Abstract
Objective
To investigate the incidence and risk factors for emergence agitation in the postanaesthetic care unit (PACU), in adult patients undergoing urological surgery.
Methods
Medical records were retrospectively reviewed. Preoperative, intraoperative and postoperative variables were evaluated. Emergence agitation was defined as a Riker sedation–agitation score ≥5. Logistic regression analysis was used to determine independent risk factors for emergence agitation.
Results
Emergence agitation was observed in 48/488 (9.8%) patients. Chronic lung disease (odds ratio [OR] 2.72, 95% confidence interval [CI] 1.03, 7.17), duration of surgery (OR 1.01, 95% CI 1.00, 1.01), history of social drinking (OR 2.48, 95% CI 1.25, 4.93), postoperative pain score (OR 1.32, 95% CI 1.14, 1.53), voiding urgency (OR 2.20, 95% CI 1.01, 4.77) and presence of gastric tube (OR 2.85, 95% CI 1.07, 7.54) were independent risk factors for emergence agitation.
Conclusions
Adequate postoperative pain management and prevention of catheter-related bladder discomfort may be helpful in reducing the incidence of emergence agitation in urology patients.
Introduction
Emergence agitation in the postanaesthesia care unit (PACU) is a common complication after general anaesthesia, and can necessitate physical or chemical restraint in order to avoid serious consequences for the patient (e.g. physical injury, increased pain, haemorrhage, removal of catheters). 1 Considerable effort by the entire medical team is required to ensure the safety of both the patient with emergence agitation and their attending staff members. 2 Postoperative emergence agitation is associated with cognitive deficit, and increased physical dependence, duration of hospital stay and in-hospital mortality. 3
Few studies have examined emergence agitation in adults, and there are no diagnostic criteria or clear definitions for this condition due to its heterogeneous clinical presentation. 4,5 Identified risk factors for emergence agitation in adults include the use of doxapram, premedication with benzodiazepines, anaesthetic induction with etomidate, severe postoperative pain, oral cavity and otolaryngological surgery, breast surgery, abdominal surgery, extended surgical duration and presence of a tracheal tube or urinary catheter. 1,4,5 The nature of urological surgery requires the postoperative insertion of urinary catheters in the majority of patients, and such devices are associated with complications including catheter-related bladder discomfort (CRBD; defined as voiding urgency), 6,7 which can be a risk factor for emergence agitation. 1 Little data are available regarding the risk of emergence agitation after urological surgery specifically, since the majority of studies include such patients as a subgroup in a larger surgical study population. 1,5
The aim of this retrospective study was to determine the incidence of emergence agitation in adult patients after general anaesthesia for urological surgery. The association between emergence agitation and CRBD was examined, and additional perioperative variables were evaluated as risk factors for emergence agitation in this patient group.
Patients and methods
Study population
This retrospective study included adult patients (aged 20–90 years) with American Society of Anesthesiologists (ASA) physical status 1–3, admitted to the PACU after elective urological surgery under general anaesthesia with an endotracheal tube or a supraglottic airway device at Seoul National University Hospital, Seoul, Republic of Korea between March 2012 and August 2012. Patients’ electronic medical records were reviewed by two board-certified anaesthetists (E.K. and H.C.K.) and one resident anaesthetist (J.H.S.). Exclusion criteria were: neurological disease potentially associated with symptoms of agitation or intellectual disability; transfer to intensive care unit.
The study was approved by the Institutional Review Board of Seoul National University Hospital, Seoul, Republic of Korea, and the requirement for patient consent was waived.
General anaesthesia
All patients received controlled mechanical ventilation during general anaesthesia; no patient was premedicated. Anaesthesia was induced with 2 mg/kg propofol or 5 mg/kg thiopental and maintained with: (i) inhalation anaesthesia (1–4 vol% sevoflurane or 4–8 vol% desflurane, and intermittent 0.5–1 µg/kg fentanyl injection); or (ii) intravenous anaesthesia (continuous effect site concentration: 3–5 µg/ml propofol and effect site concentration: 2–5 ng/ml remifentanil infusion using a target-controlled infusion device [Orchestra; Fresenius-Vial, Brezins, France]). Intubation was facilitated via 0.8 mg/kg rocuronium intravenous injection or I-gel® (Intersurgical, Wokingham, UK; used in some patients undergoing endoscopic surgery such as transurethral resection of bladder tumour). Neuromuscular blockade was maintained via intermittenr 0.03–0.05 mg/kg vecuronium. About 20 min before the end of surgery, 30 mg ketorolac was administrated for pain relief in patients without a patient-controlled analgesia (PCA) device. The majority of patients with a PCA received 0.3 mg ramosetron (majority of patients) or 0.075 mg palonosetron (three patients) and opioid-based intravenous PCA (20–30 µg/kg fentanyl and 0.3 mg ramosetron; total volume 100 ml, loading 1 ml, basal infusion rate 1–2 ml/hr, bolus 0.5–1 ml, lockout time 15 min). At the end of surgery, residual neuromuscular blockade was reversed with 0.05 mg/kg neostigmine and 0.02 mg/kg atropine. Most patients were extubated in the operating room and transferred to the PACU. No patient received epidural catheterization for intraoperative or postoperative pain control.
Postoperative assessment
Emergence agitation was assessed in the PACU by attending anaesthetic nurses using the Riker sedation–agitation score. 8 Emergence agitation was defined as a Riker score ≥5 at any time in the PACU. Postoperative pain was assessed by attending anaesthetic nurses using an 11-point numerical rating scale (0, no pain; 10, worst imaginable pain) on admission to (and discharge from) the PACU. Pain score was also assessed whenever patients asked for pain medication. If the pain score was ≥4, 50–100 µg fentanyl was administered as rescue medicine. The maximum pain score was included in the analysis. Nausea, vomiting, shivering and dizziness were also assessed at the time of pain assessment in PACU. Patients with moderate-to-severe nausea and vomiting were given 4 mg ondansetron (patients without intraoperative ramosetron administration) or 1 mg/kg dexamethasone (patients with intraoperative ramosetron administration). Patients with moderate-to-severe shivering were given 25 mg pethidine. Haemodynamic and respiratory parameters were assessed to find correctable factors when severe dizziness was detected. Patients were discharged from the PACU if they were conscious, had a pain score of ≤3, stable vital signs for 30 min and no shivering, bleeding, nausea or vomiting.
Data collection
Data were retrieved from patient electronic medical charts and categorized into pre-, intra- and postoperative variables. Preoperative variables included: demographics; ASA physical status; history of smoking and social drinking; educational state; nil-by-mouth duration; serum albumin; and surgical history within 3 months. Intraoperative variables included: surgical site; endoscopic surgery (such as transurethral resection of ureter stones, or bladder and prostate lesions); anaesthetic induction agent (propofol or thiopental); airway instrument (endotracheal tube or I-gel®); anaesthesia maintenance agent (sevoflurane or desflurane or intravenous anaesthesia); nitrous oxide use; antiemetic use; transfusion; remifentanil continuous infusion; total intraoperative opioid dose (remifentanil dose was converted to equivalent dose of fentanyl with a ratio of 1 : 19); duration of surgery; duration of anaesthesia. Postoperative variables included: postoperative pain score; voiding urgency; nausea; vomiting; shivering; dizziness. The presence of a tracheal tube, urinary catheter and gastric tube in PACU were also noted.
Statistical analyses
The incidence of emergence agitation in patients undergoing urological surgery has been reported as 4.1% in. 1 The present study required a minimum of 467 patients to reproduce the incidence of emergence agitation, with a two-sided 95% confidence interval width of 3.6%.
Data were presented as mean ± SD, n (%) or median (interquartile range [IQR]). Patients were stratified according to the presence or absence of emergence agitation. Student’s t-test and χ2 or Fisher’s exact test (if the number in a cell was less than five) were used for between-group comparisons of continuous and categorical variables, respectively. Variables with P-values <0.1 in the primary test were included in binary logistic regression to identify independent risk factors for emergence agitation. Statistical analyses were performed using SPSS® version 19.0 (SPSS Inc., Chicago, IL, USA) for Windows®. P-values <0.05 were considered to indicate statistical significance.
Results
Distribution of Riker sedation–agitation scores 8 in 488 adults after general anaesthesia for urological surgery. Emergence agitation is defined as a score ≥5.
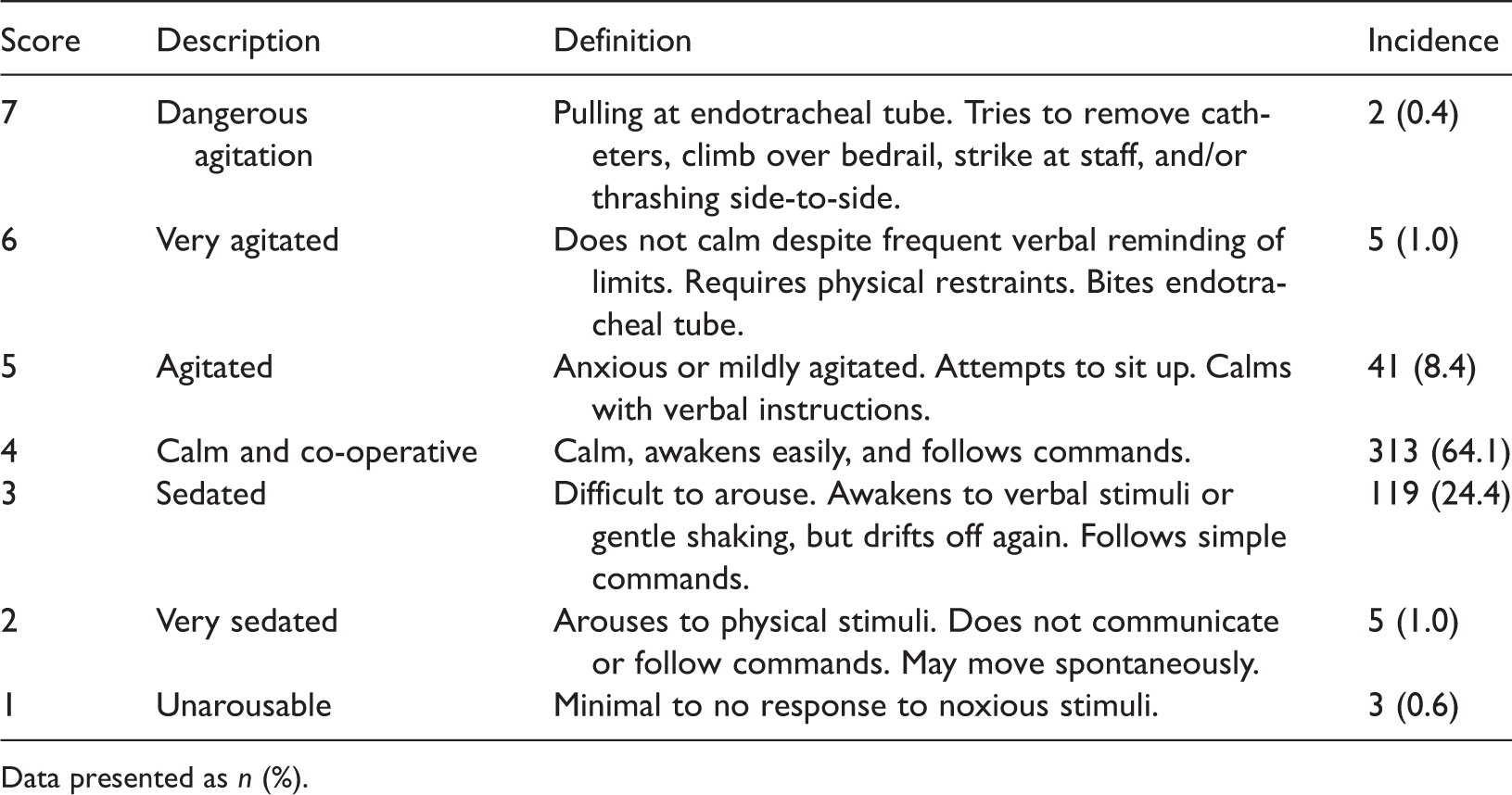
Data presented as n (%).
Demographic and clinical characteristics of adults included in a study investigating the incidence of emergence agitation following general anaesthesia for urological surgery, stratified according to the presence of emergence agitation (n = 488).

Data presented as mean ± SD or n (%).
Student’s t-test for continuous variables; χ2 or Fisher’s exact test (if the number in a cell is less than five) for categorical variables.
BMI, body mass index; ASA, American society of Anesthesiologists.
Intraoperative parameters in adults undergoing general anaesthesia for urological surgery, stratified according to the presence of emergence agitation (n = 488).
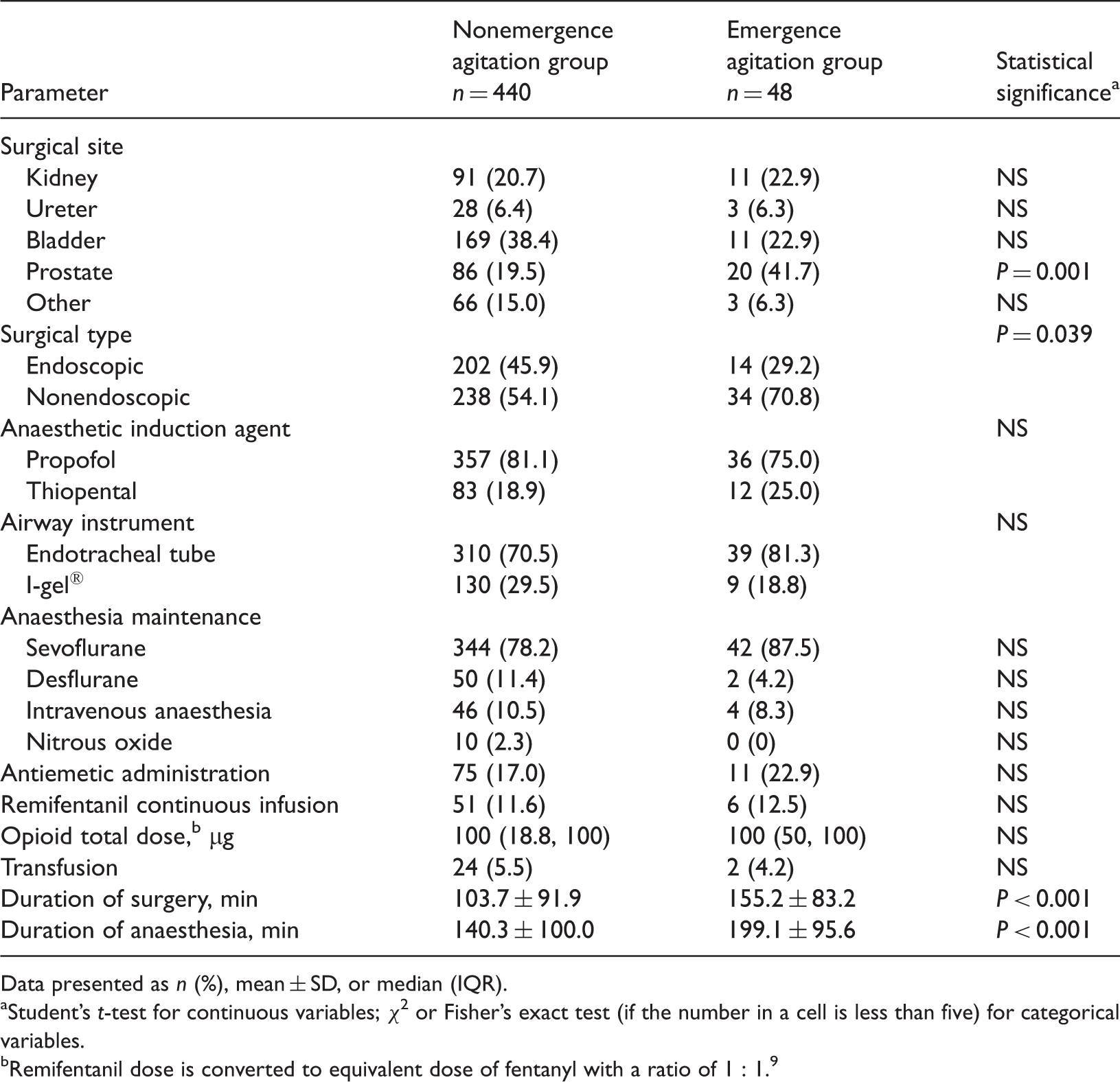
Data presented as n (%), mean ± SD, or median (IQR).
Student’s t-test for continuous variables; χ2 or Fisher’s exact test (if the number in a cell is less than five) for categorical variables.
Remifentanil dose is converted to equivalent dose of fentanyl with a ratio of 1 : 1.9
Postoperative parameters in adults undergoing general anaesthesia for urological surgery, stratified according to the presence of emergence agitation (n = 488).

Data presented as mean ± SD or n (%).
Student’s t-test for continuous variables; χ2 or Fisher’s exact test (if the number in a cell is less than five) for categorical variables.
Assessed using an 11-point numerical rating scale (0: no pain, 10: worst imaginable pain).
Chronic lung disease (odds ratio [OR] 2.72, 95% confidence interval [CI] 1.03, 7.17), duration of surgery (OR 1.01, 95%CI 1.00, 1.01), history of social drinking (OR 2.48, 95%CI 1.25, 4.93), postoperative pain score (OR 1.32, 95% CI 1.14, 1.53), voiding urgency (OR 2.20, 95% CI 1.01, 4.77) and gastric tube (OR 2.85, 95% CI 1.07, 7.54) were independent risk factors for emergence agitation. Age, male sex, chronic renal disease, bladder or prostate surgery, endoscopic surgery and endotracheal tube were not independent risk factors for emergence agitation.
Discussion
The present study found that emergence agitation in the PACU occurred in around 10% of patients undergoing urological surgery, and was associated with voiding urgency (CRBD). A long duration of surgery, history of social drinking, presence of a gastric tube, chronic lung disease and severe postoperative pain were predictive of emergence agitation.
Studies regarding postoperative emergence agitation have evaluated paediatric patients in the operating room or the PACU, 10–12 and few have examined adult patients in either setting. The overall incidence of emergence agitation was reported to be 5% in two large-scale PACU-based prospective studies using the Riker sedation–agitation scale and Richmond agitation and sedation scale, respectively. 1,4 Subgroup analyses indicated the incidence of emergence agitation was 4.1% in adult urology patients. 1 A further study that evaluated emergence agitation using a three-point scale reported that 21.3% of patients developed postoperative emergence agitation in the PACU, 5 with subgroup analysis indicating the incidence in urology patients to be 13.6%. 5 These differences may be explained by variations in surgical site, the scale used for patient assessment, interobserver agreement and possible underestimation as a result of retrospective study design.
The presence of a urinary catheter is a known risk factor for emergence agitation. 5 All patients who experienced postoperative emergence agitation in the present study had a urinary catheter, and CRBD was associated with an increased risk of emergence agitation. The incidence of postoperative CRBD has been reported at 58–90% in urology patients, with 45–60% of these patients experiencing moderate-to-severe symptoms. 13,14 Male sex and a large diameter Foley catheter are independent risk factors for CRBD. 15 The majority of patients in the present study were male and had a Foley catheter inserted postoperatively, putting them at high risk of CRBD. It is necessary to reduce CRBD in these patients in order to prevent emergence agitation. Early removal of the Foley catheter will reduce patient discomfort. Pharmacological prevention or treatment (using gabapentin, tramadol, ketamine or butylscopolamine) decreases the incidence of CRBD in the PACU. 6,7,13,16
In a finding consistent with others, 4,5,17 postoperative pain was a risk factor for emergence agitation in the present study. Poor postoperative pain management is known to increase postoperative morbidity. Pain can be managed by various means, 18–20 and adequate postoperative pain control is necessary to reduce emergence agitation.
In contrast to others, 1,5 we found that chronic lung disease was an independent risk factor for emergence agitation. Studies in patients with lung cancer, however, demonstrated that pneumonia is associated with hyperactive delirium. 21 Acute hypoxia or hypercapnia due to lung disease may result in additional postoperative stress, increasing the risk of emergence agitation.
Alcohol use is a risk factor for postoperative delirium. 22–24 This is consistent with our finding that a history of social drinking was a risk factor for emergence agitation. This may be due to acute withdrawal of alcohol during the perioperative period.
Duration of surgery was associated with emergence agitation in the present study and those of others. 1 Although it is considered a nonpreventable factor, using a surgical training programme that can increase the skill of the associated medical staff may reduce the duration of surgery. Moreover, close communication and co-operation among medical teams may be necessary to reduce any unnecessary delays during surgery. The presence of a gastric tube was also associated with increased postoperative emergence agitation in the present study. Similar to a urinary catheter, a nasogastric tube can distress a patient during anaesthetic emergence by stimulating the gag reflex. Therefore, to diminish patient discomfort, the nasogastric tube should be removed as early as possible.
Despite the significantly higher incidence of emergence agitation in male patients than female patients in the present study, male sex was not an independent risk factor. The role of sex as a risk factor for emergence agitation remains unclear, with some studies reporting that male sex is a risk factor 5 and others that it is not. 1,4 The high proportion of male patients in the present study (81.8%) limits the usefulness of our findings.
In our hospital, large Foley catheters are used in transurethral resection of bladder and prostate lesions to facilitate postoperative bladder irrigation and prevent obstruction of the catheter due to blood clots. We expected that the incidence of emergence agitation would be higher in patients undergoing endoscopic surgery because a Foley catheter with large diameter and continuous bladder stimulation with irrigation fluids is associated with CRBD. In contrast to our expectation, patients undergoing endoscopic urological surgery had a lower incidence of emergence agitation than those undergoing nonendoscopic surgery, although surgery type was not an independent risk factor in multivariate analysis.
Postoperative emergence agitation can result in serious complications, and self-extubation can lead to arterial hypoxaemia or aspiration pneumonia. In this study, one patient required reintubation in the PACU because of desaturation after self-extubation. It is most important to protect patients from self-injury, even before the cause of emergence agitation is identified. Once the cause is identified it should be immediately resolved. If this is not possible (e.g. removal of a urinary catheter in patients needing postoperative urine output monitoring), reassurance and pharmacological treatment can reduce the incidence of emergence agitation related to CRBD.
Our study has several limitations. First, the retrospective nature of the investigation meant that there were limitations in the data available. There was insufficient information related to preoperative anxiety, preoperative benzodiazepine administration for premedication or the use of intravenous anaesthetic agents for anaesthetic maintenance to allow us to evaluate their effects on emergence agitation in full. Preoperative anxiety, premedication with benzodiazepines and intraoperative use of an inhalation agent are known risk factors for emergence agitation. 1,4,5,17 Secondly, all study participants were undergoing urological surgery and CRBD is associated with an increased risk of emergence agitation. Thus, our findings may not be applicable to other surgical patients, particularly those who do not require intraoperative urinary catheterization. Thirdly, we used the Riker sedation–agitation score to evaluate emergence agitation. To our knowledge, there is no gold standard for diagnosis and other parameters such as the Richmond agitation–sedation scale can be used instead. In addition, there was no causal relationship between postoperative variables and emergence agitation. Rescue medications (such as fentanyl for postoperative pain relief, antiemetics for moderate-to-severe nausea and vomiting, and pethidine for shivering) can alter the incidence of emergence agitation. Caution is therefore needed in interpreting our results. Finally, this study focused on postoperative emergence agitation, not hypoactive emergence. Eight (1.6%) of our patients were highly sedated or unarousable in the PACU: these might be considered to be cases of hypoactive emergence. Risk factors for hypoactive emergence were not analysed in this study because of the small number of cases.
In conclusion, emergence agitation occur in around 10% of adult patients undergoing urological surgery. Chronic lung disease, a history of social drinking, voiding urgency due to an indwelling Foley catheter, long duration of surgery, inadequate postoperative pain control and presence of a gastric tube were associated with emergence agitation. Adequate postoperative pain management and prevention of CRBD may be helpful in reducing the incidence of emergence agitation in urology patients.
Footnotes
Declaration of conflicting interest
The authors declare that there are no conflicts of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Acknowledgements
We thank Dr Hyun Kang, Department of Anaesthesia and Pain Medicine, Chung-Ang University Hospital, Chung-Ang University College of Medicine, Seoul, Republic of Korea for his help with statistical analyses.
