Abstract
Objectives
To study the effects of diet on disease progression and activity levels of adenosine monophosphate-activated protein kinase (AMPK), and its downstream targets, in an amyotrophic lateral sclerosis (ALS) animal model.
Methods
AMPK activity was measured in cerebral cortex, spinal cord, cerebellum and hindlimb muscle tissue using immunohistochemistry in transgenic mice overexpressing human superoxide dismutase-1 (SOD1G93A) fed a high-fat (HFD), standard ad libitum (AL) or calorie-restricted (CR) diet; AMPK activity was also measured in wild-type (SOD1WT) mice. Activity of AMPK and phospho-AMPK, acetyl coenzyme-A carboxylase (ACC), phospho-ACC and heat shock protein-70 (Hsp70) were also measured using Western blot. Food intake and grip strength were recorded; body composition was analysed using dual energy X-ray absorptiometry. Motor neuron survival was observed using Nissl staining.
Results
AMPK activity increased and Hsp70 expression decreased in AL SOD1G93A mice compared with SOD1WT mice in spinal cord and hindlimb muscle. Compared with AL SOD1G93A mice, CR SOD1G93A mice showed increased AMPK activity, downregulated Hsp70 expression, reduced motor neuron survival in spinal cord and hindlimb muscle and reduced lifespan; HFD SOD1G93A mice showed opposite effects.
Conclusions
In this mouse model, increased AMPK activity seems to play a negative role in motor neuron survival, possibly through a novel mechanism involving Hsp70 downregulation. These changes can be modified by diet. Inhibition of AMPK may provide a therapeutic strategy for ALS.
Keywords
Introduction
Amyotrophic lateral sclerosis (ALS) is a neurodegenerative disease that selectively involves the spinal anterior horn cells, brainstem motor nuclei and pyramidal tracts. 1 A dominant mutation in the gene that encodes for the enzyme superoxide dismutase-1 (SOD1) accounts for 20% of familial ALS cases, 2 therefore it is possible to model ALS using transgenic mice that overexpress a human SOD1 transgene with a pathogenic glycine to alanine substitution (SOD1G93A).
Pathophysiological mechanisms underlying the development of ALS remain unclear; however, ALS patients often display a series of metabolic abnormalities including hypermetabolism, abnormal glucose tolerance and hyperlipidaemia.3–5 Similarly, SOD1G93A mice also show hypermetabolism and postprandial low-fat hyperlipidaemia in the presymptomatic stages of ALS. 6 Therefore, elucidating the mechanisms behind the energy deficiencies that affect disease progression in SOD1 mutant mice could yield new insights into the causes of human ALS.
Adenosine monophosphate-activated protein kinase (AMPK) is a sensor of cellular stress that is involved in maintaining energy homeostasis. AMPK is activated during energy stress by various upstream kinases via phosphorylation of threonine 172 within the catalytic α2 subunit. 7 Downstream consequences of AMPK activation include increased glucose uptake and fatty acid oxidation, and the inhibition of cholesterol, lipid and protein synthesis. 8
Since neurons are sensitive to energy fluctuations as they execute energy-demanding functions without efficiently storing nutrients, AMPK is essential for neuronal integrity and survival. 9 AMPK activation protects primary neuronal cultures from excitotoxicity and other insults; 10 however, some experimental work has suggested that AMPK activation could be detrimental in animal models of stroke, 11 Alzheimer’s disease12,13 and Huntington’s disease. 14 In the present study, activity levels of AMPK and its downstream targets acetyl coenzyme-A carboxylase and heat shock protein-70 were compared in SOD1G93A and wild-type SOD1 mice, and the effects of calorie restrictive and high-fat diets were assessed. In addition, the effects of diet on disease progression were studied.
Materials and methods
Animals
All animals were purchased from the Model Animal Research Centre of Nanjing University, Nanjing, China (originally obtained from The Jackson Laboratory, Bar Harbor, ME, USA) and were reared in the experimental animal department of Peking University Health Science Centre, Beijing, China. The strains B6SJL-TgN(SOD1G93A)1Gur and B6SJL-TgN(SOD1)2Gur were used for the mutant transgenic and wild-type SOD1 transgenic mice, respectively. All experiments were performed on male mice. Three to five mice were housed per cage under a 12-h light/dark cycle. All procedures were approved by the Ethics Committee for Experimental Animals, Peking University Health Science Centre, Beijing, China.
Diet
Up to postnatal day 39 (P39), all mice were fed ad libitum a standard rodent diet containing 4% fat and 5% protein. From P40, the wild-type SOD1 (SOD1WT) mice (n = 15) continued to receive a standard rodent diet ad libitum, but the SOD1G93A mice were fed either an identical standard rodent diet (AL; n = 28), a calorie-restricted diet providing 60% of the average daily food intake of the corresponding ad libitum group (CR; n = 26), or a high-fat diet containing 21% fat and 0.15% cholesterol (HFD; n = 30).
Assessment
Food intake measurements were initiated at P43 and recorded until P120 in all the SOD1G93A mice.
To determine disease onset, grip strength was assessed twice a week in all the SOD1G93A mice by measuring their ability to hold on to the cage bar for 20 s while they were upside down, as described previously. 15 Survival was also recorded in the SOD1G93A mice, with death being defined as the point at which the mouse could no longer roll back to a normal position within 10 s of being placed on its back.
Body weight and composition
Body weight was logged twice a week from P43 until P120 in all the SOD1G93A mice. Body composition was analysed using dual energy X-ray absorptiometry in isoflurane-anaesthetized SOD1G93A mice at P120, as described previously. 16
Immunohistochemistry
Motor cortex, cerebellum and lumbar spinal cord samples were obtained from SOD1WT and AL, HFD and CR SOD1G93A mice at P90 (n = 5 for all groups) and P120 (n = 5, n = 5, n = 6 and n = 5, respectively) for immunohistochemical analysis of phospho-AMPK (pAMPK) activity. Mice were deeply anaesthetized with 7% chloral hydrate in saline (350 mg/kg intraperitoneally) and perfused transcardially with 0.9% NaCl. Brain and spinal cord were carefully dissected and tissue samples were fixed in 4% paraformaldehyde in 0.1 mol/l phosphate buffer (pH 7.4) for 24 h, then submersed in increasing concentrations of sucrose overnight to provide cytoprotection. All the tissue samples were then embedded in paraffin wax and sectioned at 6 µm.
Immunohistochemical staining of the motor cortex, cerebellum and lumbar spinal cord sections was performed as described previously. 17 Anti-pAMPK rabbit monoclonal antibody (Cell Signaling Technology, Danvers, MA, USA) diluted to 1 : 200 in blocking buffer (pH 7.4) was used as the primary antibody; horseradish peroxidase-labelled goat antirabbit antibody (Zhongshan Biotechnology, Beijing, China), diluted to 1 : 2000, was used as the secondary antibody. Sections were stained using diaminobenzine and haematoxylin. Immunoreactivity was quantified by measuring the mean staining densities (sum of grey values of all pixels in the selected area divided by the number of pixels within the area) in five randomly selected areas from each specimen, using Scion Image software (National Institutes of Health, Bethesda, MD, USA).
Nissl staining
The number of motor neurons in the anterior horns of lumbar spinal cord tissue samples obtained as described above from AL (n = 5), CR (n = 5) and HFD (n = 6) SOD1G93A mice was assessed at P120 by Nissl staining using Cresyl Violet (Sigma-Aldrich, St Louis, MO, USA), following a standard method. Cells with diameters > 25 µm, which were considered to be motor neurons, were then counted in six tissue sections from each mouse. 18
Western blot analysis
Activity levels of AMPK and pAMPK, and the downstream targets of AMPK acetyl coenzyme-A carboxylase (ACC), ACC phosphorylated at serine 79 (pACC) and heat shock protein-70 (Hsp70) in SOD1WT and AL, CR and HFD SOD1G93A mice were assessed in spinal cord, cerebral cortex and cerebellum tissue lysates at P90 and P120; they were also assessed in hindlimb muscle tissue at P120 using Western blot analysis (n = 5 for each group).
Mice were deeply anaesthetized with 7% chloral hydrate in saline (350 mg/kg intraperitoneally) and decapitated. The relevant tissues were removed, frozen in isopentane at −70℃ and stored in a deep freezer at −80℃ for 24–48 h. All tissue samples were homogenized on ice with lysis buffer (210 mM mannitol, 70 mM sucrose and 10 mM HEPES, pH 7.2), 1 mM ethylene glycol tetra-acetic acid, freshly supplemented with 0.5% w/v fatty-acid free bovine serum albumin, and protease and phosphatase inhibitors. Lysates were sonicated, separated from particulate matter by centrifugation (10 min at 12 000
Western blot was performed as previously described 19 using AMPKα, pAMPK, ACC, pACC and Hsp70 antibodies (all from Cell Signaling Technology). β-Actin antibody (Epitomics, Burlingame, CA, USA) was used as the loading control.
Statistical analyses
All experiments were repeated at least three times. Results were expressed as the mean ± SEM of at least three independent experiments unless stated otherwise. Between-group differences were determined using a two-tailed Student’s t-test. For comparisons of multiple groups, one-way analysis of variance was performed. For disease onset and survival analyses, the Kaplan–Meier method with log-rank test was used. A two-tailed P-value < 0.05 was considered to be statistically significant. All statistical analyses were performed using SPSS software version 13.0 (SPSS Inc., Chicago, IL, USA).
Results
Activity levels of AMPK and its downstream targets in SOD1WT and SOD1G93A mice
Motor cortex, cerebellum and lumbar spinal cord sections from SOD1WT and AL SOD1G93A mice exhibited analogous levels of pAMPK immunoreactivity at P90 (data not shown). At P120, increased expression of pAMPK was seen in the lumbar spinal cord (Figure 1A and 1B) but not in the cerebellum or the motor cortex (data not shown) of AL SOD1G93A mice compared with SOD1WT controls.
Phospho-adenosine monophosphate-activated protein kinase (pAMPK) immunoreactivity in lumbar spinal cord (LSC) tissue samples at postnatal day 120 (P120), in a murine model of amyotrophic lateral sclerosis. (A) pAMPK immunoreactivity in mice overexpressing a human superoxide dismutase-1 transgene (G93A) and wild-type mice (WT). (B) Quantification of pAMPK immunoreactivity in G93A and WT mice (n = 5 per group). (C) pAMPK immunoreactivity in G93A mice fed a high-fat (HFD), standard ad libitum (AL) or calorie-restricted diet (CR). (D) Quantification of pAMPK immunoreactivity in HFD (n = 6), AL (n = 5) and CR (n = 5) mice. Data presented as mean ± SEM. *P < 0.05, **P < 0.01 (Student’s t-test). Original magnification of images × 40. The colour version of this figure is available at: http://imr.sagepub.com
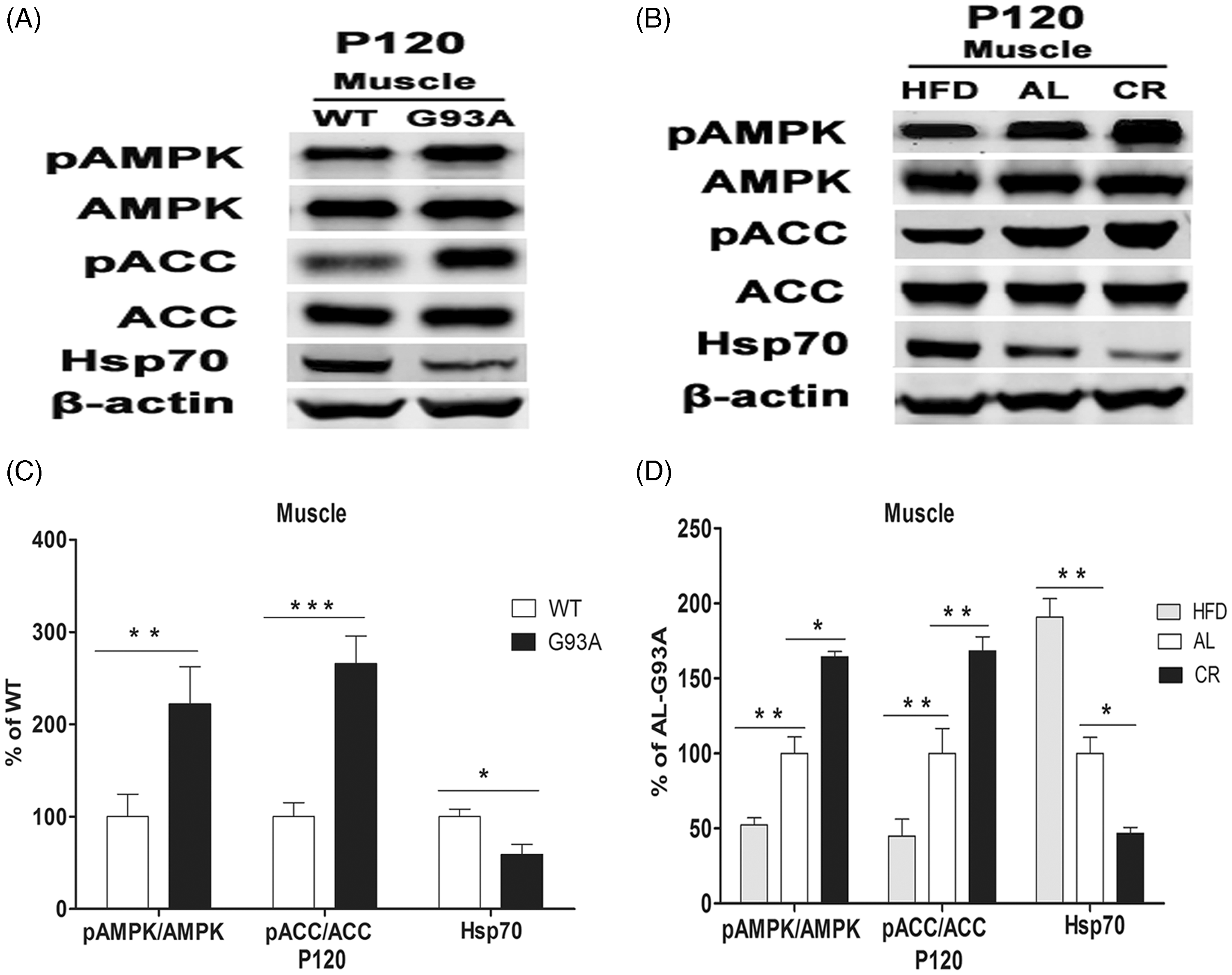
On Western blot analysis, there was a 64% increase in the pAMPK/AMPK ratio, a 110% increase in the pACC/ACC ratio and a 31% decrease in Hsp70 expression in spinal cord tissue lysates from AL SOD1G93A mice compared with SOD1WT controls at P90 (Figure 2A and 2C). These changes are consistent with an increase in AMPK activity in AL SOD1G93A mice. Such differences were even more apparent in spinal cord tissue at P120 (Figure 2B and 2D). Similar differences were seen in hindlimb muscle samples from AL SOD1G93A mice compared with SOD1WT controls at P120 (Figure 3A and 3C), but not in cerebellum or cerebral cortex tissue samples at P90 or P120 (Figure 2A and 2B).
Activity of adenosine monophosphate-activated protein kinase (AMPK) and phospho-AMPK (pAMPK), acetyl coenzyme-A carboxylase (ACC), phospho-ACC (pACC) and heat shock protein-70 (Hsp70) in cerebral cortex (CC), spinal cord (SC) and cerebellum (Cer) tissue samples in a murine model of amyotrophic lateral sclerosis. β-Actin was used as a control. (A) Activity on Western blot in mice overexpressing a human superoxide dismutase-1 transgene (G93A) and wild-type mice (WT) at postnatal day 90 (P90). (B) Activity on Western blot in G93A and WT mice at postnatal day 120 (P120). (C) Quantification of the pAMPK/AMPK ratio, pACC/ACC ratio and Hsp70 activity in G93A and WT mice at P90. (D) Quantification of the pAMPK/AMPK ratio, pACC/ACC ratio and Hsp70 activity in G93A and WT mice at P120. Data presented as mean ± SEM; n = 5 per group. *P < 0.05, **P < 0.01, ***P < 0.001 (Student’s t-test). Activity of adenosine monophosphate-activated protein kinase (AMPK) and phospho-AMPK (pAMPK), acetyl coenzyme-A carboxylase (ACC), phospho-ACC (pACC) and heat shock protein-70 (Hsp70) in hindlimb muscle at postnatal day 120 (P120) in a murine model of amyotrophic lateral sclerosis. β-Actin was used as a control. (A) Activity on Western blot in mice overexpressing a human superoxide dismutase-1 transgene (G93A) and wild-type mice (WT). (B) Activity on Western blot in G93A mice fed a high-fat (HFD), standard ad libitum (AL) or calorie-restricted diet (CR). (C) Quantification of the pAMPK/AMPK ratio, pACC/ACC ratio and Hsp70 activity in G93A and WT mice. (D) Quantification of the pAMPK/AMPK ratio, pACC/ACC ratio and Hsp70 activity in HFD, AL and CR mice. Data presented as mean ± SEM; n = 5 per group. *P < 0.05, **P < 0.01, ***P < 0.001 (Student’s t-test).
Effect of diet on neurodegeneration in SOD1G93A mice
For CR SOD1G93A mice, the mean ± SEM food intake was 2.2 ± 0.1 g/day, corresponding to 60% of the food intake for AL SOD1G93A mice (3.7 ± 0.1 g/day); the food intake for HFD SOD1G93A mice was 2.7 ± 0.2 g/day (Figure 4B). At P120, CR SOD1G93A mice weighed 21% less than AL SOD1G93A mice (15.6 ± 1.5 g versus 19.8 ± 2.0 g) (Figure 4A and 4C), while HFD SOD1G93A mice weighed 26% more than AL SOD1G93A mice (24.9 ± 1.8 g versus 19.8 ± 2.0 g) (Figure 4C). Fat body mass was increased in HFD SOD1G93A mice relative to AL SOD1G93A mice (6.3 ± 0.7 g versus 2.2 ± 0.4 g) (Figure 4D), but there were no differences in lean body mass (18.4 ± 0.5 g versus 17.8 ± 0.4 g) (Figure 4E). CR SOD1G93A mice had significantly lower fat body mass and lean body mass relative to AL SOD1G93A mice (Figure 4D and 4E).
Food intake and body weight and composition in a murine model of amyotrophic lateral sclerosis. Mice overexpressing a human superoxide dismutase-1 transgene were fed a high-fat (HFD), standard ad libitum (AL) or calorie-restricted diet (CR). (A) Body weight. (B) Food intake. (C) Total body mass at postnatal day 120 (P120). (D) Fat body mass at P120. (E) Lean body mass at P120. (F) Percentage fat body mass at P120. Data presented as mean ± SEM; n = 10 per group. *P < 0.05, **P < 0.01 (Student’s t-test). The colour version of this figure is available at: http://imr.sagepub.com
Compared with AL SOD1G93A mice, disease onset was delayed by 7 days in HFD SOD1G93A mice (P = 0.001), but was significantly hastened by 10 days in CR SOD1G93A mice (P = 0.003) (Figure 5A). Similarly, HFD SOD1G93A mice had an extended lifespan (P < 0.001) and CR SOD1G93A mice had a decreased lifespan (P < 0.001) compared with AL SOD1G93A mice (Figure 5B). Median survival times for HFD, AL and CR SOD1G93A mice were 148, 128 and 116 days, respectively.
Effects of a high-fat (HFD), standard ad libitum (AL) or calorie-restricted (CR) diet on neurodegeneration in a murine model of amyotrophic lateral sclerosis. Mice were overexpressing a human superoxide dismutase-1 transgene (G93A). (A) Onset of symptoms in CR, AL and HFD mice (n = 12 per group). (B) Percentage survival in CR (n = 15), AL (n = 14) and HFD (n = 14) mice. (C) Nissl staining of lumbar spinal cord (LSC) tissue sections in CR, AL and HFD mice at postnatal day 120 (P120; original magnification × 20). (D) Quantification of the number of motor neurons in LSC in CR (n = 5), AL (n = 5) and HFD (n = 6) mice at P120. Data presented as mean ± SEM. *P < 0.05 using Student’s t-test. The colour version of this figure is available at: http://imr.sagepub.com
Effect of diet on motor neuron loss in SOD1G93A mice
Compared with AL SOD1G93A mice, HFD SOD1G93A mice had significantly more spared motor neurons (P < 0.05), whereas CR SOD1G93A mice showed a marked reduction in motor neuron number at P120 (P < 0.05) (Figure 5C and 5D).
Effect of diet on activity levels of AMPK and its downstream targets in SOD1G93A mice
Levels of pAMPK immunoreactivity in the lumbar spinal cord were significantly reduced in HFD SOD1G93A mice and markedly increased in CR SOD1G93A mice compared with AL SOD1G93A mice at P120 (Figure 1C and 1D), whereas pAMPK immunoreactivity in the cerebellum and motor cortex were similar in all three groups (data not shown).
On Western blot analysis, there was a 40% decrease in the pAMPK/AMPK ratio, a 45% decrease in the pACC/ACC ratio and a 35% increase in Hsp70 expression in spinal cord tissue from HFD SOD1G93A mice compared with AL SOD1G93A mice at P90 (Figure 6C). Opposite changes were seen in CR SOD1G93A mice compared with AL SOD1G93A mice (Figure 6C). Similar differences were seen in spinal cord tissue and hindlimb muscle at P120 (Figure 6D and Figure 3B and 3D). No differences were observed in the activity of AMPK and its downstream targets between the three groups in cerebral cortex or cerebellum tissue at P90 (data not shown) or P120 (Figure 6B).
Activity of adenosine monophosphate-activated protein kinase (AMPK), phospho-AMPK (pAMPK), acetyl coenzyme-A carboxylase (ACC), phospho-ACC (pACC) and heat shock protein-70 (Hsp70) in cerebral cortex (CC), spinal cord (SC) and cerebellum (Cer) of mice overexpressing a human superoxide dismutase-1 transgene (G93A), in a murine model of amyotrophic lateral sclerosis. Mice were fed a high-fat (HFD), standard ad libitum (AL) or calorie-restricted diet (CR). β-Actin was used as a control. (A) Activity on Western blot in HFD, AL and CR G93A mice in SC at postnatal day 90 (P90). (B) Activity on Western blot in HFD, AL and CR G93A mice in CC, SC and Cer at postnatal day 120 (P120). (C) Quantification of the pAMPK/AMPK ratio, pACC/ACC ratio and Hsp70 activity in HFD, AL and CR G93A mice in SC at P90. (D) Quantification of the pAMPK/AMPK ratio, pACC/ACC ratio and Hsp70 activity in HFD, AL and CR G93A mice in SC at P120. Data presented as mean ± SEM; n = 5 per group. *P < 0.05, **P < 0.01, ***P < 0.001 (Student’s t-test).
Discussion
Several defects of energy metabolism occur in ALS, including weight loss, hypermetabolism and hyperlipidaemia. 5 Recurrent reports have indicated that these changes are potentiated by the co-occurrence of a mitochondrial defect. 5 SOD1G93A mice have been shown to have dysfunctional mitochondria and reduced levels of adenosine triphosphate (ATP) production, compared with wild-type mice. 20 AMPK is activated by reduced ATP production; once activated, AMPK can directly and indirectly regulate transcriptional processes through phosphorylation.
In the present study, AMPK activity was increased and Hsp70 expression was downregulated in spinal cord and hindlimb muscle samples in AL SOD1G93A mice compared with SOD1WT mice. Increased AMPK phosphorylation in AL SOD1G93A mice compared with wild-type controls has been confirmed in spinal cord tissue, with accumulation of pAMPK seen in cytoplasmic granules within motor neurons, but also in peripheral tissues, liver and kidney. 21 The involvement of AMPK in the modulation of heat shock protein gene expression is a novel mechanism of metabolic stress; several studies have confirmed that AMPK activation produces negative effects by inhibiting the expression of Hsp70, while AMPK inhibition produces neuroprotective effects via the enhancement of Hsp70 expression in stroke models. 22 The mechanisms that connect AMPK activation to Hsp70 expression require further investigation. Heat shock proteins are molecular chaperones that assist in the proper folding of newly synthesized proteins and proteins subjected to stress-induced denaturation. 23 In contrast to other heat shock proteins, Hsp70 has been identified in almost all of the intracellular compartments. 24 Motor neurons have a weakened capacity to upregulate Hsp70 under stress conditions, making them more susceptible to cell damage. 25 Overexpression of Hsp70 reduced apoptosis in cerebral ischaemia and increased expression of the anti-apoptotic protein Bcl-2. 23 Exogenous delivery of Hsp70 extended the lifespan of SOD1G93A mice by reducing mutant SOD1 aggregate formation, indicating an important protective effect for this protein in ALS. 26 In the present study, AL SOD1G93A mice had significantly lower expression of Hsp70 than SOD1WT mice, indicating that they were less protected from metabolic stress. Therefore, AMPK activation, resulting in Hsp70 downregulation, may be harmful in ALS.
The main purposes of the present study were to establish whether AMPK activation and the resulting detrimental effects could be altered by dietary changes in SOD1G93A mice. CR SOD1G93A mice showed reduced weight and fat body mass, increased AMPK activity, downregulated Hsp70 expression, increased loss of motor neurons, hastened disease onset and decreased lifespan compared with AL SOD1G93A mice. After long-term calorie restriction, increased AMPK activity has been identified in skeletal muscle in mice; 27 in the present study a similar change was noted in spinal cord tissue, suggesting that calorie restriction-induced AMPK activation may have adverse effects in ALS. Others have reported that calorie restriction shortens lifespan through reductions in mitochondria, protein oxidation and stress responses (Hsp70) in skeletal muscle in SOD1G93A mice. 28 The present study also documented downregulation of Hsp70 expression in CR SOD1G93A mice, suggesting that AMPK activation resulting in Hsp70 downregulation may be the mechanism behind the accelerated clinical onset and disease progression associated with calorie restriction.
In contrast, in the present study HFD SOD1G93A mice showed increased weight and fat body mass, inhibition of AMPK activity, upregulated Hsp70 expression, decreased loss of motor neurons, delayed disease onset and extended survival compared with AL SOD1G93A mice. Others have reported that a high-fat diet inhibited AMPK activity and that this corresponded to significant increases in leptin and glucose levels. 29 The present study results are consistent with those of a report showing that a high-fat diet increased fat storage, improved motor neuronal function and extended lifespan in SOD1G93A mice. 30 The present results also showed that a highf-at diet was associated with decreased activity of AMPK and increased Hsp70 expression. As reduced AMPK activity has been shown to be protective in genetic models of motor neuron disease, 31 this may be part of the mechanism by which a high-fat diet protects motor neurons in SOD1G93A mice but not in mutant TDP-43 mice; 21 this difference is likely to be due to different energy metabolism characteristics in these models.
In summary, the present study showed that AMPK activity is increased and Hsp70 expression is downregulated in spinal cord and hindlimb muscle of SOD1G93A mice compared with SOD1WT mice, in a manner that can be predictably altered by diet. A high-fat diet inhibited AMPK activity and motor neuron loss, upregulated Hsp70 expression, delayed disease onset and extended the lifespan of SOD1G93A mice, but calorie restriction produced opposite results. These findings suggest AMPK-induced Hsp70 downregulation is a novel mechanism in the pathogenesis of ALS. The underlying mechanisms responsible for AMPK activity and motor neuron loss require further study. Inhibition of AMPK may provide a therapeutic strategy for ALS.
Footnotes
Declaration of conflicting interest
The authors declare that there are no conflicts of interest.
Funding
This study was supported by grants from the National Natural Sciences Foundation of China (81030019).
