Abstract
Objective
To compare the treatment effects of Novolin® 30Ra, versus Lantus®a combined with acarbose (Glucobay®), in elderly patients with type 2 diabetes mellitus.
Methods
Patients (aged > 60 years) with type 2 diabetes mellitus were randomized to receive either Novolin® 30Ra (initial dose 0.5 IU/kg) or Lantus®a (initial dose 0.2 IU/kg) combined with 50 mg acarbose. After a 32-week treatment period, the following parameters were measured: blood glucose control; blood lipid levels; body mass index; proportion of patients achieving a glycosylated haemoglobin (HbA1c) level <7.5%; rate of hypoglycaemic events; change in fasting blood glucose levels from baseline in patients stratified according to their baseline HbA1c level.
Results
A total of 188 patients were enrolled in the study. After 32 weeks’ treatment, compared with baseline levels, there were significant reductions in FBG, 2 h-postprandial blood glucose during an oral glucose tolerance test, HbA1c, total cholesterol, triglycerides and low-density lipoprotein cholesterol values in both groups. Although there were fewer hypoglycaemic events in the Lantus® combined with Glucobay® group compared with the Novolin® 30R group, the difference was not significant.
Conclusion
Novolin® 30R and Lantus® combined with acarbose both had beneficial effects on blood glucose control and blood lipid levels in elderly patients with type 2 diabetes mellitus.
Introduction
Diabetes mellitus is one of the most common chronic diseases throughout the world and is rapidly becoming one of the most challenging medical issues facing healthcare professionals. For example, in mainland China, 9.7% of the adult population has diabetes mellitus. 1 Given its large population, China may have the largest number of people living with diabetes in the world. China has experienced rapid economic development, and as a consequence, life expectancy is likely to increase over time, which will result in the concomitant increase in the proportion of elderly individuals (>60 years old) in the population. Currently, the high proportion of elderly patients with type 2 diabetes mellitus in China leads to a heavy burden on medical resources, both via type 2 diabetes itself and the long-term complications associated with this disease, such as chronic kidney disease. Thus, effective treatment of elderly patients with type 2 diabetes has become a major concern for healthcare professionals in China.
As type 2 diabetes mellitus is a progressive disease and glycaemic control is usually unstable when only oral hypoglycaemic agents are used, most patients will eventually require insulin treatment. 2 However, the injection of excessive amounts of exogenous insulin via subcutaneous tissue can cause peripheral hyperinsulinaemia, hypoglycaemia and weight gain. 3 It has been suggested by an increasing body of evidence that a combination of oral hypoglycaemic agents and insulin injections could be a more-effective treatment option for diabetes management than either agent alone, which would also prevent the unwanted side-effects of insulin injections in patients with type 2 diabetes mellitus. 4 Acarbose (Glucobay®) is a potent α-glucosidase inhibitor that delays the intestinal digestion of starch, resulting in a reduction of postprandial blood glucose levels. 5 Novolin® 30Ra is isophane protamine biosynthetic human insulin (premixed 30R), 6 while Lantus®a is insulin glargine, 7 both of which are widely used in the clinic. However, investigations comparing the efficacy of Novolin® 30R versus Lantus® combined with acarbose are limited. This current randomized, prospective study investigated the clinical effectiveness of Novolin® 30R versus Lantus® combined with acarbose treatment, in elderly patients with type 2 diabetes mellitus.
Patients and methods
Patients
This randomized, prospective study enrolled patients with type 2 diabetes mellitus from the Department of Geriatric Medicine, East Hospital, Tongji University School of Medicine, Shanghai, China between 1 January 2011 and 31 March 2013. The inclusion criteria were: aged >60 years (i.e. elderly); diagnosed with type 2 diabetes mellitus according to the diagnostic classification recommended by the American Diabetes Association in 2013; 8 fasting blood glucose (FBG) ≥7.0 mmol/l (fasting was defined as no calorie intake for ≥8 h prior to the test) or a 2 h-postprandial blood glucose (2hPBG) during an oral glucose tolerance test ≥11.1 mmol/l or a random blood glucose ≥11.1 mmol/l in a patient with classic symptoms of hyperglycaemia or in a hyperglycaemic crisis. More specifically, as this study aimed to enrol patients with poorly controlled blood glucose levels, patients with type 2 diabetes mellitus were required to have an FBG ≥11.0 mmol/l, a 2hPBG ≥16.0 mmol/l or a glycosylated haemoglobin (HbA1c) ≥8.5%, even after having oral hypoglycaemic agent therapy for >6 months. Patients were excluded if they had significant hepatic or renal impairment, had received prior insulin injection treatment, or had any contraindications to the study drugs.
The study protocol was approved by the Ethics Committee of East Hospital, Tongji University School of Medicine, Shanghai, China. All of the patients entering this study provided written informed consent before undergoing any 2.
Baseline clinical characteristics and biochemical analyses
The baseline demographic and clinical characteristics of the patients with type 2 diabetes including age, sex, duration of diabetes, FBG, 2hPBG, HbA1c, triglycerides (TG), total cholesterol (TC), low-density lipoprotein cholesterol (LDL-C), high-density lipoprotein cholesterol (HDL-C) and body mass index (BMI) were recorded at the start of the study. Venous blood samples (5 ml) were obtained from each patient, using standard procedures. After clotting at room temperature for 15 min and centrifugation at 2000
Changes in FBG, 2hPBG, HbA1c, BMI, and blood lipid levels from baseline to the study endpoint and the proportion of patients who achieved an HbA1c level <7.5% were recorded for all patients in the two treatment groups. Patients were further divided into three subgroups according to their baseline HbA1c value and changes in their FBG from baseline to the study endpoint were compared within each treatment group. This was to investigate whether individuals with different baseline HbA1c levels might respond differently to the same study drug.
Study treatment
All patients were instructed to maintain their normal balanced diet and exercise regimens, and to discontinue their current oral hypoglycaemic agents immediately prior to the start of the study. Patients were randomized using a computer-generated randomization schedule to receive either an initial dose of 0.5 IU/kg Novolin® 30R (Novo Nordisk, Bagsvaerd, Denmark), via subcutaneous (s.c.) injection twice daily before breakfast and supper, or an initial dose of 0.2 IU/kg of Lantus® (Sanofi-Aventis, Paris, France) via s.c. injection once daily prior to breakfast in combination with 50 mg acarbose (Bayer, Leverkusen, Germany), administered orally during each of three meals per day. Randomized treatment was scheduled to last for 32 weeks and the treatments were not blinded to either the patients or healthcare professionals involved in the study.
Patients were followed-up at weeks 2, 4, 8, 12, 16, and 24 and at the study endpoint. Dosage modifications were made for the exogenous insulin component of each regimen according to each patient’s FBG level, with the aim of maintaining the following: FBG 6.0–8.5 mmol/l; 2hPBG 8–11 mmol/l; HbA1c 6.5–8.0%. At each follow-up, detailed inquiries were made about the frequency and severity of any hypoglycaemic events. A hypoglycaemic event was defined as an FBG ≤ 3.9 mmol/l; a severe hypoglycaemic event was defined as an FBG ≤ 2.8 mmol/l with conscious disturbance or a requirement for the assistance of others.
Statistical analyses
All statistical analyses were performed using the SPSS® statistical package, version 15.0 (SPSS Inc., Chicago, IL, USA) for Windows®. Two sample t-test was used to compare the baseline data between the two groups. Data were presented as mean ± SD. The proportion of patients who achieved an HbA1c level <7.5% is described as the n of patients (%). Changes in FBG, 2hPBG, HbA1c, BMI and blood lipid levels from baseline to the study endpoint, and the percentages of patients who achieved an HbA1c level <7.5%, were compared between the two treatment groups using Student’s t-test. All statistical tests were two-sided and performed at significant level of α = 5%. A P-value < 0.05 was considered statistically significant.
Results
A total of 188 patients with type 2 diabetes mellitus were enrolled and received the randomized study treatments, completing the seven follow-up examinations: 94 patients (50.0%) were randomized to Novolin® 30R treatment; 94 patients (50.0%) were randomized to receive treatment with Lantus® combined with acarbose for 32 weeks (Figure 1). Baseline clinical and demographic data for the study participants in the two treatment groups were not significantly different from each other (Table 1).
Flow chart showing progress through enrolment, randomization and analysis of elderly patients with type 2 diabetes mellitus, enrolled in a study investigating the efficacy of two treatment regimens (n = 188) administered for 32 weeks. Novolin® 30R (Novo Nordisk, Bagsvaerd, Denmark) is a premixed r-DNA/GE/GM human insulin mix consisting of 30% R/neutral insulin and 70% NPH/isophane protamine biosynthetic human insulin. Lantus® (Sanofi Aventis, Paris, France) is insulin glargine [DNA origin] injection. Baseline clinical and demographic characteristics of elderly patients with type 2 diabetes mellitus, enrolled in a randomized study investigating the efficacy of two treatment regimens (n = 188) administered for 32 weeks. Data are presented as mean ± SD or n of patients. No statistically significant between-group differences (P ≥ 0.05); two-sample t-test. HbA1c, glycosylated haemoglobin. Novolin® 30R (Novo Nordisk, Bagsvaerd, Denmark) is a premixed r-DNA/GE/GM human insulin mix consisting of 30% R/neutral insulin and 70% NPH/isophane protamine biosynthetic human insulin. Lantus® (Sanofi Aventis, Paris, France) is insulin glargine [DNA origin] injection.

Changes in blood glucose levels, glycosylated haemoglobin (HbA1c), lipid levels and body mass index (BMI) in patients with type 2 diabetes mellitus, randomized to receive either Novolin® 30R or Lantus® combined with acarbose for 32 weeks (n = 188).

Data are presented as mean ± SD.
aComparisons made between baseline values and values after 32 weeks’ treatment; Student’s t-test.
FBG, fasting blood glucose; 2hPBG, 2 h-postprandial blood glucose during an oral glucose tolerance test; TG, triglycerides; TC, total cholesterol; HDL-C, high-density lipoprotein cholesterol; LDL-C, low-density lipoprotein cholesterol; NS, no statistically significant difference (P ≥ 0.05).
Novolin® 30R (Novo Nordisk, Bagsvaerd, Denmark) is a premixed r-DNA/GE/GM human insulin mix consisting of 30% R/neutral insulin and 70% NPH/isophane protamine biosynthetic human insulin. Lantus® (Sanofi Aventis, Paris, France) is insulin glargine [DNA origin] injection.
Changes in fasting blood glucose (FBG) levels from baseline to after 32 weeks’ treatment, in patients randomized to either Novolin® 30R or Lantus® combined with acarbose (n = 188). Subgroups of patients were stratified according to baseline glycosylated haemoglobin (HbA1c).
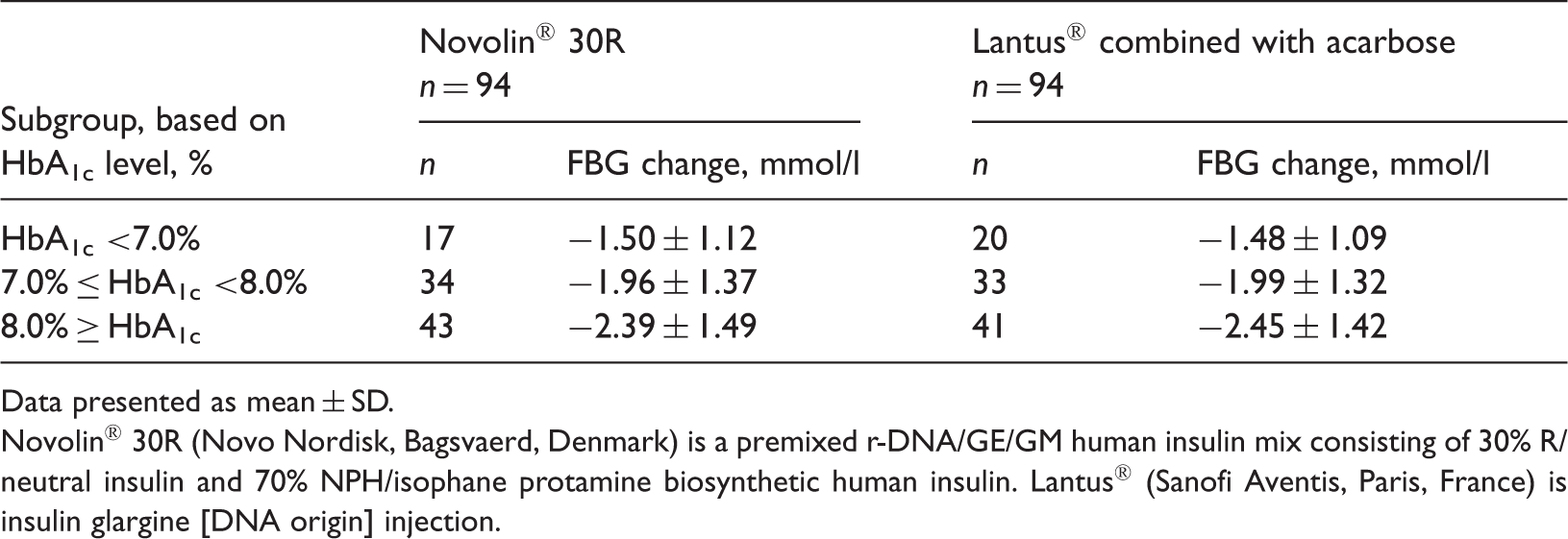
Data presented as mean ± SD.
Novolin® 30R (Novo Nordisk, Bagsvaerd, Denmark) is a premixed r-DNA/GE/GM human insulin mix consisting of 30% R/neutral insulin and 70% NPH/isophane protamine biosynthetic human insulin. Lantus® (Sanofi Aventis, Paris, France) is insulin glargine [DNA origin] injection.
With respect to blood lipid levels, TG, TC and LDL-C were significantly reduced compared with baseline values in both treatment groups (P < 0.05 for all comparisons) (Table 2). There was no significant change in HDL-C in either treatment group. BMI increased slightly in both treatment groups, but the increases were not significant.
None of the patients enrolled in the study discontinued treatment due to adverse effects associated with the study drugs. A total of 18 patients in the two treatment groups experienced hypoglycaemic events (FBG ≤ 3.9 mmol/l) for a total of 23 times, 10 of which were verified using portable home-use glucose meters by the patients. Hypoglycaemic events occurred in 11 patients in the Novolin® 30R group for a total of 13 times and in seven patients in the Lantus® combined with acarbose group for a total of 10 times. However, the difference between the Novolin® 30R and Lantus® combined with acarbose groups in terms of the rate of hypoglycaemic events, 13.8% (13/94) and 10.6% (10/94), respectively, was not significant. There were no occurrences of severe hypoglycaemia in either treatment group.
Discussion
Elderly patients with type 2 diabetes mellitus are more likely to have the additional burden of diabetes-related complications and asymptomatic hypoglycaemic events due to the decline in their ability to adjust their physiology. 9 Therefore, adequately controlling blood glucose and reducing complications should be emphasized, in the clinical management of type 2 diabetes mellitus in elderly patients.
In addition to making lifestyle changes, including diet control and exercise, oral hypoglycaemic agents are the optimum therapies for patients with type 2 diabetes mellitus whose blood glucose levels are well controlled. 10 A combination of acarbose and exogenous insulin could reduce the frequency and dosage of insulin required. The results of this present study suggest that acarbose in combination with Lantus® reduces TC, TG and LDL-C levels; the value of acarbose monotherapy has also showed similar effects on blood lipid levels. 11 High postprandial blood lipid levels are a risk factor for atherosclerosis. 12 Together, these results suggest that treatment with acarbose could ameliorate the disturbances observed in lipid metabolism that are observed in patients with diabetes mellitus.
Type 2 diabetes mellitus is a complex metabolic disorder that partly results from impaired insulin secretion by pancreatic β-cells. 13 As a result of the progressive decline in β-cell function leading to a deterioration in glycaemic control, patients with diabetes often ultimately require exogenous insulin therapy. 14 Novolin® 30R is widely used as an effective antidiabetes treatment in China, but its slow mechanism of action is not consistent with the normal physiological pattern of insulin secretion. Thus, such treatment easily leads to postprandial blood glucose variations, and therapy-related complications strongly correlate with these variations. Lantus® (insulin glargine) has useful medical properties: compared with Novolin® 30R, Lantus® offers a slower rate of uptake, a longer duration of action and has no peak plasma concentration, which is in line with the physiological pattern of human insulin secretion. 15 Lantus® is currently regarded as a useful basal analogue insulin, 15 particularly because it is associated with a low risk of inducing overnight hypoglycaemic events. 16 In addition, Lantus® is considered to be suitable for elderly patients with type 2 diabetes mellitus. The Asian diet is a high-carbohydrate diet, which is more likely to increase postprandial blood glucose levels than a low-carbohydrate diet. 17 As acarbose is an effective drug for controlling postprandial blood glucose levels, it might be a suitable candidate oral drug for combination with a basal analogue insulin.
The aim of the present study was to investigate a potentially improved therapeutic method for treating elderly patients with type 2 diabetes mellitus, which is associated with fewer negative side-effects and achieves better patient adherence than injected insulin alone. Hence, this present study compared the clinical effectiveness of two therapeutic methods; Novolin 30R® versus Lantus® combined with acarbose, in elderly patients with type 2 diabetes mellitus, over a 32-week treatment period. Both study regimens had good hypoglycaemic effects in elderly patients with type 2 diabetes mellitus: after 32 weeks’ treatment, the FBG levels in these two treatment groups significantly decreased from baseline.
Physiological function can gradually become impaired in individuals aged >60 years. Thus, the awareness of hypoglycaemic events in elderly patients with type 2 diabetes can become a problem. This problem can become serious problem if the patient has concomitant renal insufficiency, which can lead to the accumulation of oral hypoglycaemic agents and insulin in the body. All of these factors can lead to an increasing rate of asymptomatic hypoglycaemic events in elderly patients, to which more attention should be paid during the clinical management of this disease.
In the present study, the incidence of hypoglycaemic events was slightly lower in the Lantus® combined with acarbose group than in the Novolin 30R® group (10.6% versus 13.8%, respectively), but the difference was not statistically significant. The occurrence of hypoglycaemia and the frequency of daily injections are two main concerns for patients when they are considering insulin therapy. In the present study, patients received either Novolin 30R®, injected twice daily (before breakfast and supper), or Lantus®, injected once daily before breakfast. Compared with Novolin 30R®, Lantus® would appear to have a more convenient regimen, and therefore might be more likely to be accepted by patients.
The present study had a number of limitations. First, the sample size was not large enough to provide strong conclusions. Secondly, the 32-week duration was probably too short to investigate the long-term effects of the two treatment interventions. Further research with a larger sample size and a more rigorous approach is required. No significant safety differences were observed between the two groups, the reason for which may lie in the limited sample size.
In conclusion, both Novolin 30R® and Lantus® combined with acarbose provided good hypoglycaemic effects in elderly patients with type 2 diabetes mellitus; both regimens were associated with similar levels of unwanted hypoglycaemic events. Further research, with a larger sample size, is required to investigate the potential benefits of using oral hypoglycaemic agents with insulin injections in elderly patients with type 2 diabetes mellitus.
Footnotes
Declaration of conflicting interest
The authors declare that there are no conflicts of interest.
Funding
This research was supported by grants from the National Natural Science Foundation of China (no. 30600294), Natural Science Foundation of Shanghai (no. 11ZR1429500), Science and Technology Funds from Pudong New Area (no. PKJ2012-Y07; no. PKJ2012-Y09), and the Academic Leaders Training Programme of Pudong Health Bureau of Shanghai (no. PWRd2011-01).
