Abstract
Objectives:
To observe sex determining region Y-box 11 (SOX11) gene expression in cutaneous malignant melanoma and its effect on tumour cell proliferation.
Methods:
Clinicopathological data and tissue samples from patients with cutaneous malignant melanoma, together with tissue samples from healthy volunteers (controls), were retrospectively reviewed. Protein levels of SOX11 and the antigen identified by monoclonal antibody Ki-67 (Ki-67) in skin lesions were analysed using immunohistochemistry. The correlation between protein levels and clinipathological parameters was investigated.
Results:
Out of 40 patient samples, 25 (62.5%) were positive for SOX11 protein in malignant melanoma tissue. This was significantly higher than in 40 control tissue samples, in which no SOX11 protein was detected. Presence of SOX11 protein was positively related to the proliferation index of cutaneous malignant melanoma tumour cells. Presence of SOX11 protein in cutaneous malignant melanoma was related to tumour type, tumour location, lymph node metastasis and 5-year survival rate.
Conclusion:
Human cutaneous malignant melanoma tissues expressed high levels of SOX11 compared with healthy controls, suggesting that SOX11 may be a new prognostic marker for malignant melanoma.
Keywords
Introduction
Malignant melanoma – which is a type of malignant tumour derived from the nevus cells and melanocytes1,2 – has a high mortality rate and a poor prognosis, due to a high degree of malignancy. Malignant melanoma is associated with early blood and lymphatic metastases that require intensive chemotherapy and radiotherapy.3 Sex determining region Y-box 11 (SOX11) protein is a transcription factor that participates in the regulation of neuronal maturation during embryonic neurogenesis; SOX11 is present in neural tumours, immature neurons and gliomas, but not in normal adult neurones.4,5 Neuronal cells and melanocytes are derived from the neural crest of the ectoderm during embryonic development.6,7 As neural tumour cells express SOX11 and melanocytes are precursor cells for malignant melanoma, SOX11 might be expressed in cutaneous malignant melanoma. The present retrospective study examined SOX11 protein levels in cutaneous malignant melanoma and healthy control tissue samples.
Patients and methods
Study population and tissue samples
Tissue samples and 5-year follow-up survival data from patients diagnosed with cutaneous malignant melanoma between January 2000 and December 2007 were retrieved from the files of the Departments of Dermatology and Pathology at Qilu Hospital, Shandong University, Jinan, China. Tissue samples that had been collected (prior to surgery or any other treatment) were studied. Patients with typical clinical manifestations and a pathological diagnosis of cutaneous malignant melanoma were eligible for inclusion, and were excluded if they refused permission for their data to be included. There were no other specific inclusion/exclusion criteria for the study. All samples were reviewed for histopathological classification of malignant melanomas according to the World Health Organization 2006 Classification of skin tumours. 8 Control samples of healthy tissue were used for comparison. These samples were obtained from people attending the Qilu Hospital, for routine examination. Control samples came from healthy people who had volunteered to provide samples for study purposes.
The study was approved by the Ethics Committee of Qilu Hospital, Shandong University, No. 2010QL0126. All patients provided written informed consent.
Immunohistochemistry analysis of Ki-67
The tissue samples were 3 µm-thick sections that had been previously fixed in 10% buffered formalin and embedded in paraffin wax. Following an antigen retrieval step, these tissue sections were incubated with primary antibodies, rabbit antihuman polyclonal primary antibody to SOX11 protein (ab42853, Abcam®, Cambridge, MA, USA; 1 : 200 dilution) and mouse antihuman monoclonal antibody to Ki-67 (ZM-0167, Jinqiao Biotechnology Company, Beijing, China; 1 : 200 dilution), for 1 h at 37℃.
The slides were washed three times for 2 min in 0.01 M phosphate-buffered saline (PBS, pH 7.25) then incubated with undiluted horseradish peroxidase conjugated goat antirabbit and antimouse secondary antibodies (ZSGB-BIO Beijing, China) for 15 min at 37℃. The slides were washed a final three times for 2 min in 0.01 M PBS, then immunohistochemical staining was visualized using EnVision™ two stage 3,3′-diaminobenzidine (DAB) staining (DAKO, Glostrup, Denmark) according to the manufacturer’s instructions.
Following DAB staining. Geimsa dye was used to counterstain sections to mask endogenously occurring yellow pigmentation in the melanoma cells: this process avoids the confusion with yellow DAB-stained granules representing the immunohistochemical signal.
Using a standard light microscope, the presence of SOX11 and antigen KI-67 proteins was observed as yellow (lower protein levels) or brown (higher protein levels) granular staining in the nuclei of the tumour cells. To evaluate the SOX11 staining, five randomly selected fields of view (within areas where ameloblastoma neoplastic cells predominated) were used to quantify positive cells. The number of positive cells in 200 tumour cells was calculated and the percentage of cells staining positive out of the total number of tumour cells counted was scored as follows: 1, ≤5%; 2, 6–25%; 3, 26–50%; 4, ≥50% of cells stained positive. Of these, a score of 1 was regarded as negative; scores of 2–4 were regarded as positive. The proliferation index was calculated as a percentage, as described previously: 9
number of tumour cells staining positive for antigen Ki-67/total number of tumour cells × 100.
Positive staining was evaluated using one photomicrograph per field (magnification × 40), and 10 fields of view were analysed per slide or case.
Statistical analyses
Statistical analyses were performed using the SPSS® software package, version 16.0 (SPSS Inc., Chicago, IL, USA) for Windows®. Between-group analysis of SOX11 levels in samples of skin lesions from patients with malignant melanoma and those from control subjects (healthy skin), and the relationship between clinicopathological parameters and SOX11, were performed using the χ2-test. The relationship between SOX11 expression and the proliferation index was analysed using Spearman’s rank correlation coefficient. The cumulative patient survival rate was determined using the Life Table method. 10 Univariate survival analysis was performed using the Kaplan–Meier method. Between-group differences were assessed using the log-rank test. Logistic regression was used to perform multivariate analysis of survival by SOX 11 protein scoring score. A P-value < 0.05 was considered to be statistically significant.
Results
In total, 40 patients with cutaneous malignant melanoma (age range 25–70 years) were enrolled into the study: 28 males and 12 females. There were 30 cases with, and 10 cases without, lymph node metastasis. Samples of healthy tissue were also included in the study. These were provided by 40 healthy volunteers (age range 22–60 years): 25 males and 15 females.
Incidence of sex determining region Y-box 11 (SOX11) expression in skin lesion tissue samples from 40 patients with cutaneous malignant melanoma.

Data presented as n (%) of patients.
SOX11 staining score relates to no. of cells staining positive for SOX11 protein, expressed as a percentage of total cells counted: 1, ≤5%; 2, 6–25%; 3, 26–50%; 4, ≥50%.
Antigen Ki-67 was observed as sporadic nuclear staining in the basal cell layer of healthy skin tissue and diffuse staining in cutaneous malignant melanoma tissue. The mean ± SD proliferation index was significantly higher in the malignant melanoma tissue samples compared with the healthy control tissue samples (72.7 ± 11.31 versus 3.06 ± 1.02, respectively, t = –38.79, P < 0.001). According to Spearman’s rank correlation coefficient, the presence of SOX11 correlated positively with the proliferation index (r = 0.672, P = 0.008).
Clinicopathological parameters and sex determining region Y-box 11 (SOX11) protein levels in 40 patients with cutaneous malignant melanoma.
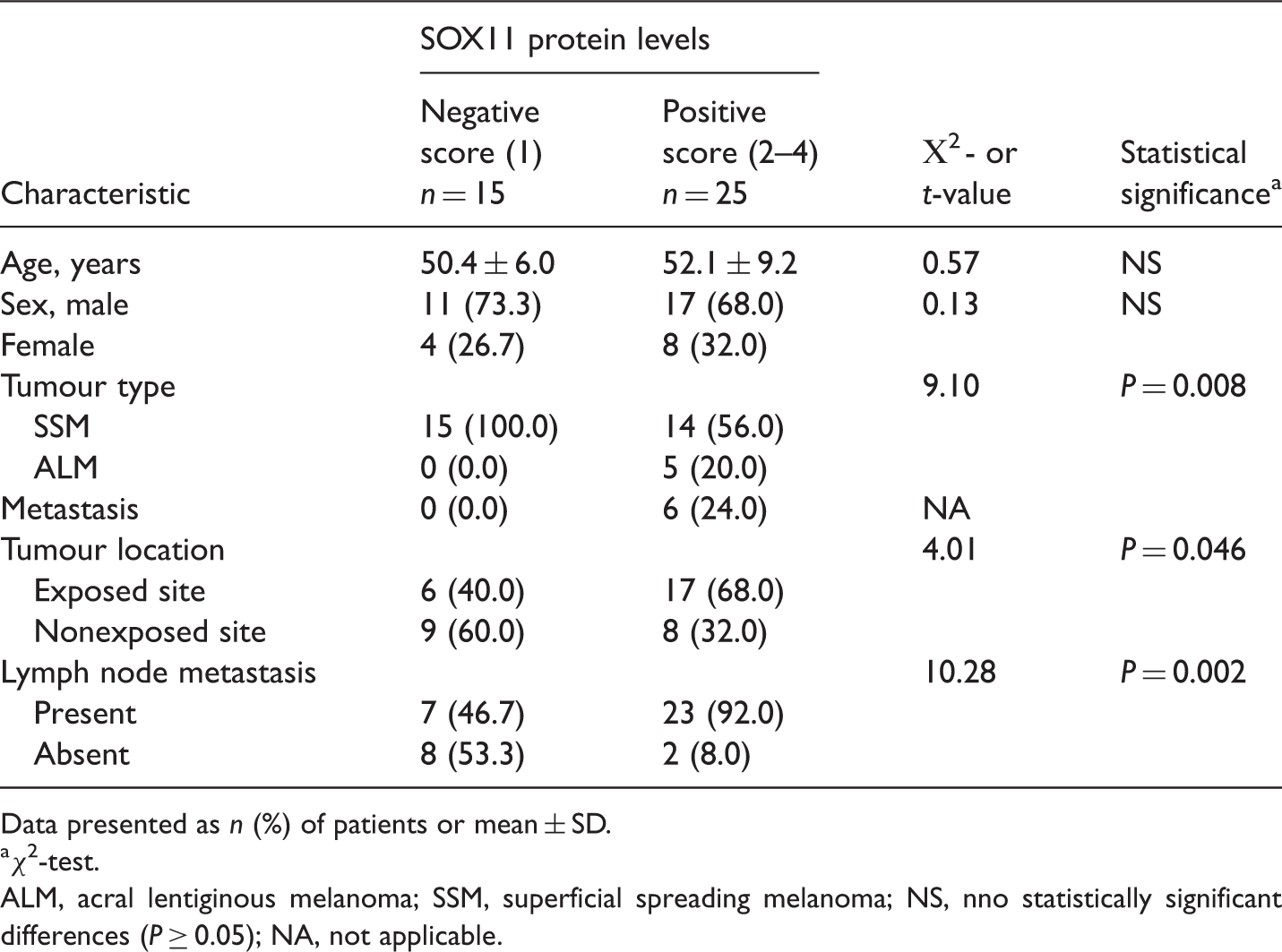
Data presented as n (%) of patients or mean ± SD.
χ2-test.
ALM, acral lentiginous melanoma; SSM, superficial spreading melanoma; NS, nno statistically significant differences (P ≥ 0.05); NA, not applicable.
Complete follow-up data were available for 40 patients with cutaneous malignant melanoma. Kaplan–Meier survival analysis demonstrated that the presence of SOX11 protein correlated with the 5-year survival rate. The survival rate in the SOX11 protein positive group was significantly lower than that in the SOX11 protein negative group (χ2 = 5.12, P = 0.024) (Figure 1).
Survival curves for 40 patients with cutaneous malignant melanoma, either positive (n = 25) or negative (n = 15) for sex determining region Y-box 11 (SOX11) protein expression; the 5-year survival rate was significantly different between the two patient groups (χ2 = 5.12, P = 0.024); univariate Kaplan–Meier survival analysis.
Discussion
Sex determining region Y-box 11 is one of a group of transcription factors with high activity within a nuclear protein superfamily, and is normally present in embryonic or developmental neurons; it plays an important part in the regulation of neuronal cell survival and neurite growth.11,12 Increased research into the SOX11 gene has revealed it to be expressed in several haematopoietic and lymphoid neoplasms and solid tumours, as well as in central nervous system malignancies, and has found that it may have some effect on tumour-cell survival, growth and transformation.13,14
The extent of involvement of SOX11 expression in the development of tumour pathogenesis is largely unknown.15–17 During maturation of the adult brain, SOX11 expression is gradually downregulated, although its expression is reactivated during tumourigenesis. 4 It has been suggested, therefore, that SOX11 represents a promising novel molecular target for adjuvant therapy of malignant glioma. 4 Increased expression of SOX11 has been shown in epithelial ovarian carcinoma: 18 multivariate analysis confirmed that SOX11 was an independent predictor of improved recurrence-free survival, after controlling for cancer stage and grade. 18 Studies have shown that SOX11 protein is also upregulated in lymphoproliferative disorders including B-cell lymphoma and mantle cell lymphoma.19,20 Little is known about the role of SOX11 in signal transduction, although some research has indicated that it is involved in tumourigenesis by combining with p53 and upregulating its transcriptional activity. 21 Epigenetic studies into the mechanisms by which SOX11 influences tumour pathogenesis have confirmed methylation of the SOX11 promoter in lymphoma and ovarian cancer.22,23 Moreover, de novo expression of SOX11 is associated with some aggressive lymphoid malignancies, which appear to be mediated by a shift from inactivating to activating histone modifications. 24 In the present study, SOX11 protein levels were increased in cutaneous malignant melanoma tumour cells, and the presence of SOX11 protein correlated positively with the proliferation index. Reactivation of SOX11 expression may, therefore, be involved in the pathogenesis of cutaneous malignant melanoma tumour-cell proliferation. Whether SOX11 is involved in cell-cycle regulation (by interaction with p53, or the presence of epigenetic regulatory mechanisms) requires further investigation.
There were some limitations to the present study. For example, because malignant melanoma is rare in China, the number of patients included in the study was small. Furthermore, this was a retrospective analysis of archived patient data. Larger, prospective studies are needed to clarify these initial study findings.
In conclusion, the present study findings indicate that the incidence of SOX11 protein may correlate with tumour type, tumour location, lymph node metastasis and 5-year survival rate in cutaneous malignant melanoma. This suggests that SOX11 may be a useful new prognostic marker for human cutaneous malignant melanoma and its possible role in cell proliferation may reveal new targets for biological treatment strategies.
Footnotes
Declaration of conflicting interest
The author declares that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
