Abstract
Objective
To investigate changes in standard base excess (SBE) when administering two different infusion regimens for elective hip replacement within a goal-directed haemodynamic algorithm.
Methods
This prospective, double-blind, randomized, controlled study enrolled patients scheduled for primary hip replacement surgery, who were randomized to receive either an unbalanced crystalloid (chloride: 155.5 mmol/l) or a 1 : 1 mixture of a balanced crystalloid and a balanced colloid (6% w/v hydroxyethyl starch 130/0.42; chloride: 98 and 112 mmol/l, respectively). Fluid management was goal-directed to optimize stroke volume using oesophageal Doppler.
Results
A total of 40 patients (19 female/21 male) participated in the study. After surgery, median (25–75% percentiles) SBE was significantly lower in the unbalanced group compared with the balanced group: −2.0 mmol/l (−3.1 to −1.1) versus −0.4 mmol/l (−1.2 to 0.7), respectively. This difference was mainly due to greater plasma chloride concentrations in the unbalanced group. The amount of study medication required to reach haemodynamic stability (median 1200 ml) did not differ between the two groups.
Conclusion
SBE decreased in the unbalanced group without influence on fluid requirements and haemodynamic stability.
Keywords
Introduction
Every year, an estimated 234.2 million operative procedures are performed worldwide. 1 The overall mortality rate after surgery is 4% and varies between countries.1,2 Intraoperative fluid and volume therapy is a cornerstone of perioperative care, and goal-directed therapy has been shown to substantially reduce postoperative complication rates and hospital length of stay in different types of surgery. 3
There is controversy about the kind of intravenous solutions (unbalanced versus balanced; crystalloids versus colloids) that should be used for fluid replacement and haemodynamic optimization.4,5 Compared with 0.9% (w/v) sodium chloride (NaCl; containing 154 mmol/l Na+ and 154 mmol/l Cl−), balanced solutions have an electrolyte composition closer to plasma and contain bases such as acetate and lactate that can be metabolized to bicarbonate, so they are therefore also referred to as buffered solutions. However, although in acute trauma a decrease of the base excess presumably due to hyperlactataemia is a strong indicator for an increased morbidity and mortality,6–9 unbalanced solutions are still widely used despite an additional decreasing effect on base excess through hyperchloraemic acidosis.10–13
For short-lasting elective (trauma) surgery, crystalloids are generally recommended for perioperative fluid and volume replacement;14,15 in contrast to this, goal-directed haemodynamic management in trauma surgery is regularly performed with colloid solutions.16,17
To date, there have not been any studies that have compared the usual standard, i.e. an unbalanced crystalloid, with an infusion strategy that uses a mixture of both balanced crystalloids and colloids within a goal-directed haemodynamic algorithm. Earlier studies researching this issue could not exclude interference with occult hypoperfusion or hypervolaemia as they neither measured circulatory flow nor did they guide the administration of study fluids by advanced target controlled haemodynamic monitoring.12,13 With regard to the current concept of infusion therapy, hydroxyethyl starch (HES) should only be used for acute blood loss in noncritically ill patients without kidney disease. 18
The primary objective of this present study was to investigate changes in the standard base excess (SBE) as a key parameter of the metabolic acid–base status when applying two different infusion regimens for elective hip replacement within a goal-directed haemodynamic algorithm. In one group, an unbalanced chloride rich crystalloid solution was infused, whereas in the other group a balanced crystalloid mixed 1 : 1 with a balanced colloid was infused. Both concepts are considered standard treatments in different medical communities around the world. As secondary outcome measures, further acid–base parameters, the amount of perioperative fluid infusions applied, haemodynamic parameters and renal function were investigated.
Patients and methods
Study design
The study was conducted as a single-centre, randomized, double-blind, parallel-group study, registered with Clinicaltrials.gov (NCT01117519) and EudraCT (2009-016043-19) and approved by the Bundesinstitut für Arzneimittel und Medizinprodukte (BfArM-Nr. 4036063). Ethics approval was obtained from the Landesamt für Gesundheit und Soziales (LaGeSo), Ethical Committee of Berlin (chairman of board 6: Dr J. Hamann), Germany, on 26 April 2010 (ZS EK 15 100/10).
Eligible patients were ≥60 years old and scheduled for primary elective hip replacement surgery in the Centre of Musculoskeletal Surgery, University Hospital Charité, Campus Charité Mitte, Berlin, Germany, between May 2010 and November 2011. Written informed consent was obtained from each patient at least 1 day prior to surgery.
The patients were stratified preoperatively according to the American Society of Anesthesiologists Physical Status Classification System (ASA 1 and 2 versus ASA 3 to rule out confounding due to an impaired health status). 19 To determine demographic and clinical characteristics beside ASA, the risk for postoperative nausea and vomiting (APFEL score), 20 the Physiological and Operative Severity Score for the enUmeration of Mortality and Morbidity (POSSUM) score,21,22 the Mini Mental State Exam (MMSE) points, 23 the health status (EQ-5D) of the EuroQol group on admission, 24 and the postoperative cognitive deficit (POCD) using the Cambridge Neuropsychological Test Automated Battery (CANTAB) and International Study of Post-Operative Cognitive Dysfunction (ISPOCD) neuropsychological batteries were measured. 25 Complications were documented according to the Clavien–Dindo classification. 26 There was an extensive list of exclusion criteria, which is available from the corresponding author upon request. The study ended according to schedule.
Study groups
Composition of the study fluids that were investigated in a randomized study that included patients scheduled to undergo hip replacement surgery.

Values were measured by the manufacturer. For internal consistency, some of the values are rounded.
Randomization into the two study groups was accomplished using computer-generated random numbers and performed in blocks of four. For blinding, consecutively numbered (randomization numbers) closed envelopes were used, which contained the coded assignment to the therapy.
Buffer-free Ringer’s solution is characterized by its high chloride concentration (155.5 mmol/l), which resembles the chloride concentration of 0.9% (w/v) saline (154 mmol/l). Although it does not contain any buffer such as lactate or acetate, buffer-free Ringer’s solution contains sodium, potassium and calcium as cations and not just sodium as in saline solution. For simplicity, in this study, the group who received the unbuffered, chloride-rich Ringer’s solution is called the unbalanced group. All patients underwent general anaesthesia and were operated upon by one of the authors (C. P.) or by another surgeon under his direct supervision.
Goal-directed therapy
During surgery, all patients were treated according to a goal-directed therapy (GDT) algorithm as previously published. 3 Briefly, in addition to standard monitoring, the fluid responsiveness of all patients was monitored by oesophageal Doppler (CardioQ–ODM system; Deltex Medical, Chichester, UK). The guidance parameter for fluid management was stroke volume (SV). If the SV increased by ≥10% after infusion of 200 ml study fluid over 5 min, volume responsiveness was assumed and the fluid challenge was repeated until the SV failed to rise by 10%. Thereafter, no further boluses of the study fluid were given until a 10% decrease in SV occurred. After fluid optimization, mean arterial pressure (MAP) and cardiac index (CI) served as safety parameters within this algorithm. In this context, norepinephrine, dobutamine or enoximon were allowed to keep the MAP between 70–90 mmHg and CI >2.5 l/min per m2 body surface area. 3 Previous routine data from the Department of Anaesthesiology and Intensive Care Medicine, Charité – Universitätsmedizin Berlin, obtained without goal-directed treatment showed that at least 2 l of intravenous solutions were administered during hip replacement.
An arterial line was inserted if this could be accomplished within no more than two attempts; otherwise blood pressure was measured noninvasively. In both cases, measurements were carried out using standard monitoring equipment (Philips IntelliVue MP80™ patient monitor; Philips Healthcare, Hamburg, Germany). After induction of anaesthesia, arterial, or in cases where arterial catheter insertion was not feasible, peripheral venous blood gas analyses (BGA) were performed every 15 min until the end of surgery, and every 30 min postoperatively until discharge from the recovery room (ABL700 blood gas analyser; Radiometer Medical, Brønshøj, Denmark).
Despite the different origins, all blood samples were included in the analyses. According to Toftegaard et al.27,28 it was assumed in patients in this study (elective surgery, no critical illness, GDT to rule out hypoperfusion) that the change of base excess across the tissue side (from arterial to venous) was close to zero. During the course of the operation, the mean values for the haemodynamic parameters were calculated for all measured values over a 10-min period and for the acid–base parameters for all measured values over a 20-min period.
Acid–base calculations
As end expiratory CO2 was kept within the normal range (35–45 mmHg), the pH was calculated with regard to the different origins (venous or arterial) of the BGA samples by the Henderson–Hasselbalch equation as [pHcalc] = 6.1 + log ([
Kidney function parameters
Neutrophil gelatinase-associated lipocalin (NGAL) was determined using an NGAL Test™ kit (BioPorto, Gentofte, Denmark). Serum creatinine was measured by the Institut für Laboratoriumsmedizin from an arterial or venous blood sample collected at the same time-point.
Statistical analyses
Results are expressed as median (25% and 75% percentiles) or frequencies (%), unless otherwise specified. After checking the distributions for normality, differences between the two groups were tested using nonparametric exact Mann–Whitney tests for independent groups or exact Wilcoxon tests for pairwise comparisons. Frequencies were compared using the exact Mantel–Haenszel test (ordered categories) or the exact χ2-test in contingency tables. Changes in clinical outcomes with regard to time were analysed using multivariate nonparametric analysis of longitudinal data in a two-factorial design (1st [independent] factor: groups; 2nd [dependent] factor: repetitions in time). Therefore, all the time-points were simultaneously compared on the corresponding response curves. In the multivariate analysis, three hypotheses were tested: (i) differences in groups (Group); (ii) differences in time (Time); and (iii) one hypothesis about interactions between the two factors (Group × Time). Lastly, each group was checked for differences in time, separately. Moreover, a nonparametric multivariate analysis of covariance with the baseline as the covariate was applied to adjust for possible baseline influences in some of the preceding analyses. After global testing, post hoc analyses were carried out to detect specific differences with respect to groups for fixed times (Mann–Whitney tests). A two-tailed P-value <0.05 was considered statistically significant. All tests have to be understood in the area of exploratory data analysis. Therefore, no adjustments for multiple testing have been made. All numerical calculations were performed using the SPSS® statistical package, version 20.0 (SPSS Inc., Chicago, IL, USA) for Windows®, and the SAS® statistical package, version 9.1 (SAS Institute, Cary, NC, USA). Power calculations were performed by nQuery Advisor® version 7.0 (Statistical Solutions, Cork, Ireland). All graphs were created by ‘R – The project for statistical computing’, version 2.14.2 (2012-02-29). 34
Results
Of the 152 patients assessed for eligibility, 41 patients who agreed to general anaesthesia including the insertion of the oesophageal Doppler probe were enrolled in the present study (Figure 1). One of these patients had to be excluded before receiving the study medication because of a planned extension of the operation, so 40 patients received study fluids and their data were collected for this study. All patients received cementless hip replacement in the supine position using a minimally invasive technique. The demographic and clinical characteristics of the patients did not differ significantly between the two groups (Table 2).
Flow of participants through a study that compared two different fluids within a goal-directed therapy protocol for fluid replacement during hip replacement surgery showing screening, enrolment, randomization and analysis of patients. Baseline demographic and clinical characteristics, surgical data, outcomes and serious adverse events for patients who participated in a randomized study that investigated the use of two different fluids within a goal-directed therapy protocol for fluid replacement during hip replacement surgery (n = 40). Data presented as median (25–75% percentiles) or n of patients (%) unless specified. No statistically significant between-group differences (P ≥ 0.05); using the exact Mann–Whitney test for independent groups or exact Wilcoxon test for pairwise comparisons; frequencies were compared using the exact Mantel–Haenszel test (ordered categories) or the exact χ2-test in contingency tables. ASA, American Society of Anesthesiologists; NYHA, New York Heart Association; ACE, angiotensin-converting enzyme; AT1, angiotensin II receptor; POSSUM, Physiological and Operative Severity Score for the enUmeration of Mortality and Morbidity; PACU, postanaesthesia care unit; POD, postoperative day; POCD, postoperative cognitive dysfunction.

The SBE and pHcalc were significantly lower (Figure 2; P < 0.05 for all comparisons) and plasma chloride concentrations were significantly greater (Table 3; P < 0.05 for all comparisons) in the unbalanced group during (intraoperative course of chloride not shown) and immediately after surgery compared with the balanced group.
Intraoperative course and values at arrival in the recovery room for standard base excess (SBE) (a) and calculated pH (pHcalc) (b) for patients who were treated with one of two different fluids within a goal-directed therapy protocol for fluid replacement during elective hip replacement surgery (n = 40). White circles: unbalanced group; black circles: balanced group. Data presented as median (25% and 75% percentiles). *P < 0.05, **P < 0.005, for between-group comparisons for SBE and pHcalc. P-values were confirmed by a multivariate nonparametric analysis for longitudinal data and nonparametric exact Mann–Whitney tests for independent groups. NS, no statistically significant between-group difference (P ≥ 0.05). Plasma electrolyte concentrations, variables of acid–base status, haemoglobin concentration, fluid management data, transfusion data, blood loss through Redon drainage and parameters of kidney function for patients who participated in a randomized study that investigated the use of two different fluids within a goal-directed therapy protocol for fluid replacement during hip replacement surgery (n = 40). Data presented as median (25–75% percentiles). Parameters were recorded after induction (baseline), at arrival in the recovery room, 1 h PO, 6 h PO and POD 1; except for oral and intravenous intake and parameters of renal function, which were recorded after induction (baseline), 1 h PO, 6 h PO and POD 1. P < 0.05 for between-group difference; ***P < 0.001 for between-group difference; using the exact Mann–Whitney test for independent groups or exact Wilcoxon test for pairwise comparisons. PO, postoperative; POD, postoperative day; NGAL, neutrophil gelatinase-associated lipocalin.

Analyses using the Stewart–Fencl approach showed that the changes in SBE were the result of changes in both chloride concentration and albumin concentration, whereas free water and serum lactate had no influence on SBE in this setting (Figure 3).
Influence of free water (a), serum chloride (b), serum albumin (c) and serum lactate (d) on standard base excess (BE) according to Stewart–Fencl corrections. White box-whisker plot: unbalanced group; black box-whisker plot: balanced group. The heavy central horizontal lines for each box show the median; the extremities of each box are the 25th and 75th percentiles; the error bars represent the minimum and maximum outliers; and the small circle(s) represent extreme outliers. *P < 0.05; P-values were confirmed using nonparametric exact Mann–Whitney tests for independent groups.
In this context, there was a significantly lower serum albumin concentration 1 h after surgery in the balanced group compared with the unbalanced group (P < 0.05) (Table 3). Time courses of plasma lactate, pCO2, and plasma potassium (data not shown), as well as plasma sodium (Table 3), were uneventful.
At 1 h postoperatively, there was still a slight increase in plasma chloride in the unbalanced group compared with baseline and the concentration was significantly greater compared with the balanced group (P < 0.05) (Table 3); serum albumin and serum protein concentrations were significantly greater in this group compared with the balanced group (P < 0.05 for all comparisons) (Table 3). A 6 h postoperatively, there were no longer any significant differences in serum protein and serum albumin concentrations between the groups (Table 3). The intraoperative time course of the haemoglobin concentrations was similar between the two groups (data not shown), whereas at arrival in the recovery room, the haemoglobin concentrations in the balanced group were significantly lower compared with the unbalanced group (P < 0.05). This difference in the haemoglobin concentration between the two groups had disappeared at postoperative day 1 (Table 3).
As a result of GDT, which was guided by SV optimization, equal volumes of study fluids were administered to the two groups (Figure 4a). The SV (Figure 4b), as well as MAP, cardiac output and flow time corrected (FTc) (data not shown), did not differ significantly between the two groups during surgery.
Cumulative volumes of study fluid administered (a) and intraoperative course of stroke volume measured by oesophageal Doppler (b). White circles or white box-whisker plot; unbalanced group; black circles or black box-whisker plot: balanced group. Data presented as median (25% and 75% percentiles). The heavy central horizontal lines for each box show the median; the extremities of each box are the 25th and 75th percentiles; the error bars represent the minimum and maximum outliers. NS, no statistically significant between-group differences (P ≥ 0.05); P-values were confirmed using a multivariate nonparametric analysis for longitudinal data and using nonparametric exact Mann–Whitney tests for independent groups.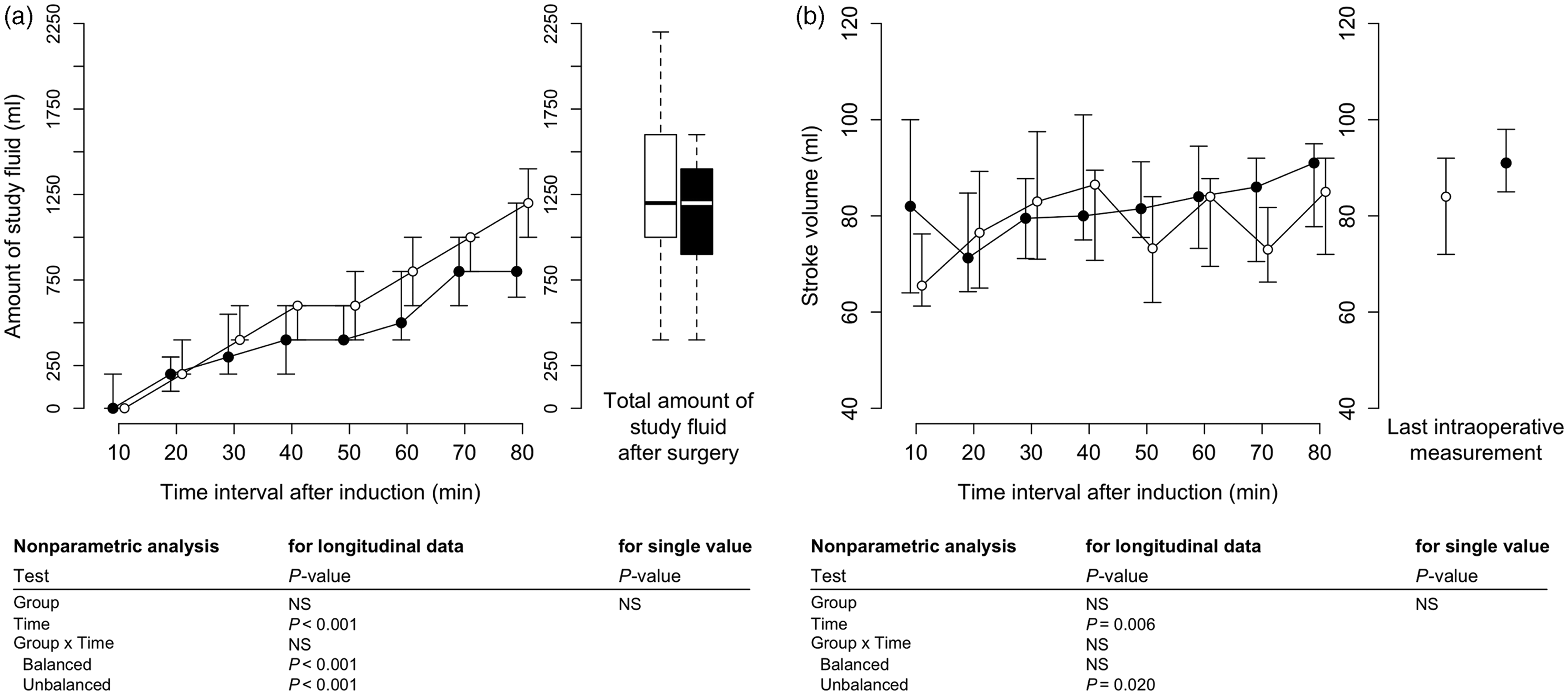
Patients in both groups received norepinephrine. Neither the number of patients (unbalanced group: 5 of 21 [23.8%]; balanced group: 3 of 19 [15.8%]) that received norepinephrine nor the median doses (median [25%, 75% percentiles]: 0.05 [0.02–0.06] versus 0.03 [0.02–0.04] µg/kg per min for the unbalanced group versus balanced group, respectively), nor the duration of treatment (data not shown) were significantly different between the two groups. One patient in the balanced group received dobutamine (maximum dose of 5 µg/kg per min).
At 1 h postoperatively, there were no significant differences between the two groups with regard to mean blood pressure and heart rate (data not shown). No patient received fresh frozen plasma, packed red blood cells or thrombocyte concentrates during surgery and/or in the recovery room. Within the first hour in the recovery room, patients from both groups received the same amounts of a balanced infusion solution (i.e. Balanced crystalloid – cumulative, ml; Table 3).
Patients in both groups showed no significant difference in the amount of fluid solutions, transfusions (red packed blood cells and fresh frozen plasma), oral fluid intake and urine output until the first postoperative day (Table 3). During the follow-up (until the fifth postoperative day), there was no significant difference in the cumulative amount of red packed blood cell transfusions administered between the two groups (unbalanced versus balanced: three versus five patients, respectively).
There was no significant difference in serum creatinine at 1 h and 6 h after surgery between the two groups (Table 3), as well as on the first postoperative day. The NGAL concentration showed no significant difference between the two groups at any time-point (Table 3). No patient needed renal replacement therapy perioperatively or during the follow-up period.
No significant differences were found between the two groups for length of hospital stay, incidence of POCD on postoperative day 5, in the MMSE score on postoperative day 5 and regarding the occurrence of serious adverse events (SAEs) (Table 2). In both groups, five SAEs with no accumulation of one entity were reported (Table 2).
No significant differences were found with regard to delirium scores and postoperative pain level, as well as the incidence of nausea and vomiting, between the two groups at 1 h and 6 h after the arrival in the recovery room (data not shown).
Discussion
This present study is the first to examine the perioperative changes in acid–base status, fluid requirements and haemodynamics, comparing the combined infusion of a balanced crystalloid and balanced colloid (1 : 1 mixture) with the infusion of an unbalanced crystalloid alone, using a goal-directed haemodynamic algorithm that guided fluid administration through optimization of stroke volume during elective hip surgery. Apart from using a goal-directed algorithm, at the time that this present study was started, these two regimens of intraoperative fluid administration were common practice in anaesthesiology all over the world.
The main finding of the present study was that the administration of 1200 ml (median) of an unbalanced crystalloid solution with an unphysiologically high chloride concentration was able to induce acid–base disturbances as demonstrated by a significant decrease in SBE compared with a balanced crystalloid mixed 1 : 1 with a balanced colloid. Surprisingly, although a colloid was infused in the balanced group, this had no effect on the amounts of fluids needed to maintain haemodynamic stability and normovolaemia in this setting.
The use of balanced fluids within a GDT protocol prevented the decrease in SBE that was encountered when unbalanced fluids were used during elective hip replacement surgery in the present study. Since pCO2 and plasma lactate remained within the normal range, the developing metabolic acidosis was most likely due to the administration of a high chloride load through the unbalanced infusion solution. This notion is also in accordance with the calculations on the contribution of chloride, albumin, lactate and free water to base excess (Figure 3). These small deviations in acid–base status were still detectable even after 500 ml of a balanced crystalloid infusion had postoperatively been infused in the recovery room but resolved within the next few hours.
When preparing for this present study, the amounts of fluids usually infused during elective hip surgery were measured and were found to be approximately 2 l. However, using this GDT regime, this present study demonstrated that only 1200 ml were required during an intraoperative period lasting a mean of 1:40 h (i.e. approximately 720–750 ml/h).
Analysing the data by the Stewart–Fencl approach revealed that the decrease of the SBE in the unbalanced group seemed to be mainly associated with the change of plasma chloride whereas the change of serum albumin, especially in the balanced group, seems to have almost no influence on SBE.
The idea behind the concept of combining an HES with a crystalloid (1 : 1) in one of the study groups in the present study was to provide sufficient hydration and to obtain a better haemodynamic profile.35–37 However, both groups showed no differences in the intraoperative course of SV, cardiac output, FTc, or MAP or plasma lactate concentration. In a study comparing a balanced crystalloid with a balanced colloid solution within a goal-directed haemodynamic algorithm, there was significantly less haemodynamic stability in the crystalloid group compared with a colloid group. 38 This instability was not seen within the first hour of surgery; however, it became apparent after 1–1.5 h. In the present study, the haemoglobin level was significantly reduced in the balanced group at the end of surgery compared with the unbalanced group. This may indicate that the circulating blood volume was higher in the balanced group, but since both groups showed no differences in the amounts of the study fluids administered, the use of 6% (w/v) HES 130/0.42 in the context of short-lasting elective hip replacement surgery within a GDT regimen does not seem to offer a benefit in terms of fluid volume as long as blood loss is small. At the same time, there was no clinically relevant bleeding tendency or increased blood loss and no increase in registered SAEs when 6% (w/v) HES 130/0.42 was used in this clinical setting. In addition, no deterioration in renal function measured by urine output, serum creatinine and NGAL was seen when 6% (w/v) HES 130/0.42 was administered within this goal-directed haemodynamic algorithm. These current findings were consistent with a study in patients undergoing major gynaecological surgery in which balanced colloids were administered within the same goal-directed haemodynamic algorithm. 38 It has to be noted, however, that this present study was not powered to investigate tolerability or safety issues of HES.
The equality of the amounts of fluids infused was unexpected, since the volume of distribution for crystalloids is the extracellular space, whereas the (physiological) volume of distribution for colloids is the plasma volume. The extracellular space is about five times the plasma volume. Accordingly, to replace a blood loss of 500–600 ml, crystalloids in a volume of 2500 ml should be necessary 39 and not 1200 ml as used in the present study. The reason for this remarkable difference may be due to the volume kinetics of crystalloids. 40 According to Hahn et al., 41 the initial fraction of a crystalloid that remains in the intravascular space may reach 50–70% in normovolaemic patients and the shift to the interstitium may take several minutes. This notion is supported by the intraoperative and postoperative course of haemoglobin concentrations in the present study. Intraoperatively, haemoglobin concentrations did not significantly differ between the two groups in the present study, while in the recovery room, the haemoglobin concentrations in the balanced crystalloid/colloid group were found to be significantly lower than in the unbalanced crystalloid group, which might be explained by the delayed shift of the crystalloid from the intravascular to the interstitial compartment, whereas the colloid remained primarily in the intravascular compartment.
Explanations for these findings cannot be derived from studies on sepsis,42,43 blunt multiple trauma 44 or states of hypervolaemia, 45 where a leakage into the interstitial space by destruction of the endothelial surface layer might be assumed as the mechanism responsible. Therefore, the results of this present study cannot be extrapolated to major trauma cases where there is greater volume turnover due to the inflammatory responses and destruction of the endothelial surface layer is regularly present.
This present study had two limitations. First, no significant differences were observed between the two groups regarding length of hospital stay, mortality and delirium/POCD. However, to address these outcome parameters adequately, randomized controlled trials with larger numbers of patients are necessary, especially with regard to delirium and POCD, and possibly more severe derangements in acid–base status induced by greater volumes of infused unbalanced solutions. Secondly, the present study was performed in patients treated within a highly standardized clinical pathway undergoing surgery with rather low blood loss. Therefore, it is not clear whether these results would also be valid or even more pronounced in major and/or longer lasting operations with a greater volume turnover. A timely application of colloids may be advantageous in such a setting with regard to interstitial oedema and haemodynamic stability, especially if guided by GDT. 38 In contrast, this cannot be extended to unstable proinflammatory situations (e.g. sepsis).
Based on the results of the present study, during short-lasting (1–1.5 h) hip replacement surgery highly standardized within a clinical pathway, even small amounts of an unbalanced, chloride-rich solution caused a significant fall in SBE. These effects were seen even without major blood loss or volume turnover and within a goal-directed haemodynamic algorithm to avoid hypo- or hypervolaemia. In the described setting and during short-lasting operations, this present study has neither observed clinical benefits nor disadvantages when infusing 6% (w/v) HES 130/0.42 for reasons of volume replacement.
Footnotes
Declaration of conflicting interest
Aarne Feldheiser received travel support from Deltex Medical. Willehad Boemke received payments for lectures from B. Braun, Melsungen, Germany, and MSD Sharp & Dohme GmbH, Germany, and advisory board honoraria from Baxter Healthcare and MSD Sharp & Dohme GmbH. Claudia Spies received grants from Abbott, Aspect, Baxter, Bispebjerg Hospital, Care Fusion, Deltex, Essex Pharma, Fresenius, Grünenthal, GlaxoSmithKline, Hutchinson, Köhler Chemie, MSD Sharp & Dohme, MCN Medizinische Congressorganisation Nürnberg, Novartis, Pajunk, Pulsion, Roche, Sysmex, and the University Hospital Stavanger, as well as payments for lectures from Abbott, Essex Pharma and GlaxoSmithKline. Holger Krebbel, Aarne Feldheiser, Olga Müller, Willehad Boemke, Michael Sander, Carsten Perka, and Klaus-Dieter Wernecke and Claudia Spies declare that they have no conflicts of interest.
Funding
This was an investigator initiated trial supported by institutional grants of the Medical Faculty of the Charité – Universitätsmedizin Berlin and Baxter Healthcare Corporation, Baxter Deutschland GmbH, Edisonstrasse 4, 85716 Unterschleissheim, Germany.
Acknowledgements
The authors are indebted to Velizara Pavlova for recruiting patients; Katharina Berger for her participation in designing this study; and Kathrin Scholtz for her invaluable help in the preparation of the Institutional Review Board proposal and registration of this study. We also thank Karin Weimann and Andreas Weimann for their support in performing the measurements of neutrophil gelatinase-associated lipocalin. In addition, we thank Ali Coskuner and Benedikt Hoffmann for their help in collecting the data as a part of their doctoral theses.
