Abstract
Objective
To perform a retrospective comparison of the clinical and radiological features of Klebsiella pneumoniae (KP)-associated and non-KP-associated pyogenic liver abscesses (PLA) in Chinese patients.
Methods
Patients with confirmed diagnoses of bacterial liver abscess at three Beijing hospitals were enrolled. Clinical isolates from liver abscesses were used to determine serology and expression of hypermucoviscosity genes. Basic clinical, ultrasonographic (US) and computed tomography (CT) data were recorded and compared between patients with KP- and non-KP-associated PLA.
Results
A total of 101 (77.10%) and 30 (22.90%) cases were due to KP and non-KP pathogens, respectively. Compared with the non-KP cohort, the KP cohort demonstrated a significantly higher incidence of underlying diabetes mellitus, and more gas-forming abscesses, as demonstrated by US and CT examinations. Prior abdominal surgery or chemoradiation therapy was significantly associated with non-KP cases. The non-KP group had a higher chance of a clear edge, compared with the KP group, on pre-contrast CT images.
Conclusion
KP and non-KP-associated PLA have distinctive risk factors and unique US and CT features, in Chinese patients.
Keywords
Introduction
Liver abscesses can be caused by intra-abdominal infection due to bacterial, fungal or parasitic organisms. 1 It was initially believed that Escherichia coli was the single most important pathogen in the aetiology of pyogenic liver abscess (PLA). 1 Klebsiella pneumoniae is an enteric Gram-negative nosocomial and/or community-acquired human pathogen, commonly associated with pneumonia or urinary tract infections. 2 In the last two decades, it has also emerged as the major pathogen causing PLA. K. pneumoniae-mediated PLA was first detected in Taiwan3–5 and has now assumed the status of an emerging global disease, with anecdotal accounts and case series reported worldwide. 6
A hypermucoviscous phenotype is associated with serotypes K1 and K2 K. pneumoniae strains, and this is largely responsible for causing invasive liver abscesses.2,7 Several virulence factors have been described for K. pneumoniae including presence of: the capsular serotype; mucoviscosity-associated gene A (magA); regulator of mucoid phenotype gene A (rmpA); aerobactin.8–10 Approximately 50% of reported patients with PLA have diabetes mellitus (type 1 or type 2: the type is not relevant to the development of PLA), while the remainder of patients do not appear to exhibit any other underlying disease. 11 Worldwide, 43 (5.28%) of the 813 invasive PLA cases reported in the past decade have died. 2 The high mortality rate can be attributed to the high antibiotic resistance profile of K. pneumoniae, which is further compounded by its unique capacity to cause invasive and metastatic complications.12–14
It is imperative to understand the differences observed between patients with K. pneumoniae- or non-K. pneumoniae-associated PLA. Such an understanding may help to guide the development of comprehensive recommendations regarding the early diagnosis and treatment of K. pneumoniae-associated PLA. The present study, therefore, investigated the distinctive clinical and radiological features found among patients with K. pneumoniae- or non-K. pneumoniae-associated PLA.
Patients and methods
Patient population
Consecutive patients with bacterial liver abscesses, admitted to Beijing Shijitan Hospital, Capital Medical University, Chinese PLA General Hospital, and Peking University First Hospital (all in Beijing, China) between January 2008 and December 2011 were recruited into this study. Patients were included if they had: (i) clinical symptoms of liver abscess including fever, chills and liver pain; (ii) one or more area(s) of echolucency in the liver on ultrasonography (US); (iii) undergone computed tomography (CT)-guided or US-guided percutaneous liver abscess pus puncture therapy; (iv) a culture of liver aspirates positive for bacteria (single pathogenic strain).
The study was approved by the Institutional Review Board at the Chinese PLA General Hospital and patients provided written informed consent.
Outcome measures
Basic clinical data including patient information, evidence of underlying disease, clinical manifestations, laboratory tests, and US and CT imaging characteristics were obtained from all enrolled patients by a blinded independent researcher (X-Y.X).
Culture and aetiology identification
Clinical isolates from liver aspirates (obtained from enrolled patients with a confirmed diagnosis of PLA) were identified in the clinical microbiology laboratories of each participating hospital, using the API® 20E test kit system (bioMérieux, Marcy I'Etoile, France). 15 All cultures were collected using a culture collection and transport system (BBL™ CultureSwab™, Becton Dickinson and Co., Franklin Lakes, NJ, USA). Isolates were stored in brain–heart infusion broth at −80℃ for a mean of 2 years until use. Liver isolates were collected from the same patient only once during each episode of infection (nonduplicate isolates) and were serotyped using a countercurrent immunoelectrophoresis method, as previously described. 16
String test
The hypermucoviscosity phenotype of K. pneumoniae can be identified by a positive string test. 7 Briefly, MacConkey agar plates (Thermo Fisher Scientific, Rockford, IL, USA) were inoculated with the cultures and incubated at 35℃ without CO2. The plates were examined the day after inoculation for the presence of hypermucoviscous, lactose-fermenting Gram-negative rods, using an automated bacterial identification system (VITEK®2, bioMérieux, Hazelwood, MI, USA). Identified isolates were subcultured on a 5% sheep blood agar plate, to ensure purity. Any isolate yielding a ≥ 90% probability of K. pneumoniae was further analysed using the string test: a bacterial loop was touched to a suspect colony on the agar plate and withdrawn slowly; bacteria forming a mucoid ‘string’ of ≥ 5 mm were considered to be positive.
Molecular analyses
Sequences of primers used for amplification of target genes, serotype analysis and virulence genetic testing of hypermucoviscous isolates of Klebsiella pneumoniae, obtained from patients with pyogenic liver abscesses.

Statistical analyses
All statistical analyses were performed using SPSS® statistical software, version 16.0 (SPSS, Inc. Chicago, IL, USA) for Windows®. Data are expressed as mean ± SD. Student’s t-test was used to determine any significant differences in gene expression and outcome measures between patients with K. pneumoniae-associated PLA and those with non-K. pneumoniae-associated PLA. A P-value of < 0.05 was considered statistically significant.
Results
A total of 131 patients with a definitive diagnosis of PLA were included in the present study; clinical isolates from liver aspirates were positive for K. pneumoniae in 101 (77.10%) cases and negative for K. pneumoniae in 30 (22.90%) cases. Escherichia coli was the next most-common pathogen (accounting for 20 [15.27%] of cases), followed by Pseudomonas aeruginosa (3 [2.29%] cases), Enterococcus (2 [1.53%] cases), Citrobacter freundii (2 [1.53%] cases), Enterobacter cloacae (1 [0.76%] case), Staphylococcus aureus (1 [0.76%] case) and coagulase-negative Staphylococci (1 [0.76%] case).
Virulence gene expression profile associated with hypermucoviscosity (HMV) in 101 cases of Klebsiella pneumoniae, detected over 2 consecutive years (during the period between 2008 and 2011) in patients with pyogenic liver abscesses.

Data presented as n (%) of patients.
magA, mucoviscosity-associated gene A; rmpA, regulator of mucoid phenotype gene A.
Clinical characteristics and laboratory test results in 131 patients with Klebsiella pneumoniae- or non-K. pneumoniae-associated pyogenic liver abscesses (PLA).
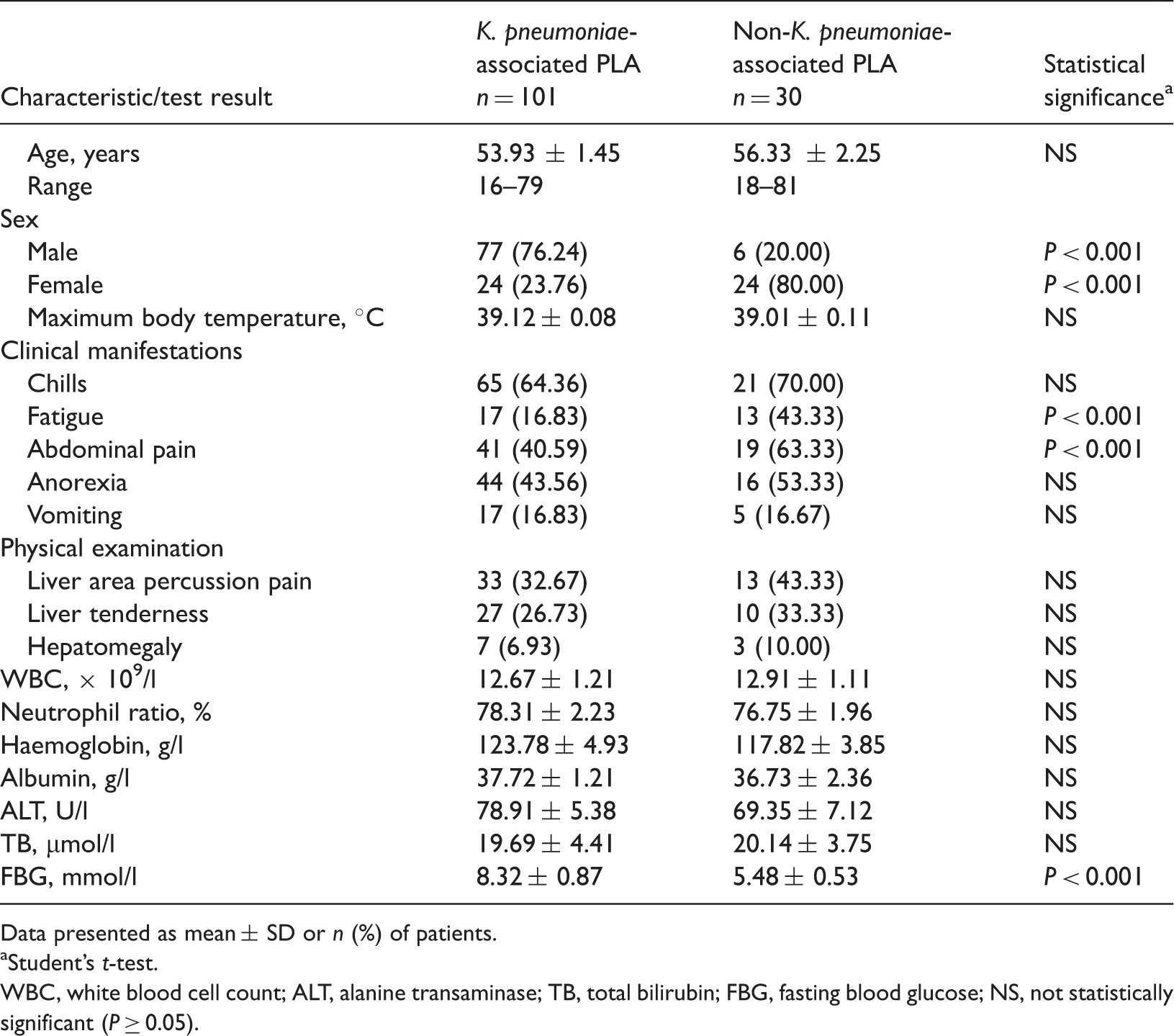
Data presented as mean ± SD or n (%) of patients.
Student’s t-test.
WBC, white blood cell count; ALT, alanine transaminase; TB, total bilirubin; FBG, fasting blood glucose; NS, not statistically significant (P ≥ 0.05).
Underlying diseases in 131 patients with Klebsiella pneumoniae- or non-K. pneumoniae-associated pyogenic liver abscesses (PLA).
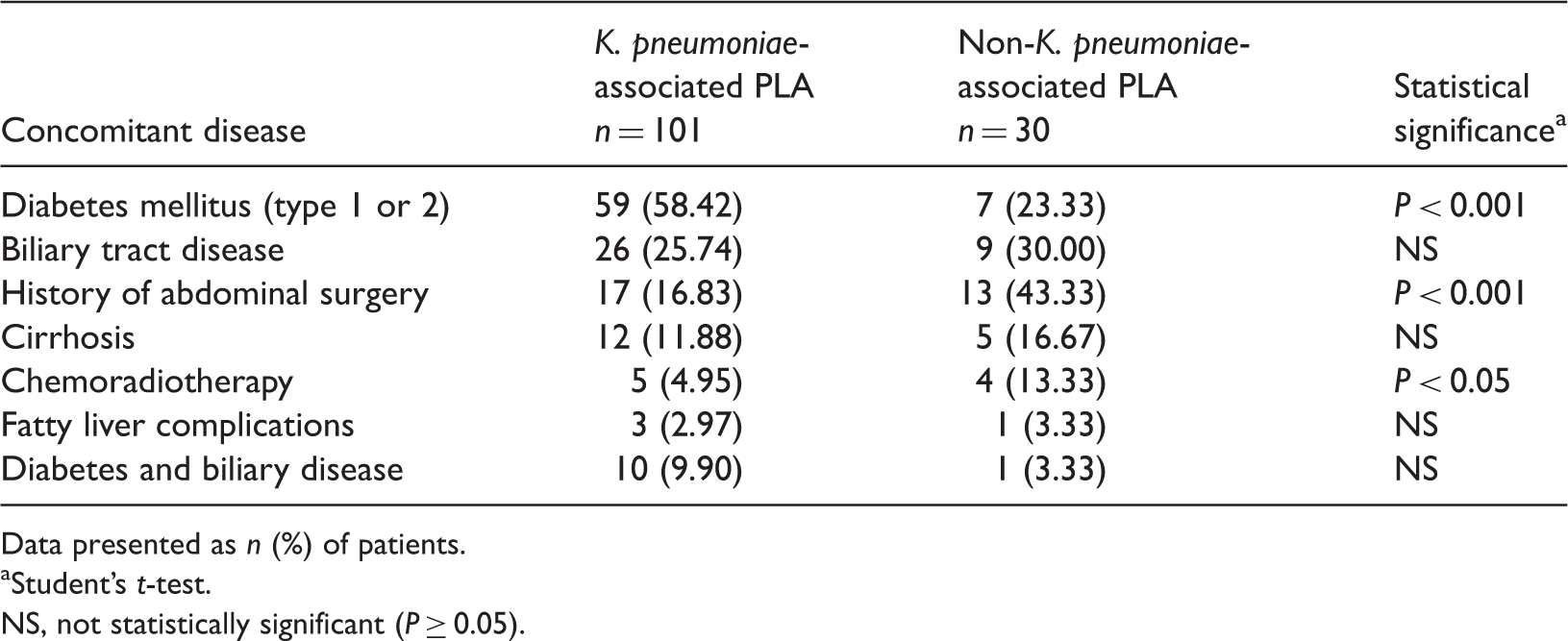
Data presented as n (%) of patients.
Student’s t-test.
NS, not statistically significant (P ≥ 0.05).
B-mode ultrasonography (US) and computed tomography (CT) scanning examination results in patients with Klebsiella pneumoniae- or non-K. pneumoniae-associated pyogenic liver abscesses (PLA).

Data presented as mean ± SD or n (%) of patients.
US examination performed in all enrolled patients (K. pneumoniae-associated PLA, n = 101; non-K. pneumoniae-associated PLA, n = 30); CT examination performed in 98/131 patients (K. pneumoniae-associated PLA, n = 80; non-K. pneumoniae-associated PLA, n = 18).
Student’s t-test.
NS, not statistically significant (P ≥ 0.05).
Computed tomography examination was performed in 98/131 patients (80 K. pneumoniae cases and 18 non-K. pneumoniae cases). Pre-contrast CT images demonstrated that there was a greater chance of a clear edge in the non-K. pneumoniae group compared with the K. pneumoniae group (P < 0.001; Table 5). Presence of gas in the abscess was significantly higher in the K. pneumoniae group (P < 0.05; Table 5). Rates of pneumatosis in the biliary system or pleural effusion were not significantly different between the two study groups.
Discussion
Klebsiella pneumoniae-mediated community-acquired PLA is an emerging infectious disease worldwide.3–6 Results from the present study corroborate this observation, demonstrating that the main bacterial liver abscess pathogen was K. pneumoniae in 77.10% of the 131 cases examined. Furthermore, it was shown that ∼80% of the K. pneumoniae serotypes were K1 and K2, and that 89.11% of the strains exhibited the hypermucoviscosity phenotype, which is consistent with other published studies. 2
Molecular analyses undertaken in the present study indicated that expression of rmpA and aerobactin was high in K. pneumoniae-positive isolates, whereas magA expression was low. These data suggest that rmpA and aerobactin expression may be closely related to K. pneumoniae-mediated invasion of the liver and the subsequent development of liver abscesses. Siderophores, including enterochelin and aerobactin, are intracellular iron chelators that allow bacteria to compete for iron sources in the host.8–10 In combination with rmpA, aerobactin plays an important part in the virulence and hypermucoviscosity phenotype of K. pneumoniae isolates other than those of serotype K1 and K2. 2 In fact, it has been shown that the nonserotype K1 and K2 isolates (21 cases in the present investigation) that express aerobactin genes display comparative virulence to serotype K1 and K2 isolates from patients with liver abscesses. 17 As such, the increased virulence and invasiveness of the K. pneumoniae isolates in the present study may be explained by the high levels of rmpA and aerobactin expression that were found in the majority of these isolates. magA has been described as the causative gene for K. pneumoniae liver abscess and septic metastatic complications.18–21 K1 serotypes were detected more often than K2 serotypes in the present study, however, which may be indicative of the involvement of other genes in the regulation of K1. rmpA, which aids capsule synthesis, has also been proposed as a virulence factor, in addition to magA and capsular serotypes K1/K2.17,22,23
A high blood-glucose level is conducive to bacterial growth and inhibition of phagocyte chemotaxis, phagocytosis and bactericidal function.24,25 In addition, the negative impact of diabetes mellitus on haemodynamics hinders immune-cell mobilization. Half of reported patients with PLA also have diabetes mellitus.9,11,12 Data from the present study are consistent with these findings: patients with K. pneumoniae-associated PLA had higher fasting blood-glucose levels and this was corroborated by the finding that diabetes was significantly more common in these patients compared with non-K. pneumoniae cases. These results suggest that diabetes mellitus may be one of the most important risk factors for K. pneumoniae-associated PLA.
Interestingly, the present study also established that prior history of abdominal surgery and chemoradiation therapy are the most important risk factors for non-K. pneumoniae-associated PLA. These data suggest that non-K. pneumoniae-associated PLA largely occurs as a nosocomial infection. In accordance with previous findings, the present study showed that prior hepatobiliary disease did not seem to be a risk factor in either study group. 2
Research has indicated that PLA has no sex predilection but most commonly involves patients ≥ 65 years of age,26,27 although the mean ages of the patients in our study were younger than this. In the present study, the K. pneumoniae cohort consisted of significantly higher numbers of male patients than females, compared with the non-K. pneumoniae cohort. It is of note, however, that there were a higher number of cases in general, in the K. pneumoniae cohort compared with the non-K. pneumoniae cohort, which may have biased the observed difference in the male : female ratio.
Others have reported that solitary abscesses were more frequently found in patients with K. pneumoniae-associated PLA compared with non-K. pneumoniae-associated PLA.28,29 The present study did not, however, reveal any between-group differences in this regard. Our findings replicate previous observations that the size of abscess was similar in both groups, that most cases had single abscesses and that abscesses were often situated in the right lobe of the liver.30,31 Other studies have also reported that gas is not commonly present in CT images obtained from patients with K. pneumoniae-associated PLA32–34 and that there was no significant difference in the presence of gas between K. pneumoniae and non-K. pneumoniae cases. 1 We observed significantly more gas in liver abscesses from patients with K. pneumoniae compared with non-K. pneumoniae, using either US or CT imaging techniques. In addition, pre-contrast CT imaging showed that the non-K. pneumoniae group had a higher chance of a clear abscess edge than the K. pneumoniae group, in the present study.
Bacterial culture is affected by objective conditions, such as time and environment. The positive culture rate may also decrease significantly when using empirical antibiotics, which in turn can delay diagnosis and subsequent treatment. Incorporation and interpretation of improved clinical and imaging data, combined with detection at the molecular level, is required to make fast and accurate diagnosis of pathogens (particularly in acute-onset cases or severe infections). The present study showed that K. pneumoniae and non-K. pneumoniae-associated PLA have distinctive risk factors and unique US and CT features. Taken together, these data suggest that rapid and accurate diagnosis of K pneumoniae infection in invasive liver abscesses could be achieved by analysis of the presenting symptoms, culture reports and radiological investigations, which may ultimately prevent severe metastatic complications. Further elucidation of the genomic basis of the higher risk of K. pneumoniae infection and the pathological mechanism of PLA in an Asian population are warranted.
Footnotes
Declaration of conflicting interest
The authors declare that there are no conflicts of interest.
Funding
This study was supported by the Capital Health Research and Development of Special Capital fund (Grant No. 2011-2008-07).
Acknowledgements
The authors wish to thank everyone who helped with this study.
