Abstract
Objective:
Studies have demonstrated the protective effects of biomaterials against myocardial infarction (MI), but the relationship between their physical characteristics and their function is incompletely understood. This study investigated such relationships for a hydrogel preparation.
Methods:
Two types of poly(N-isopropylacrylamide) (PNIPAAm) hydrogel with different degradation times (Gel A and Gel B) were synthesized. In vivo hydrogel formation and maintenance were observed and confirmed in mice. The solutions were also injected into the infarct area immediately after MI induction in rats.
Results:
PNIPAAm hydrogel exhibited a three-dimensional structure resembling native extracellular matrix (ECM). Compared with phosphate-buffered saline, Gel A and Gel B increased contractility of isolated infarcted myocardium, reduced collagen deposition, increased neovascularization, inhibited left ventricle expansion and improved cardiac function. Myocardial contractility was greater with Gel B than with Gel A.
Conclusions:
Intra -myocardial injection of PNIPAAm hydrogel provides structural support and some functional repair of damaged ECM, suggesting that it might be useful for cardiac tissue engineering.
Keywords
Introduction
Although new drugs and innovative devices have improved the health-related quality of life for patients who have experienced myocardial infarction (MI), MI is still one of the most common causes of morbidity and mortality worldwide. 1 Progressive left ventricle architectural remodelling is independently associated with dysfunction and heart failure. 2 Heart transplantation is the last therapeutic option for terminal heart failure. However, the demand for organs far outweighs the number of available donor organs. New therapeutic strategies are therefore urgently needed.
Over the last 10 years, biomaterial technology has shown great promise as a potential treatment for MI. 3 Synthetic polymers are advantageous materials for cardiac tissue engineering because of the ease of controlling their final properties such as structure, pore size and orientation. 4 Poly(N-isopropylacrylamide) (PNIPAAm) 5 and polycaprolactone 6 are polymers used for engineered myocardial constructs, providing improved cardiac function, reduced infarct size and increased neovascularization.7,8 Most researchers have focused on the effectiveness of synthetic polymers as a delivery scaffold for cells and/or other bioactive factors.9 – 11 However, a previous study by our research group demonstrated good performance of PNIPAAm hydrogel alone in the inhibition of ventricular remodelling and protection of cardiac function after MI in rabbits. 7 Similar results were reported by two studies using rat infarction models.12,13 These findings showed that, even without cells or bioactive factor transplantation, biomaterials alone had a biological impact on animal hearts.7,12,13
The present study investigated how the physical properties of biomaterials may contribute to their effectiveness in the treatment of MI, because little research has been undertaken on this subject. Two types of temperature-sensitive PNIPAAm hydrogels, with different theoretical biodegradation times, were synthesized and their effectiveness was investigated in a rat MI model.
Materials and methods
Study Animals
This study was conducted in the Department of Cardiology, Renmin Hospital of Wuhan University, Wuhan, China. Animals used in the study received humane care in accordance with the Revised Guide for the Care and Use of Laboratory Animals, published by the US National Institutes of Health. 14 The study protocol was approved by the Ethics Committee of Wuhan University.
Male Wistar rats (weight, 180 – 220 g) and male KM mice (weight, 15 – 25 g) provided by the Disease Prevention and Control Centre of Hubei Province, Wuhan, China, were used in the experiments. Animals were housed in clean laboratory rooms with a temperature-controlled environment, under a 12-h light/12-h dark cycle, with free access to food and water.
Materials
Dextran (molecular weight 60 000 – 90 000 g/mol), NIPAAm and ε-caprolactone (CL) were purchased from Sigma (St Louis, MO, USA). 2-Hydroxeylthyl methacrylate (HEMA) was obtained from Shanghai Chemical Reagent (Shanghai, China).
Synthesis of Pnipaam Hydrogel
Two types of PNIPAAm hydrogel (designated Gel A and Gel B) were synthesized, as previously described. 15 First, a dextran– poly(CL-HEMA) macromonomer was synthesized by the chemical precipitation of CL (6.84 g, 0.06 mol), HEMA (0.03 g, 0.003 mol) and dextran (4.86 g, 0.03 mol glucopyranose residues), and subsequently dried in a vacuum. Next, a NIPAAm monomer and dextran–poly(CL-HEMA) macromonomer with a weight ratio of 19 : 1 were dissolved in 1 mM phosphate-buffered saline (PBS; pH 7.4) to form Gel A. Gel A was then dialysed for 2 days and subsequently freeze-dried. The lyophilized Gel A product was then dissolved in 1 mM PBS (pH 7.4) to obtain a 3% (w/v) solution of Gel A, which was prepared before the experiment and stored at 4 °C until being prepared for injection at 20 °C.
Gel B was synthesized by the same procedure as Gel A, except that twice the amount of dextran was used (9.72 g, 0.06 mol glucopyranose residues). A 3% (w/v) solution of Gel B was prepared and stored using the same procedure as for the Gel A solution. Both Gel A and Gel B shift from solution to gel at ∼37 °C within 10 s because of the temperature sensitivity of the NIPAAm monomer, and this shift reverses when the temperature returns to 4 °C. Except for the theoretical degradation time, which is based on the adjusted ratio of dextran to CL, the other physical properties of Gel A and Gel B are the same.
Scanning Electron Microscopy of Pnipaam Hydrogel
Two millilitres of 3% (w/w) Gel B solution were injected into a 2 × 2 cm plate and formed into the gel state at 37 °C, and the plate was then placed in a dessicator. The dried Gel B formed a membrane, which was coated with platinum and examined by scanning electron microscopy.
Pnipaam Hydrogel Biodegradation In Vivo
Sixty-one male KM mice (6 – 8 weeks old) were used in this investigation. Mice were used because the lower amount of subcutaneous fat in mice, compared with rats, facilitates the use of a 27-gauge needle for injection into the space between the back muscles and subcutaneous tissues. In addition, it is relatively easy to detect visually any eminences formed around the hydrogel injection site; mice have a smaller body surface area than rats, making it easier to find any remaining hydrogel several days after injection. Previously, we have undertaken research on biomaterial degradation in KM mice (unpublished). Before the test, the mice were anaesthetized with 0.5% sodium pentobarbital solution (30 mg/kg body weight) given by intraperitoneal injection. The level of anaesthesia was considered to be adequate when the corneal, righting and pain reflexes of the mice had disappeared. An aqueous solution of 3% (w/v) Gel A (400 μl) or 3% (w/v) Gel B (400 μl) was then injected subcutaneously into the back of each mouse, using a syringe with a 27-gauge needle.
At designated time intervals, the mice were killed by intraperitoneal injection of an overdose of pentobarbital solution (50 mg/kg body weight), and the hydrogel injection site was carefully cut open. The surrounding tissue was removed as quickly as possible and the remaining hydrogel was extracted and weighed. The biodegradation rate of the hydrogel was calculated as Mt/M0 × 100%, where M0 is the initial weight of the injected hydrogel and Mt is the weight of the remaining hydrogel at the designated euthanization time.
In the Gel A group, one mouse was euthanized every day for the first 5 days, thereafter one mouse was euthanized every 3 days. In the Gel B group, one mouse was euthanized every day. The different schedules for the two gels were based on differences in predicted degradation rates. The in vivo hydrogel biodegradation test was continued until complete degradation of the hydrogel was observed.
Myocardial Infarction Model
Seventy Male Wistar rats (aged 5 – 6 weeks) were anaesthetized by intraperitoneal injection of 0.5% sodium pentobarbital (30 mg/kg body weight). Additional small doses were given to maintain deep anaesthesia when needed. Lidocaine (2%, 0.2 ml per rat) was injected around the incision for local infiltration anaesthesia. A small-animal ventilator (TKR 400H; Teli Anesthesia and Breathing Equipment, Chengdu, China) was used during surgery for respiratory support by tracheal intubation.
The heart of each rat was exposed by thoracotomy; the left anterior descending coronary artery (LAD) was then ligated 2 mm below the left atrial appendage using 7-0 polypropylene sutures. Occlusion was confirmed by observing pallor and abnormality of regional motion of the left ventricle wall. Five minutes after LAD ligation, the rats were assigned, using a table of random numbers, to receive either intramyocardial injections of 100 μl 1 mM PBS (pH 7.4; MI + PBS group), 100 μl Gel A solution (MI + Gel A group) or 100 μl Gel B solution (MI + Gel B group). The temperature of the PNIPAAm solution was ∼20 °C at the time of injection; the solution shifted from the liquid to the gel state under the effect of body temperature within 10 s after injection. A suture was placed along the infarct region to mark the injection site. Rats in a sham-operated group were subjected to the same procedure as described above, except that the polypropylene suture around the LAD was loose and no injection of solution was performed. All rats were maintained under standard conditions for 90 days after the operation.
Echocardiography and Haemodynamic Analyses
All rats treated as described in the previous section were anaesthetized again in preparation for echocardiography. Dimensional and functional measurements of the left ventricle (LV) were determined with an echocardiograph equipped with a 3 – 7 MHz linear transducer (Acuson Sequoia™ 512 ultrasound system; Siemens Medical Solutions USA, Mountain View, CA, USA). Baseline echocardiography was performed 2 days after the operation. Rats with a visual infarct size < 20% at the papillary level in the short-axis view were excluded. Ninety days after surgery, LV end-systolic diameter (LVESD), LV end-diastolic diameter (LVEDD) and thickness of the infarcted wall were measured in at least three consecutive cardiac cycles. Fractional shortening (FS) was calculated using the following formula: FS (%) = ([LVEDD - LVESD]/ LVEDD) × 100.
After echocardiography, an additional small dose of 0.5% pentobarbital sodium was given to maintain deep anaesthesia. Supplementary 2% lidocaine was used for local infiltration anaesthesia around the incision site (0.2 ml per rat). For the haemodynamic measurements, a catheter was inserted into the ascending aorta through the right carotid artery, and the catheter was then further inserted into the LV to record the LV systolic and diastolic curves. The systolic and diastolic functions, LV +dP/dtmax and LV -dP/dtmax (where P is the pressure at time t), respectively, were calculated with a computer system (Lead 2000B, JinJiang, Chengdu, China). These two parameters represent the minimum and maximum rate of pressure change in the ventricle.
Contractility of Isolated Cardiac Muscle In Vitro
Eight rats from each group were used to measure the contractility of isolated cardiac muscle in vitro. After the echocardiogram and haemodynamic measurements had been completed, the rats were euthanized by intraperitoneal injection of an overdose of pentobarbital solution (50 mg/kg body weight). The heart was immediately removed and placed in cold Tyrode's solution (pH 7.38; NaCl 135 mmol/l, KCl 5.4 mmol/l, MgCl2.6H2O 1 mmol/l, NaH2PO4.2H2O 0.33 mmol/l, HEPES 10 mmol/l, glucose 10 mmol/l and CaCl2 1.8 mmol/l). The infarct or sham-operated area from the left ventricle was dissected into an isolated 1.0 × 0.5 cm area of cardiac muscle.
The excised cardiac muscle was then mounted horizontally in a constant-temperature bath of Tyrode's solution (pH 7.38) at 37°C aerated with 100% oxygen. All samples were submitted to a resting tension of 1 g before electrical stimulation. A constant voltage stimulator (RM 6240 multichannel physiological signal acquisition and processing system; Chengdu Instrument, Chengdu, China) was programmed to provide a constant positive voltage stimulus with a square-wave impulse of 10 ms and a frequency of 2 Hz, to stimulate the isolated cardiac muscle via two platinum electrodes. The intensity of the stimulus was 120% of the threshold, which was confirmed by pretesting to be 5 V.
Measurements were taken for 30 min and the subsequent contraction curve was recorded by computer image analysis (Lead 2000B). The contraction force and contraction time of the isolated cardiac muscle from ≥ 10 consecutive contraction cycles were measured. The mean time needed to reach the peak contraction force during one contraction was recorded as the half contraction time; the time required for a complete contraction was recorded as the whole contraction time.
Western Blot Analysis
Western blot analysis was conducted using lysates from infarcted and sham-operated myocardium 90 days after operation. Protein samples were extracted with G2001 and G2002 protein extraction kits (Guge Biotechnology, Wuhan, China). The concentrations of protein samples were estimated with a Bradford protein assay kit (Beyotime, Wuhan, China). Samples containing 40 μg protein were separated by 10% sodium dodecyl sulphate–polyacrylamide gel electrophoresis and transferred to polyvinylidene difluoride (PVDF) membranes. After blocking with 5% skimmed milk for 1 h, the membranes were probed with primary antibodies against collagen I (1 : 2000, ab292; Abcam®, Cambridge, MA, USA), collagen III (1 : 2000, ab7778; Abcam®) and transforming growth factor (TGF)-β1 (1 : 2000, ab92486; Abcam®) overnight at 4°C. After washing three times for 5 min each using 1 × Tris-buffered saline (TBS, pH 7.5), the membranes were probed with secondary antibody (074-1506 horseradish peroxidase [HRP]-labelled goat antirabbit, 1 : 3000; Guge Biotechnology) for 30 min at room temperature. Membranes were then washed three times with 1 × TBS (pH 7.5). Blots were visualized by chemiluminescence (Amersham ECL; Beyotime) and the signals were quantified by densitometry. The ratios of collagen I to collagen III and the amount of TGF-β1 protein in the infarct and sham-operated myocardium were normalized to β-actin, which was used as the internal control.
Infarct Size and Collagen Volume Fraction
After completing the echocardiographic and haemodynamic analyses, the rats were anaesthetized (all processes as described above). The numbers of animals surviving at this stage were as follows: sham-operated group, 13; MI + PBS, 14; MI + Gel A, 19; MI + Gel B, 19. A table of random numbers was used to reduce the number of animals analysed in each group to 13. The hearts were explanted and the infarcted parts were cut into two parts; one part was used for Western blot analysis and the other part was immediately fixed in 4% paraformaldehyde at room temperature for 24 h, dehydrated with a graded ethanol series and embedded in paraffin wax. Specimens were cut from apex to base to produce five transverse sections (5 μm thick). Sections were mounted on gelatine-coated glass slides and stained with haematoxylin and eosin (H&E) or Masson's trichrome.
The H&E-stained sections were used to measure infarct size. Infarct length and left ventricular circumference were measured by automated computer image analysis (Image-Pro® Plus version 6.0; Media Cybernetics, Silver Spring, MD, USA). Infarct size was calculated by the following formula: infarct size (%) = (epicardial infarct length + endocardial infarct length) / (epicardial left ventricular circumference + endocardial left ventricular circumference) × 100.
The areas of collagen and myocardium in the sections stained with Masson's trichrome were also measured by automated computer image analysis (Image-Pro® Plus, version 6.0). The collagen volume fraction (CVF) was calculated by the following formula: CVF (%) = collagen area/myocardium area × 100.
Immunohistochemical Staining
Five rats from each group were used for histological and immunohistochemical staining. Infarcted and sham-operated hearts were fixed in 4% paraformaldehyde for 24 h and paraffin wax-embedded sections 5 μm thick were prepared. Antigen was retrieved by the microwave method to optimize staining quality. The slides were incubated overnight at 4°C with anti-α-smooth muscle actin primary antibody (dilution 1 : 50; Boster, Wuhan, China). The slides were washed and incubated for 50 min at 4 °C with the secondary antibody HRP-labelled goat antimouse (REAL™ EnVision™+/HRP Rabbit/Mouse Kit; Dako, Copenhagen, Denmark) according to the manufacturer's instructions. The chromogen solution used was that included in the REAL™ EnVision™+/HRP kit. The slides were then washed three times (5 min each time) in 1 mM PBS, pH 7.4. Immunohistochemical staining was photographed through a microscope (XSP-C204; Chongqing Photoelectric Instrument Co., Ltd, Chongqing, China). Five images of tissue sections from each group under × 100 high-power fields (HPFs) were selected using a random number table. The number of blood vessels in each infarct or sham-operated area was counted. The number of vessels was then used to calculate vessel density per HPF.
Statistical Analyses
All statistical analyses were performed using the SPSS® statistical package, version 13.0 (SPSS Inc., Chicago, IL, USA) for Windows®. All data were expressed as mean ± SD. The data were compared, and intergroup differences were analysed by one-way analysis of variance and an unpaired Student's t-test. Differences were considered statistically significant if the P-value was < 0.05.
Results
Structure and Biodegration of Pnipaam Hydrogel
A scanning electron micrograph of PNIPAAm hydrogel (Gel B) is shown in Fig. 1A. The image shows that the three-dimensional structure of PNIPAAm hydrogel was that of a skeleton-like network with an abundance of pores.
Scanning electron micrograph showing the three-dimensional and cross-linked structure of the poly(N-isopropylacrylamide) hydrogel (Gel B) (A). In vivo biodegradation curves for Gel A (B) and Gel B (C) in KM mice
The biodegradation curves of Gel A and Gel B obtained in the in vivo biodegradation tests in KM mice are shown in Figs 1B and C, respectively. The time required for complete biodegradation of Gel B was 28 days; biodegradation was rapid within the first 4 days, became slow during the following 2 weeks, and then became rapid again until biodegradation was complete. The biodegradation curve of Gel A was similar to that of Gel B, but the rate of biodegradation of Gel A was slower than that of Gel B, and complete biodegradation of Gel A took 85 days.
Animal Survival Rate
A total of 61 male KM mice and 70 male Wistar rats were analysed in this study. All mice that were used in the in vivo biodegradation test survived until the scheduled euthanasia time. One rat died during endotracheal intubation. A total of 10 rats died within 24 h (four in the MI + PBS group, three in the MI + Gel A group and three in the MI + Gel B group) and three rats died within 7 days (all in the MI + PBS group) after infarct induction. Four rats died from chronic heart failure (two in the MI + PBS group, one in the MI + Gel A group and one in the MI + Gel B group). The number of rats remaining 90 days after surgery was 13 in each group.
Echocardiography and Haemodynamic Analyses
Two days after operation, baseline echocardiography showed no significant difference in LVEDD, LVESD and FS among the three experimental MI groups of rats (data not shown). Ninety days after surgery, LVEDD and LVESD were significantly greater in the MI + PBS group than in the sham-operated group (P < 0.05 for both comparisons) (Table 1). LVEDD, FS and the thickness of the infarcted wall were significantly smaller in the MI + PBS group than in the sham-operated group (P < 0.05 for all comparisons). Echocardiography showed that, compared with the MI + PBS control group, Gel A and Gel B injection treatments both significantly reduced left ventricular expansion, thickened the infarcted wall and improved cardiac FS (P < 0.05 for all comparisons). Although there were no significant differences between the MI + Gel A and MI + Gel B groups, the results suggested an advantage for the Gel B-treated hearts.
Parameters of left ventricular (LV) function derived from echocardiography 90 days after surgery In rats that had undergone sham operation (sham-operated group) or Induction of myocardial Infarction (Ml), followed by treatment with phosphate-buffered saline (Ml + PBS group) or poly(N-lsopropylacrylamlde) hydrogel preparations (Ml + Gel A group and Ml + Gel B group)

Data presented as mean ± SD.
P < 0.05 compared with sham-operated group.
P < 0.05 compared with MI + PBS group.
P < 0.05 compared with MI + Gel A group; one-way analysis of variance and unpaired Student's t-test.
LVEDD, left ventricular end-diastolic diameter; LVESID, left ventricular end-systolic diameter; LV thickness, thickness of myocardium in the infarct zone of the left ventricle; +dP/dtmax’ represents systolic function; -dP/dtmax’ represents diastolic function.
Systolic function (LV +dP/dtmax) was also significantly reduced 90 days after infarction in the MI + PBS group compared with the sham-operated group (P < 0.05) (Table 1). Compared with infarcted hearts treated with PBS (MI + PBS group), Gel A or Gel B treatment significantly increased the value of LV +dP/dtmax (P < 0.05 for both comparisons). Moreover, Gel B injection therapy showed significant benefit compared with Gel A in improving cardiac systolic function (P < 0.05). Similar differences were found for diastolic function (LV -dP/dtmax).
Contractility Analysis of Infarcted Myocardium
The mean contraction force of isolated cardiac muscle was significantly reduced after MI compared with the sham operation (P < 0.05 for all comparisons) (Fig. 2A). Both the Gel A and the Gel B injection treatment significantly improved the mean contraction force compared with the MI + PBS group (P < 0.05 for both comparisons). Furthermore, treatment with Gel B resulted in significantly better cardiac performance compared with Gel A (P < 0.05).
Contractility of isolated cardiac muscle. (A) Mean contraction force and (B) mean contraction time of isolated cardiac muscle from each experimental group of rats. Sham, sham-operated group; MI + PBS, phosphate-buffered saline (PBS)-treated myocardial infarction (MI) group; MI + Gel A, Gel A-treated MI group; MI + Gel B, Gel B-treated MI group; Half, mean time needed to reach peak contraction force during one contraction; Whole, mean time required for one complete contraction. aP < 0.05 versus sham-operated group; bP < 0.05 compared with MI + PBS group; cP < 0.05 versus MI + Gel A group; one-way analysis of variance and unpaired Student's t-test
The half and whole contraction times of isolated infarcted myocardium showed similar patterns (Fig. 2B). The mean half and whole times were significantly greater in the MI + PBS group than in the sham-operated group (P < 0.05 for both comparisons), but were significantly shortened by hydrogel (both Gel A and Gel B) injection compared with the MI + PBS group (P < 0.05 for both comparisons). The half and whole contraction times exhibited by the MI + Gel A and MI + Gel B groups were similar to the normal values demonstrated by the sham-operated group, but there was no significant difference between the MI + Gel A and MI + Gel B groups.
Western Blot Analysis
Western blot analysis of lysates from infarcted myocardium showed elevated amounts of collagen I, collagen III and TGF-β1 protein 90 days after infarction compared with sham-operated myocardium (Fig. 3). The Gel A and Gel B treatments both significantly reduced upregulation of the collagen I / collagen III ratio compared with the MI + PBS group (P < 0.05 for both comparisons), but there was no significant difference between the MI + Gel A and MI + Gel B groups. The hydrogel and PBS injection treatments did not alter the upregulation of TGF-β1 after infarction.
(A) Western blot analysis showing collagen I, collagen III and transforming growth factor (TGF)-β1 protein quantities 90 days after induction of myocardial infarction (MI) in rats. Quantification of the western blot yielded (B) amount of TGF-β1 protein (normalized to β-actin) and (C) amounts of collagen I and collagen III proteins (normalized to β-actin) expressed as collagen I / collagen III ratio. Sham, sham-operated group; MI + PBS, phosphate-buffered saline (PBS)-treated MI group; MI + Gel A, Gel A-treated MI group; MI + Gel B, Gel B-treated MI group; aP < 0.05 compared with sham-operated group; bP < 0.05 compared with MI + PBS group; oneway analysis of variance and unpaired Student's t-test
Infarct Size and Collagen Volume Fraction Analysis
Microscopic examination showed that the Gel A- and Gel B-treated hearts had significantly smaller infarcts than the PBS-treated hearts (P < 0.05 for both comparisons) (Fig. 4), but the infarct size was not significantly different between the MI + Gel A and MI + Gel B groups.
Haematoxylin and eosin (H&E)-stained sections and quantification of infarct size of the left ventricle of experimental groups 90 days after induction of myocardial infarction (MI) in rats. H&E-stained sections from (A) sham-operated group, (B) phosphate-buffered saline (PBS)-treated MI group, (C) Gel A-treated MI group and (D) Gel B-treated MI group were used to quantify (E) infarct size, calculated as described in the text, in each group. Black arrows in B, C and D show infarct area of the left ventricle. aP < 0.05 compared with sham-operated group; bP < 0.05 compared with MI + PBS group; one-way analysis of variance and unpaired Student's t-test
The collagen volume fraction of the infarct area was significantly higher in the MI + PBS group than in the sham-operated group (P < 0.05), whereas both the Gel A and Gel B treatments significantly reduced the value compared with the MI + PBS group (P < 0.05 for both comparisons) (Fig. 5). Gel A and Gel B showed no significant difference with respect to their ability to inhibit collagen hyperdeposition.
Masson's trichrome-stained sections and quantification of the collagen volume fraction of the infarct area in the experimental groups of rats 90 days after induction of myocardial infarction (MI) in rats. Masson's trichrome-stained sections for (A, B) sham-operated group, (C, D) phosphate-buffered saline (PBS)-treated MI group, (E, F) Gel A-treated MI group and (G, H) Gel B-treated MI group. (I) Quantification of collagen volume fraction. Black arrows in A, C, E and G show sham-operated or infarcted myocardium. aP < 0.05 versus sham-operated group; bP < 0.05 compared with MI + PBS group; one-way analysis of variance and unpaired Student's t-test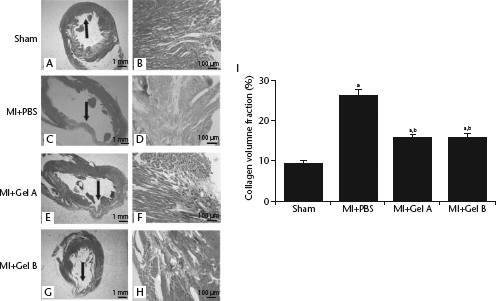
Neovasculature Formation
Anti-α-smooth muscle actin immunohistochemical staining was used to label microvessels in the infarct or sham-operated areas of the rat myocardium (Fig. 6). Infarcted hearts treated with Gel A or Gel B had significantly greater vessel density in the infarct areas than infarcted hearts treated with PBS (P < 0.05 for both comparisons). The Gel A- and Gel B-treated groups both exhibited a vessel density similar to that of the control sham-operated myocardium.
Neovasculature formation in sham-operated and infarct areas of myocardium 90 days after induction of myocardial infarction (MI) in rats, as identified by immunohistochemical staining for α-smooth muscle actin. (A) Sham-operated group; (B) phosphate-buffered saline (PBS)-treated MI group; (C) Gel A-treated MI group; (D) Gel B-treated MI group (scale bars, 100 μm). Immunohistochemical staining was used to quantify (E) blood vessel density per high-power field (HPF) in each group. Black arrows show neovasculature formed in sham-operated or infarcted myocardium. aP < 0.05 compared with sham-operated group; bP < 0.05 compared with MI + PBS group; one-way analysis of variance and unpaired Student's t-test
Discussion
Ventricular remodelling after MI plays an important role in progression to heart failure and is associated with worsened survival. 16 Previous findings demonstrated the effectiveness of biomaterials in cardiac protection,3,4,7– 10,12 but data on the possible role of biomaterials in MI treatment are still scarce.
Researchers have suggested that the ideal biomaterial for MI treatment would be biodegradable and biocompatible, and would need to restore the structure and composition of the native myocardium.4,17 Jugdutt observed that myocytes were supported by an extracellular matrix (ECM) consisting of an intricate, three-dimensional, organized collagen fibre network. 18 Scanning electron microscopy images showed that the three-dimensional honeycomb-like network of the PNIPAAm hydrogel was similar in structure to human heart tissue after removing cardiac myocytes. 19 Because the ECM plays a significant role in development, remodelling and signalling in the cardiovascular system, 20 designing and fabricating biomaterial scaffolds with a high level of biocompatibility to replace damaged ECM is an interesting potential treatment strategy for MI. The PNIPAAm hydrogel appears to be an ideal biomaterial for the structural replacement of damaged ECM following infarction because of the similarity of its three-dimensional structure to that of native ECM.
Although many methods of cell transplantation for MI treatment have been established, research has indicated that the therapeutic effects of implanted cells are affected by the harsh microenvironment of the infarct region. 21 Well-designed biomaterials not only provide good structural support but may also ameliorate the local environment and initiate a degree of functional repair of the damaged heart tissue. In the present study, intramyocardial injection of PNIPAAm hydrogel downregulated the elevation of the collagen I/collagen III ratio and inhibited collagen deposition, without affecting the expression of TGF-β1. Collagen is the predominant ECM protein, and the abnormal metabolism and deposition of collagen after MI is well known.18,22 The present results therefore indicate that a PNIPAAm hydrogel may regulate collagen metabolism by providing an exogenous supply of supplementary collagen, and we believe the ameliorative microenvironment of this hydrogel contributes to the repair of ischaemic myocytes.
A study in cardiomyocytes showed that biomaterials could influence cell function, such as contractility. 23 In addition, the use of highly controlled hyaluronic acid increased capillary density and normalized left ventricular function in rat hearts. 24 In the present study, there was a significant improvement in the electrically stimulated contraction force of isolated infarcted myocardium after hydrogel injection compared with PBS-treated infarcted myocardium. Although the precise mechanism underlying the improved contractility of isolated myocardium is unknown, some researchers suggest that neovascularization plays a role; 25 others consider that alignment of cells and increased gap junction proteins contribute to the regulation of synchronous contractions.26,27 In contrast with previous reports, the present study found that the contraction time of isolated myocardium was similar in the hydrogel-treated and sham-operated groups. Alcon et al. 28 point out that the continuous asynchronous contraction and relaxation of damaged myocardium could result in poor heart function. Thus, the enhancement of contractility observed in the present study was probably due to the improvement of myocardial systolic–diastolic co-ordination.
The present study also investigated the biodegradation times of the two preparations of PNIPAAm hydrogel. The results demonstrated distinct effects of the two preparations on cardiac protection, although both hydrogels showed good performance compared with the PBS-treated group. Biomaterial with a suitable degradation time is needed, as it is highly desirable that the rate of degeneration of the biomaterial should match the rate of tissue regeneration at the site of the defect. However, there is controversy about the appropriate degradation time of biomaterials.18,29,30 Contrary to previous findings, the present study did not find any apparent harmful effects from the hydrogel used in the myocardium during the 90 days following its injection. The present findings demonstrated that the effectiveness of cardiac protection provided by PNIPAAm hydrogel may peak at 4 weeks and subsequently continue. On this basis, it is our opinion that 4 – 8 weeks may be an appropriate degradation time for PNIPAAm hydrogel in the treatment of MI.
In 1993, Langer and Vacanti reported that tissue regeneration could be directed by artificially constructed biomaterials. 31 Since then, a growing body of evidence has shown that hydrogel can be used for numerous biomedical applications, especially for cardiac tissue engineering.8,32– 35 The high biocompatibility, biodegradability and porosity of PNIPAAm hydrogel (and the lack of immunogenic reactions to it) make it ideal for tissue engineering applications. However, more studies are needed to understand fully how biomaterials function during the MI process, in order to enable the development of improved design parameters with the goal of changing the functional performance of these materials in vivo. More studies are needed to assess the functional effects of the administration of engineered biomaterials in a more chronic model of MI.
Footnotes
Acknowledgements
We thank Junlian Xing, who provided technical assistance on behalf of the Physiology Laboratory of the Basic Medical College, Wuhan University of Science and Technology, Wuhan, China. This work was supported by the National Key Basic Research Programme of China (grant no. 2011CB606202), the National Natural Science Foundation of China (grant no. 81170307/H0222) and the Fundamental Research Funds for the Central Universities of China.
Conflicts of interest: The authors had no conflicts of interest to declare in relation to this article.
