Abstract
Objective
To analyse brain microbleed (BMB) progression, its possible underlying factors, and the influence of inter-observer differences, in older individuals with none or mild vascular pathology.
Methods
This study analysed magnetic resonance images, cognitive, demographic and laboratory data from all individuals from the Alzheimer’s Disease (AD) Neuroimaging Initiative database who had the required sequences for identifying BMBs over three consecutive years at the time the database was accessed (January 2016). BMBs were assessed independently by two observers with similar levels of experience.
Results
A total of 291 patients were included in the study. The number of individuals with BMBs and the number of BMBs per individual slightly and nonsignificantly increased across three consecutive years (Y1: 55/291 [19%]; Y2: 61/291 [21%]; Y3: 66/291 [23%]) with 1–2 BMBs and (Y1: 11/291 [4%]; Y2: 12/291 [4%]; Y3: 14/291 [5%]) with ≥ 3 BMBs. Both observers identified a similar pattern of BMB prevalence and progression in each cognitive group (normal < early/late mild cognitive impairment (MCI) > AD patients) despite inter-observer differences (1.5 BMBs, 95% confidence interval –3.7, 6.2], κ=0.543), which were mainly in the cortex. Serum cholesterol was the main predictor of change in BMB count between time-points but did not predict overall progression.
Conclusions
Inter-observer differences are always present and it is difficult to ascertain their influence in the analysis of BMB progression, which was observed in cognitively normal and MCI individuals, but not in AD patients. This should be confirmed in further studies.
Keywords
Introduction
Cognitive decline is an ever-increasing challenge to health and social care systems worldwide due to the growing elderly population. The availability of biomarkers and improved understanding of factors that contribute to heterogeneity in cognitive decline are required to improve diagnosis and prognosis. Small chronic brain haemorrhages occurring with impaired small vessel wall integrity, known as brain microbleeds (BMBs), 1 are commonly found in elderly individuals and have been linked to key hypotheses in the neuro-pathogenesis of the most common form of dementia (i.e. Alzheimer’s Disease [AD]), namely the ‘amyloid cascade’ 2 and the ‘vascular hypothesis’. 3 However, the clinical effect of BMBs on cognition is an evolving research field. 4 The prevalence of BMBs at different stages within the continuum of cognitive decline, reported by a recent review of multiple studies using different magnetic resonance imaging (MRI) sequences and different magnet strengths, 1 are shown in Figure 1.

Schematic representation of the continuum of cognitive decline: from cognitively normal to dementia, as defined by the American Psychiatric Association, and the prevalence of brain microbleeds (BMBs) at its different stages. 1 The colour version of this figure is available at: http://imr.sagepub.com.
A further exploratory analysis on the progression pattern of brain mineral deposition, including BMBs, as a differential indicator of cognitive decline in a cross-sectional sample of 200 individuals aged 55–90 years old (58 cognitively normal [CN], 127 with mild cognitive impairment [MCI] and 15 with AD) observed a similar trend, of a higher count in the number of BMBs in individuals with MCI compared with those with AD. 5 Indeed, a number of studies have identified a lack of association between BMBs and cognitive function in AD patients,6,7 indicating that BMBs may not be present in patients with severe AD at all 6 and that cognitive decline in AD may be independent of BMB number. 8 MCI could be manifested on different neurocognitive disorders. However, as MCI is a stage in the continuum of cognitive decline that leads to AD, exploring the manifestations at each stage of this continuum would certainly lead to a better understanding of the AD pathogenesis. Few longitudinal studies have considered the predictive value of BMB detection on the progression of cognitive decline.9,10 A study found MCI participants with BMBs were twice as likely to progress to dementia than those without BMBs. 10 Another study investigated AD patients and found that patients with BMBs were more cognitively impaired than patients without BMBs, despite having similar disease duration. 9 Although these studies suggest that BMBs may have a potential predictive value for progression of cognitive decline, the evidence is limited. The variability in the outcome of the assessment of BMBs when magnetic field strengths and sequence parameters differ is known; 11 and it has been considered the main limitation in the analyses of BMB progression. To the best of our knowledge, and in agreement with a recent review on the reproducibility and variability of MRI markers in cerebral small vessel disease, 11 no study has assessed the reproducibility of any longitudinal measurement for BMBs.
This study investigated BMB progression over three consecutive years in groups of individuals with different cognitive status: CN, early MCI (EMCI), late MCI (LMCI), and AD, with the hypothesis that BMB prevalence and count progress in CN and MCI individuals, but not in AD patients. The study also evaluated putative risk factors for BMB progression at each time-point and analysed the influence of inter-observer differences on the assessment of BMB progression. Of note, the use of the term ‘progression’ not only refers to whether BMB prevalence (i.e. the proportion of patients with BMBs) and count (i.e. the individual number of BMBs per patient) increased (i.e. progressed), but also if it decreased (i.e. regressed), or remained stable from one year to the next.
Patients and methods
Clinical data extraction from the ADNI database
Data used in the preparation of this article were obtained from the Alzheimer’s Disease Neuroimaging Initiative (ADNI) database. 12 The ADNI was launched in 2003 as a public-private partnership, led by Principal Investigator Michael W Weiner, MD. The primary goal of ADNI has been to evaluate whether serial MRI, positron emission tomography (PET), other biological markers, and clinical and neuropsychological assessments can be combined to measure their progression through the process of normal ageing to EMCI, to LMCI, to dementia or AD. 13
Relevant clinical information for all patients, selected based on clinical plausibility and/or previous research, was extracted from the ADNI database as per Table 1.14–39 Data collection was approved by the corresponding review boards of the United States National Institutes of Health and the Canadian Institutes of Health Research. 12 Written informed consent was obtained from the ADNI participants or their next-of-kin.
Past medical history of cardiovascular risk factors refers to smoking, other risk factors mentioned in the participant’s medical history, and previous medical reports of having (or not) any cardiovascular disease. The latter referred to/included the presence of coronary or peripheral artery disease, mild stroke, hypertensive or rheumatic heart disease, cardiomyopathy, carditis, heart arrhythmia, or thromboembolic disease. The most common risk factors described in the participant’s medical history are hypertension and hypercholesterolaemia.
Study participants
Patients were identified from the ADNI database, accessed in January 2016, using the following selection criteria: (i) three consecutive study visits at 12 months apart; (ii) structural MRI acquisitions at each visit, including T1-weighted (MPRAGE) and T2*-weighted (GRE) sequences, to assess BMBs. Patient selection was done blind to any clinical, demographic or cognitive data.
MRI acquisition
The T2*-weighted (GRE) sequence, which was the primary sequence used for identifying BMBs, was consistently acquired in axial orientation, 2D, with TE/TR = 20/650 ms, flip angle 20°, at 3T, and with an in-plane resolution of 256x256 voxels in all cases. The T1-weighted (MPRAGE) sequence was acquired 3D, with TE/TR = 3.16/6.8 ms, flip angle 8°, voxel size of 1×1×1.2 mm3. Although the scanner vendors and coil models were not the same in all centres, all sequence parameters were consistently equal among scanning waves and across centres.
Image analysis
All (291×3) T2*-weighted (GRE) scans, obtained from each annual visit, were visually assessed by one observer (Observer 1 [A.H.]) for BMBs within the software programme MRIcron®, version 12.12.2012. The Brain Observer Microbleed Scale was used to determine presence, number and location of BMBs. 40 Any black dots initially rated as uncertain were re-rated definitively after considering symmetry, blood vessel anatomy and checking for presence in scans from other visits. Vessel mineralization was also assessed added by the T1-weighted (MPRAGE) sequence. A cross-sectional subsample of 150, and a longitudinal subsample of 20 patients, both randomly selected, were equally assessed by a second observer (Observer 2 [LC]) with a similar level of experience and following the same guidelines. All assessments were done by each observer blind to each other and to any clinical, demographic or cognitive data. The time difference between each observer’s assessments was 1 year. A third observer (M.C.V.H.) checked each observer’s results at the time of their respective assessments, simultaneously viewing all sequences from the three yearly scans of the same patient, to guarantee consistency in the output and decide on ‘uncertain’ cases.
Statistical analyses
All statistical analyses were performed using the SPSS® statistical package, version 24.0 (SPSS Inc., Chicago, IL, USA) for Windows® and MATLAB® R2015a (MathWorks® Inc., Natick, MA, USA).
In order to analyse the progression of BMBs, the Mann–Whitney U-test was used to determine differences in the distribution of BMB prevalence (i.e. the proportion of patients with BMBs) between cognitive groups each year and the median number of BMBs in each cognitive group each year to evaluate BMB progression (as defined previously).
In order to analyse the risk factors for BMB progression, one-way analyses of covariance (ANCOVA) were performed to evaluate candidate variables (clinical data) associated with potential change in BMB count at each time-point. Since BMB counts were obtained at three time-points (year 1 [Y1], year 2 [Y2] and year 3 [Y3]), the evaluation was performed for potential change from Y1 to Y2, Y2 to Y3 and Y1 to Y3. Candidate variables were selected a priori based on clinical plausibility and/or previous research (Table 1).14–39 Data exploration suggested three ANCOVA analyses for bio-specimens (ANCOVA 1), brain measurements (ANCOVA 2) and cognition (ANCOVA 3) should be performed at each time-point. Prior to conducting ANCOVAs, collinearity was assessed through Belsley collinearity diagnostics 41 and any covariates found to have statistically significant associations with other covariates included in each ANCOVA were excluded. Independence between each covariate and the independent variable and homogeneity of regression slope assumptions were also tested and any statistically significant associations excluded from the analysis. Finally, principal component analysis was performed prior to conducting the cognition ANCOVA to restrict cognitive test scores to four principal components that explained 78.38% of the variance. Finally, statistically significant predictors for potential change in BMB count at each time-point from the ANCOVAs were then used to calculate odds ratios to determine whether each predictor was a factor for BMB progression, i.e. an increase in BMB count from Y1 to Y2, Y2 to Y3 or Y1 to Y3. For this, significant predictors from ANCOVAs were dichotomized appropriately; presence or absence for binary factors and clinical criterion used for continuous factors.
Brain microbleed counts reported by each observer for a subsample of 150 patients (cross-sectional subsample) were used to assess inter-observer variability on BMB prevalence per cognitive group. A Bland-Altman plot was constructed to evaluate inter-observer differences. 42 The Mann–Whitney U-test was used to determine whether differences in BMB prevalence between cognitive groups were significant. Cohen’s kappa for BMB presence/absence and for BMB count were also calculated, using an online tool (®Richard Lowry 2001–2015) 43 and the MATLAB® function kappa, version 1.3 by Giuseppe Cardillo. 44
Brain microbleed counts reported by each observer for a subsample of 20 patients (longitudinal subsample) were used to assess inter-observer variability on BMB progression per cognitive group. The Mann–Whitney U-test was used to determine significance in the differences in average (i.e. mean or median depending on whether the data were normally distributed or not) BMB prevalence between cognitive groups. The median number of BMBs reported by each observer, in each cognitive group each year, was also calculated.
Differences in BMB counts reported by each observer were further investigated. Reported BMB location was considered to ascertain whether either observer was consistently identifying more BMBs in a particular location. An experienced observer also re-assessed scans where inter-observer differences were reported to investigate factors that may have confounded BMB assessments.
Results
In general, BMB progression remained stable across the three consecutive years in all cognitive groups despite a BMB count increase that was observed in some CN and MCI individuals. As a consequence, it was not possible to determine whether BMB progression in such a short time frame could be considered an indicator of cognitive decline, as detailed below.
Sample characteristics
A total of 291 patients were identified from the ADNI database using the selection criteria. The descriptive statistics for each group’s baseline characteristics and number of BMBs counted by Observer 1 are presented in Table 2. Prevalence of BMBs in the sample slightly increased over the three consecutive years (Y1: 66/291 [22.68%]; Y2: 73/291 [25.09%]; Y3: 80/291 [27.49%]); as did the individual BMB count per patient (Y1: 55/291 [18.90%]; Y2: 61/291 [20.96%]; Y3: 66/291 [22.68%]) with 1–2 BMBs and (Y1: 11/291 [3.78%]; Y2: 12/291 [4.12%]; Y3: 14/291 [4.81%]) with ≥ 3 BMBs. This slight increase was observed in CN and MCI individuals, but not in AD patients. Of note, 93/291 patients that comprised this sample were part of the cross-sectional sample analysed in a previous study. 5
Descriptive statistics of baseline characteristics for the total sample and by cognitive group.

Data presented as described in the table. IQR = QR3 – QR1.
*Amended sample size where data were unavailable.
CN, cognitively normal; EMCI, early mild cognitive impairment; LMCI, late mild cognitive impairment; AD, Alzheimer’s Disease; GPO, glycerol phosphate oxidase; BMB, brain microbleed.
BMB progression pattern in the total sample
Brain microbleed prevalence overall was higher in the CN group than in the EMCI, LMCI and AD groups each year, but these group differences were not significant. The median number of BMBs per patient per year (Y1, Y2, Y3) was greater in the AD group (2.5, 2.5, 2.5) compared with the CN (1.0, 1.0, 1.0), EMCI (1.0, 1.0, 2.0) and LMCI (1.0, 1.0, 1.0) groups, but these group differences were not significant.
In general, from Y1 to Y2, BMB count increased in 16/291 individuals and decreased in 8/291 individuals. From Y2 to Y3, BMB count increased in 21/291 individuals and decreased in 7/291 individuals. The median increase/decrease counted was 1 BMB. Only 4/291 individuals experienced a consistent yearly increase in the number of BMBs.
Factors for BMB progression
Statistically significant predictors for potential change in BMB count at each time-point identified by ANCOVA analyses are shown in Table 3. Total cholesterol was the main predictor (B = 0.002, P < 0.044) when included in ANCOVA 1. Number of years in education (B = –0.029, P = 0.028) and family history of dementia (B = – 0.10, P = 0.028) were also found to be predictors when included. Family history of AD (B = 0.060, P = 0.016) and endocrine-metabolic risk factors (B = 0.10, P = 0.044) were found to predict potential change in BMB count from Y1 to Y2 in ANCOVAs 2 and 3, respectively. However, as Table 3 shows, significant predictors for potential change in BMB count at each time-point could not be considered significant factors that could potentially increase the likelihood of BMB to occur.
One-way analyses of covariance (ANCOVA) results showing statistically significant predictors for potential change in brain microbleed (BMB) count at each time-point and odds ratios (OR) and 95% confidence intervals (95% CI) of whether the statistically significant predictors are or are not risk factors for BMB progression.
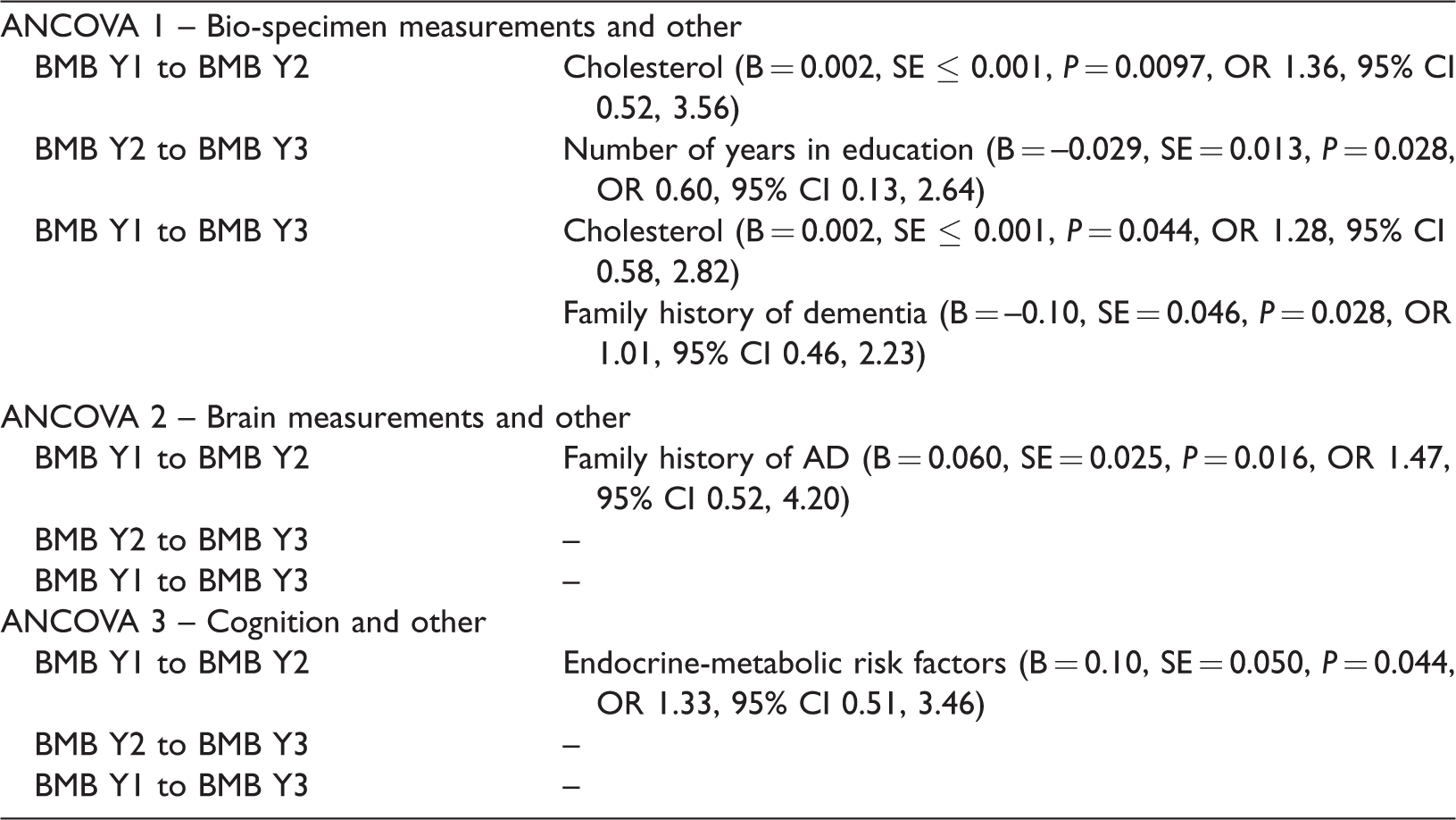
BMB Y1, BMB count in year [Y] 1 (first visit) brain scan; BMB Y2, BMB count in Y2 (second visit) brain scan; B, un-standardized regression coefficient; SE, standard error; BMB Y3, BMB count in Y3 (third visit) brain scan; AD, Alzheimer’s Disease.
Analyses of inter-observer variation on BMB progression pattern assessment
Cross-sectional inter-observer differences
Figure 2a shows the Bland-Altman plot. Both observers had perfect agreement on 68/150 cases. The mean difference of BMB count between the two observers was 1.47, suggesting a bias toward Observer 2 reporting a higher count. Inter-observer differences increased with the increase in the number of BMBs counted. Observer 2 had a tendency to report a higher BMB count in the cortex, a likely explanation for the bias and increasing difference. Observer 1 found BMBs present in 9/50 CN, 15/54 EMCI, 6/38 LMCI, and 1/8 AD patients (Figure 2b) and reported a BMB prevalence lower in CN than EMCI patients. However, BMB prevalence was higher in CN than LMCI and AD patients. None of these differences were significant. Observer 2 found BMBs present in 22/50 CN, 28/54 EMCI, 21/38 LMCI and 4/8 AD patients (Figure 2b) and reported a BMB prevalence lower in CN than EMCI, LMCI and AD patients, but these differences were not significant. While the median number of BMBs found by Observer 1 was slightly greater in the LMCI group (1.7) than the CN (1.0), EMCI (1.0) and AD (1.0) groups, the median number of BMBs found by Observer 2 was slightly greater in the EMCI group (3.5) than the AD (2.5), CN (2.0) and LMCI (2.0) groups.

Baseline inter-observer variability and brain microbleed (BMB) prevalence. (a) Bland-Altman plot for BMB count. The difference in BMB count between observers is plotted against the mean BMB count for 150 patients. It increased with the increase of the mean number of BMB counted by both observers. The mean difference of BMB count between observers is 1.47 (95% confidence interval [–3.7, 6.2], suggesting a bias towards Observer 2 reporting a higher count. Larger circles in the graph represent greater numbers of observer comparisons for these data points. (b) Percentage of patients with BMBs in each cognitive group reported by each observer in the same subsample of 150 patients. CN, cognitively normal; EMCI, early mild cognitive impairment; LMCI, late mild cognitive impairment; AD, Alzheimer’s Disease. The colour version of this figure is available at: http://imr.sagepub.com.
The overall agreement between Observers 1 and 2, of having versus not having BMBs was κ = 0.543. However, there was only a slight agreement between both observers in the exact number of BMBs counted: unweighted Cohen’s kappa κ = 0.058 (95% confidence interval –0.077, 0.194), with a maximum possible kappa given the marginal frequencies κ = 0.424.
Longitudinal inter-observer differences
Observer 1 found BMBs present in 2/3 CN, 2/12 EMCI and 2/5 LMCI patients (Figure 3) and reported an average BMB prevalence higher in CN than EMCI and LMCI patients, over each year although these differences were not significant. Observer 2 found BMBs present in 1/3 CN, 5/12 EMCI and 3/5 LMCI patients (Figure 3) and reported an average BMB prevalence higher in LMCI compared with EMCI and CN patients, over each year. However, the differences in BMB prevalence between cognitive groups were also not significant. The median number of BMBs identified by each observer in each cognitive group per year is detailed in Table 4. Both observers identified BMB progression in the CN group each year. Similarly, for EMCI patients, both observers found BMB progression from Y1 to Y2 and then regression from Y2 to Y3. For LMCI patients, both observers identify BMB progression from Y1 to Y2, but Observer 2 reported a regression from Y2 to Y3 whereas Observer 1 reported a progression (Figure 3).

Longitudinal inter-observer differences per cognitive group in 20 patients. Each patient is represented by a different colour and the patient colours correspond for both observers. *Representative of more than one patient. Brain microbleed (BMB) count refers to the number of BMBs that each observer counted on each scan. CN, cognitively normal; EMCI, early mild cognitive impairment; LMCI, late mild cognitive impairment. The colour version of this figure is available at: http://imr.sagepub.com.
Median number of brain microbleeds (BMBs) identified by each observer in each cognitive group each year on a subsample of 20 patients.

CN, cognitively normal; EMCI, early mild cognitive impairment; LMCI, late mild cognitive impairment.
Factors that affected inter-observer reliability and location of the inter-observer differences
Observer 2 had a tendency to report a higher BMB count in the basal ganglia in the subsample of 20 patients, and in the cortex in the subsample of 150 patients. Inter-observer differences were mainly found in these two locations (i.e. the basal ganglia and cortex). However, in the subsample of 150 patients, median differences of 1 BMB were found in the grey/white matter junction (three cases), brain stem (six cases), thalami (two cases), cerebellum (eight cases) and subcortical white matter (three cases). Vessel calcifications (Figure 4a), basal ganglia mineralization (Figure 4b) and reduced cortical thickness (Figure 4c) hampered identification of ‘true’ BMBs and hence affected inter-observer reliability.

Factors that affected inter-observer variability as seen on magnetic resonance imaging T2*-weighted gradient-recalled echo images (arrows): (a) vessel calcifications; (b) basal ganglia mineralization; (c) reduced cortical thickness.
Discussion
The findings of the current study suggest that the number of BMBs may increase in cognitively normal and mild cognitively impaired individuals but not in patients with Alzheimer’s disease. However, a progression pattern was not present, and none of the factors reported to date to be associated with BMB prevalence appeared to predict these results, although total serum cholesterol was identified as the main potential predictor of the change in BMB count at each time-point. Despite equal assessment conditions, inter-observer variations can be considered problematic in the assessment of BMB progression; their variation seems to have little influence in the analysis of BMB progression in the sample, not so in individual cases.
Contrary to our hypothesis, progression of BMBs in CN and MCI groups of individuals could not be ascertained, as it was observed only in a small proportion of individuals from each group. BMB prevalence remained stable in all AD patients as hypothesized. However, the AD group of patients had higher median number of BMBs than the other three groups. The lack of progression maybe due to the low prevalence of vascular risk factors in AD patients compared with the other groups of individuals. Despite considerable heterogeneity within the existing literature, these current findings contrast with the general trend reported. 1 For example, the Rotterdam study investigated elderly individuals without dementia and reported progression in BMB prevalence from 24.4% at baseline to 28.0% after three years. 21 Although a percentage of CN individuals with BMBs in this current study showed similar progression, increasing from 25.51% to 28.57% three years later, in our case this increase was not significant in the overall sample. Possible explanations for this discrepancy include differences in sample sizes, study population characteristics, MRI sequences and imaging parameter choices, inter-observer variation and/or observer experience. Many studies described in a review 1 used small sample sizes,10,45–47 particularly for CN controls, which may not be representative of BMB prevalence per cognitive group. MRI sequence and imaging parameter choice can also affect BMB identification with susceptibility-weighted images enhancing the susceptibility-related contrasts, and higher magnetic fields increasing the contrast-to-noise ratio and the BMB size. 48 Different study sample characteristics may also have influenced BMB prevalence. The current sample was not representative of the general population, as individuals that have baseline MRI scans with evidence of infarction or other focal lesions, presence of multiple lacunes or lacunes in a critical memory structure, and/or other brain structural abnormalities are excluded from ADNI, hence only individuals with absence or mild presence of vascular pathology are included. Moreover, in the current sample, the mean age in the CN group (74 years) was slightly higher than the EMCI and LMCI groups (70 and 71 years, respectively); and the proportion of CN individuals with two cardiovascular risk factors (29.59%) was also greater than for the EMCI and LMCI groups (26.00% and 21.05%, respectively) and AD (5.88%) patients. BMB prevalence is known to increase with age and cardiovascular risks.20,22,23 Finally, accurate BMB identification is crucial to reporting BMB prevalence, and to our knowledge, this has not been analysed in any of the previous longitudinal studies of BMB progression.
No specific clinical risk factors predicted BMB progression. The association of specific clinical risk factors with BMBs is somewhat conflicting in the existing literature. For example, a study found low total cholesterol to be associated with BMB prevalence, 22 whereas this study found elevated total cholesterol to predict potential change in BMB count from Y1 to Y2 and Y1 to Y3. This latter finding is more consistent with that reported in a study of dementia patients, with BMB prevalence increasing with hyperlipidaemia and with MCI patients with hyperlipidaemia having increased BMB counts. 23 Similarly, conflicting findings have been reported for endocrine-metabolic risk factors, specifically diabetes.21,23 There is a dearth of information on factors and neuropathological markers of cognitive decline and dementia progression, which include years in education, family history of AD and BMBs.14,35,36 The present study is specific to factors that contribute to potential change in BMB count and therefore not directly comparable with research on cognitive decline per se.
Odds ratios were small with confidence intervals overlapping one in the current study. Predictors derived from ANCOVA models may not have translated into significant odds ratios due to sample size and/or small effect size of putative risk factors, and/or dichotomization. Change of the number of BMBs counted in the period analysed was only observed in less than 30 patients. Such sample sizes may have been too small to produce significant findings. Furthermore, the predictors determined by ANCOVA had to be dichotomized to calculate odds ratios. Such dichotomization may have resulted in loss of statistical power. Indeed, two studies investigating risk factors associated with BMBs used much larger samples sizes and logistic regression models to avoid limitations associated with dichotomization.20,22 They still reported odds ratios and confidence intervals quite close to (and often overlapping) one. The progression of other markers of cerebral small vessel disease like white matter hyperintensities could predict the progression of BMBs. However, only visual ratings of white matter hyperintensities and perivascular spaces at Y1 were available for the analyses. Future longitudinal studies require larger sample sizes, quantitative measures of small vessel disease indicators at each time-point and/or alternative statistical approaches to fully investigate risk factors for BMB progression.
Variation in inter-observer differences was observed in the current study. Bland-Altman analysis found a bias towards Observer 2 reporting a higher BMB count, with an increased difference as BMB count increased. However, the analysis of the overall BMB progression in the sample yielded similar results for both observers. Despite recognition of large ranges of inter-observer reliability within the existing literature, 40 there are no guidelines on what constitutes clinically acceptable bias/limits of agreement. Most studies have reported kappa statistics; however, this parameter is limited in interpreting the extent of agreement, with the quantitative value being qualitatively described as slight, fair, moderate etc. For consistency in the way results are reported and to allow comparability with other studies, this current study also reported kappa statistics. Bland-Altman methodology, though, is more appropriate to parametric data, permitting the meaningfulness of change to be determined within the context of the measurement scale (i.e. the 95% limits of agreement are placed in ‘test score’ units in contrast to kappa).
Factors found to affect inter-observer reliability have previously been reported as those confounding BMB detection.11,49,50 Basal ganglia mineralization is likely to explain Observer 2’s tendency to report a higher BMB count in the basal ganglia, and vessel calcifications and reduced cortical thickness, hampering differentiation between BMBs and vessel calcification, explaining the higher BMB count reported for the cortex. A previous study on 100 CN community-dwelling individuals aged 73 years showed a positive and strong correlation and considerable overlap between calcified regions and other regions with iron accumulation in its different forms.52,51 Others have suggested a shared pathway for the age-related deposition of both minerals in the brain, occasionally attributed to the primary accumulation of one followed by the other; 53 or to a shared transport mechanism that manifests in the deposition of both minerals in the vicinities of the blood vessels. 54 These may partly explain why inter-observer differences increased with the increase of the BMB count and the consistency in the overall progression pattern of BMBs found by both observers despite the influence that the inter-observer differences had on the assessment of the BMB progression in individual cases. Future studies should seek to reduce inter-observer variation and there should be a clinical consensus on acceptable bias/limits of agreement.
This study had a number of strengths and limitations. To the best of our knowledge, this is the first study that assesses the reproducibility of longitudinal measurements of BMBs, as such contributing to the emerging field of research on BMBs as potential pathological markers within the spectrum of cognitive decline. Strengths of this study also include the large number of individuals assessed under the same MRI protocol using well-validated BMB guidelines and the wide variety of relevant participant data available. The observers were blinded to clinical data, including cognitive status, throughout image analysis. A limitation of this study was the relatively small AD group. Longitudinal study designs and the nature of AD may impede obtaining comparatively sized AD groups; 55 however, future studies should try to secure larger AD populations to facilitate valid comparisons. This study was also limited by the inclusion/exclusion criteria of the ADNI database, a cohort not entirely representative of the general population. 56 Individuals with substantial small vessel disease are excluded from ADNI and therefore AD patients are likely to be more genetically prone to the disease. Clinical study data included were also restricted to available ADNI database parameters collected across multiple sites. Although a general protocol is in place, variations in techniques and data collection methodologies are likely. The location of BMBs was not statistically analysed in this study. Lobar BMBs have been previously found to be associated with longitudinal cognitive decline, whereas BMBs located in the deep grey matter and infratentorial brain revealed no significant associations. 57 BMBs in all regions were included in this study, potentially diluting findings. A further limitation is the failure to apply a Bonferroni correction to the ANCOVA models that would have resulted in a more stringent criterion for statistical significance. Finally, intra-observer variation was not considered here due to time constraints; such measurements are also important to determine, although might reasonably be expected to be less than inter-observer variations.
Brain microbleed prevalence and count appeared to progress in CN and MCI individuals but not in AD patients; however, differences between cognitive groups were not significant and the median number of BMBs tended to remain stable in each group over time. No significant progression pattern was found in the sample analysed, although as noted above this sample may not be representative of the general ageing population. Although total cholesterol, years in education, family history of dementia and AD, and endocrine-metabolic risk factors were weakly predictive of potential change in BMB count at certain time-points, they were not significant risk factors for BMB progression. Finally, despite equal conditions, inter-observer differences were found and inter-observer variation is likely to have influenced BMB progression pattern assessment. Therefore, this report cannot exclude BMB progression in AD patients.
While this study highlights the importance of reporting inter-observer variation, future studies should seek to reduce inter-observer differences. Similar inter-observer variation analyses could be performed for computational rather than visual assessments of BMBs, to determine whether computational assessment methods are more reliable. Repeating this study in a few years may yield different results as the cognitive status of individuals enrolled in ADNI is likely to advance along the continuum of cognitive decline and more patients are likely to have had three consecutive study visits, minimizing AD group limitations. A similar study could also be performed in a larger sample of patients, adjusting for baseline characteristics, and across a wider spectrum of cognitive decline. Also, analysing a wider interval between assessments and/or increasing the number of yearly assessments (i.e. analysing progression in 4 or more years) would be clinically important. Analysis of BMB location may also be informative. Such a study is likely to provide useful information on BMB progression and associated risk factors, confirming or otherwise the potentially valuable role of BMBs as a differential indicator of cognitive decline.
Footnotes
Acknowledgements
Authors thank Dr Francesca Chappell, Medical Statistician at the Centre for Clinical Brain Sciences in The University of Edinburgh for comments on the statistical analyses.
Declaration of conflicting interests
The authors declare that there are no conflicts of interest.
Funding
M.C.V.H. is funded by the Row Fogo Charitable Trust (grant no. BRO-D.FID3668413), and the work was carried out at the Centre for Clinical Brain Sciences of The University of Edinburgh, part of the UK Dementia Research Institute at The University of Edinburgh. Funds from European Union Horizon 2020, PHC-03-15, project No 666881, ‘SVDs@Target’ and Foundation Leducq Network for the Study of Perivascular Spaces in Small Vessel Disease, ref no. 16 CVD 05 are also gratefully acknowledged.
Data collection and sharing for this project was funded by the Alzheimer's Disease Neuroimaging Initiative (ADNI) (National Institutes of Health Grant U01 AG024904) and DOD ADNI (Department of Defense award number W81XWH-12-2-0012). ADNI is funded by the National Institute on Aging, the National Institute of Biomedical Imaging and Bioengineering, and through generous contributions from the following: AbbVie, Alzheimer’s Association; Alzheimer’s Drug Discovery Foundation; Araclon Biotech; BioClinica, Inc.; Biogen; Bristol-Myers Squibb Company; CereSpir, Inc.; Cogstate; Eisai Inc.; Elan Pharmaceuticals, Inc.; Eli Lilly and Company; EuroImmun; F. Hoffmann-La Roche Ltd and its affiliated company Genentech, Inc.; Fujirebio; GE Healthcare; IXICO Ltd.; Janssen Alzheimer Immunotherapy Research & Development, LLC.; Johnson & Johnson Pharmaceutical Research & Development LLC.; Lumosity; Lundbeck; Merck & Co., Inc.; Meso Scale Diagnostics, LLC.; NeuroRx Research; Neurotrack Technologies; Novartis Pharmaceuticals Corporation; Pfizer Inc.; Piramal Imaging; Servier; Takeda Pharmaceutical Company; and Transition Therapeutics. The Canadian Institutes of Health Research is providing funds to support ADNI clinical sites in Canada. Private sector contributions are facilitated by the Foundation for the National Institutes of Health (![]() ). The grantee organization is the Northern California Institute for Research and Education, and the study is coordinated by the Alzheimer’s Therapeutic Research Institute at the University of Southern California. ADNI data are disseminated by the Laboratory for Neuro Imaging at the University of Southern California.
). The grantee organization is the Northern California Institute for Research and Education, and the study is coordinated by the Alzheimer’s Therapeutic Research Institute at the University of Southern California. ADNI data are disseminated by the Laboratory for Neuro Imaging at the University of Southern California.

