Abstract
Objective:
To investigate the effect of the Intrafix® SafeSet infusion apparatus on the incidence of phlebitis in patients being intravenously infused in a neurological intensive care unit (ICU).
Methods:
Patients aged > 12 years, with no history of diabetes mellitus and no existing phlebitis, requiring a daily peripheral intravenous infusion of ≥ 8 h with the total period lasting ≥ 3 days, were enrolled. Infusions were performed using the Intrafix® SafeSet or normal infusion apparatus. Incidence of phlebitis (scored according to the Infusion Nursing Standards of Practice of the American Infusion Nurses Society) was analysed.
Results:
Patients (n = 1545) were allocated to Intrafix® SafeSet (n = 709) or normal infusion (n = 836) groups, matched for age, gender and preliminary diagnosis. Incidence of phlebitis was significantly higher using normal infusion apparatus compared with the Intrafix® SafeSet (23.4% versus 17.9%, respectively).
Conclusion:
Intrafix® SafeSet infusion apparatus significantly reduced the incidence of phlebitis in patients in the neurological ICU, compared with normal infusion apparatus, and may be suitable for use in routine clinical practice.
Keywords
Introduction
Nursing care for intravenous infusion is associated with technical risks and underlying safety issues, despite providing benefits to the patient.1,2 Each patient in the neurological intensive care unit (ICU) is in a critical condition and requires continuous infusion of a variety of medications. Intravenous infusion apparatus, infusion pumps or improper catheter nursing care can induce phlebitis and drug extravasation, which increases patient suffering and medical costs, and can even be life-threatening. 3 Numerous reports have suggested that phlebitis is related to multiple factors such as indwelling catheter location and duration, material and length of the catheter, replacement interval, infusion filtration apparatus, type of medication infused and concomitant diseases.4 – 7 The effect of the infusion apparatus on phlebitis appears to be primarily dependent on the pore size of the filter. The present case–control study analysed the incidence of phlebitis in patients in a neurological ICU, undergoing intravenous infusion performed using the Intrafix® SafeSet or normal infusion apparatus.
Patients and methods
Study Population
This case–control study included patients aged > 12 years who were admitted to the neurological ICU at West China Hospital, Sichuan University, Chengdu, China, between July 2010 and January 2011. Patients were sequentially enrolled in the study if they had: a requirement for peripheral intravenous infusion in the neurological ICU; a phlebitis score of 0 (according to the Phlebitis Scale developed by the American Infusion Nurse Society, 2006); 6 no history of diabetes mellitus; a daily infusion duration ≥ 8 h with the total period lasting ≥ 3 days. Patients were excluded from study participation if: their initial phlebitis score was > 0; the fat emulsion of the total infused solution was ≥ 20%; they required infusion therapy through a central venous catheter or a peripherally-inserted central catheter (PICC); the daily infusion duration was < 8 h; the total infusion duration was < 3 days.
Patient data (gender, age, diagnosis, foam rubber allergies, phlebitis [scoring grade and treatment of phlebitis] and mannitol administration) were collected by trained staff. Phlebitis was scored at intervals of 1 h (or at intervals of < 1 h in the case of any exceptional circumstance, such as venous blockage or malfunctioning of the infusion apparatus). Patients were closely monitored by nursing staff during the study and clinicians were notified as soon as possible of the emergence of phlebitis, so that the patient could recieve assessment and treatment. All details of patient care were handed over to the successive healthcare professional working the next shift in the neurological ICU.
A total of 26 beds were available in the neurological ICU. Patients were divided into two groups (according to their bed number), to receive infusions via normal infusion apparatus (patients in odd-numbered beds [control group]) or Intrafix® SafeSet infusion apparatus (patients in even-numbered beds [study group]). Eligible patients were treated according to the trial protocol, until discharge from the neurological ICU or completion of the study.
The study protocol was approved by the Ethics Review Committee of the West China Hospital, Sichuan University, Chengdu, China. All patients or their next of kin provided written informed consent.
Materials
The Intrafix® SafeSet infusion apparatus (B Braun Melsungen AG, Melsungen, Germany) and normal infusion apparatus (Chengdu Likang Industry Ltd, Chengdu, China) were used in this study. Both sets of apparatus complied with the quality standards of disposable infusion apparatus.
Phlebitis Scoring
Phlebitis was assessed according to the Infusion Nursing Standards of Practice set by the American Infusion Nurses Society in 2006: 6 Grade 0 phlebitis was defined as having no symptoms; grade 1 as redness at the infusion site in the presence or absence of pain; grade 2 as pain at the infusion site accompanied by redness and/or oedema; grade 3 as pain at the infusion site accompanied by redness and/or oedema, and formation of cord-like substances or cord-like veins that can be palpated; grade 4 as pain at the infusion site accompanied by redness and/or oedema, formation of cordlike substances and cord-like veins > 2.5 cm in length that can be palpated, and pus discharged.
Treatment of Phlebitis
Patients developing phlebitis received the following treatment regimens, according to their symptoms: (i) wet compress with 50% magnesium sulphate in grade 1 – 2 phlebitis; (ii) external application of a traditional Chinese medicine, Liuhedan (formulated in the West China Hospital Pharmacy), away from the puncture point in grade 2 – 3 phlebitis where the skin was not ulcerated; (iii) hydrocolloid dressing (Comfeel® Plus Transparent [Coloplast, Humlebaek, Denmark]) in grade 1 – 4 phlebitis (such treatments were chosen by the clinician, according to the individual patient's condition and the extent of skin swelling or broken skin); (iv) antagonist plus block therapy (2 ml procaine [0.5%] and 5 mg dexamethasone in 7 ml normal saline) in cases where there was leakage of highly irritable drugs from the infusion apparatus.
Statistical Analyses
All statistical analyses were performed using the SPSS® statistical package, version 18.0 (SPSS Inc., Chicago, IL, USA) for Windows®. Numerical data were analysed using Pearson's χ2-test. Between-group differences in age, gender and preliminary diagnosis were determined using the two-sample t-test. A P-value < 0.05 was considered statistically significant.
Results
A total of 836 patients were infused using normal infusion apparatus and 709 patients were infused using Intrafix® SafeSet infusion apparatus. No differences in sex, age, mannitol administration or the incidence of foam rubber allergy were noted between the two groups (Table 1). There were no significant differences between the two groups in terms of the preliminary diagnoses (Table 2). The incidence of phlebitis was significantly higher in the control group compared with the study group (P = 0.008, Table 3).
Clinical and demographic data for patients intravenously infused in the neurological intensive care unit with either normal infusion apparatus (control group) or Intrafix® SafeSet infusion apparatus (study group)

Data presented as mean ± SD or n of patients.
No statistically significant between-group differences (P ≥ 0.05) as determined by two-sample t-test (age and gender) or Pearson's χ2-test (use of mannitol and foam rubber allergy).
Preliminary diagnoses for patients intravenously infused in the neurological intensive care unit with either normal infusion apparatus (control group) or Intrafix® SafeSet infusion apparatus (study group)
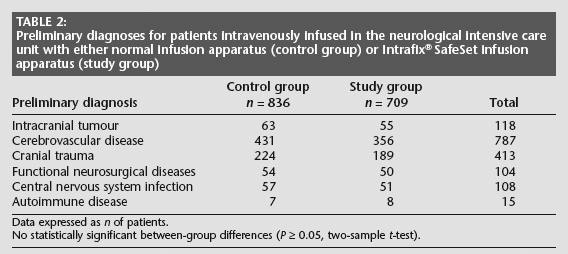
Data expressed as n of patients.
No statistically significant between-group differences (P ≥ 0.05, two-sample t-test).
Incidence of phlebitis in patients intravenously infused in the neurological intensive care unit with either normal infusion apparatus (control group) or Intrafix® SafeSet infusion apparatus (study group)

Data expressed as n (%) of patients.
Pearson's χ2-test.
Discussion
Phlebitis is an inflammation of the veins in which the vascular intima proliferates, leading to narrowing of the vascular cavity and slowing of the blood flow. 8 Congestive erythema accompanied by oedema sometimes appears in the peripheral skin, but fades over time and is replaced by pigmentation. After the occurrence of phlebitis, a minority of patients present with general phlebitis symptoms (including a decrease in skin temperature, fever and raised white blood cell counts), and complain of pain and swelling. Pathogenesis of phlebitis results from stimulation of the blood vessels by chemical, physical and infectious factors. The release of inflammatory mediators in the vessel causes damage to the vascular intima and increased vasopermeability. Consequently, local tissue fluids increase, causing redness, oedema, fever and pain. Phlebitis is more likely to occur with the use of irritant medications, and in the presence of inflammatory cells and necrotic endothelial cells. It can occur in both superficial and deep veins and is categorized as biomechanical, chemical, bacterial or thrombotic, according to the aetiology. The primary manifestations of infusion-related phlebitis are local skin redness, oedema, pain, fever, phlebosclerosis and substantial red cords. Phlebitis can prolong the duration of hospitalization and increase medical costs. 9
Infusion irritation is the most common cause of phlebitis. Numerous studies have investigated approaches to the prevention and treatment of phlebitis,4,5,7,9–11 although the effect of the infusion apparatus (in particular, use of the Intrafix® SafeSet infusion apparatus) on phlebitis has not been investigated. Di-(2-ethylhexyl) phthalate (DEHP) is a commonly used plasticizer in medical devices such as infusion apparatus. Concern has been growing worldwide regarding the toxic potential of DEHP, which has been confirmed to cause thrombophlebitis. 12 The link between the incidence of infusion-related phlebitis and the type of infusion apparatus does, however, require further investigation.
In the present study, the incidence of phlebitis was 17.9% and 23.4% in the study and control groups, respectively. Both of these values are within the standard range of phlebitis incidence (2.5 – 45%) reported by the Chinese Infusion Nurses Society Standards Committee, 13 but are above the 5% incidence reported in the Infusion Nursing Standards of Practice from the American Infusion Nurses Society. 6 The discrepancy between the incidence rates reported in the present study and those cited by the American Infusion Nurses Society may be due to several reasons. First, a large proportion of patients included in the current study were treated (as is common in patients with critical neurological diseases) with high-dose mannitol (1319 [85.4%] of patients; 607 and 712 in the study and control groups, respectively), which has been demonstrated to be a high-risk factor for phlebitis. 14 Secondly, the Standards of Practice from the American Infusion Nurses Society focus on general patients, but the current study included patients with critical neurological disease, treated without the use of an infusion solution containing ≥ 20% lipids, and who did not require infusion through a central venous catheter or PICC in the neurological ICU. Furthermore, eligible patients in the present study had undergone repeated puncture of blood vessels that could induce a high incidence of mechanical and thrombotic phlebitis. Thirdly, patients in the neurological ICU are characterized by their critical and rapidly changing conditions; consequently often they require various antibiotics, hypertonic glucose and emergency drugs, all of which also greatly increase the incidence of chemical phlebitis. In general, patients in a specialized ICU such as the neurological ICU are subject to almost all the known high-risk factors for phlebitis, and determination of the standard range within this population requires further investigation.
A significant reduction in the incidence of phlebitis in the study group compared with the control group was demonstrated in the present study, and it is speculated that this was due to the use of the Intrafix® SafeSet infusion apparatus. This device is made of a novel material and contains no DEHP, which is known to be harmful to the human body. 15 In addition, it is fitted with three special devices: (i) the Air Stop, which effectively blocks the entry of air into the infusion tubes and the patient's body to prevent aeroembolism and allows for the retention of air within the device when the fluid has run out, reducing venous return; (ii) the Prime Stop, which causes the infusion apparatus to exhaust the air automatically and form a closed system, reducing infection risk and avoiding drug waste; (iii) the filter, which decreases infusion-related phlebitis.16,17
In conclusion, the present study demonstrated a significantly reduced incidence of phlebitis associated with the use of the Intrafix® SafeSet infusion apparatus, compared with normal infusion apparatus, in patients admitted to a neurological ICU. The type of phlebitis (e.g. chemical, bacterial or thrombotic) and the level of reduction in the incidence of phlebitis, however, require further investigation. These data suggest that the Intrafix® SafeSet infusion apparatus may be suitable for use in routine clinical practice.
Footnotes
Conflicts of interest: The authors had no conflicts of interest to declare in relation to this article and have no commercial interests in the Intrafix® SafeSet infusion apparatus.
