Abstract
Objectives
To investigate the association between polymorphisms of aromatase (encoded by the CYP19A1 gene and a key enzyme in biosynthesis of oestradiol) and the risk of lung cancer, and whether there were differences stratified by sex and smoking history.
Methods
This case–control study included consecutive, nonselected and pathologically-confirmed lung cancer patients and healthy people. Participants were classed as nonsmokers or smokers by questionnaire. Peripheral blood samples from all participants were genotyped for three single-nucleotide polymorphism (SNPs; rs727479, rs730154 and rs10046); allelic frequencies were compared across genotype and clinical records.
Results
A total of 529 patients with lung cancer and 567 age- and sex-matched controls were included. After adjustment for age, sex and smoking history, rs727479 was significantly associated with the incidence of lung cancer (for alleles AC vs AA). There was also a significant difference between patients and controls in haplotype CCA, while haplotype ACA was only significantly associated with nonsmokers and female nonsmokers.
Conclusions
Polymorphisms of CYP19A1 may be related to the increased risk of lung cancer; in particular, haplotype ACA may contribute to lung-cancer progression in nonsmokers. Further validation with larger populations is required.
Introduction
Lung cancer has the highest death rate worldwide among malignancies, with prevalence and mortality rates seemingly increasing more rapidly in females than in males.1,2 Lung cancer in women is more likely to be adenocarcinoma (ADC); women who have never smoked have a higher risk of lung cancer than men who have never smoked. 3 The proportion of lung cancers occurring among people who have never smoked is 15–25% among women, but only 10% among men. 4 These facts are attracting research attention to risk factors for lung cancer in nonsmokers, especially the involvement of the oestrogen system.
Studies indicate that the oestrogen system is involved in the biological behaviour of lung cancer. A higher level of oestrogens in the blood is found in females with nonsmall-cell lung cancer (NSCLC) compared with healthy women of the same age. 5 Epidemiological and laboratory-based studies have also suggested that tumour expression of oestrogen may influence NSCLC pathogenesis, and the prognosis of all lung cancer patients.1, 6 Furthermore, there is a positive correlation between high tumour aromatase and oestrogen levels: tumour aromatase levels may predict survival in women with early-stage NSCLC. 7
The cytochrome P (CYP) 450, family 19, subfamily A, polypeptide 1 (CYP19A1) encodes aromatase, which is a key enzyme in oestradiol biosynthesis8–9 and a member of the CYP 450 superfamily of enzymes. This protein localizes in the endoplasmic reticulum and catalyses three successive hydroxylations of the ring of androgen, which is the last step of oestrogen biosynthesis. Mutations in the CYP19A1 gene can result in either increased or decreased aromatase activity. 10
The present study explored genetic risk factors in lung-cancer development by analysing three single-nucleotide polymorphisms (SNPs) of the CYP19A1 gene, in consecutive lung-cancer cases and healthy controls from the same geographic area during the same time period, paying particular attention to smoking status and sex.
Patients and methods
Study population
Consecutive, nonselected, pathologically-confirmed lung cancer patients attending the Shanghai Cancer Hospital, Fudan University, Shanghai, China, between January 2005 and December 2008, were included in the study. All cases were diagnosed as ADC, squamous cell carcinoma or small-cell lung cancer by histological examination, according to the World Health Organization 2004 tumour classification system; 11 diagnoses were confirmed by two independent pathologists. There were no other inclusion or exclusion criteria in this study. Healthy residents with no history of cancer, living in 11 communities in Pudong New Area and Baoshan District, Shanghai, China, were recruited as controls during the same time period.
Patients and controls provided written informed consent and the study was approved by the Ethics Committee of the Shanghai Institute for Biological Sciences, Shanghai, China.
Smoking history of patients and controls was obtained via questionnaire interview during clinic visits or at hospital admission. The smoking level of each participant was expressed as ‘cigarettes per day’, i.e. the number of cigarettes smoked per day on most days. Smoking exposure was expressed as ‘pack-years’, defined as number of packs per day (i.e. cigarettes per day divided by 20) multiplied by years of smoking. ‘Smokers’ were defined as those who had smoked regularly for ≥12 months at any time in their life; ‘nonsmokers’ were defined as those who had never smoked or who had stopped smoking for a period of 12 months at some time, and were not current smokers.
Genotyping
Three SNPs of CYP19A1 (rs727479, rs730154, and rs10046) were used in the study: these had minor allele frequencies (MAFs) > 10% in the Chinese Han population, according to the dbSNP database (http://www.ncbi.nlm.nih.gov/SNP/). Two SNP (rs727479, rs730154) have been reported previously. 12 Genotype frequencies for each polymorphism were calculated for patients and controls, stratifying by the three SNPs.
Standard blood samples were collected from all participants during clinic visits or at hospital admission. Samples were treated with 1.8 mg/ml ethylenediamine tetra-acetic acid to prevent coagulation and stored at −80℃ until further analysis.
Genomic DNA was extracted from 2-ml peripheral blood samples using TIANamp Blood DNA Kit (DP318; TIANGEN Biotech [Beijing] Co. Ltd, Beijing, China) according to the manufacturer’s instructions. Genomic DNA was amplified and the CYP19A1 polymorphisms were genotyped, using a gene-specific TaqMan® real-time PCR assay kit (Invitrogen, Carlsbad, CA, USA) and the ABI Prism® 7900HT Sequence Detection System (Applied Biosystems, Foster City, CA). Primers for genotyping and the cycling programme were undertaken, as recommended by the manufacturers.
Genotyping was performed by laboratory personnel who were blinded to patient status, and a random 5% of the samples were repeated to validate genotyping procedures. The authors independently reviewed genotyping results, data entry and statistical analyses. Genotype frequencies were calculated using the Hardy–Weinberg equilibrium test, which was performed using the SHEsis software (available at http://analysis2.bio-x.cn/myAnalysis.php). Calculation of linkage disequilibrium (D′ and r2 values), as well as haplotype estimation, was undertaken using the expectation–maximization algorithm of the same software.
Statistical analyses
Data were compared across genotypes and clinical records using Pearson’s χ2-tests (for categorical variables) and Kruskal–Wallis tests (for continuous variables) where appropriate. The strength of association of genotypes was measured as odds ratios (ORs) with 95% confidence intervals (95% CIs), using an unconditional logistic regression analysis adjusted for sex, age and smoking status. These statistical analyses were performed using SPSS® software, version 15.0 (SPSS Inc., Chicago, IL, USA). A level of P < 0.05 was considered statistically significant.
Results
Clinical characteristics and smoking status of Chinese patients with lung cancer and healthy controls, included in a study assessing the risk of lung cancer associated with polymorphisms of the cytochrome P450, family 19, subfamily A, polypeptide 1 (CYP19A1, aromatase) gene.
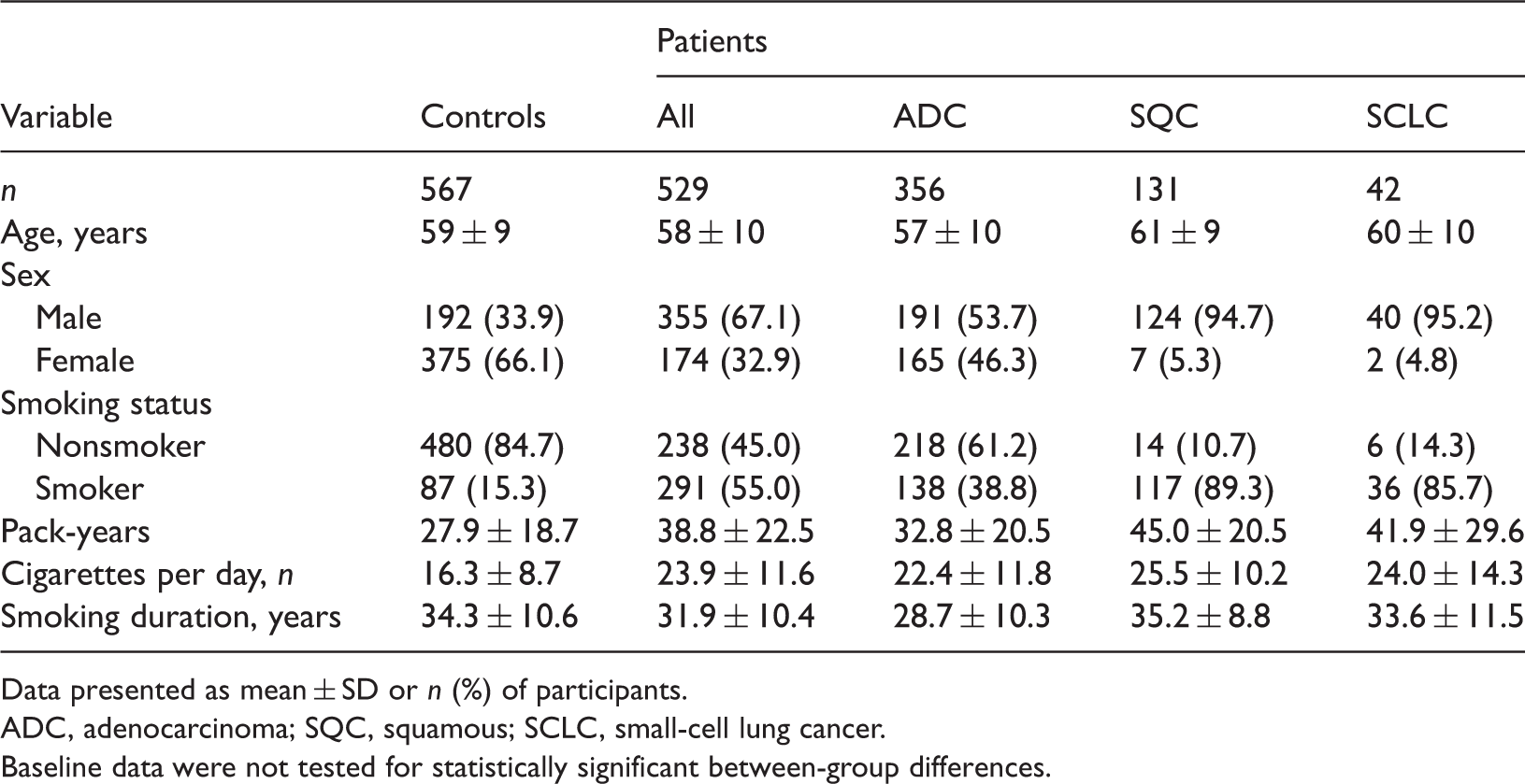
Data presented as mean ± SD or n (%) of participants.
ADC, adenocarcinoma; SQC, squamous; SCLC, small-cell lung cancer.
Baseline data were not tested for statistically significant between-group differences.
Allele frequency of three single nucleotide polymorphisms (rs727479, rs730154, rs10046) of the cytochrome P450 (CYP), family 19, subfamily A, polypeptide 1 (CYP19A1, aromatase) gene, in Chinese patients with lung cancer (n = 529) and healthy controls (n = 567).
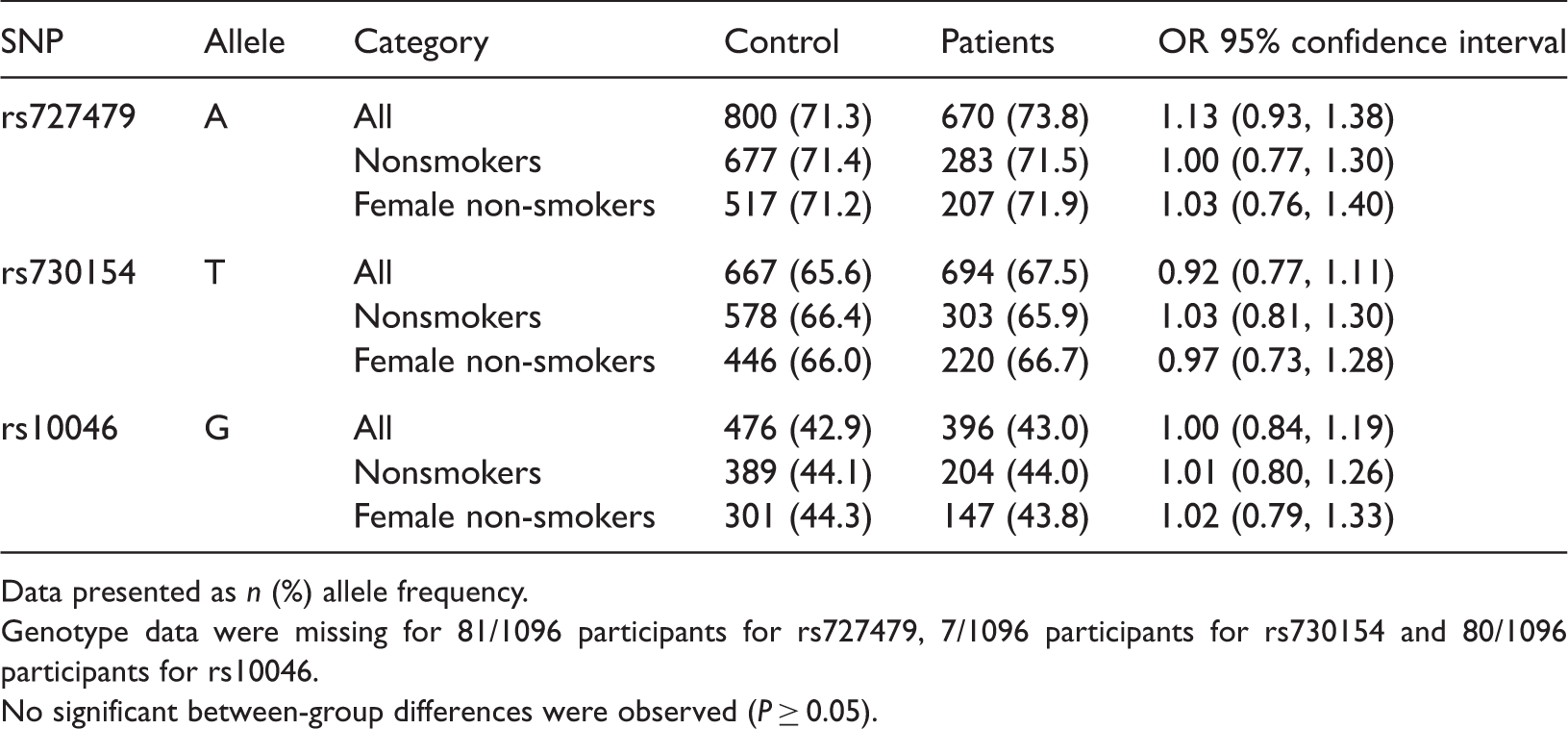
Data presented as n (%) allele frequency.
Genotype data were missing for 81/1096 participants for rs727479, 7/1096 participants for rs730154 and 80/1096 participants for rs10046.
No significant between-group differences were observed (P ≥ 0.05).
Estimated haplotype frequencies of three single nucleotide polymorphisms (rs727479, rs730154, rs10046) of the cytochrome P450 (CYP), family 19, subfamily A, polypeptide 1 (CYP19A1, aromatase) gene, in Chinese patients with lung cancer (n = 529) and healthy controls (n = 567).
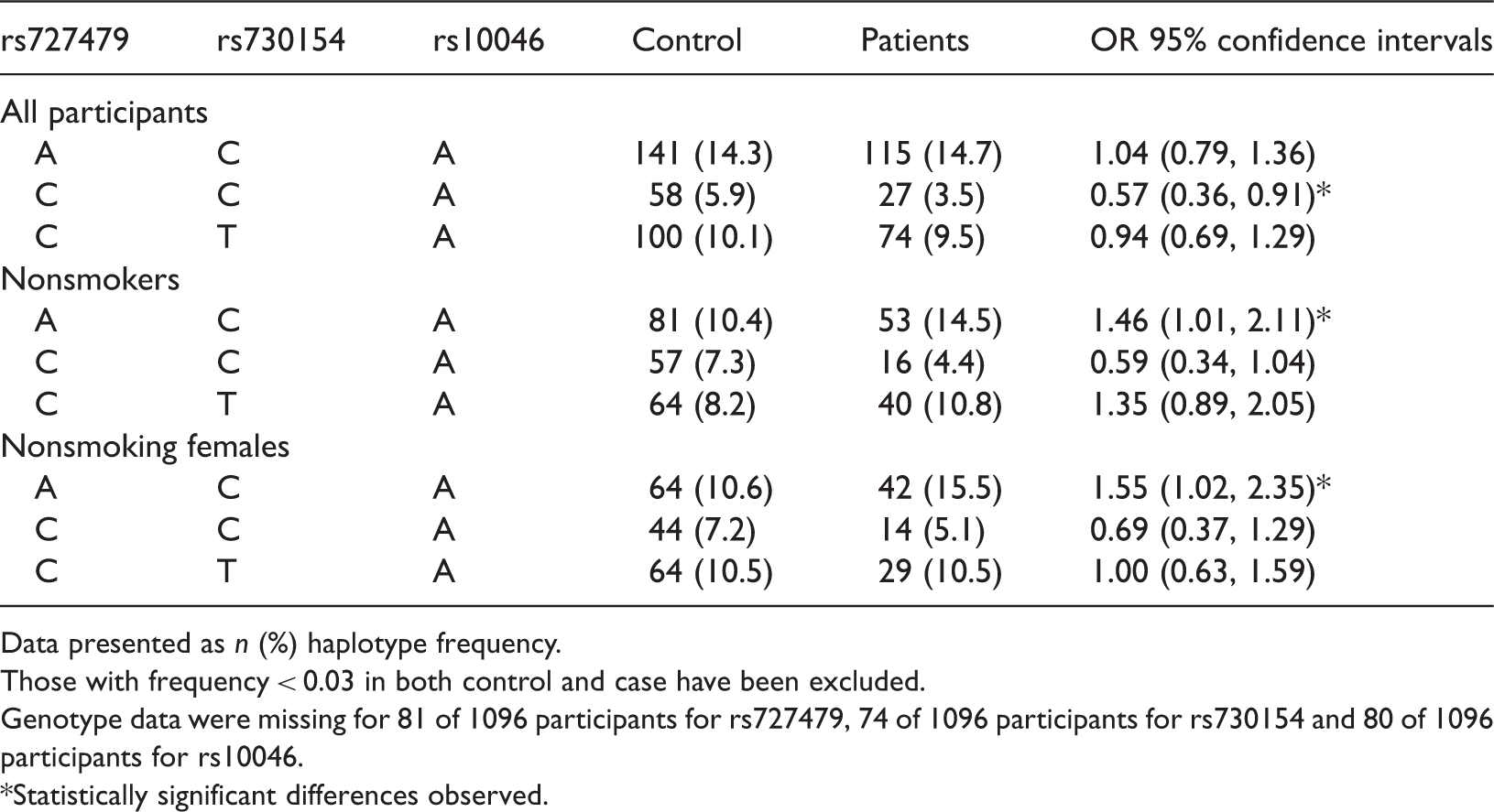
Data presented as n (%) haplotype frequency.
Those with frequency < 0.03 in both control and case have been excluded.
Genotype data were missing for 81 of 1096 participants for rs727479, 74 of 1096 participants for rs730154 and 80 of 1096 participants for rs10046.
Statistically significant differences observed.
Genotype frequency of three single nucleotide polymorphisms (rs727479, rs730154, rs10046) of the cytochrome P450 (CYP), family 19, subfamily A, polypeptide 1 (CYP19A1, aromatase) gene in Chinese patients with lung cancer (n = 529) and healthy controls (n = 567).

Data presented as n (%) genotype frequency.
Adjusted for: aage, sex and smoking status; bage and sex; cage.
Genotype data were missing for 81/1096 participants for rs727479, 74/1096 participants for rs730154 and 80/1096 participants for rs10046.
Statistically significant differences observed.
Discussion
It has been reported that higher levels of aromatase may predict prognosis among female patients with lung cancer.13,14 Additionally, an aromatase inhibitor, exemestane, has already been shown to reduce proliferation effectively in NSCLC cell lines. 14
The present study assessed the possible association between SNPs of the CYP19A1 gene and the risk of lung cancer by evaluating the allele, haplotype and genotype frequencies of three SNPs (rs727479, rs730154, and rs10046), in smoking and nonsmoking lung cancer patients and healthy controls. The haplotype CCA was associated with the presence of lung cancer whereas the haplotype ACA was significantly associated with lung cancer in all non-smokers, and in nonsmoking females. The study also showed that one locus rs727479 at 15q21.1 may be regarded as a potential disease candidate in lung cancer. It was hypothesized that, when mutation of the three SNPs presented as haplotype ACA, expression of aromatase would be upregulated. This would directly contribute to the upregulation of oestrogen in the patient and contribute to the nonsmoking pathway of lung cancer. The haplotype CCA also possibly contributes to the progression of lung cancer, but has no strong relationship with lung cancer in nonsmokers. As the statistical significance of the correlations between CYP19A1 gene polymorphism and the risk of lung cancer were often marginal, this study needs further validation with larger populations.
Existing epidemiological studies that examine whether oestrogen is a risk factor for lung cancer are inconsistent.15,16 Associations between SNPs related to oestrogen metabolism and lung cancer have been investigated in numerous studies, with variable results.17–19 It has been suggested that such inconsistency is possibly due to linkage disequilibrium across these loci. In other words, different choices of SNP locus result in the conflicting findings. Race differences (and the use of different techniques, such as genome-wide association studies) can also contribute to inconsistencies.
In summary, the present study suggests that polymorphisms of CYP19A1 may be associated with an increased risk of lung cancer. In particular, the haplotype ACA may contribute to the progression of lung cancer in nonsmokers.
Footnotes
Declaration of conflicting interests
The authors declare that there are no conflicts of interest.
Funding
This work is supported, in part, by the Pujiang Talent grant, Longhua Medicial Project, Young Investigator grant from Shanghai Municipal Health Bureau, and the Zhuo Shi grant from Fudan University, Ministry of Education Returned Scientific Research Foundation (to J. Z.).
