Abstract
Objective
This study was performed to investigate the effect of
Methods
Two 2- × 2-cm full-thickness wounds, one on each side of the midline, were made on the back of 12 rats. One wound was covered with Vaseline gauze soaked in normal saline, whereas the other was covered with Vaseline gauze and N-CWS. Wound dressings were changed every other day from day 0 (wound creation) to day 11. Four of the 12 rats were killed on day 7, and biopsy samples were obtained for biochemical and histopathological analyses. The expression levels of CD31, CD68, and F4/80 in the tissues were examined immunohistologically. The expression of transforming growth factor (TGF)-β1 in the wound was determined by western blot.
Results
N-CWS increased the wound healing rate, reduced the complete wound healing time, and increased the expression levels of CD31, CD68, and F4/80 on day 7. The TGF-β1 expression level in the wound was significantly higher in the N-CWS group than in the control group on day 7.
Conclusions
N-CWS can activate macrophages, increase TGF-β1 expression, and enhance angiogenesis and thus accelerate cutaneous wound healing.
Introduction
The inflammatory response is the earliest process that occurs after injury, and macrophages are the principal cellular components that exhibit multiple functions during the healing phase. Macrophages originate from blood monocytes and are found in all tissues. These cells respond to tissue injury to repair any damage and restore the healthy state of tissues. In the injury zone, macrophages perform phagocytosis of pathogens and cell debris and secrete growth factors, chemokines, and cytokines. After being stimulated by pro-inflammatory cytokines, interferon, or other activators, macrophages release many mediators and cytokines, including IL-1, IL-6, IL-12, TNF-α, and transforming growth factor (TGF)-β1. 8 N-CWS has been shown to accelerate the wound repair process in women with cervical erosion. 9 The present study was performed to investigate the effect of N-CWS on cutaneous wound healing. The effect of N-CWS on wound healing was evaluated by measuring macrophage activation and TGF-β1 expression in tissues. The effect of N-CWS on formation of new blood vessels was also assessed.
Materials and methods
Experimental design and cutaneous wound procedure
Twelve healthy adult male Sprague–Dawley rats weighing 200 to 250 g were obtained from the animal laboratory center of Anhui Medical University (Hefei, China). The rats were housed in a controlled environment and provided with standard animal chow and water. Food and water were accessible ad libitum during the procedure. This study was performed in strict accordance with the regulations outlined by the Animal Protection Committee and was approved by the Animal Experimental Ethics Committee of Anhui Medical University in Hefei, China.
The rats were anesthetized by an intraperitoneal injection of chloral hydrate (200 mg/kg), and the dorsum of each rat was clipped free of hair. Two square full-thickness wounds of 2 × 2 cm were excised using a surgical blade on both sides of the back. These two wounds were managed through the self-control test in which the experimental wound was on the left side and the control wound was on the right side.
Immediately after surgical excision, the wounds were randomly divided into the control and N-CWS-treated groups. In the control group, the wounds were covered with Vaseline gauze soaked in normal saline, followed by a bandage. N-CWS was obtained from Fujian Shanhe Pharmaceutical Co. Ltd. (Fujian, China) and was provided as a foam agent at a concentration of 0.02% N-CWS. In the N-CWS-treated group, the wounds were treated topically with 2 mL of N-CWS foam and covered with Vaseline gauze and a bandage. In both groups, the dressing was changed and topical agent was applied every other day from day 0 (wound creation) until the wound was completely healed. The wounds were digitally photographed at each dressing change.
The 10-day healing rate and complete healing time were recorded. Photoshop software (Adobe Systems Inc., San Jose, CA, USA) was used to compute the shadow area and calculate the wound healing rate. The formula used to determine the wound healing rate was (already healed area)/(total injury area) × 100%. The wound healing time was subsequently recorded. The results were summarized by two researchers.
Four of the 12 rats were killed on day 7, and wound samples were harvested for immunohistochemical analysis and western blot.
Immunohistochemical analysis for CD68, CD31, and F4/80 expression levels on wound macrophages
The expression levels of F4/80 and CD68 on wound macrophages were immunohistochemically assessed. Wound angiogenesis was assessed by determining CD31 expression.
First, skin biopsy samples were harvested from the wound tissues, fixed in formalin, and embedded in paraffin. The samples were subjected to immunohistochemical staining with antibodies to CD68, CD31, and F4/80. Biotinylated secondary antibody was used to detect the primary antibodies and was subsequently stained with the avidin–biotin–peroxidase complex system as a chromogenic substrate. Finally, the sections were counterstained with hematoxylin. After antibody staining, the sections were visualized using a high-resolution slide scanner at 100× and 400× magnification. The integrated optical density of CD68, CD31, and F4/80 in the entire section was quantified with Image-Pro Plus software (Media Cybernetics, Rockville, MD, USA).
Western blot for TGF-β1 expression in wound tissues
Wound tissues were homogenized in ice-cold radioimmunoprecipitation assay lysis buffer with 1 mg/mL of protease inhibitor cocktail (Roche, Basel, Switzerland). The homogenates were centrifuged at 1400 ×
Statistical analysis
The results are shown as mean ± standard deviation. Data were statistically analyzed using a paired t-test and Bonferroni correction. A
Results
Effect of N-CWS on cutaneous wound healing
As shown in Figure 1, gross examination of the wounds on days 7, 9, and 11 indicated that wound acceleration was markedly accelerated in the N-CWS group compared with the control group. The 10-day wound healing rate was significantly higher in the N-CWS than control group (89.55% ± 5.68% vs. 86.36% ± 3.23%, respectively;

Gross examination of cutaneous wounds. (a–l) Representative photographs of wounds from control group and N-CWS-treated group. N-CWS,

Effect of N-CWS on cutaneous wound healing rate and time. (a) N-CWS treatment significantly increased the 10-day wound healing rate of cutaneous wounds. (b) Administration of N-CWS significantly reduced the complete wound healing time of cutaneous wounds. Results are expressed as mean ± standard deviation (n = 8). *
Effect of N-CWS on wound macrophage infiltration
Macrophages play a critical role in cutaneous wound healing; these cells are recruited to the wound sites and exhibit pleiotropic function during healing. The macrophage-specific markers CD68 and F4/80 were detected by immunohistochemistry to investigate the effect of N-CWS on macrophage infiltration during wound healing. The wounds in the N-CWS group showed a higher density of macrophages in the granulation tissue on day 7 post-injury than those in the control group (Figures 3(a)–(d) and 4(a)–(d)). Based on the results from Image-Pro Plus software, the N-CWS group had significantly higher expression levels of CD68 (
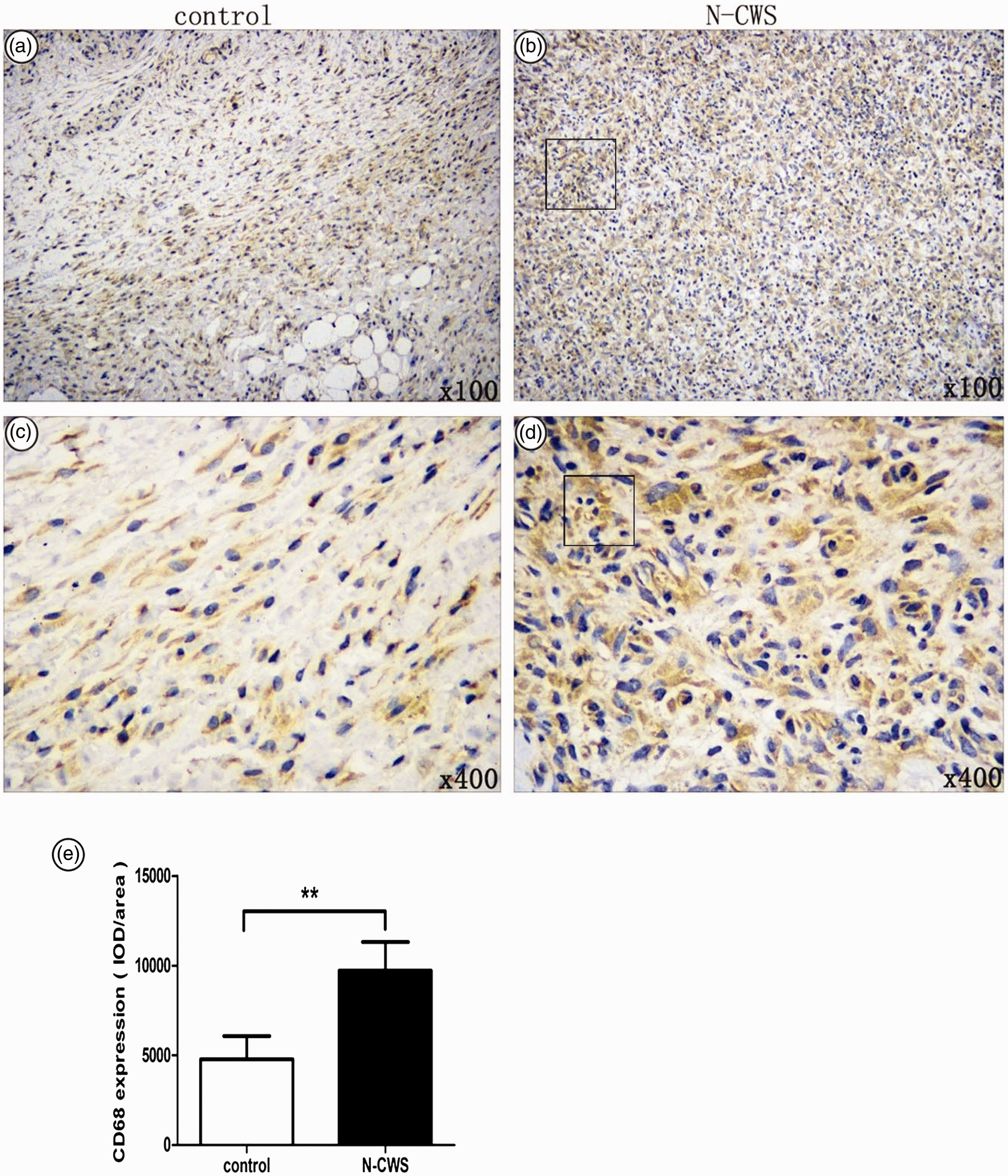
Effect of N-CWS on wound CD68 expression on day 7 post-injury. (a–d) Representative immunohistochemical staining of wound CD68 on day 7. (e) The integrated optical density of CD68 was analyzed with Image-Pro Plus software. N-CWS significantly increased the 7-day wound CD68 expression level. Results are expressed as mean ± standard deviation (n = 4). **

Effect of N-CWS on wound F4/80 expression on day 7 post-injury. (a–d) Representative immunohistochemical staining of wound F4/80 on day 7. (e) The integrated optical density of F4/80 was analyzed with Image-Pro Plus software. N-CWS significantly increased the 7-day wound F4/80 expression level. Results are expressed as mean ± standard deviation (n = 4). *
Effect of N-CWS on TGF-β1 protein expression at 7 days post-injury
TGF-β1 is a growth factor that is released by macrophages and platelets and initiates the healing response. TGF-β1 protein expression in the wounds 7 days post-injury was determined by western blot. The TGF-β1 protein expression was significantly higher in the N-CWS than control group on day 7 (

Effect of N-CWS on wound TGF-β1 expression on day 7 post-injury. (a) Representative results. (b) Densitometric analysis of TGF-β1 by western blot. N-CWS significantly increased 7-day wound TGF-β1 expression. Results are expressed as mean ± standard deviation (n = 4). **
Wounds of using N-CWS exhibit high number of angiogenesis
Wound tissue angiogenesis plays an important role in wound healing. The expression of the endothelial-specific marker CD31 was examined on day 7 to quantify the differences in wound vascularization of the granulation tissues between the two groups. Immunohistological micrographs showed markedly more intense staining for CD31 in the granulation tissue of the N-CWS than control group (Figure 6(a)–(d)). Based on the results obtained from Image-Pro Plus software, the CD31 expression was significantly higher in the N-CWS than control group on day 7 (
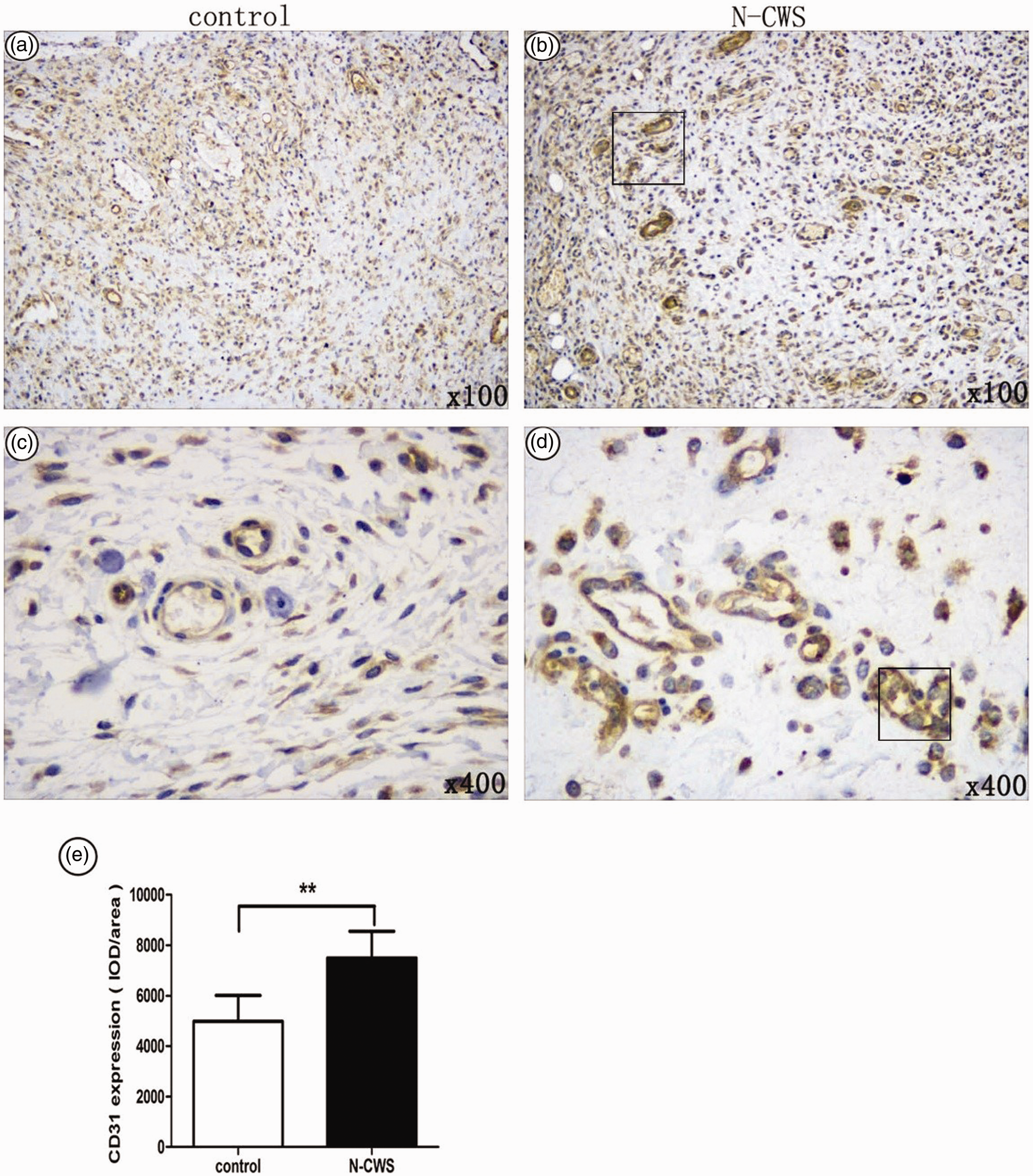
Effect of N-CWS on wound CD31 expression of day 7 after the injury. (a–d) Representative immunohistochemical staining of wound CD31 on day 7. (e) The integrated optical density of CD31 was analyzed with Image-Pro® Plus software. N-CWS significantly increased the 7-day wound CD31 expression level. Results are expressed as mean ± standard deviation (n = 4). **
Discussion
Endothelial cells, macrophages, and fibroblasts participate in tissue formation, epithelialization, and formation of new granulation tissues to restore tissue integrity. 10 This phenomenon is a complicated process encompassing the interactions of extracellular matrix molecules, soluble mediators, various resident cells, and infiltrating leukocyte subtypes. 11 The highly coordinated remodeling and deposition of extracellular matrix molecules is essential for progression to the healing phase. 11
Wound healing involves three overlapping but distinct phases: inflammation, tissue formation, and tissue remodeling. Inflammation is the first stage of wound repair and occurs immediately after injury-induced damage. 16 After 2 to 3 days, monocytes are observed in the wound and differentiate into macrophages, which are vital for later events in injury response. 17 During wound healing, macrophages have at least three major functions: antigen presentation, phagocytosis, and immunomodulation. 18 Two main macrophage phenotypes, classically activated (M1) and alternatively activated (M2), have been identified. M1 macrophages promote the killing of pathogens, drive the inflammatory response, and hold host defense by enhancing the inflammatory environment. M2 macrophages can upregulate the secretion of anti-inflammatory cytokines and activate downstream tissue repair.18,19 M1 and M2 macrophages have different functions and transcriptional profiles. The balance of M1/M2 macrophage polarization governs the fate of an organ in inflammation or injury. In the initial inflammatory phase, neutrophils and monocyte-derived macrophages accumulate in the wound and create pro-inflammatory cytokines. M1 macrophages play a major role in this process by releasing cytokines that participate in the regulation of immune/inflammatory responses, such as IL-1, IL-6, and TNF-α. 20 However, continuation of M1 activity can cause tissue damage secondary to chronic inflammation. Therefore, M2 macrophages secrete high amounts of IL-10 and TGF-β to suppress the inflammation; contribute to tissue repair, remodeling, and angiogenesis; and retain homeostasis. 20
As a glycosylated glycoprotein, CD68 is highly expressed in macrophages and other mononuclear phagocytes. 21 Assessment of CD68 is valuable in histochemical analysis of tumor tissues and inflamed tissues as well as in other immunohistopathological applications. 22 F4/80 is another macrophage marker that is expressed on various macrophage subsets and is widely used to identify macrophages under normal or pathological conditions.23,24 Both of these proteins are common detectors of macrophages and can thus indirectly reflect the condition of wound healing. In the present study, the wounds treated with N-CWS displayed a higher density of macrophages in the granulation tissues than did the untreated wounds. This result is consistent with a study by Mine et al., 25 who found that intraperitoneal N-CWS increased the number of peritoneal macrophages in mice. These findings suggest that N-CWS can increase the quantity of macrophages in cutaneous wounds and thus accelerate healing.
TGF-β1 exhibits biological functions, including embryonic development, angiogenesis, and immune regulation. 26 Released by platelets and macrophages, TGF-β1 conversely influences macrophages during the healing response. 27 TGF-β1 plays an important role in every phase of wound healing. 28 In general, TGF-β1 regulates the transition of inflammation to proliferation in the healing wound. 8 TGF-β1 enhances fibroblast and smooth muscle cell chemotaxis and modulates collagen and collagenase expression.8,29 Our results show that TGF-β1 protein expression in the wound was significantly enhanced in the N-CWS group compared with the control group on day 7. An increased number of macrophages leads to increased TGF-β1 release. N-CWS may indirectly increase the expression of TGF-β1 protein that affects wound healing.
The formation of new blood vessels, termed angiogenesis, plays an important role in wound healing. Angiogenesis results in the formation of a new substrate for keratinocyte migration during the later stages of repair. Angiogenesis is vital for increasing the blood flow to the damaged area. 30 Angiogenesis is regulated by extracellular matrix proteins. TGF-β1 is a major regulator of the extracellular matrix, inducing cellular change and enhancing angiogenesis. 31 A recent study suggested that TGF-β1 promotes angiogenesis by upregulating the composition of the extracellular matrix. 32 CD31 is expressed by endothelial cells, platelets, granulocytes, macrophages, dendritic cells, T and B cells, and natural killer cells. 33 CD31 is always used to detect the occurrence of angiogenesis. In the present study, angiogenesis in the N-CWS group was significantly enhanced compared with that in the control group. Our results suggest that N-CWS may increase the occurrence of angiogenesis by activating TGF-β1 expression.
This study has two main limitations. First, the study sample was relatively small. A larger sample will be very helpful for a better understanding of the role of N-CWS in wound healing. Although the results are statistically significant, more data may strengthen the findings of our study. Second, fibroblasts were not evaluated. Fibroblasts also play a major role in would healing, and the effects of N-CWS on fibroblast migration and activation warrant further research.
In conclusion, these results suggest that N-CWS can activate macrophages, increase TGF-β1 expression, and enhance angiogenesis and thereby accelerate cutaneous wound healing. Our findings show that N-CWS has therapeutic potential and can be used to improve the healing of cutaneous wounds.
Footnotes
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This study was supported by the National Natural Science Foundation of China (Grant nos. 81372050, 81671877).
