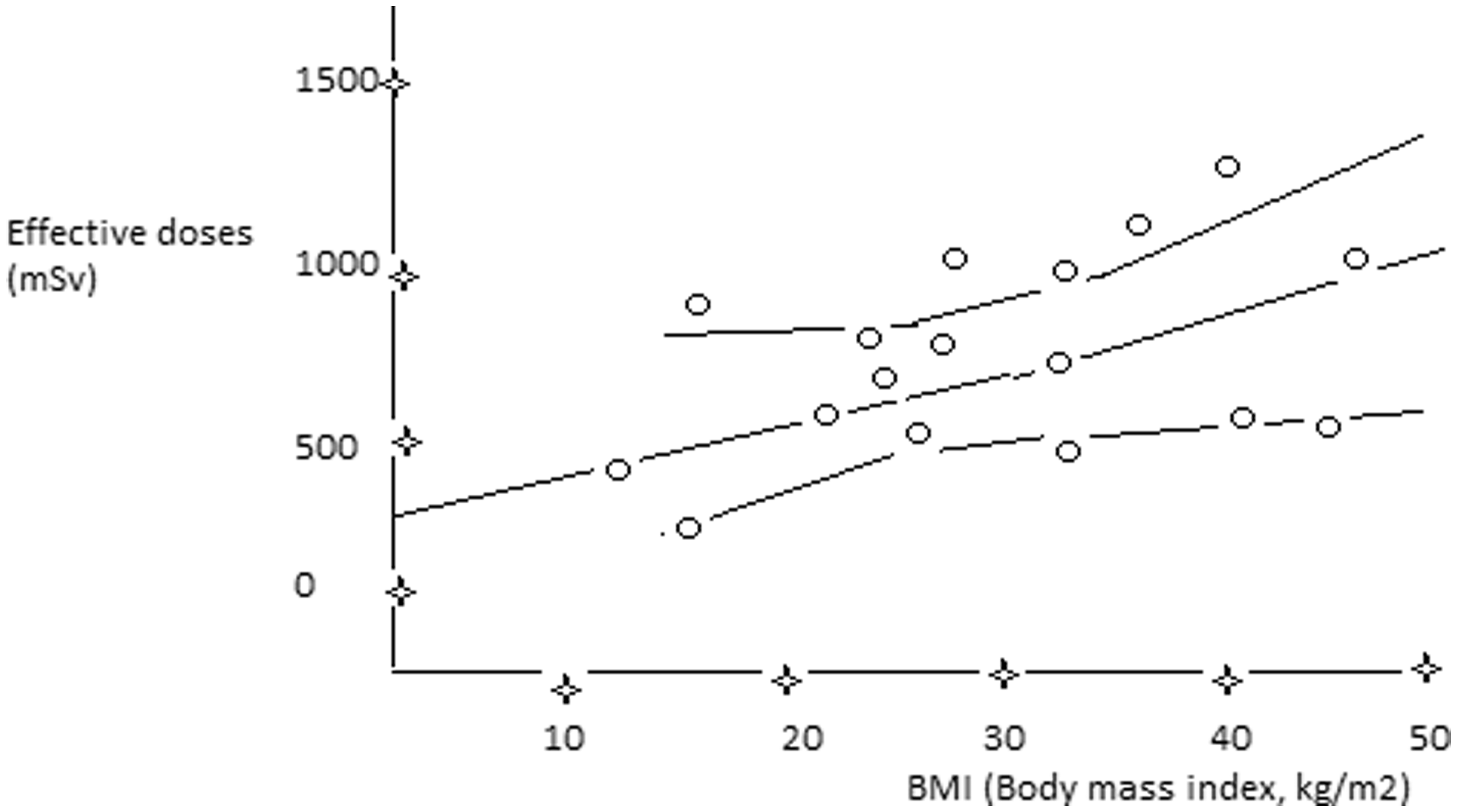
Correlation between effective dose and body mass index.
The altered endocrine regulation in the adipose tissue of morbidly obese patients may also interfere in the mechanisms of drug distribution via release of proinflammatory cytokines, therefore augmenting the local inflammatory response and potentially leading to subtherapeutic drug concentrations. Although comorbidities associated with obesity, such as diabetes mellitus and other factors, may interfere with the eradication rate,25,26 this connection was not specifically followed in our study. However, we found that 54.7% of H. pylori-positive patients had OSA. Although the mechanisms linking OSA and H. pylori infection have not been clearly elucidated, the pathogenesis of OSA (reactive oxygen species, hyperactivation of the sympathetic system, and systemic inflammation) and H. pylori infection might dampen gastric and duodenal mucosa defense via a common cytokine network, hence favoring the occurrence of peptic ulcer bleeding. 27 Zhang 20 evaluated the effect of various types of disease on H. pylori eradication therapy and found that patients with duodenal ulcers exhibited significantly lower H. pylori eradication rates than patients with non-ulcer dyspepsia (21.9% vs. 33.7%, respectively; (p < 10−6). These data led us to speculate that H. pylori infection not only affects the eradication rate but also elevates the risk of postoperative complications in morbidly obese patients with OSA.
In general, however, the clinical outcomes of H. pylori eradication in terms of postoperative complications after bariatric surgery are not clear. While some studies have revealed a reduced prevalence of postsurgical lesions (reduced incidence of viscus perforations 28 and postoperative marginal ulcers 9 ) in patients undergoing LRYGB, others have shown the rates of marginal ulcers or stomal stenosis after LRYGB to be independent of the H. pylori status 29 or attributed to the specific surgical procedures (LRYGB 30 or LSG5,31). Nonetheless, the high prevalence of H. pylori infection found in our cohort of morbidly obese patients was not associated with a higher rate of postoperative complications (2.2%) regardless of the type of operation. This is consistent with the observations in previous studies showing a 1% to 7% incidence of leakage 32 and 2% to 12% incidence of marginal ulcers after LRYGB. 33
Apart from active H. pylori colonization, the operation technique, the type of stapler, and the use of reinforcement, 34 other preoperative factors may also affect the rate of postoperative complications. In the present study, we observed no correlation of the type of surgery and postoperative %EWL with the incidence of early or late postoperative complications. Our patient-tailored preoperative and postoperative regimen was based on lifestyle intervention and nutrition counseling with assessment of the %EWL, which ranged from 17% to 82% between the preoperative period and 1 year postoperatively. Additionally, the eradication rate in our patients remained stable 1 year postoperatively. Available data show an association between increased preoperative weight loss and a reduced frequency of complications in patients undergoing open and laparoscopic gastric bypass, 35 potentially due to the improvements in metabolic parameters (i.e., glycemia, hyperlipidemia) and concomitant reduction in the incidence of comorbidities (irrespective of the performance of bariatric surgery). 36 These findings were also observed in our study, although the latter was not clearly followed.
With respect to the influence of bacterial eradication on body weight among Slovenian bariatric candidates, no significant differences were observed between H. pylori-positive and H. pylori-negative patients, thereby confirming the data of several previous studies.37,38 In contrast to these findings, some studies of adults39,40 and children 41 showed an inverse correlation between H. pylori infection and BMI. A significant increase in BMI after H. pylori eradication might result from increased secretion of ghrelin from the gastric mucosa, which may in turn affect the coordination of eating behavior and body mass regulation, therefore supporting the protective role of H. pylori infection against obesity. 42 However, contrary to a recent review showing that non-obese H. pylori-positive people have lower circulating ghrelin levels than non-obese H. pylori-negative people, 43 morbidly obese patients have been reported to have lower 44 or higher 45 numbers of immunoreactive ghrelin cells. Several other factors may contribute to this discrepancy, such as the lack of classification of H. pylori-related gastritis patterns, 42 dietary changes after eradication, 7 and the relationship between ghrelin gastric mucosal expression and its plasma concentration. 46 However, none of these factors were followed in our study.
Besides gastrointestinal manifestations, several studies have shown a correlation between H. pylori and disorders outside of the gastrointestinal tract; i.e., extraintestinal manifestations. 47 Such manifestations include hematologic, cardiopulmonary, metabolic, neurologic, and dermatologic disorders, but to date only iron deficiency anemia and immune thrombocytopenic purpura are reportedly associated with H. pylori infection. 47 Unexpectedly, postoperative iron deficiency was found in seven patients who had undergone LSG and one patient who had undergone LRYGB in the present study. This was contrary to our expectations because after bypass, patients have excluded sites of iron absorption (duodenum and proximal jejunum). However, previous studies have shown no differences in the risk of iron deficiency between LRYGB and LSG, 48 potentially because of the adaptational change of iron transfer in the intestinal tract after ileojejunal transposition. 49 Additionally, the obese state upregulates hepcidin expression, which is associated with a diminished response to oral iron therapy in patients with iron deficiency anemia, 50 thereby partly explaining the persistent anemia despite iron supplementation therapy in our study. The persistence of anemia also suggests a contributive effect of H. pylori, as studies have shown improved responsiveness to iron supplementation 51 after anti-H. pylori therapy. In the present study, recommendations for supplementation starting in the same week as sleeve and bypass surgery were clearly introduced to patients, but adherence to the suggested supplementation schema was not obtained. Further studies are needed to confirm these positive effects of eradication therapy on extragastric manifestations. Nevertheless, obesity is accompanied by chronic low-grade inflammation, and proinflammatory adipocyte-derived and -released cytokines, especially tumor necrosis factor-alpha and interferon-gamma, negatively affect erythropoiesis and iron homeostasis both in vitro and in vivo.52,53 It is therefore tempting to speculate that a combination of dysfunctional adipocytes and H. pylori infection may also augment the local and systemic immune-angiogenic response systems and contribute to the pro-tumorigenic gastric microenvironment and cancer initiation through the bidirectional cross-talk mechanisms between adipocytes and gastric mucosa. 54
At the time of routine preoperative UGE, approximately 80% of morbidly obese patients are asymptomatic 55 ; additionally, H. pylori is associated with several pathologies, including peptic ulcers, gastric mucosa-associated lymphoid tissue, and gastric cancer (by acting as a class I carcinogen).4,7 Therefore, current European guidelines recommend routine use of UGE in all morbidly obese patients, whether symptomatic or asymptomatic, prior to bariatric surgery in order to treat potential lesions and H. pylori infection 1 and reduce the risk of postoperative complications. To date, however, high-quality evidence indicating the advantages of preoperative H. pylori screening and eradication is lacking. Prospective well-designed randomized controlled trials are necessary to establish the real clinical outcomes of H. pylori positivity and negativity after bariatric surgery.
The limitations of our study are its small sample size, lack of detailed data regarding socioeconomic status and dietary factors, the lack of detailed follow-up regarding comorbidities, and the low sensitivity of the RUT.
In summary, our study indicates that a patient-tailored H. pylori eradication protocol prior to bariatric and metabolic surgery procedures improves the eradication rate and reduces the incidence of postoperative complications in mostly asymptomatic H. pylori-positive bariatric candidates. These findings imply that dose augmentation might be optimally based on the relationship between the BMI and CrCl as shown by the high prevalence of H. pylori-infected candidates for bariatric surgery in our study. Based on our results, well-designed randomized controlled trials are necessary to evaluate the suggested approach and its impact on the postoperative complication rate in bariatric candidates. Importantly, an individualized approach is highly recommended to improve the eradication rate in H. pylori-positive patients and to reduce the risk factors in obese patients undergoing bariatric surgery.
Footnotes
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
The study was funded by the internal research program at UMC Ljubljana.
