Abstract
Objective
To investigate whether levels of asymmetric dimethylarginine (ADMA), as a measure of endothelial dysfunction, are higher in patients with rheumatoid arthritis compared with healthy control subjects. The relationships between ADMA and surrogate measures of arterial stiffness were evaluated.
Methods
Patients with rheumatoid arthritis and healthy control subjects were recruited. ADMA was quantified via enzyme-linked immunosorbent assay. Arterial stiffness was evaluated using pulse wave analysis.
Results
There was no significant difference in plasma ADMA concentration between patients with rheumatoid arthritis (n = 30) and healthy controls (n = 30). Aortic augmentation pressure was significantly higher in patients than in controls. C-reactive protein and Health Assessment Questionnaire score were independent predictors of arterial stiffness in patients. There was no relationship between ADMA concentration and aortic augmentation pressure in the study population as a whole.
Conclusions
Arterial stiffness appears to be increased in rheumatoid arthritis and independently associated with systemic inflammation and physical disability. ADMA concentration was not increased in this small group of patients with rheumatoid arthritis compared with healthy controls; nor was it associated with arterial stiffness.
Keywords
Introduction
Arterial stiffness is influenced by endothelial dysfunction in both healthy individuals and patients with cardiovascular disease.1,2 Little is known regarding the prevalence of endothelial dysfunction and its relationship with arterial stiffness in patients with rheumatoid arthritis. The aim of the present study was to investigate whether levels of asymmetric dimethylarginine (ADMA), as a measure of endothelial dysfunction, 3 are higher in patients with rheumatoid arthritis compared with healthy control subjects. In addition, the relationships between ADMA and surrogate measures of arterial stiffness were evaluated.
Patients and methods
Study population
Consecutive patients with rheumatoid arthritis who met the criteria of the American College of Rheumatology 4 and had no history of cardiovascular events were enrolled from the outpatient clinic of the Rheumatology Unit, Department of Clinical and Experimental Medicine, Azienda Ospedaliero-Universitaria of Sassari and University of Sassari, Sassari, Italy, between January and May 2007. Collected data included: presence of conventional cardiovascular risk factors; treatment with aspirin, or antihypertensive or cholesterol-lowering drugs; duration of rheumatoid arthritis; steroid treatment; type and dosage of antitumour necrosis factor (TNF)-α inhibitor treatment; C-reactive protein (CRP) concentration; erythrocyte sedimentation rate; immunoglobulin M rheumatoid factor; anticyclic citrullinated peptide; Disease Activity Score (DAS); 5 Ritchie Index; 6 and Health Assessment Questionnaire (HAQ). 7
Control subjects strictly matched for conventional cardiovascular risk factors (using echo colour Doppler carotid ultrasound), who were attending routine check-ups at the outpatient clinic of the Cardiology Unit, Department of Clinical and Experimental Medicine, Azienda Ospedaliero-Universitaria of Sassari and University of Sassari, were also enrolled. The protocol was approved by the ethics committee of the University of Sassari and all participants provided written informed consent prior to enrolment.
Study parameters
Whole blood was collected into sterile tubes containing ethylenediaminetetra-acetic acid (EDTA)-potassium, then centrifuged in a Jouan centrifuge at 1000g for 10 min, and the resulting plasma was stored at −20°C until analysis. Plasma ADMA was quantified using enzyme-linked immunosorbent assay (ELISA; DLD Diagnostika GmbH, Hamburg, Germany), according to the manufacturer’s instructions. Arterial stiffness was evaluated by pulse wave analysis as described, 8 using a high-fidelity hand-held tonometry probe (Millar pressure tonometer, PWV Medical, Sydney, Australia). Registered radial pulse waveforms were recorded using a computer-based pulse wave analysis system (SphygmoCor®, PWV Medical, Sydney, Australia).
Statistical analyses
Data were presented as mean ± SD or n (%) of participants. Between-group comparisons were made using Student’s t-test, Kolmogorov–Smirnov test or χ2-test, as appropriate. Bivariate relationships were analysed using nonparametric Spearman’s test. A two-sided P-value ≤ 0.05 was considered statistically significant. Data were analysed using SPSS® version 11.0 (SPSS Inc., Chicago, IL, USA) for Windows®.
Results
Demographic and clinical characteristics of patients with rheumatoid arthritis and healthy control subjects included in a study to investigate the relationships between asymmetric dimethylarginine concentrations and surrogate measures of arterial stiffness.

Data presented as mean ± SD or n (%) of patients.
ESR, erythrocyte sedimentation rate; HDL, high density lipoprotein; LDL, low density lipoprotein.
P = 0.04 versus healthy controls; χ2-test.
Plasma asymmetric dimethylarginine concentration and arterial stiffness parameters in patients with rheumatoid arthritis and healthy control subjects.
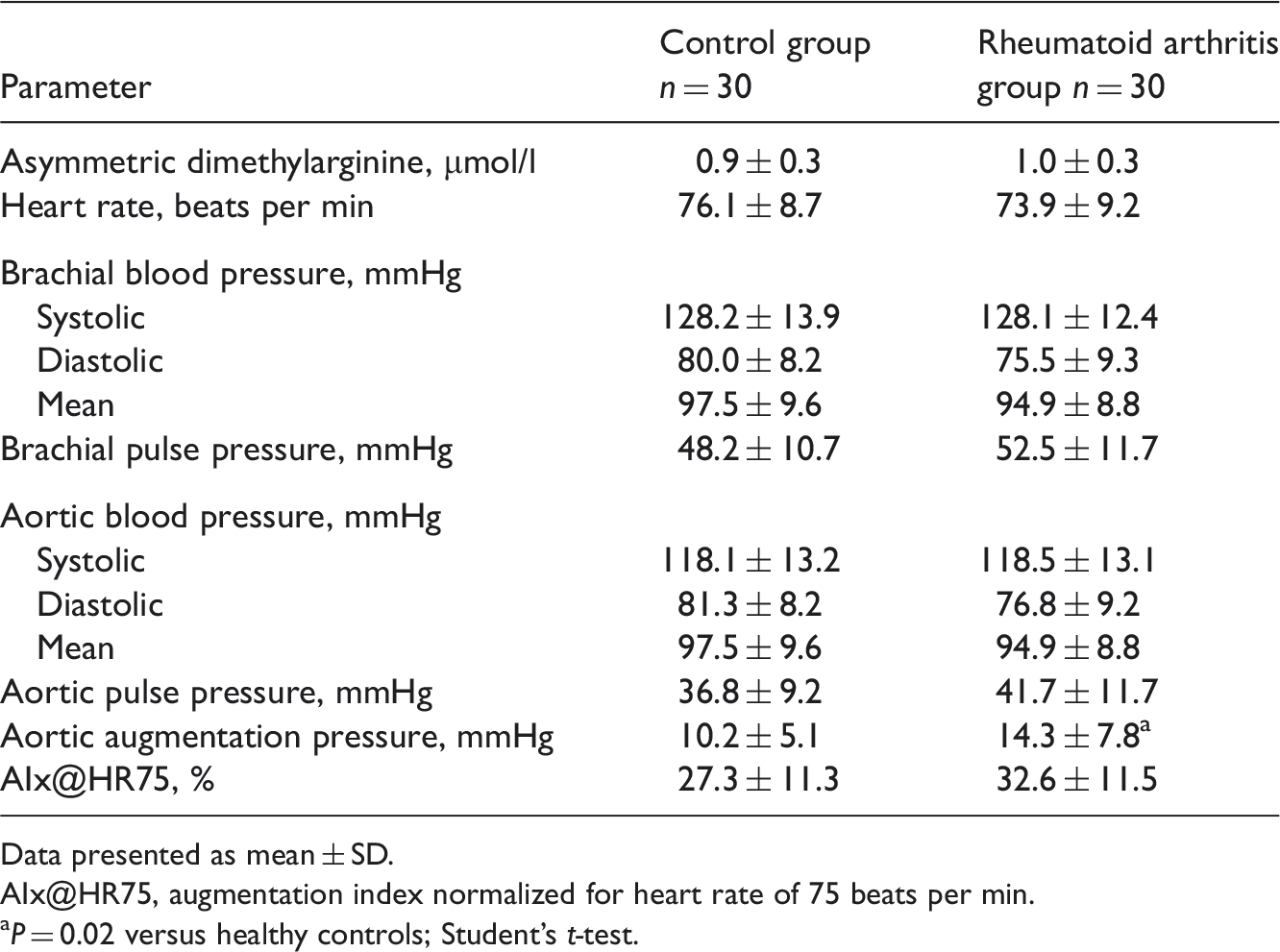
Data presented as mean ± SD.
AIx@HR75, augmentation index normalized for heart rate of 75 beats per min.
P = 0.02 versus healthy controls; Student’s t-test.
Bivariate analysis of the study population as a whole found significant associations between plasma ADMA concentration and age (P = 0.002) and aortic augmentation pressure (P = 0.015). Bivariate analysis of the patient group showed significant associations between ADMA concentration and cumulative steroid dose (P = 0.025), Ritchie Index (P = 0.018) and DAS (P = 0.025), and significant associations between aortic augmentation pressure and age (P = 0.013), CRP concentration (P = 0.0001) and HAQ score (P = 0.004). Multiple linear regression analysis found that CRP concentration and HAQ score alone were independently associated with aortic augmentation pressure in the patient group (P = 0.0001).
Discussion
Plasma ADMA concentrations were not elevated in patients with rheumatoid arthritis compared with control subjects in the present study. This is in contrast to the findings of other studies, which have reported significantly higher ADMA concentrations in treatment-naïve patients with rheumatoid arthritis, compared with controls. 9 A small prospective study found a significant reduction in ADMA concentrations after 3 months’ anti-TNF-α treatment. 10 It is therefore possible that the absence of a significant between-group difference in ADMA concentration in the present study may be a result of long-term treatment with anti-TNF-α drugs in our patient group. It should be noted that, as the range of biological variation of ADMA is extremely narrow both in health and disease, 11 the sample sizes of our study and other studies might be too small to draw firm conclusions regarding ADMA concentrations in rheumatoid arthritis. Furthermore, heterogeneity in ADMA quantification methods and in enrolled patient populations may further explain these apparently contradictory results.
Arterial stiffness is related to systemic inflammation and is elevated in patients with rheumatoid arthritis compared with healthy controls. 12 There was a statistically significant increase in arterial stiffness in patients compared with controls in the present study. In addition, the inflammatory marker CRP was a major predictor of arterial stiffness in our patient group. We found no relationship between ADMA concentration and aortic augmentation pressure (as a measure of arterial stiffness) in either the study population as a whole or in the patient group alone. This may be due to the small sample size of our study.
In conclusion, arterial stiffness appears to be increased in rheumatoid arthritis and is independently associated with systemic inflammation and physical disability. ADMA concentration was not increased in patients with rheumatoid arthritis; nor was it associated with arterial stiffness.
Footnotes
Declaration of conflicting interest
The authors declare that there are no conflicts of interest.
Funding
Editorial assistance was provided by Gayle Robins on behalf of HPS–Health Publishing and Services Srl and funded by Pfizer Italia.
