Abstract
Congenital insensitivity to pain with anhidrosis (CIPA) is a rare autosomal recessive heterogeneous disorder mainly caused by mutations in the neurotrophic tyrosine receptor kinase 1 gene (NTRK1) and characterized by insensitivity to noxious stimuli, anhidrosis, and intellectual disability. We herein report the first north Han Chinese patient with CIPA who exhibited classic phenotypic features and severe intellectual disability caused by a homozygous c.851-33T>A mutation of NTRK1, resulting in aberrant splicing and an open reading frame shift. We reviewed the literature and performed in silico analysis to determine the association between mutations and intellectual disability in patients with CIPA. We found that intellectual disability was correlated with the specific Ntrk1 protein domain that a mutation jeopardized. Mutations located peripheral to the Ntrk1 protein do not influence important functional domains and tend to cause milder symptoms without intellectual disability. Mutations that involve critical amino acids in the protein are prone to cause severe symptoms, including intellectual disability.
Keywords
Introduction
Congenital insensitivity to pain with anhidrosis (CIPA, OMIM #256800), also known as hereditary sensory and autonomic neuropathy IV, is an autosomal recessive disorder caused by loss-of-function mutations in the neurotrophic tyrosine receptor kinase 1 gene (NTRK1, OMIM *191315), 1 nerve growth factor beta gene (NGFβ, OMIM *162030),2,3 and necdin gene (NDN, OMIM *602117). 4 The vast majority of mutations are observed in NTRK1. 5 CIPA is characterized by insensitivity to noxious stimuli, anhidrosis, and intellectual disability.2,6 Classic symptoms of CIPA include recurrent unexplained fevers without sweating, self-mutilating behavior (e.g., biting of the tongue, lips, and distal extremities), recurrent bone fractures and Charcot arthropathy, nonhealing ulcers and injuries, and psychomotor retardation.5,7,8 The pain insensitivity in patients with CIPA is caused by an absence of small-diameter thinly myelinated primary afferent or unmyelinated neurons, and the anhidrosis is caused by a deficiency of sympathetic postganglionic neurons.5,9
Phenotypic heterogeneity in the occurrence of intellectual disability has been reported in patients with CIPA. Ntrk1-knockout mice lack basal forebrain cholinergic neurons and striatal cholinergic neurons. 9 No autopsy data in humans with CIPA are available to validate these animal observations. Additionally, cranial magnetic resonance imaging has revealed no structural brain alterations in patients with CIPA. 10 We herein present a case involving a patient with CIPA with classic phenotypic features and severe intellectual disability caused by a previously reported homozygous c.851-33T>A mutation of NTRK1 gene. We performed a literature review and in silico analysis to determine the association between mutations and intellectual disability in patients with CIPA.
Methods
Patient
This study was approved by the Ethical Review Board of the Peking Union Medical College Hospital. Written informed consent was obtained from the patient’s parents.
A 27-year-old Han Chinese woman was referred to our hospital in 2013 and diagnosed with CIPA. Her clinical history was reviewed and a physical examination was performed. Her intelligence quotient was measured with the Wechsler Adult Intelligence Scale (WAIS IV, 2008 version). A nerve biopsy was not conducted because of the minimal benefit to the patient.
Genetic analysis
After obtaining informed consent, we drew 4 ml of peripheral blood from the patient and her parents. Genomic DNA was extracted with the TIANamp Genomic DNA Kit (Tiangen Biotech, Beijing, China) according to the manufacturer’s protocol.
All 17 exons and intron–exon boundaries were amplified by polymerase chain reaction (PCR) with primers used in previous reports.6,11,12 Sanger sequencing was conducted by Berry Genomics Co., Ltd. (Beijing, China). The sequences were analyzed with Chromas software (version 2.1.1) and BLAT to UCSC Human Genome (Feb 2009, GRCh37/hg19, http://genome.ucsc.edu/) to call mutations.
The oligonucleotide-based comparative genomic hybridization (CGH) microarray is an efficient and reliable assay for copy number variation studies. The Agilent 1x1M Human Genome CGH microarray (Agilent, Santa Clara, CA, USA) was employed in this study. DNA processing, microarray handling, and data analysis were conducted by following the Agilent oligonucleotide array CGH protocol (version 6.0).
Literature review
The MeSH terms “congenital insensitivity to pain with anhidrosis” and “ntrk1 receptor” were used to search for articles pertinent to CIPA caused by NTRK1 mutation. Title and abstract screening was conducted to retrieve genetic studies, which elucidated genetic mutations causing CIPA. All mutations reported up to October 2017 were reviewed. Furthermore, using the known Ntrk1 protein structure (available at the RCSB Protein Data Bank, http://www.rcsb.org/pdb/home/home.do) and PyMOL software (Version 4.3.0), we attempted to interpret the heterogeneity of the intellectual disability in patients with CIPA.
Results
Clinical findings
The patient in the present case was a 27-year-old Han Chinese woman (Figure 1(a), III:3) born to nonconsanguineous parents (Figure 1(a), II:6 and II:7). Her mother had a history of a terminated pregnancy (Figure 1(a), III:1) and fetal death at 7 months of gestation (Figure 1(a), III:2). The proband had a 27-year history of anhidrosis and insensitivity to pain and had experienced recurrent unexplained fevers (maximum body temperature, 42°C) during her first year of life. Bilateral dislocation of the hip joints had been present since age 2 years. Although her body development was normal, she showed cognitive impairment at a young age. Self-mutilating behavior was first observed at age 6 years and resulted in finger, ankle, tibia, hip, and femur fractures. She developed osteomyelitis due to poor healing following trauma or surgical treatment of fractures. She required a wheelchair for ambulation because of deformation of her right hip joint, right knee, and left ankle (Figure 1(b)). Her intelligence quotient was about 44 (WAIS IV, 2008 version). She showed emotional instability and communication difficulty. The clinical characteristics of patients with CIPA with the hotspot c.851-33T>A mutation are summarized in Table 1.

Patient’s pedigree and clinical and imaging characteristics. Panel A: Pedigree of the patient (P), who is indicated by an arrow. Panel B: Clinical characteristics and imaging of the proband. (a, b) Hypoplasia of the distal phalanges of both hands due to self-mutilation and osteomyelitis. (c) Deformity of the hip joint. (d–f) Malunion of the right knee. (g–i) Deformity and multiple fractures of the ankle and tibia. Panel C: Sequencing of NTRK1 reveals the homozygous c.851-33T>A mutation in the patient (P) and heterozygous c.851-33T>A mutation in her father (F) and mother (M).
Clinical characteristics of patients with CIPA with the c.851-33T>A mutation

All patients had the c.851-33T>A mutation. Pt., patient; F, female; M, male; Homo., homozygous; Co.Het., compound heterozygous; NA ,not available.
Genetic analysis
PCR followed by Sanger sequencing of NTRK1 was completed. Because previous reports have indicated that point mutations in the intron cause abnormal splicing and resultant CIPA, we repeated the analysis and confirmed the presence of a homozygous c.851-33T>A mutation in intron 7 in the patient and a heterozygous c.851-33T>A mutation in both of her nonconsanguineous parents (Figure 1, Panel C). The CGH microarray showed no evidence of copy number variation in her genome (data not shown), validating the homozygosity of the c.851-33T>A mutation. This mutation was previously verified to result in aberrant splicing and a reading frame shift and can reportedly cause CIPA.13–19 To our knowledge, this is the first reported patient homozygous for the c.851-33T>A mutation in the north Han Chinese population.
Literature review
Structure of Ntrk1 protein
Crystal structures of the extracellular and intracellular parts of the Ntrk1 protein were identified by Wehrman et al. 20 and Bertrand et al., 21 , respectively (Figure 2). The Ntrk1 protein contains 796 amino acids and consists of three leucine-rich repeats, two immunoglobulin-like domains (Ig-C1 and Ig-C2), a transmembrane domain, an adenosine triphosphate (ATP) binding site, and a tyrosine kinase domain (TKD). Once the nerve growth factor (NGF) homodimer binds to Ntrk1, it causes Ntrk1 dimerization and autophosphorylation of tyrosine residues 675, 679, and 680. 22 This initiates the cytoplasmic signaling cascade that is essential for the survival and growth of neurons. 23
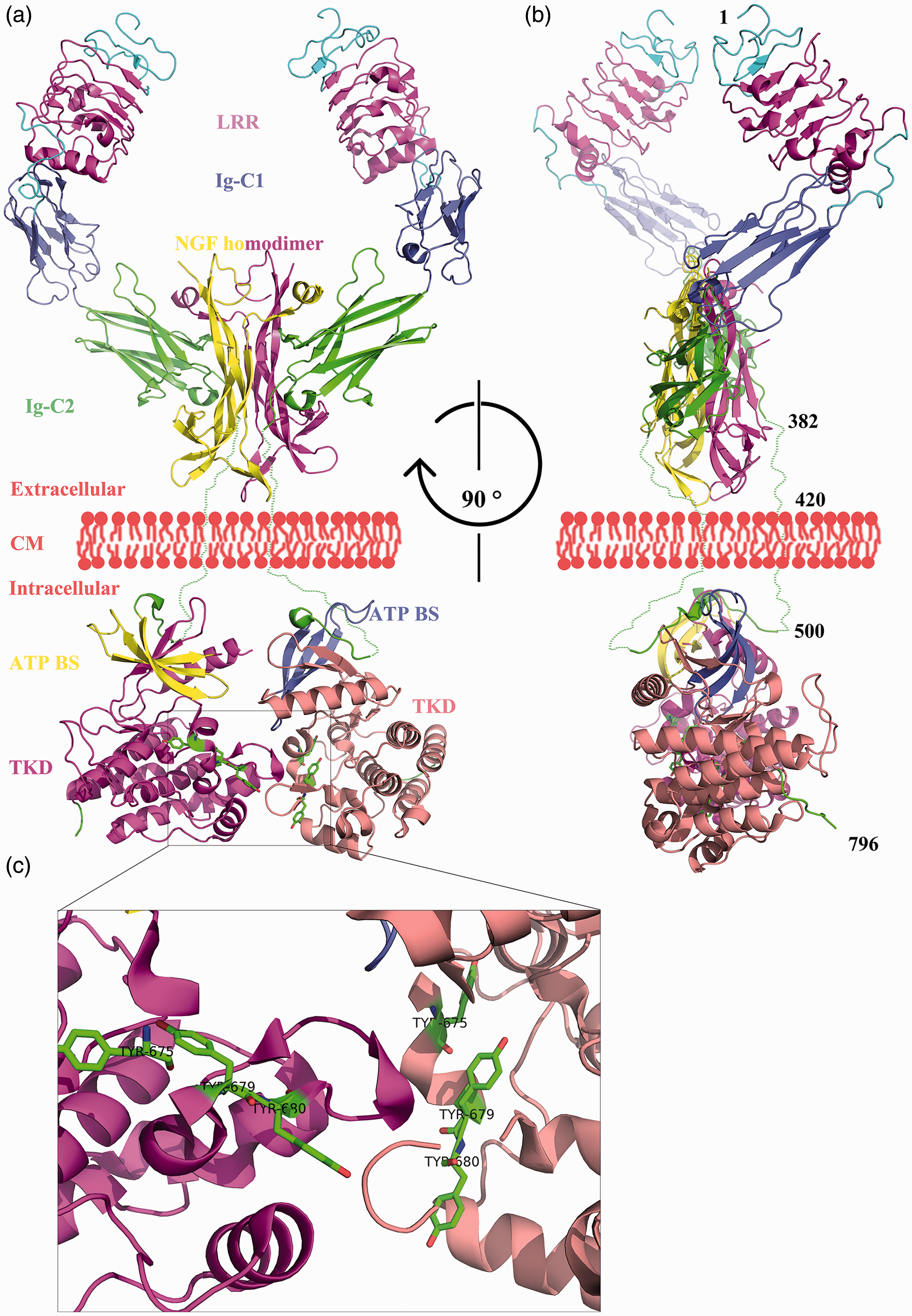
Whole structure of Ntrk1 dimer binding with NGF homodimer. Panel A: Front view. Panel B: Lateral view. Panel C: Details of the interfaces of the cytoplasmic domains and three tyrosine residues phosphorylated during signaling transduction. Stereo visualization is realized by shading the further parts of the molecule. Residue numbers were labeled approximately in Panel B. Dotted lines were drawn manually to connect the extracellular and intracellular portions because no exact crystal structure for the transmembrane domain is available. LRR, leucine-rich repeats; Ig-C1 and Ig-C2, immunoglobulin-like domains; NGF, nerve growth factor; CM, cytomembrane; ATP BS, adenosine triphosphate binding site; TKD, tyrosine kinase domain.
Heterogeneity of intellectual disability in patients with CIPA
According to our literature review, 90% (99/110) of previously reported patients with CIPA had documented intellectual disability. However, about 10% (11/110) of the patients did not have intellectual disability (44 additional patients with CIPA whose intellectual disability status was not mentioned in the literature were excluded from the statistical analysis). We attempted to determine whether an association exists among CIPA mutation, its effect on Ntrk1 protein, and the presence of intellectual disability (Table 2).
NTRK1 mutations reported in patients with CIPA with or without intellectual disability

CIPA, congenital insensitivity to pain with anhidrosis; Pt., patient; Homo., homozygous; Co.Het., compound heterozygous; Pr.D, probably damaging; Po.D, possibly damaging; D, damaging; T, tolerated; B, benign
Notes: Patients who were Homo. or Co.Het. with frameshift mutations, premature stop mutations, and verified splice site mutations were not included in this table because of their highly deleterious effect with all patients exhibiting intellectual disability.
*Polyphen2 and SIFT scores and predictions (Pr.D, Po.D, D, T, and B) are given only for missense mutation. Discordances (patients without intellectual disability who had highly deleterious missense mutations or those with intellectual disability who had at least one possibly damaging or tolerable mutation) are in italics.
Because of the alternative splicing of NTRK1, different nomination systems were employed among the literature. Here, we mapped these mutations to the protein structure, in which the residue number may differ from that in the corresponding article.
-Data not available for frameshift or intron mutations.
In silico investigation of heterogeneity
We hypothesized that if the mutation on one of the two NTRK1 alleles is benign or tolerable, the patient may have less severe symptoms. We used the Polyphen2 (Harvard Medical School, Boston, MA, USA; http://genetics.bwh.harvard.edu/pph2/) and SIFT (J. Craig Venter Institute, La Jolla, CA, USA; http://sift.jcvi.org/) scores to predict the influence of a mutation. As shown in Table 2, these predictions were not correlated with the patients’ phenotype with respect to intellectual disability, as shown in italics. We then examined whether any observable association was present between the mutations and intellectual disability with an available Ntrk1 protein structure.
Figure 3(a) shows missense mutations (Table 2, Patients 1–5) in patients with CIPA without intellectual disability. As shown in the table, the point mutations p.Leu213Pro, Arg760Trp, and p.Pro767Leu were all located peripherally without apparent involvement in NGF binding, ATP binding, or interfaces of the TKD. In addition, leucine and proline are of similar structure with respect to their nonpolar side chains, which may have less effect on protein function. Because CIPA is a recessive disease, one copy of near fully functional genes will relieve the patient’s symptoms. The premature stop mutation p.Tyr756Ter caused Ntrk1 protein truncation of 40 amino acids at the C-terminal. This part of the Ntrk1 protein is located at a region distal to the kinase interface, which may be another reason that these children were not intellectually compromised.

Observable association between mutations and the presence of intellectual disability. Panel A: At least one mutation reported in patients with CIPA without intellectual disability involved peripheral amino acids of the Ntrk1 protein. Panel B: Missense mutations in patients with CIPA with intellectual disability altered critical domains of the Ntrk1 protein. (Rotated by about 20º clockwise from the front view.)
Figure 3(b) shows missense mutations (Table 2, Patients 9–20) in patients with CIPA with apparent intellectual disability. All mutations were located in the functional domains; i.e., NFG binding site, ATP binding site, and interfaces of the TKD. Thus, we conclude that mutations located in functional domains of NTRK1 contribute to the observed intellectual disability.
In the present case, the c.851-33T>A mutation of NTRK1 caused a frame shift after the 283rd amino acid and a premature stop codon at the 319th codon, 41 deleting the whole transmembrane domain, ATP binding site, and TKD. This mutation is highly deleterious and would be expected to cause intellectual disability in this patient.
Discussion
To our knowledge, this is the first case of CIPA in a north Han Chinese patient caused by a homozygous c.851-33T>A mutation that resulted in aberrant splicing and an open reading frame shift. We also reviewed all published Ntrk1 CIPA mutations in the literature and postulate that the presence of intellectual disability is determined by the location of the Ntrk1 mutation with respect to the functional protein domain. Missense mutations located peripheral to the Ntrk1 protein that do not jeopardize important domains (i.e., NGF binding site, ATP binding site, and TKD) tend to cause milder symptoms, usually without intellectual disability. Frame shift mutations, premature stop mutations, splice site mutations, and missense mutations that involve critical amino acids in the protein all cause severe intellectual disability.
However, several reports appear to be discordant. Sarasola et al. 28 described a 6-year-old Spanish girl with c.574 + 1G>A and c.2206-2A>G compound heterozygous mutations (Table 2, Patient 6) who was cognitively normal. The author verified the influence of the two splice site mutations with reverse transcription PCR. The c.574 + 1G>A mutation caused exon 5 skipping and predicted Ntrk1 protein truncation after residue 164. The c.2206-2A>G mutation caused two alternative splicings, and the predicted proteins were Ala736_Gln742 interstitial deletion and truncation after residue 735, respectively. Tuysuz et al. 10 reported three Turkish patients with CIPA with a homozygous c.1633–1G>T mutation, one 2.5-year-old boy without psychomotor retardation, and one 14-year-old boy and one 8-year-old girl with psychomotor retardation (Table 2, Patients 7 and 8). This discrepancy indicates that there may be other factors modifying the phenotypes of CIPA. The functional impact of the variant, including dimerization, autophosphorylation, PLCγ activity, and toxicity of the mutated protein to the cell should be validated to confirm the pathogenicity of the variant.
Conclusions
In this study, we observed that intellectual disability in patients with CIPA appears to be determined by which Ntrk1 protein domain a mutation has jeopardized. Although this observation method is qualitative, it provides another alternative to evaluate the function of a missense mutation in addition to the Polyphen2 and SIFT scores. We are awaiting the delineation of additional cases of CIPA to provide additional evidence supporting our observations.
Footnotes
Acknowledgments
The authors thank Dr. Uluc Yis from the Division of Child Neurology, Department of Pediatrics, School of Medicine, Dokuz Eylul University, Izmir, Turkey for sharing their full-text article, which added rationality to our analysis.
Declaration of conflicting interests
The authors declare that there is no conflict of interest.
Funding
This research was funded by the National Natural Science Foundation of China (81501852, 81472046, 81472045), Beijing Natural Science Foundation (7172175), Beijing Nova Program (Z161100004916123), Beijing Nova Program Interdisciplinary Collaborative Project (xxjc201717), 2016 Milstein Medical Asian American Partnership Foundation Fellowship Award in Translational Medicine, The Central Level Public Interest Program for Scientific Research Institute (2016ZX310177), PUMC Youth Fund & the Fundamental Research Funds for the Central Universities (3332016006), CAMS Initiative Fund for Medical Sciences (2016-I2M-3-003, 2016-I2M-2-006), the Distinguished Youth Foundation of Peking Union Medical College Hospital (JQ201506), and the 2016 PUMCH Science Fund for Junior Faculty (PUMCH-2016-1.1).
