Abstract
Objective
To evaluate the efficacy of bicyclol in preventing chemotherapy-induced liver damage.
Methods
Patients ≥60 years of age with cancer were equally randomized into control (chemotherapy alone) or prophylactic (chemotherapy supplemented with 75 mg bicyclol, oral, daily) groups. Liver function indices were assessed immediately before treatment, during each therapy cycle and following treatment.
Results
Of 306 patients enrolled, 300 patiets completed the study (n = 147 and n = 153; prophylactic and control groups, respectively). Incidence of grade I–IV elevation of serum transaminase and/or bilirubin was significantly lower in the prophylactic group (17.1%) compared with the control group (47.1%). Incidence of grade II–IV hepatic injury was also significantly lower in the prophylactic group (0.7%) than in the control group (12.4%).
Conclusions
Prophylactic bicyclol (75 mg daily) could significantly reduce the incidence and degree of chemotherapeutic agent-induced liver damage in elderly patients with cancer. Further studies are recommended with larger sample sizes and long-term follow up.
Introduction
Chemotherapeutic agents are metabolized and eliminated mainly in the liver, and most are capable of causing hepatic toxicity.1–3 Presence of underlying hepatic injury can alter the metabolism and excretion of chemotherapeutic drugs and can result in higher or more persistent plasma drug levels; this in turn can cause increased systemic toxicity (particularly myelosuppression) or worsening of chemotherapy-induced hepatotoxicity.4,5 Such events often affect the disease response, and are associated with treatment-course delays, discontinuations and dose reductions.6,7 Patients undergoing cytotoxic chemotherapy require careful assessment of their liver function before, during and after chemotherapy. The liver-reserve functions tend to decrease with increasing age, which could increase the incidence of chemotherapeutic agent-induced liver injury among elderly people.8–10 Thus, it is of clinical importance to aim to prevent chemotherapeutic agent-induced liver injury among older patients with cancer.
The compound 4, 4′-dimethoxy-5, 6, 5′, 6′-dimethylene-dioxy-2, 2′-dicarboxylate biphenyl (DDB) is an effective liver protectant; bicyclol (4,4′-dimethoxy-5,6,5′,6′-dimethylene-dioxy-2- hydroxymethyl-2′-carbonylbiphenyl) – a derivative of DDB, widely used for treating liver diseases in China and other countries – has been shown to reduce liver toxicity and liver fibrosis significantly in experimental and clinical studies, and to possess significant anti-inflammatory activity.11–15 A self-case–control study (evaluating the effect of bicyclol as a liver protectant in 27 patients) showed that bicyclol 75 mg/day could effectively treat chemotherapeutic agent-induced liver toxicity, and significantly decrease the frequency and degree of liver damage, when administered in combination with chemotherapy compared with chemotherapy alone. 16 There are several relatively small published studies that verify the efficacy of hepatoprotective drugs against chemotherapeutic agent-induced liver injury,17–21 but it remains unclear whether bicyclol, as an orally administered drug, plays a role in reducing levels of such injury.
The present randomized, prospective, controlled clinical study was conducted to explore the incidence of chemotherapeutic agent-induced liver injury among patients ≥60 years of age undergoing chemotherapeutic treatment for cancer, and to evaluate the role of bicyclol in preventing such injuries.
Patients and methods
Study population and design
This randomized, prospective, controlled clinical study was conducted in the department of Medical Oncology, Peking Union Medical College Hospital, Peking, China between January 2009 and September 2012. Patients with cancer were equally randomized into one of two groups: a prophylactic group (chemotherapy supplemented with 75 mg bicyclol, oral, daily) and a control group (chemotherapy alone). Inclusion criteria were: patients with cancer ≥60 years of age; normal baseline liver functions; no use of chemotherapeutic agents in the previous 1 month. Exclusion criteria were: history of progression of previous liver metastasis following chemotherapy; reactivation of viral hepatitis.
A review of the literature indicated that the incidence of chemotherapeutic agent-induced grade I–IV liver injury was ∼35%, which was higher than the 20% incidence observed when chemotherapy was combined with hepatoprotective drugs.16,17 To conduct a two-sided test with an α-level of 0.05 and power of 0.80, ≥135 patients in each group were required. To allow for losses to follow-up and other incidents, the study aimed to enroll a total of 153 patients into each of the prophylactic and control groups, respectively. A randomization table was created using SPSS® version 16.0 (SPSS Inc., Chicago, IL, USA).
Investigations undertaken in this study were conducted in accordance with the ethical recommendations of the Declaration of Helsinki (World Medical Association) and Good Clinical Practice. The study was approved by the Ethics Committee of Peking Union Medical College Hospital. Written informed consent was obtained from each patient following a detailed description of the potential benefits and risks of the study.
Drug administration
Patients in the prophylactic group received 25 mg bicyclol (Beijing Union Pharmaceutical Factory, Beijing, China), oral tablet form, three times per day, on initiation of cancer chemotherapy. Bicyclol was administered until the end of all cycles of the studied chemotherapeutic regimen. Patients in the control group received chemotherapeutic agents alone (without bicyclol supplementation).
Monitoring of liver function and toxicity
Levels of alanine transaminase (ALT), aspartate aminotransferase (AST) and total bilirubin (TBIL) were monitored immediately prior to treatment, at 1 week following initiation of chemotherapy and immediately following the end of each chemotherapy cycle. Liver toxicity assessment continued until the end of the studied chemotherapy regimen or the occurrence of grade II–IV liver injury according to the National Cancer Institute Common Terminology Criteria for Adverse Events (CTCAE; version3.0). In both groups, when patients experienced grade II–IV liver injury (defined as >2.5 times the upper limit of normal [ULN] for serum transaminase or >1.5 times ULN for bilirubin, hepatoprotective drugs (including glycyrrhizic acid drugs or bicyclol) were administered and the prophylactic assessment was stopped. The primary endpoint was the occurrence of grade I–IV liver injury (elevation of serum transaminases and/or bilirubin above ULN) according to the CTCAE.
Statistical analyses
Statistical analyses were performed using SPSS® version 16.0 for Windows®. Between-group differences in categorical data were analysed using χ2-test; between-group differences in continuous data were analysed using independent-samples t-test. To assess the independency of bicyclol in preventing liver injury, unconditional logistic regression was used for multivariate analysis, adjusting for variables considered in the subgroup analysis (age, chemotherapy course, chemotherapy regimen, hepatitis B virus [HBV] carrier, previous chemotherapy, liver metastasis, stage of cancer, sex). Differences in between-group distribution of variables were assessed using Wilcoxon’s rank sum test. A P value <0.05 was considered to be statistically significant. Data were presented as mean (±SD), unless otherwise stated.
Results
Patient demographics and cancer treatment
Demographic and baseline data for patients ≥60 years of age with cancer, who received chemotherapy combined with 25 mg bicyclol, orally, three times a day (prophylactic group) or chemotherapy alone (control group).

Data presented as n (%) or mean (range).
American Joint Committee on Cancer staging system: 6th edition for patients enrolled before April 2011; 7th Edition for patients enrolled during or after April 2011.
XELOX, oxaliplatin and capecitabine; FOLFOX, oxaliplatin, leucovorin, and 5-fluorouracil; SOX, oxaliplatin and S-1; CHOP, cyclophosphamide, doxorubicin, vincristine, and prednisone; HBV, hepatitis B virus.
No statistically significant between-group differences were observed (P > 0.05; χ2-test).
Incidence of liver injury
Incidence of liver injury in patients ≥60 years of age with cancer who received chemotherapy combined with 25 mg bicyclol, orally, three times a day (prophylactic group) or chemotherapy alone (control group).
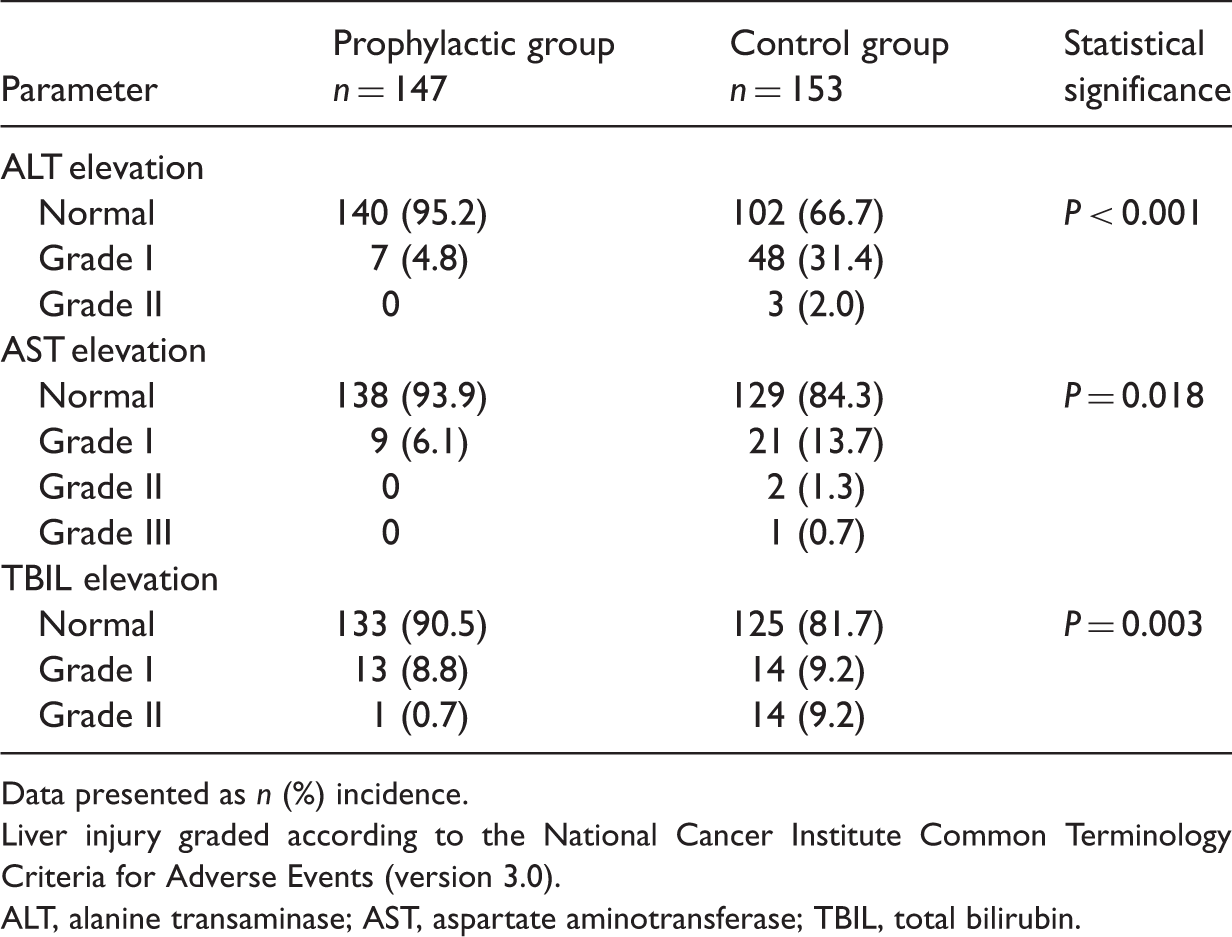
Data presented as n (%) incidence.
Liver injury graded according to the National Cancer Institute Common Terminology Criteria for Adverse Events (version 3.0).
ALT, alanine transaminase; AST, aspartate aminotransferase; TBIL, total bilirubin.
Subgroup analyses
Subgroup analyses of prevention of liver injury using bicyclol in patients with cancer ≥60 years of age who received chemotherapy combined with 25 mg bicyclol, orally, three times a day (prophylactic group) or chemotherapy alone (control group).

Data presented as n incidence of raised liver enzymes/n total incidence (%).
CT, chemotherapy; FP, fluoropyrimidine; HBV, hepatitis B virus; OR, odds ratio; CI, confidence interval.
NS, no statistically significant between-group difference (P ≥ 0.05; χ2-test).
Multivariate analysis of prevention of liver injury using bicyclol, in patients ≥60 years of age with cancer, who received chemotherapy combined with 25 mg bicyclol, orally, three times a day (prophylactic group) or chemotherapy alone (control group).

CT, chemotherapy; HBV, hepatitis B virus; OR, odds ratio; CI, confidence interval.
NS, no statistically significant difference (P ≥ 0.05).
Discussion
The present study, investigating the role of bicyclol in preventing liver injury among patients ≥60 years of age with cancer, found that the incidence of chemotherapeutic agent-induced liver injury in the control group (47.1%) was higher than the incidence (36.6%) reported in a published retrospective study. 10 This may be due to differences in sample size and frequency of liver function tests between the two studies. A lower incidence (17.1%) and lower rate of higher-grade liver injury (0.7%) were both observed in the bicyclol-prophylactic compared with control group in the present analysis, which concurred with the findings of similar published studies.17,20,21 Further subgroup analyses in the present study showed that the lower incidence of liver damage was statistically significant in the prophylactic group, but not in the subgroups of patients who were positive for HBV or who had a previous history of chemotherapy. The lack of statistical significance between the prophylactic and control groups might have been because there were only two and four HBV carriers in the prophylactic and control groups, respectively, thus too few to hold an enough power. Similarly, in the subgroup analysis among patients with previous history of chemotherapy, the lack of statistical significance might have been due to the small sample size. Multivariate analysis showed that use of bicyclol was statistically significant in preventing liver injury after adjusting for factors considered in the subgroup analysis. These results indicated that administration of bicyclol for prophylaxis in preventing liver injury was an independent factor in liver protection. The incidence of grade II–IV hepatic injuries was significantly lower in the prophylactic group (0.7%) compared with the control group (12.4%). In many clinical trials, liver toxicity more severe than grade II (as per the CTCAE) is not usually considered as an inclusion criteria, and in clinical practice, a course of chemotherapy might be delayed when liver toxicity is more severe than grade II. Thus, supplementary administration of bicyclol to reduce the incidence of grade II–IV hepatic injury has important clinical relevance.
Several trials with a limited number of cases have reported the use of hepatoprotective drugs in patients at risk of chemotherapeutic agent-induced liver injury.17,20,21 Tiopronin was reported to be effective in the prevention of hepatotoxicity caused by chemotherapy in patients with malignant tumours: 17 incidence of liver injury was lower in a prophylactic group treated with tiopronin compared with a control group (13.5% versus 37.5%, respectively). Two studies investigating the hepatoprotective effects of S-adenosyl methionine administrated as a supplemental therapy included 105 patients with colorectal cancer receiving adjuvant FOLFOX regimen and 74 patients with metastatic colorectal cancer receiving bevacizumab plus XELOX regimen, respectively.20,21 S-adenosyl methionine was administrated as a supplemental therapy in 60 and 32 patients, respectively. Both studies reported that patients supplemented with S-adenosyl methionine experienced a significantly lower incidence of liver damage (30.0% and 34.4% in supplemented groups, respectively, versus 66.7% and 58.7% in control groups, respectively),20,21 lower grade of liver toxicity, reduced need for course delays and chemotherapy dose reductions, compared with patients receiving chemotherapy alone.
The present study explored the incidence of chemotherapeutic agent-induced liver injury in older patients and the role of bicyclol in protecting the liver against such toxic effects. The results showed that bicyclol had a strong effect in protecting the liver against chemotherapeutic agent-induced injury, and the findings were consistent with published studies.17,20,21
Chemotherapy-course delays may involve heterogeneous factors other than liver injury. 22 The present research is limited by the fact that differences in course delays between the two study groups were not investigated. Because of the limited study design, differences in chemotherapy response rates and performance status between the two groups were also not been investigated. Further research is required to understand the mechanisms of bicyclol in preventing chemotherapeutic agent-induced liver injury and explore chemotherapy course delays, discontinuations and dose reductions due to liver toxicity. The present study included patients with different forms and grades of cancer undergoing different chemotherapy regimens, however, the cases were well balanced between the two study groups. Multicentre trials focusing on specific cancer types and chemotherapy regimens would provide further understanding regarding the hepatoprotective role of bicyclol.
In conclusion, the results of the present study suggest that there is a high incidence of chemotherapy-induced liver injury in patients ≥60 years of age with cancer, and that bicyclol may be an efficient liver protectant. Supplementary intake of bicyclol may help patients to tolerate chemotherapy and significantly reduce the liver-toxicity effects in clinical practice. Due to the limited number of patients and relatively short duration of treatment in the present study, further clinical trials are needed to verify the efficacy of bicyclol in chemotherapy agent-induced liver injury.
Footnotes
Declaration of conflicting interest
The authors have no potential conflicts of interest that are directly relevant to the content of this study.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
