Abstract
Objective
To investigate the cardiometabolic effects of a severe hypothyroid state induced by withdrawal of thyroid hormone replacement before radioactive iodine therapy.
Methods
Patients with thyroid cancer who were scheduled to receive radioactive iodine ablation were enrolled. Cardiometabolic parameters were measured using blood samples taken immediately before levothyroxine withdrawal, 4 weeks following withdrawal (on radiotherapy day), and 4 weeks following reinstitution of levothyroxine.
Results
Out of 48 patients (age 49.4 ± 10.5 years; 77.1% [37/48] female), the severe hypothyroid state induced by levothyroxine withdrawal significantly aggravated the majority of lipid parameters, particularly in patients with a greater number of metabolic syndrome components. Fasting plasma glucose levels and homeostatic model assessment values for insulin resistance and β-cell function significantly decreased following levothyroxine withdrawal. Serum high-sensitivity C-reactive protein, fibrinogen and cystatin C levels significantly decreased, and homocysteine levels increased during the severe hypothyroid state. All of these changes were reversed by levothyroxine reinstitution.
Conclusions
Severe hypothyroid state induced pronounced changes in cardiometabolic parameters. Further studies should identify the long-term effects of changes in these parameters on cardiovascular morbidity and mortality in relation to thyroid disease.
Introduction
Hypothyroidism adversely affects cardiovascular morbidity and mortality,1,2 as thyroid hormone regulates lipid metabolism and cardiovascular haemodynamics. Hypothyroidism leads to lipid abnormalities that are characterized by hypercholesterolaemia with increased levels of low-density lipoprotein (LDL) and apolipoprotein B, due to reduced numbers of LDL receptors in the liver and subsequent decrease in LDL clearance. 3 Furthermore, hypothyroidism increases systemic vascular resistance and impairs cardiac contractility and diastolic function. 4 These changes in lipid profiles and haemodynamics are potentially associated with atherosclerosis and an increased risk of cardiovascular disease and mortality among patients with subclinical or overt hypothyroidism.5–7
Thyroid hormone status has also been demonstrated to affect various adipocytokines8–10 and atherogenic inflammatory markers,11–15 which may lead to additional harmful effects that promote the progression of cardiovascular disease. In addition, patients with hypothyroidism exhibit elevated serum levels of high-sensitivity C-reactive protein (hs-CRP),11,15 homocysteine,11–13 and fibrinogen,13,14 which are well-known biomarkers and independent risk factors for atherosclerosis. Serum levels of cystatin C are a marker for renal function and cardiovascular disease, and have been reported to exhibit changes according to thyroid function.16–18 Whether these markers are independently correlated with thyroid function and whether they can be reversed following treatment of thyroid dysfunction, however, remains controversial. In addition, the abovementioned studies have included heterogeneous populations with different severities, durations, and aetiologies of thyroid dysfunction, which might confound any analyses of the effects of thyroid hormone on these markers.
During radioactive iodine therapy following thyroidectomy for differentiated thyroid cancer, patients experience a severe hypothyroid state after thyroid hormone treatment withdrawal, and they subsequently recover once thyroid hormone treatment is reinstated.9,10 Such serial changes in thyroid hormone status may cause drastic changes in cardiometabolic parameters, and produce a representative and well-controlled group of patients with homogeneous thyroid hormone status.
The present study aimed to evaluate the effects of the severe hypothyroid state following thyroid hormone withdrawal on cardiometabolic and vascular inflammatory parameters among patients who underwent radioactive iodine therapy after thyroidectomy, and to identify any patient subgroups who experienced more pronounced changes in these parameters.
Patients and methods
Study population and design
This prospective cohort study enrolled consecutive patients with differentiated thyroid cancer who were scheduled to receive radioactive iodine ablation following total thyroidectomy at Konkuk University Medical Center, Seoul, Korea between May 2012 and May 2013. Patients with chronic disease (including kidney, liver, and heart failure), acute infection or inflammatory conditions, or who were receiving drugs for diabetes mellitus, hypertension, dyslipidaemia, or other conditions that might affect metabolic parameters were excluded.
Blood samples were obtained at three time points to identify serial changes in various metabolic parameters (Figure 1): Visit 1, immediately before withdrawal of levothyroxine in preparation for radioactive iodine ablation (a mild thyrotoxic state due to receiving levothyroxine for thyroid stimulating hormone [TSH] suppression); Visit 2, day of radioactive iodine ablation following 4 weeks of levothyroxine withdrawal (a severe hypothyroid state with TSH levels ≥30 mU/l); and Visit 3, at 4 weeks following reinstitution of levothyroxine (recovery to a mild thyrotoxic state).
Schematic of study design in patients with differentiated thyroid cancer who received radioactive iodine ablation following total thyroidectomy. LT4, levothyroxine; RAI, radioactive iodine; TSH, thyroid stimulating hormone.
The study protocol was reviewed and approved by the institutional review board of Konkuk University Medical Center (KUH1010507), and this study was registered in the ClinicalTrial.gov registry (NCT01744769). All participants provided written informed consent.
Outcome measures
Blood pressure and heart rate were recorded three times between 7:00 and 9:00 h after the patients had been in a relaxed state for ≥10 min, and a 5-min rest period was provided between each measurement. The mean of three readings was used for analysis.
Between 7:00 and 9:00 h, and following a 14 h overnight fast, 20 ml of venous blood was drawn from the antecubital vein of each patient into tubes without anticoagulant and tubes containing ethylenediaminetetra-acetic acid-2Na (1 mg/ml blood). Plasma was immediately separated by centrifugation at 2000
Biochemical measurements of cardiometabolic parameters were conducted immediately following sample preparation. Fasting plasma glucose (FPG), serum triglycerides, free fatty acid, homocysteine, cystatin C, fibrinogen, hs-CRP, total cholesterol, high-density lipoprotein cholesterol (HDL-C), LDL cholesterol (LDL-C), and apolipoprotein A and B levels were measured using a Toshiba 200FR automatic analyser (Toshiba Medical System Co., Ltd, Tokyo, Japan) according to the manufacturer’s instructions. Plasma insulin levels were determined using a MODULAR analytics E170 module (Roche Diagnostics, Manheim, Germany) and glycosylated haemoglobin (HbA1c) levels were measured using a VARIANT II TURBO 2.0 kit (Bio-Rad Laboratories, Hercules, CA, USA), both according to the manufacturer’s instructions. Free thyroxine and TSH were measured using an automatic SR300 radioimmunoassay analyser (STRATEC Biomedical AG, Birkenfeld, Germany) and associated reagents, according to the manufacturer’s instructions.
Primary outcome measures were changes in cardiometabolic parameters, comprising blood lipids (total cholesterol, LDL-C, HDL-C, triglycerides, free fatty acids, and apolipoproteins A and B), glycaemic parameters (FPG, fasting insulin, HbA1c, and homeostatic model assessment values for insulin resistance and β-cell function [HOMA-IR and HOMA-B], and vascular inflammation and atherosclerosis markers (uric acid, hs-CRP, fibrinogen, homocysteine, and cystatin C), according to thyroid hormone status.
Secondary outcome measures were the correlation between TSH levels and metabolic parameters, and the baseline characteristics that predicted changes in metabolic parameters. The signs and symptoms of hypothyroidism were assessed using the Zulewski score as previously described, 19 and metabolic syndrome was defined according to the National Cholesterol Education Program (NCEP) Adult Treatment Panel III report. 20
Statistical analyses
All normally distributed continuous variables are presented as mean ± SD, and categorical variables as presented as
Results
Baseline characteristics
Baseline characteristics of patients with differentiated thyroid cancer who were scheduled to receive radioactive iodine ablation following total thyroidectomy.

Data presented as mean ± SD or
Changes in metabolic parameters according to changes in thyroid hormone status
Change of metabolic parameters in patients with differentiated thyroid cancer who received radioactive iodine ablation following total thyroidectomy (

Data presented as mean ± SD.
ALT, alanine aminotransferase; AST, aspartate aminotransferase; HDL, high-density lipoprotein; LDL, low-density lipoprotein; HbA1c, glycosylated haemoglobin; HOMA-IR, homoeostasis model assessment for insulin resistance; HOMA-B, homoeostasis model assessment for β-cell function; hs-CRP, high-sensitivity C-reactive protein.
Changes metabolic syndrome components during a severe hypothyroid state
Most metabolic syndrome components were aggravated following levothyroxine withdrawal, with the exception of glycaemic profiles during the severe hypothyroid phase. Thus, the proportions of patients who fulfilled the metabolic syndrome criteria for each related component, based on the NCEP Adult Treatment Panel III definition of metabolic syndrome,
20
were analysed according to changes in thyroid hormone status (Figure 2). Among the metabolic syndrome components, proportions of male and female patients with high triglyceride levels (≥150 mg/dl) increased significantly during the severe hypothyroid state (Visit 2), however, proportions of other metabolic syndrome components, such as high blood pressure, high FPG levels (among female patients), and high HDL-C levels decreased during the hypothyroid state ( Serial changes in the proportion of patients with metabolic syndrome components associated with withdrawal and reinstitution of thyroid hormone in patients with differentiated thyroid cancer who received radioactive iodine ablation following total thyroidectomy (
Correlation between TSH levels and metabolic parameters following thyroid hormone withdrawal
The correlation between TSH levels and the metabolic parameters that changed during the severe hypothyroid state were further analysed. During the severe hypothyroid state (Visit 2) TSH levels exhibited a significant positive correlation with LDL-C levels ( Correlation between thyroid stimulating hormone (TSH) levels and metabolic parameters in patients with differentiated thyroid cancer who received radioactive iodine ablation following total thyroidectomy (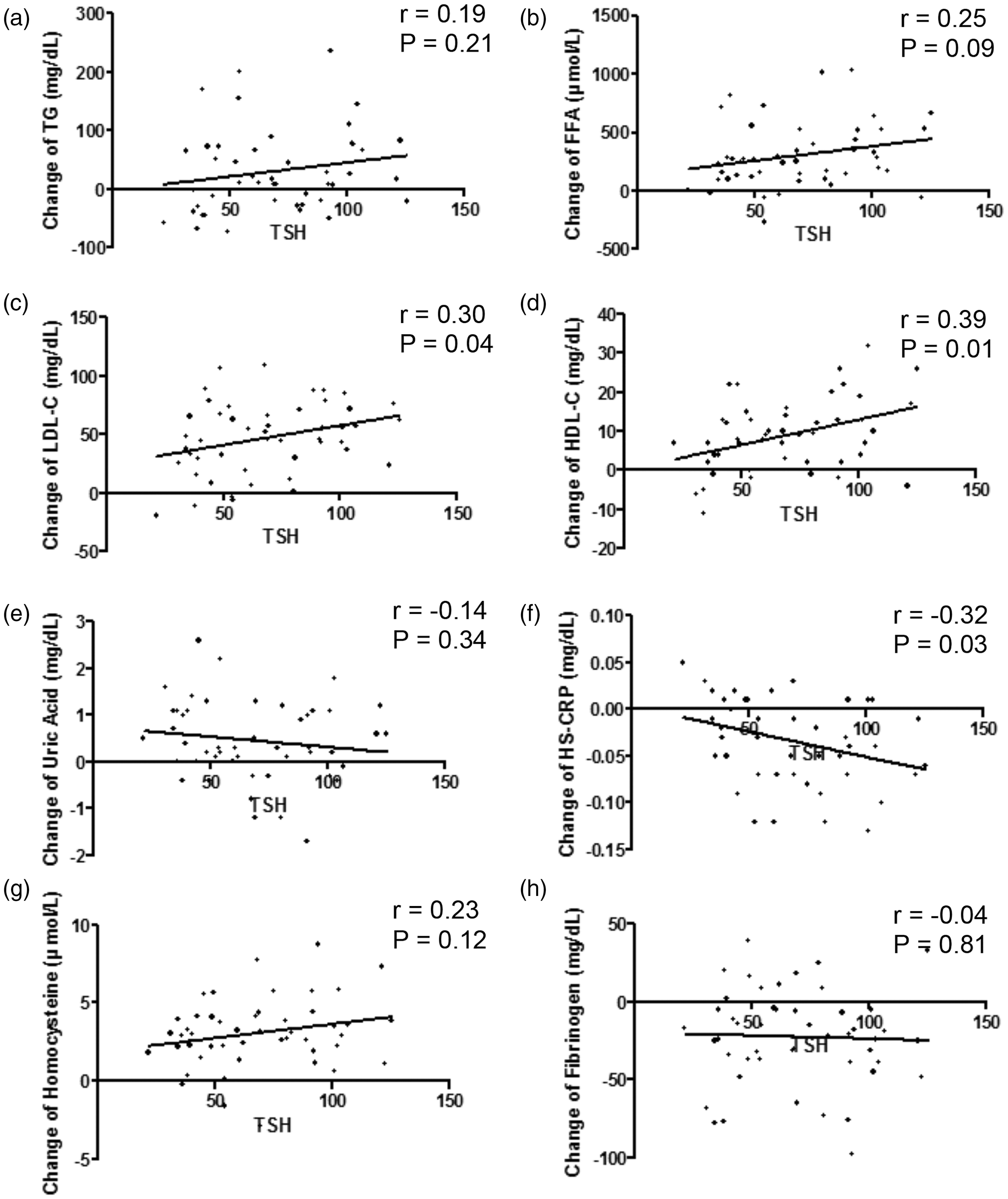
Baseline characteristics of patients with different metabolic responses following thyroid hormone withdrawal
Comparison of baseline characteristics between upper and lower tertiles grouped according to change of lipid profiles during levothyroxine withdrawal in patients with differentiated thyroid cancer who received radioactive iodine ablation following total thyroidectomy (

Data presented as mean ± SD or
TSH, thyroid stimulating hormone; ALT, alanine aminotransferase; AST, aspartate aminotransferase; HDL, high-density lipoprotein; LDL, low-density lipoprotein; hs-CRP, high-sensitivity C-reactive protein.
Discussion
In the present study among patients who underwent total thyroidectomy for differentiated thyroid cancer, severe hypothyroidism due to thyroid hormone withdrawal produced noticeable changes in various cardiometabolic parameters, particularly among patients with underlying metabolic syndrome. Interestingly, these changes were fully reversed following reinstitution of thyroid hormone treatment. Furthermore, the present study revealed the direction of changes in these metabolic parameters, which have been reported with conflicting results in previous studies.15,21
As expected, a severe hypothyroid state significantly aggravated the majority of lipid parameters that were evaluated in the present study, such as levels of LDL-C, triglycerides, free fatty acids and apolipoprotein B. In addition, patients who had a greater number of metabolic syndrome components prior to levothyroxine withdrawal subsequently experienced greater changes in these parameters during the severe hypothyroid state. Furthermore, changes in LDL-C levels were significantly correlated with TSH levels, although not with triglycerides and free fatty acids levels. The results of the present study concur with previously published findings. For example, differences in lipid profiles according to thyroid function have been reported, 15 with levels of total cholesterol, triglycerides, LDL-C, and apolipoprotein B increasing in patients with hypothyroidism and decreasing in patients with hyperthyroidism. TSH levels were also reported to be significantly correlated with changes in triglycerides, apolipoprotein B, and LDL-C levels, 15 although this relationship was not observed in the present study. The discrepancy between the present and published results may be related to the fact that correlations between TSH levels and lipid parameters were examined during a severe hypothyroid state in the present study, when TSH levels were 20–125 mU/l. HDL-C levels were also elevated during the hypothyroid state in the present study, and may have resulted from decreased hepatic lipase and cholesterol-ester transport protein activity, leading to decreased HDL catabolism, reduced transfer of cholesteryl esters from HDL2 to very low- and intermediate-density lipoproteins, reduced transport of HDL2 to HDL3, and ultimately increased levels of HDL2. 21
Several studies have reported that thyroid hormone replacement exerts favourable effects on lipid profiles and haemodynamic factors, which may eventually reduce the risk of cardiovascular disease morbidity and mortality. Levothyroxine replacement in patients with hypothyroidism reduces body weight 22 and improves various haemodynamic factors, such as central arterial stiffness and endothelium-dependent vasodilatation. 23 In addition, atherogenic lipid levels (e.g., total cholesterol, LDL-C, triglycerides, and apolipoprotein B) are reported to be reduced following levothyroxine replacement in patients with overt hypothyroidism. 24 Studies have also reported, however, that levothyroxine replacement does not affect these cardiometabolic parameters among patients with subclinical disease.24,25 In the present study, levothyroxine reinstitution fully reversed the changes in cardiometabolic parameters that were induced by short-term levothyroxine withdrawal, although further studies are needed to determine if levothyroxine has different effects in the acute and chronic hypothyroid states.
In addition to lipid profiles, changes in atherogenic markers (i.e., homocysteine, CRP, fibrinogen, and cystatin C levels) associated with the withdrawal and reinstitution of levothyroxine were also investigated in the present study. Plasma levels of homocysteine levels were observed to be significantly elevated during the severe hypothyroid state, and levothyroxine replacement reversed this elevation. Interestingly, homocysteine is an atherogenic factor that promotes the production of vascular inflammatory chemokines, reactive oxygen species, and oxidized LDL-C.26,27 A meta-analysis has also revealed that plasma homocysteine levels are elevated in patients with hypothyroidism, compared with those in healthy individuals, although levothyroxine replacement can reverse this elevation. 28 Another study has suggested that these hypothyroidism-induced elevated homocysteine levels may induce insulin resistance and increase the risk of cardiovascular disease. 12 In the present study, however, HOMA-IR was significantly decreased during the hypothyroid state.
Although homocysteine levels and the majority of lipid parameters exhibited unfavourable changes during the severe hypothyroid state in the present study, favourable changes in blood pressure, FPG levels, and atherosclerosis markers (e.g., hs-CRP, fibrinogen, and cystatin C levels) were observed during this state. Furthermore, HOMR-B was shown to be reduced during the severe hypothyroid state. Although several studies have reported increased insulin resistance in cases of hypothyroidism,12,29 the present results are consistent with findings of one study that showed a 7.4% decrease in FPG levels in cases of overt hypothyroidism compared with cases of subclinical disease, and that the decrease was correlated with reduced insulin resistance. 10 Interestingly, hypothyroidism induces reductions in energy expenditure and metabolic rate, which may lead to lower levels of FPG and insulin. 30
Previous studies have reported conflicting results regarding changes in CRP, fibrinogen and cystatin C levels among patients with hypothyroidism.11,13–17 These factors are well-known inflammatory markers of cardiovascular disease, and many of the previous studies have focused on the increase in these markers and their association with the risk of cardiovascular disease during the hypothyroid state.11,14,16 Thus, the favourable changes in hs-CRP, fibrinogen, and cystatin C levels observed during the hypothyroid state in the present study are difficult to interpret. Nevertheless, the authors speculate that the low-iodine diet recommended for patients undergoing radioactive iodine therapy could affect these parameters, and that acute withdrawal of levothyroxine may induce different responses in these inflammatory markers, compared with the responses observed during chronic hypothyroidism. 31
The present study has several clinical implications. First, patients with hyperlipidaemia should be tested to determine their thyroid function, and should be actively treated using thyroid hormone if hypothyroidism is detected, as levothyroxine reinstatement restored lipid profiles to their baseline state within 4 weeks. Furthermore, patients with baseline metabolic syndrome components experienced pronounced changes in their lipid profiles after levothyroxine withdrawal in the present study, which suggests that these patients should be carefully assessed before withdrawing levothyroxine in preparation for radioactive iodine therapy. Given that these changes were fully reversed following levothyroxine reinstitution, however, and that several cardiometabolic markers exhibited favourable changes during the severe hypothyroid state, short-term withdrawal of levothyroxine may not significantly affect the development of cardiovascular disease.
The results of the present study may be limited by the relatively small sample size and the fact that this was a single centre study. Further multicentre studies should be conducted, with larger sample sizes. In addition, further studies are needed to identify the long-term effects of changes in these parameters on cardiovascular morbidity and mortality in relation to thyroid disease.
In conclusion, levothyroxine withdrawal-related hypothyroidism induced pronounced changes in various cardiometabolic parameters among patients who had undergone total thyroidectomy and were preparing to undergo radioactive iodine therapy. These changes may increase the risk of atherosclerosis and cardiovascular diseases, especially among patients with metabolic syndrome components, although these changes were rapidly reversed after reinstituting thyroid hormone treatment.
Footnotes
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This study was supported by a grant from the Korean Society of Lipidology and Atherosclerosis.
