Abstract
Objective
To investigate the efficacy of the intraoperative posterior pericardial window technique in preventing pericardial tamponade following open heart surgery.
Patients and methods
Adult patients undergoing coronary and/or valve surgery were randomly divided into a control (traditional) or a pericardial window (PW) technique group. Pre-, intra-, peri- and postoperative clinical data were collected prospectively, including incidence of pericardial tamponade, cardiac arrest, drainage volume, ventilation assistance time and moderate-to-large pericardial effusion.
Results
In total, 458 patients were included: 230 controls and 228 in the PW group. The incidence of pericardial tamponade in the PW group was significantly lower than in controls. Cardiac arrest occurred in one patient (0.4%) in the PW group and five (2.2%) controls; this difference was not statistically significant. Moderate-to-large pericardial effusion after drainage extubation and new-onset atrial fibrillation were significantly more common in controls than in the PW group. After stratification by age (≤70 versus >70 years), there was no between-group difference in duration of endotracheal intubation, although in the PW group, after removal of the tracheal cannula, duration of noninvasive positive pressure ventilation was significantly longer in older patients.
Conclusions
The pericardial window procedure did not increase the rate or severity of procedure-related complications. This simple technique significantly decreased the incidence of postoperative pericardial tamponade and new-onset atrial fibrillation.
Introduction
Pericardial effusion is commonly encountered following open heart surgery, with an incidence as high as 53–85%;1,2 2% of these cases can progress to pericardial tamponade. 3 Even if the effusion is small and localized rather than circumferential, 2 it may lead to supraventricular arrhythmia and haemodynamic derangement.4–6
After surgery, patients lie in the supine position, which can easily cause localized accumulation of fluids in the posterior pericardial cavity: Kuvin et al. 7 demonstrated that 49% of pericardial effusion events were posterior. Moreover, Pepi et al. 3 reported that local effusion (57.8%) was more frequent than diffuse effusion (42.2%). However, the traditional pericardial cavity and mediastium drainage pathway, with one chest tube placed along the right atrium and the other placed retrosternally in the anterior mediastinum, hardly reaches the posterior regions. 5 Others8,9 have also experienced failure when attempting to aspirate posterior effusion by percutaneous pericardiocentesis. Although Georghiou et al. 10 and Mangi et al. 11 successfully resolved delayed posterior effusion after cardiac surgery by either video-assisted pericardial fenestration or fluoroscopic guidance, the procedures were arduous and complicated, requiring general anaesthesia and single-lung ventilation.
As a result, we have investigated a novel drainage approach, the pericardial window technique, 12 to achieve a free passage between the limited pericardial cavity and the larger pleural cavity. Theoretically, this manoeuvre facilitates the transmission of pressure from the pericardial cavity to the pleural space below. Intrapericardial pressure increases during intrapleural saline infusion in the same way as intrapleural pressure. Nevertheless, cardiac output and aortic blood pressure are better preserved, pulmonary artery systolic blood pressure is higher, and the degree of pulsus paradoxus is lower with intrapleural infusion. 13 The present study assessed the efficacy of the pericardial window technique in preventing postoperative tamponade and effusion-related complications with open heart surgery.
Patients and methods
Patient groups and characteristics
The present study was undertaken between January 2012 and January 2013 in adult patients (≥18 years old) who underwent coronary artery bypass grafting (CABG) and/or valve operation at Zhengzhou University People’s Hospital and Henan Provincial People’s Hospital (this is the same institution). Patients’ daily function and quality of life were classified according to the New York Heart Association system. Patients were prospectively randomized using a computer-generated randomization table into a control group and a pericardial window group (PW group). For each patient, the randomization result was concealed in a sealed opaque envelope which was not opened until the patient was put under general anaesthesia. Patients with hypertension (systolic level, >140 mmHg, diastolic level >90 mmHg), type 2 diabetes mellitus or chronic obstructive pulmonary disease were included in the study. Patients were excluded if they had prior sternotomy or cardiac surgery, paroxysmal atrial fibrillation, preoperative coagulant disorders that could have influenced the postoperative results, asthma, and hepatic or renal dysfunction. The Institutional Review Boards of Zhengzhou University People’s Hospital and Henan Provincial People’s Hospital approved the study, and all patients provided written informed consent.
For all patients, the following demographic, pre-, intra-, peri- and postoperative characteristics were recorded: (1) factors predisposing to the development of postoperative effusion 14 such as age, sex, body mass index, hypertension, duration of cardiopulmonary bypass and coagulation profile; (2) incidence of postoperative cardiac tamponade, cardiac arrest (excluding arrests due tocardiogenic factors and electrolyte disorders; included arrests were those caused by pericardial tamponade as identified at reoperation) and blood transfusion; (3) total drainage volume, incidence of pericardial effusion and volume of effusion collected by thoracentesis or pericardiocentesis after removal of chest tube; (4) duration of mechanical ventilation and noninvasive positive pressure ventilation; (5) occurrence of new-onset atrial fibrillation and duration of intensive care unit (ICU) stay. Some parameters (such as mechanical ventilation time and noninvasive positive pressure ventilation time) were stratified by age ≤70 years or >70 years.
Operative techniques
During the study period, the study centre’s standard clinical protocols remained unchanged and there was no alteration in the senior surgical or anaesthetic consultant staff.
Cardiac surgery
Prior to anaesthesia, continuous electrocardiogram monitoring of leads I, II and V5 with automated ST-segment analysis was initiated. The radial artery was cannulated under local anaesthesia to monitor blood pressure directly and to sample blood gases. Anaesthesia was induced with intravenous injection of fentanyl (10 µg/kg), midazolam (0.1 mg/kg) and propofol (2 mg/kg); anaesthesia was maintained with isoflurane 1% in oxygen, entanyl (3 µg/kg per h) and midazolam (0.2 µg/kg per min). Endotracheal intubation was achieved after the intravenous administration of pancuronium bromide (0.03 mg/kg per h). A triple-lumen central venous catheter was inserted into the internal jugular vein to monitor central venous pressure, vasoactive drug infusion and blood gas levels.
All patients underwent medial sternotomy. The left internal mammary artery was the graft of choice for the left anterior descending coronary artery, and saphenous vein grafting was performed for other anastomoses. In off-pump CABG patients, heparin was given (150 U/kg; activated clotting time ≥200 s; Tianjin Pharma, Tianjin, China). Proximal anastomosis was performed with aortic partial clamping; distal grafting was performed using a stabilizer (Octopus; Medtronic, Minneapolis, MN, USA). In on-pump CABG and/or valve operation patients, 300 U/kg heparin was used (activated clotting time ≥480 s; Tianjin Pharma). Cardiopulmonary bypass (CPB) was established with a roller pump (nonpulsatile flow between 2.0 and 2.4 l/m2 per min). Alpha-stat arterial carbon dioxide tension management was used, and arterial pressure was maintained at 50–80 mmHg. Single cross-clamp antegrade and retrograde blood cardioplegia was used, which was repeated every 20 min before the removal of the cross-clamp. Heparin was neutralized at the end of the CPB stage by protamine (1 mg for every 100 U of initial dose of heparin). Cell saver and autologous blood transfusion were performed as required. Blood products were not used unless haematocrit was <27%. Extubation was performed according to blood gas levels (inspired O2 40%, PaO2 >70 mmHg). Before surgery was completed, patient-controlled postoperative analgesia was initiated via intravenous infusion of 100 ml per kg of 20 mg/ml fentanyl citrate and 16 mg/8 ml ondansetron hydrochloride in 0.9% normal saline, infused continuously at a basic flow rate of 2 ml/h. If the patient experienced pain, a 0.5 ml bolus was given via an automated patient-controlled analgesia pump.
Pericardial window
Each patient in the PW group underwent the pericardial window technique under direct vision (Figure 1). First, the phrenic nerve was identified and an inverse-T incision (2.5 cm long in both dimensions) was created. Gauze was used to shield the moving (inflating) lung and, before incision, surgeons identified the phrenic nerve to avoid damaging it. The incision was made into the pleural cavity by noncontinuous electrocautery, parallel and posterior to the phrenic nerve and extending from the left inferior pulmonary vein to the diaphragm, as described by Mulay and coworkers.
5
As well as protecting the phrenic nerve, adipose tissue around the incision was staunched by electrocautery. Protocols were adjusted in the following conditions: (1) if right or bilateral pleurae were inadvertently opened during the sternotomy, a right or bilateral pericardial window technique was performed; (2) if the patient had dense adhesions to the pleurae, the patient was treated by the traditional method.
Position of the inverse-T incision relative to the phrenic nerve with the pericardial window technique.
A 28 F rubber tube was placed in anterior mediastinum and a 28 F silicone tube was inserted into the left and/or right pleural cavity, through the seventh intercostal space at the left midaxillary line. The tubes were separately connected by a waterseal drainage device. If bilateral pleurae were opened, a second 28 F silicone tube was inserted. To avoid tube-induced arrhythmia, no drain was placed retrocardially. The tubes were secured to the skin with 4-0 silk sutures and covered with a sterile dressing. After routine closure of the chest, the drains were milked and stripped at 30 min intervals to ensure patency.
Traditional drainage
In the control group, a 28 F rubber drain tube was placed along the right atrium in the pericardium and a 32 F silicone tube was placed retrosternally in the anterior mediastinum overlying the heart. The tubes were connected using a T-shaped, negative-pressure, drainage device. Tubes were secured, covered and drained as for the PW group.
Chest tube removal
In the PW group, the mediastinal drain was routinely removed on the first postoperative day and the pleural drain was maintained until the daily amount of drainage was <100 ml. In the control group, because of the T connection, both drains were maintained until the daily amount of drainage was <100 ml.
Effusion measurement and categorization
After removal of the drains, pleural effusion was measured by echogenicity of 2D-echocardiography. Echo procedures were routinely performed at 6, 24, 48 and 72 h postoperation and on the day before discharge. Patients were kept in the supine position, at an angle of 60° for each measurement, and all echo procedures were performed by the same investigator (X.Q.). Effusion was categorized by the investigator (X.Q.) according to size, as follows: small, <40 mm; moderate, 40–80 mm; large, >80 mm. Similarly, pericardial effusion was measured by 2D-echocardiography and categorized according to size and site as follows: small, effusion localized in posterior pericardial cavity, <10 mm; moderate, effusion involved with anterior wall of right ventricle, 10–20 mm; large, circumferential effusion, >20 mm.
On postoperative days 3 and 5, and before discharge, 2D-echocardiography was routinely performed to ascertain that there was no recurring effusion in the pericardial or pleural cavity.
Thoracentesis and pericardiocentesis
The indication for thoracentesis was moderate-to-large pleural effusion. Considering the high risk of pericardiocentesis, the indication for pericardiocentesis was strict: (1) under haemodynamic instability, pericardiocentesis was immediately performed; (2) if haemodynamic parameters were stable, diuretic therapy was initiated, and in cases where this was ineffective, pericardiocentesis was performed.
Statistical analyses
Characteristics were described as mean ± SD or n (%). Two-sample t-test and two-tailed Fisher’s exact test were used to compare continuous and dichotomous variables between groups, respectively. Differences were considered statistically significant at P < 0.05. Analyses were performed using SPSS® version 13.0 (SPSS Inc., Chicago, IL, USA).
Results
Comparison of demographic, preoperative and intraoperative characteristics of patients undergoing open heart surgery with effusion aspirated by either the pericardial window pathway or the traditional pericardial cavity and mediastium drainage pathway (control).
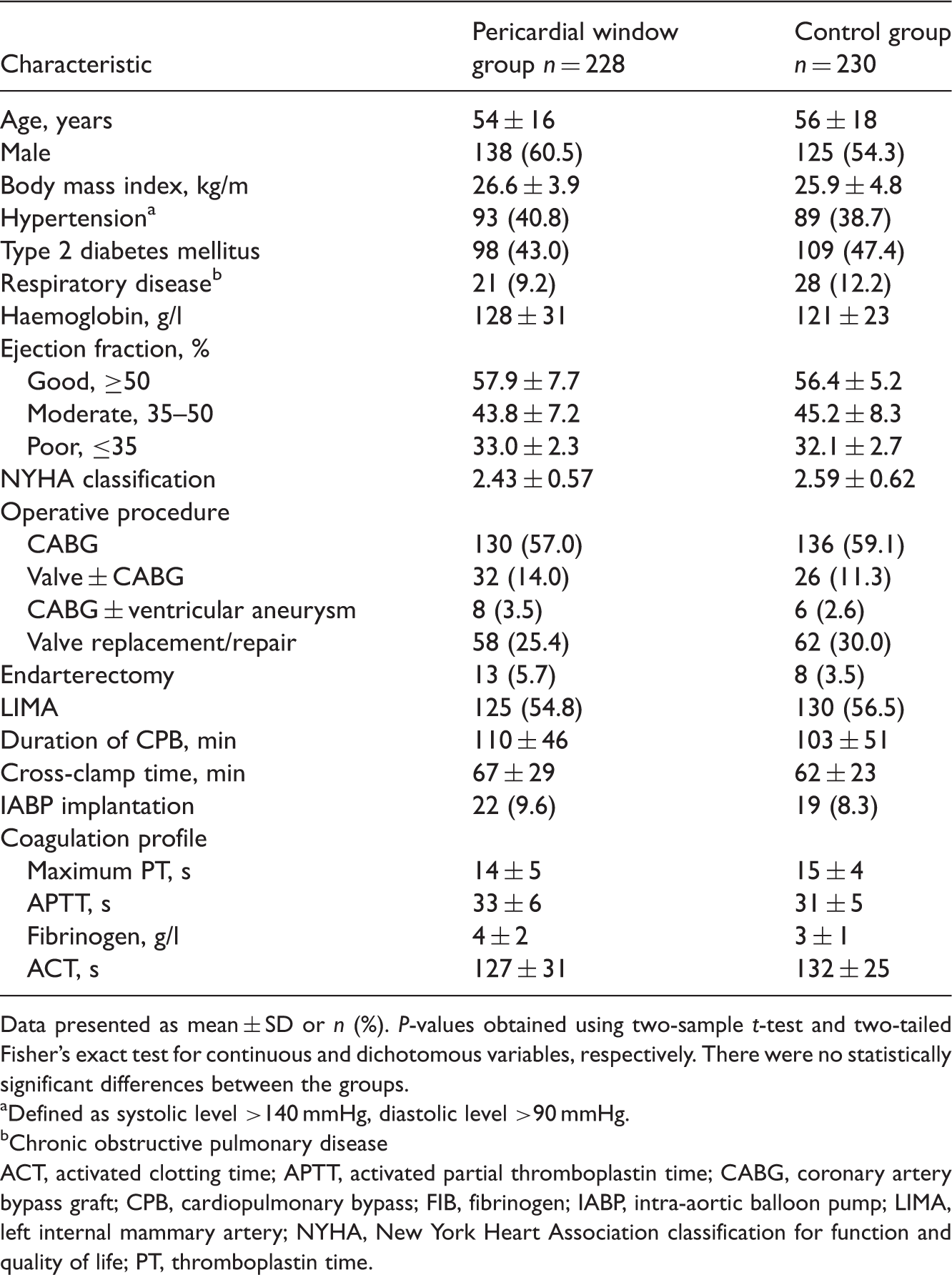
Data presented as mean ± SD or n (%). P-values obtained using two-sample t-test and two-tailed Fisher’s exact test for continuous and dichotomous variables, respectively. There were no statistically significant differences between the groups.
Defined as systolic level >140 mmHg, diastolic level >90 mmHg.
Chronic obstructive pulmonary disease
ACT, activated clotting time; APTT, activated partial thromboplastin time; CABG, coronary artery bypass graft; CPB, cardiopulmonary bypass; FIB, fibrinogen; IABP, intra-aortic balloon pump; LIMA, left internal mammary artery; NYHA, New York Heart Association classification for function and quality of life; PT, thromboplastin time.
Comparison of perioperative characteristics of patients undergoing open heart surgery with effusion aspirated by either the pericardial window pathway or the traditional pericardial cavity and mediastium drainage pathway (control).

Data presented as mean ± SD or n (%). P-values obtained using two-sample t-test and two-tailed Fisher’s exact test for continuous and dichotomous variables, respectively.
AF, atrial fibrillation; ICU, intensive care unit; NIPPV, noninvasive positive pressure ventilation.
Pleural and pericardial effusion after removal of chest tube in patients undergoing open heart surgery with effusion aspirated by either the pericardial window pathway or the traditional pericardial cavity and mediastium drainage pathway (control).

Data presented as mean ± SD or n (%). P-values obtained using two-sample t-test and two-tailed Fisher’s exact test for continuous and dichotomous variables, respectively.
Size of effusion by 2D-echocardiography: small, <40 mm; moderate-to-large >40 mm.
Site and size of effusion by 2D-echocardiography: small, localized in posterior pericardial cavity, <10 mm; moderate- to-large: involved with anterior wall of right ventricle or circumferential, >10 mm.
One case of moderate-to-large pericardial effusion showed haemodynamic stability after diuretic therapy and did not undergo pericardiocentesis.
An additional analysis was performed after stratification by age (Table 2). In patients aged ≤ 70 years, there was no significant difference in the duration of mechanical ventilation and noninvasive positive pressure ventilation between the groups. However, in patients aged >70 years, duration of mechanical ventilation was longer in the PW group than in the control group. Furthermore, in the PW group, after removal of the tracheal cannula, the duration of noninvasive positive pressure ventilation was longer. In addition, the duration of ICU stay was slightly, but not significantly, longer.
In the PW group, three cases developed pericardial tamponade with hypotensive, oliguric and peripheral vasoconstriction, which were identified and resolved by reoperation. One of these cases (with saphenous vein graft) was due to dropping of the haemoclip. At urgent bedside reoperation, active bleeding and rapidly accumulating effusion were disclosed, and, unexpectedly, a large blood clot presented a herniation at the inverse-T incision. The two other cases of tamponade occurred after standard postoperative analgesia was stopped: the patients experienced dysphoria, hypertension (systolic blood pressure >200 mmHg) and subsequently showed the clinical features of tamponade. In both patients, transthoracic echocardiography showed no evidence of localized pericardial tamponade, except for distension of the inferior vena cava. Reoperations disclosed discrete bleeding from the right atrial incision and each had a large clot (5 × 6 cm) that surrounded and compressed the right atrium and ventricle. Surprisingly, the incision still kept patency and the posterior pericardial cavity did not show any accumulation of effusion. Prompt evacuation of the blood clot led to immediate improvement in haemodynamic parameters, in both patients.
In the control group, 13 cases encountered pericardial tamponade. Two of these cases were resolved by thoracentesis instead of resternotomy: one patient underwent valve replacement and prolonged CPB; the other patient had a preoperative history of frequent chest pain, and tirofiban (0.4 µg/kg per min; Yuanda Pharma, Wuhan, China) and heparin (10 U/kg per min) were administered until sternotomy. Both patients presented with loss of blood from the wound surface at the end of operation after surgically active bleeding had ceased. After ∼4–6 h in ICU, the patients presented haemodynamic instability. Bedside transthoracic echocardiography did not reveal pericardial tamponade and only indicated a large left pleural effusion. By thoracentesis, the pleural effusion showed a projectile feature that implied high intrapleural pressure. A total of 2.2–2.5 l of incoagulable blood was drained from each patient, and their clinical signs improved immediately. The other 11 cases of pericardial tamponade in the control group were resolved by resternotomy.
Discussion
The pericardial window method was technically easy to perform during heart surgery and did not increase patient morbidity or the number of surgical procedures. There were no serious complications during surgery except for one case with bleeding from the inverse-T incision. Subsequently, the surgeons routinely cauterized the incisions and checked for bleeding, and there were no further cases of bleeding from incisions. Therefore, the experience of this study centre is that a T-shaped incision 2.5 cm long in both dimensions is appropriate for the pericardial window method. Yorgancioğlu et al. 15 reported a protrusion of a sequential bypass graft after CABG from the pericardiotomy side; theoretically, the heart could be squeezed by the pericardiotomy edges, which would increase the risk of an incisional herniation. Also, a larger incision could lead to the heart losing support from the pericardium and moving within the pericardial cavity.
Our finding of a significantly greater total drainage volume with the pericardial window technique compares favourably with the result of Farsak et al. 4 The current findings of more cases and greater volume of pleural effusion, and fewer cases of moderate-to-large pericardial effusion and postoperative new-onset atrial fibrillation in the PW group, indicate that the pericardial window technique provides an effective drainage pathway from the pleural cavity. Without the use of this technique, fluid may collect in the pericardium and compress the heart.
Overall, use of the pericardial window technique resulted in a lower rate of tamponade than the traditional drainage route. However, there were still three cases of tamponade in the PW group. In the case where dropping of the haemoclip with saphenous vein graft (leading to active bleeding, rapidly accumulating effusion and incisional herniation), it is speculated that the rapid formation and collection of the clot before blood could drain from the pericardial cavity resulted in complete occlusion of the incision and that, once the clot had occluded the incision, intrapericardial pressure rose steeply and impaired cardiac filling. For the two cases with discrete bleeding from the right atrial incision and a large clot around the right atrium and ventricle leading to tamponade, we speculate that the pericardial window technique for posterior effusion may be unreliable for localized, rapidly formed clot masses at locations other than around the right atrium and ventricle. Transthoracic echocardiography before reoperation did not disclose the localized pericardial tamponade, possibly because the early effusion with a low echogenicity gradually became a clot with higher echogenicity, which made it difficult to differentiate it from surrounding soft tissue.
In the control group, 13 cases encountered pericardial tamponade, two of which were unexpectedly relieved by simple thoracentesis. We speculate that, although these cases did not undergo the pericardial window technique, the accumulated effusion in the anterior mediastinum drained into the pleural cavity through a small undetectable gap in the pleural cavity. Transthoracic echocardiography did not reveal pericardial tamponade in the other cases because of the effect of mechanical ventilation on lung tissue. With the accumulation of pleural fluid, the increasing intrapleural pressure was transmitted to the pericardial space, resulting in tamponade-like physiology, namely ‘extrapericardial tamponade’.16–19 Vaska et al. 13 demonstrated in a canine model that right ventricular collapse is produced by intrapleural fluid infusion into the pleural space, and that there is a linear relationship between intrapericardial and intrapleural pressure. The other cases of pericardial tamponade were resolved by resternotomy because large clots in the pericardial cavity cannot be resolved by pericardiocentesis.
On the basis of the present study results, use of the pericardial window technique decreased the incidence of tamponade but did not completely prevent its occurrence, especially in patients with active bleeding in the early postoperative period. We consider that the pericardial window technique might be most suitable for patients with coagulation disorders, because the effusion can be immediately drained into the larger pleural cavity, reducing the risk of rapid accumulation of intrapericardial fluid and a steep rise in pressure around the heart.
The incidence of moderate-to-large pericardial effusion in the control group was higher than in the PW group, in the present study. Fluid accumulating in the pericardium presents a mechanical stimulus to the atria, which can trigger arrhythmia, 5 and the current study observed a decrease in the incidence of atrial fibrillation in the PW group compared with the control group, which is consistent with that observed by Farsak et al. 4 Conversely, the current study demonstrated a greater volume recovered by thoracentesis in the PW group, which implies that the pericardial window technique provides a reliable drainage pathway between the pericardial and pleural cavities. Furthermore, continuous respiratory movement with the pericardial window technique removes fibrinogen from the effusion, which results in uncoagulated blood collecting in the pleural cavity that can be easily drained out through the lateral chest tube. This contrasts with the tendency for clots to form in the drainage tubes when the traditional pathway is used. However, at postoperative day 10, there was one case of moderate effusion in the PW group, in which the pericardium was diffusely adherent to the epicardium by fibrinous adhesions; this resulted in the occlusion of the window itself, as described by Sugimoto et al. 12
Compared with those aged ≤70 years, patients aged >70 years in the PW group needed longer ventilation assistance time. The pericardial window technique damages the negative pressure and closed characteristics of the pleural cavity and leads to alveolar collapse, or even atelectasis. 20 However, the significantly lower incidences of both pericardial tamponade and atrial fibrillation observed in the present study suggest that the advantages of the pericardial window technique offset its disadvantages.
One limitation of the present study is the sample size, which was too small to find significant differences in rates of cardiac arrest between the groups. Pericardial tamponade is a rare complication and, therefore, has a low rate of occurrence. Consequently, studies with larger sample sizes may reveal statistically significant findings in relation to the rate of cardiac arrest observed with the pericardial window technique. Another limitation of the present study is that there were no infant cases, so this study could not assess the efficacy of the pericardial window technique in this patient group. On balance, in the authors’ opinions, the pericardial window technique may be valuable in infants. Although the pericardial space in infants is smaller than in adults (and consequently pericardial compliance is limited), the authors of the present study would be in favour of using the pericardial window technique in infants undergoing complicated congenital heart surgery and prolonged CPB time, which has an adverse effect on coagulation function. However, the present study findings do not support using the pericardial window technique in infants requiring simple heart surgery (such as atrial or ventricular septum defects).
In conclusion, the present study findings show that that the pericardial window technique is simple, easy to perform and without serious complications. The technique is useful in reducing not only the incidence of pericardial tamponade, but also in reducing cases of effusion-related atrial fibrillation. For patients with marked effusion due to coagulation dysfunction, the intraoperative pericardial window technique is suggested.
Footnotes
Declaration of conflicting interest
The authors declare that there are no conflicts of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
