Abstract
Objective:
To analyse the effect of chronic caffeine use on risk reduction and prognosis of diabetes mellitus.
Methods:
In this 60-day study, five groups of 11 healthy male Wistar rats were selected to receive one of four doses (37.5, 56.2, 75.0 or 93.0 mg/kg per day) of caffeine orally or no caffeine (control). The effect of caffeine on glycaemia and glucose tolerance was evaluated. After 15 days, each group was treated with 60 mg/kg of streptozotocine to induce diabetes mellitus, and glycaemia and glucose tolerance were assessed for a further 45 days.
Results:
In nondiabetic rats, caffeine had no effect on blood glucose. Compared with controls, the fasting blood glucose levels declined significantly in two caffeine-treated groups (93.0 mg/kg per day and 56.2 mg/kg per day) during the first 15 days following diabetes induction. Glucose tolerance was significantly improved 120 min after glucose loading in all caffeine-treated groups. The mean ± SE halfmaximal effective concentration of caffeine was 35.79 ± 2.44 mg/dl.
Conclusions:
Blood glucose levels decreased, and glucose tolerance improved, in diabetic rats administered increasing doses of caffeine.
Introduction
Type 2 diabetes mellitus is a chronic disease associated with high rates of morbidity, accompanied by characteristic long-term complications and premature mortality.1,2 Coffee is the most widely consumed beverage in the world and heavy coffee consumption has been associated with a lower risk of diabetes, but little is known about the mechanisms responsible for this association.3 – 5 Caffeine is one of the active biological components in coffee and is the principal source of the suggested benefits of coffee consumption.4,5 The effect of caffeine on glucose tolerance is still controversial, however. In studies conducted several years ago, blood glucose levels were reported to be higher,6,7 lower,8 – 10 or unchanged 11 after coffee or caffeine administration. In those studies, acute effects were observed in healthy humans, rats and dogs.
The present study evaluated whether blood glucose levels and glucose tolerance were modified by the long-term administration of caffeine in healthy and diabetic rats.
Materials and methods
Experimental Animals
Male Wistar rats, aged 8 – 10 weeks old and weighing a mean ± SD 252 ± 9 g, were obtained from Harlan Laboratories (Madison, WI, USA). The rats were maintained, with free access to food and water, at a constant temperature of 22 – 24°C at 55% humidity, with a 12-h light/12-h dark cycle (lights on at 07:00 h). Rats were used in accordance with the Institute for Laboratory Animal Research Guide for the Care and Use of Laboratory Animals. 12 The Ethics Committee of the University Centre for Biomedical Research, University of Colima, Colima, Mexico, approved the protocol. After the experiments, the rats were anaesthetized and underwent euthanasia.
Study Design
A total of 55 rats were divided into five groups, each consisting of 11 rats: the sample size of each group was calculated according to methods described previously.13,14 Rats received one of four doses (37.5, 56.2, 75.0 or 93.0 mg/kg per day) of caffeine administered orally, or no caffeine (control). Blood glucose levels were determined in each group for 15 days to determine the effects of caffeine in healthy rats. After 15 days diabetes was induced in each group as described below, and the effects of caffeine on blood glucose levels were assessed every 48 h until the end of the study (60 days). The weight of rats in each group was measured at the beginning of the study, at 15 days after diabetes induction and at the end of the study.
Diabetes Induction
Diabetes was induced using 60 mg/kg streptozotocine administered intra peritoneally (i.p.) in a single dose. A single dose of 230 mg/kg nicotinamide i.p. was administered 15 min before the streptozotocine, to reduce the damage to pancreatic β cells caused by streptozotocine. This technique was based on the model described by Masiello et al. 15 and it creates an experimental diabetic syndrome that is similar to human type 2 diabetes mellitus, with an insulin response to glucose increase and to the action of sulpholnylurea. 16 According to this model, ≥ 40% of the pancreatic insulin reserve is preserved, which is necessary for the development of type 2 diabetes.
Caffeine Dose and Administration
Caffeine (Sigma-Aldrich, St. Louis, MO, USA) was administered orally by catheter (Industrial Medica Plastica Silice SA de CV, Mexico). Considering the allometric scaling of rat metabolism according to Duff, 17 doses of 37.5, 56.2, 75.0 and 93.0 mg/kg per day are equivalent to the following doses in humans: 6 mg, 9 mg, 13 mg and 15 mg/kg per day, respectively. It is estimated that an adult human (mean weight 70 kg) with a moderate coffee intake consumes ∼400 mg of caffeine per day, which is 6 mg/kg, per day. 18 Furthermore, 6 mg/kg of caffeine is equivalent to three cups of coffee, and one cup is equivalent to drinking 227 g of regular coffee, which contains 137 mg of caffeine. 19
Glycaemia Assessment
Glycaemia, assessed by measuring glucose levels, was assessed every 48 h in the five groups from the beginning of the experiment to the end (day 60). Prior to every glucose measurement, each rat underwent 12 h of overnight fasting as described previously. 20 After fasting, to determine baseline glucose levels, blood samples (0.1 μl) were collected from the distal end of the tail and analysed immediately using an Accutrend® Plus glucometer with the corresponding Accutrend® test strips (Roche, Mannheim, Germany).
The oral glucose tolerance test (OGTT) was determined before diabetes induction in healthy rats and 6, 8, 10, 14, 30, 45, 50, and 60 days after diabetes induction. The OGTT was assessed, after a 12-h overnight fast, by glucose loading with 2 g/kg of oral glucose and measurement of glucose levels, as described above, at 0, 30, 60 and 120 min after glucose loading. If the fasting blood glucose level value was ≥ 126 mg/dl, a diagnosis of diabetes mellitus was confirmed.
Statistical Analyses
Statistical analyses were performed using the SPSS® statistical software, version 15.0 (SPSS Inc., Chicago, IL, USA) for Windows®. Data were presented as mean ± SE. Differences between continuous data were analysed using the paired Student's t-test and analysis of variance using a Scheffé post hoc comparison, as appropriate. A P-value < 0.05 was considered statistically significant. The dose –effect graphs were generated using Origin software (Originlab, Northampton, MA, USA) and the data were adjusted to a Hill equation of the type I = Imax/ (1+(EC50/x)h), where EC50 is the half-maximal effective concentration, I is the decrease in glycaemia, Imax is the maximum glycaemia decrease, and x is the caffeine concentration.
Results
Induction of Diabetes
In total, seven rats in the 93 mg/day caffeine group developed diabetes 24 h after disease induction, and four developed diabetes 7 days after disease induction (n = 11 per group). In contrast, all rats in the control group and in the other three caffeine-treated groups (n = 11 per group) developed diabetes 24 h after diabetes induction.
Effects of Chronic Caffeine on Blood Glucose
Over the 15 days prior to the induction of diabetes, fasting blood glucose levels following 93 mg/kg per day caffeine consumption were comparable with those in the control group, in healthy rats (Fig. 1). Over the first 15 days following the induction of diabetes, treatment with 93 mg/kg per day caffeine significantly reduced fasting blood glucose levels compared with the control group (P < 0.05 between groups, days 15 and 30). The relationship between fasting blood glucose levels and caffeine dose after the induction of diabetes was, however, unclear. There were no significant differences between the 37.5 and 75.0 mg/kg per day caffeine groups and the control group at any time point (data not shown). For the 56.2 mg/kg per day group, a significant difference was observed compared with the control group only on day 15 and day 30 (P < 0.05) (data not shown). For the remaining 30 days of the study, there were no significant differences in blood glucose levels in any of the caffeine-treated groups compared with the control group.
Fasting blood glucose levels in male Wistar rats receiving 93 mg/kg per day caffeine administered orally, or no caffeine (control), for 15 days before and 45 days after the induction of diabetes mellitus by the administration of 60 mg/kg streptozotocine (STZ) (n = 11 per group). Blood glucose measurements were carried out every 48 h for a period of 60 days. Data are mean ± SE; *P < 0.05, analysis of variance using a Scheffé post hoc comparison
Effects of Chronic Caffeine on Glucose Tolerance
On day 15 before the induction of diabetes, there was no significant difference in blood glucose levels between the 93 mg/kg per day caffeine and control groups at 30, 60 and 120 min following glucose loading (Fig. 2).
Oral glucose tolerance test (carried out before the induction of diabetes mellitus with of 60 mg/kg streptozotocine on day 15) measuring blood glucose levels at 0, 30, 60 and 120 min after loading with 2 g/kg glucose, orally, in healthy male Wistar rats that had been fasted for 12 h. The rats had received 93 mg/kg per day caffeine administered orally or no caffeine (control) for 15 days (n = 11 per group). Data presented mean ± SD; there were no significant between-group differences (P ≥ 0.05), Student's analysis of variance using a Scheffé post hoc comparison
Over the 15 days following the induction of diabetes, blood glucose levels in the 93 mg/kg per day caffeine group were significantly lower at 30, 60 and 120 min after glucose loading, versus the control group (P < 0.05; Fig. 3; results show mean data from days 6, 8, 10 and 14 after diabetes induction). There was no significant difference in blood glucose levels after glucose loading between the other caffeine-treated groups and the control group, with the following exceptions: 56.2 mg/kg per day group at 0 and 120 min, 75.0 mg/kg per day group at 120 min versus controls (P < 0.05 for all comparisons; Fig. 3). For the 37.5 mg/kg per day caffeine group, there were no significant differences at any time point after glucose loading except at 120 min (data not shown).
Oral glucose tolerance test (carried out between days 15 and 30, i.e. after the induction of diabetes mellitus with of 60 mg/kg streptozotocine on day 15) measuring blood glucose levels at 0, 30, 60 and 120 min after loading with 2 g/kg glucose, orally, in healthy male Wistar rats fasted for 12 h. The rats had received either 37.5 mg/kg per day, 56.2 mg/kg per day, 75.0 mg/kg per day or 93.0 mg/kg per day caffeine or no caffeine (control), for 15 days (n = 11 per group). Results show mean ± SE data from days 6, 8, 10 and 14 after diabetes induction. *P < 0.05 at all time points for the 93 mg/kg per day group, at 0 and 120 min for the 56.2 mg/kg per day group and at 120 min for the 56.2 mg/kg per day group versus controls; analysis of variance using a Scheffé post hoc comparison. Data for the 37.5 mg/kg per day caffeine group are not shown and only showed a statistical difference at 120 min
The effects of caffeine on glucose tolerance disappeared from 15 days after diabetes induction. No significant difference in glucose tolerance was observed between the four caffeine-treated groups and the control group from days 30 to 60 following diabetes induction (Fig. 4; results show mean data from days 30, 45, 50 and 60, after diabetes induction).
Oral glucose tolerance test (carried out between days 30 and 60, i.e. after the induction of diabetes mellitus with of 60 mg/kg streptozotocine on day 15) measuring blood glucose levels at 0, 30, 60 and 120 min after loading with 2 g/kg glucose, orally, in healthy male Wistar rats fasted for 12 h. The rats had received either 37.5 mg/kg per day, 56.2 mg/kg per day, 75.0 mg/kg per day or 93.0 mg/kg per day caffeine or no caffeine (control), for 15 days (n = 11 per group). Results show mean ± SE data from days 30, 45, 50 and 60 after diabetes induction. There were no significant differences between the four caffeine-treated groups and the control group (37.5 mg/kg caffeine group; data not shown); analysis of variance using a Scheffé post hoc comparison
Based on a dose–response curve showing the relationship between the percentage decrease in glycaemia, with respect to the control group, and caffeine concentration at 120 min postglucose loading, the mean ± SE EC50 for caffeine was calculated to be 35.79 ± 2.44 mg/dl (Fig. 5). The maximum mean ± SE effect on glycaemia calculated by the Hill equation was 46.18 ± 4.84%. The mean ± SE Hill coefficient was 1.35 ± 0.18.
Dose–response curve for caffeine. The half-maximal effective concentration (EC50) required to decrease blood glucose levels was 35.79 ± 2.44 mg/dl. Each point in the curve corresponds to the means from the four caffeine-treated groups (37.5 mg/kg per day, 56.2 mg/kg per day, 75.0 mg/kg per day or 93.0 mg/kg per day; n = 44). Curves are as mean ± SE data taken from days 6, 8, 10 and 14 after induction of diabetes with 60 mg/kg streptozotocine. Data adjusted using the Hill equation
Caffeine and Weight Loss
Weight loss was observed in each of the five groups during the first 15 days of the study and over the 60 days after diabetes induction, although there was no significant difference in weight loss over the course of the study, or between the caffeine-treated groups and the control group, at 15 or 60 days (Table 1).
Baseline body weight, and body weight 15 and 60 days after diabetes induction using 60 mg/kg streptozotocine, in healthy Wistar rats orally administered daily doses of caffeine or no caffeine for 60 days (control) (n = 11 per group)
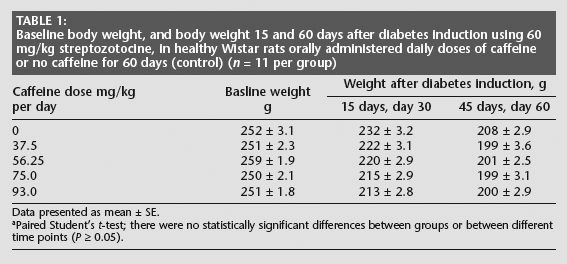
Data presented as mean ± SE.
Paired Student's t-test; there were no statistically significant differences between groups or between different time points (P ≥ 0.05).
Discussion
In the present study, Wistar rats received a chronic administration of caffeine for 60 days, during which baseline blood glucose levels (after fasting for 12 h) and glucose tolerance (after 2 g/kg glucose load) were observed to be dependent on the dose of caffeine administered. These effects were only observed during the first 15 days after diabetes induction; there was no significant difference between caffeine-treated rats and the control group from day 15 after the induction of diabetes (day 30 of study) until the end of the study. These results suggest that caffeine may have caused a delay in the appearance of diabetes.
It is known that caffeine increases the rate of glucose transport in the absence of insulin in rodent skeletal muscle,21 – 23 and enhances glucose transporter-4 (Glut-4) mRNA and protein levels in cultured myotubes. 24 In a rat model, caffeine predominantly activates the cyclic AMP-dependent protein kinase α-1 isoform (α-1 cAMP) by means of an insulin-independent mechanism, 25 which increases AMP kinase-1 to promote glucose transport. In addition, the upregulation of Glut-4 is dependent on the caffeine concentration. 25 These findings could partly explain the results observed in the present study.
An alternative mechanism could be that caffeine acts as an antagonist of adenosine A1 receptors, which are present in skeletal muscle. Antagonism of these receptors has been shown to improve glucose tolerance in rats. 26 Moreover, A1 adenosine receptor antagonism in obese Zucker rats increases glucose uptake in skeletal muscle. 27
In the present study, the effect of caffeine was observed during the first 15 days after diabetes induction with streptozotocine – a substance that acts by destroying β pancreatic cells. In addition, there was a gradual increase in glucose levels in the group receiving the highest caffeine dose (93 mg/kg per day). This did not occur in the control group, where glucose increase was immediate. It is not clear why the effects of caffeine only occurred during the first 15 days after diabetes induction. It is possible that there is an induced tolerance mechanism after longer periods of consumption. 28 Some studies in habitual coffee drinkers suggest, however, that tolerance does not develop. 9
In healthy rats, during the 15 days prior to diabetes induction, caffeine did not have a significant effect on glucose level and glucose tolerance. These results concur with a previous study. 25 There are, however, conflicting reports of an increase in coffee consumption reducing9,10 or increasing 6 blood glucose levels in healthy volunteers.
It has been suggested that the protective effect of coffee could be due to a thermogenic effect of caffeine, which could indirectly reduce the risk of obesity (a major risk factor for diabetes). 29 The thermogenic effect of caffeine and the subsequent decrease in body weight is well known. A large prospective study has previously reported that increased caffeine intake is correlated with lower weight gain. 30 In the present study, weight was not significantly different between the caffeine-treated rats and the control group at 15 days after diabetes induction (day 30) and at the end of the study (60 days).
The antioxidant effect of caffeine has also been described. 31 It has been proposed that caffeine could contribute to the protection of β pancreatic cells from free radicals produced by oxidative stress, prevent membrane damage and preserve the β pancreatic cell. 32
A study on biomarkers showed that caffeine metabolites (isoprenoids) persist in the bloodstream for 24 h and are not easily eliminated following daily coffee and caffeine intake for ≥ 15 days. 33 This could partially explain the long-term effects of caffeine.
There are only a few studies reporting a possible relationship between the consumption of caffeine and the progression of diabetes. The present study showed a tendency toward the reduction in severity of disease with daily doses of caffeine during the first 15 days following diabetes induction. This time frame corresponds to a longer period in humans, given that rats live for 2.5 – 3 years, whereas humans live for 75.7 – 80.6 years,34,35 and it represents the period of time during which the severity of disease can decrease. With a caffeine dose of 93.3 mg/kg per day, some rats did not become diabetic until day 7 after induction of diabetes with streptozotocine. This delay in disease presentation could result in a better prognosis for the diabetic rats.
There were certain limitations to the study. As this was an animal study, it would be useful to repeat the study in another experimental model of diabetes, to verify the results. Further research is required to determine the possible secondary effects of these caffeine doses on animals, to extend the time of caffeine administration in normal rats and induce diabetes prior to administering caffeine, to observe whether this effect on baseline blood glucose levels or OGTT can be modified in existing disease.
Footnotes
Acknowledgements
The authors thank the Consejo Nacional de Ciencia y Tecnologia (CONACyT) Mexico for financial support (Project 83313) for a Master in Sciences scholarship given to Z Urzúa. Portions of this work formed part of the MSc thesis of Z Urzúa at the Universidad de Colima (Mexico). The authors wish to thank Karla Vera, Ezequiel Viera, Araceli Santos-Diego, Evelyn Mancilla-Morfín, and Jesus Dueñas for their technical assistance. Z Urzúa, X Trujillo, M Huerta, and M Ríos-Silva designed and performed the experiments; B Trujillo-Hernández, C Onetti, M Ortiz-Mesina and E Sánchez-Pastor provided the discussion, and help with analysis of the results. M Huerta and X Trujillo wrote the manuscript and take responsibility for the contents of the article.
Conflicts of interest: The authors had no conflicts of interest to declare in relation to this article.
