Abstract
Objective:
This study investigated compensation for loss of the fumaryl - aceto acetate hydrolase gene (Fah) by gene therapy using the Sleeping Beauty transposon system (SBTS), in a hereditary tyrosinaemia type 1 (HT-1) mouse model (Fah-/-).
Methods:
Twenty Fah-/- study mice, five wild-type positive controls and five Fah-/- negative controls were included. All Fah-/- mice received 2-(2- nitro-4-trifluoro-methylbenzoyl)-1,3-cyclo - hexaedione (NTBC). Fah-/- study mice were randomly injected with one of two SBTS
Constructs:
Fah-SBTS (containing mouse Fah gene), or forkhead box M1b (FOXM1B)-Fah-SBTS (containing mouse Fah and human FOXM1B genes). Firefly luciferase-SBTS was injected as a trace marker. NTBC treatment stopped after construct injection; Fah-/- negative controls were kept healthy with continued NTBC. Mice were weighed daily; the luciferase signal was monitored by in vivo bioluminescence, and Fah and FOXM1B gene expression were evaluated.
Results:
The Fah gene integrated into the mouse chromosomes within 1 week of Fah-SBTS injection (mice survived without NTBC thereafter) and within 1 month of FOXM1B-Fah-SBTS injection (mice lost weight dramatically and needed additional NTBC).
Conclusion:
The shorter Fah gene had an advantage over the longer FOXM1B-Fah gene for stable integration into the host mouse chromosomes.
Keywords
Introduction
Hereditary tyrosinaemia type 1 (HT-1) is caused by a defective fumarylacetoacetate hydrolase (FAH) enzyme, which is the last enzyme of the tyrosine degradation pathway. 1 HT-1 is a common condition resulting from defects in tyrosine metabolism, 2 the main clinical features of which include liver and kidney dysfunction. Although symptoms of HT-1 are reduced in response to a phenylalanine- and tyrosine-restricted diet, many patients go on to develop hepatocellular carcinoma, for which liver transplantation is the only cure. 2,3 Currently, HT-1 is treated using a single daily dose of 2-(2-nitro-4-trifluoro-methylbenzoyl)-1,3-cyclohexaedione (NTBC), which improves metabolic control; as patients are diagnosed at birth, treatment is lifelong. 3
Inherited genetic diseases, especially gene-deficient diseases, can be corrected by gene therapy. 4 Currently available gene delivery systems include viral and nonviral types. Viral gene delivery systems are considered not to be as safe as nonviral systems, due to viral toxicity and unpredictable immune responses. Nonviral gene delivery systems include naked DNA, RNA and the Sleeping Beauty transposon system (SBTS). Nonspecificity and low efficacy are the major barriers for the naked DNA and RNA gene delivery methods.5,6 The SBTS allows gene incorporation into vertebrate chromosomes, delivers foreign genes more efficiently than the naked DNA molecules, and has a better safety profile than viral gene delivery systems. 7 Additionally, the SBTS is highly effective for transferring DNA < 5.6 kb. 8 Since the SBTS was first constructed in 1997, 9 it has been successfully used as a gene delivery tool in human hepatocytes, 10 primary T cells, 11 human embryonic stem cells 12 and human lung cells.13,14 The timing of foreign gene integration into vertebrate chromosomes has not previously been studied.
When normal liver function and mass are challenged in murine models, adult mouse hepatocytes enter the cell cycle to proliferate and replicate, in order to restore liver functional capacity.15–17 The forkhead box (FOX) genes belong to an extensive family of transcription factors18,19 and FOXM1B (also known as HFH-11B) is a proliferation-specific activator of transcription. 20 Reduced expression of Foxm1b contributes to the decline in hepatocyte proliferation in aged mice, 21 and overexpression of FOXM1B has been shown to repopulate chronically injured mouse liver cells efficiently, independent of donor age. 22 FOXM1B expression is induced at the G1/S transition phase of the cell cycle and continues throughout hepatic cell proliferation. 23
The present study investigated whether the SBTS system could efficiently deliver the mouse Fah gene into HT-1 mouse hepatocytes using two genetic constructs of different length, and whether overexpression of human FOXM1B contributed to hepatocyte repopulation.
Materials and methods
Mouse Strains and Animal Husbandry
Animal care and experiments were conducted in accordance with guidelines of the University of Minnesota Institutional Animal Care and Use Committee. Homozygous Fah–/– transgenic mice on a 129S4 background were generated to mimic HT-1 disease. Fah–/– mice (15 males and 15 females; 6 months of age, body weight ∼20 g) were chosen. Mice were kept in a pathogen-free environment under a 12-h light/12-h dark cycle, with free access to food and water. The drinking water was supplemented with 7.5 mg/ml NTBC (synthesized in the Chemical Department, University of Minnesota, Minneapolis, MN, USA) to keep the mice healthy. Five wild-type mice on a 139S4 background were used as controls (three males and two females; 6 months of age, body weight of ∼20 g). Control mice were kept under the same conditions as Fah
Sbts Constructs
The Fah-SBTS plasmid was a gift from Dr Xin Wang (University of Minnesota, Minneapolis, MN, USA); the Fah-FOXM1b-SBTS and firefly luciferase-SBTS plasmids were gifts from Dr Kirk Wangensteen (University of Minnesota). The structures of the plasmids used in the study are shown in Fig. 1. Fah-SBTS contained the mouse Fah gene (GenBank accession number: NM_010176; Pubmed 22209984); FOXM1B-Fah-SBTS contained the human FOXM1B gene (GenBank accession number: BC006192.2) and the mouse Fah gene. The total RNA used to derive the Fah gene was taken from normal mouse hepatocytes; the human FOXM1B gene was derived from the human HEK293 cell line. An internal ribosome entry site (IRES) sequence, supplied with the stock plasmid, was used to conjugate the two genes in the Fah-FOXM1b-SBTS plasmid.
Plasmid maps for the Sleeping Beauty transposon system (SBTS) constructs: the fumarylacetoacetate hydrolase (Fah)-SBTS construct contained the mouse Fah gene; the forkhead box M1b (FOXM1B)-Fah-SBTS contained the human FOXM1B gene conjugated with the mouse Fah gene using the internal ribosome entry site (IRES) sequence; the Luc-SBTS construct contained the firefly luciferase gene. Gene expression was driven by the CAGGS promoter (CaggP) in all three constructs, which all shared the same kanamycin resistance (KanR) and Sleeping Beauty 11 transposase (SB11) genes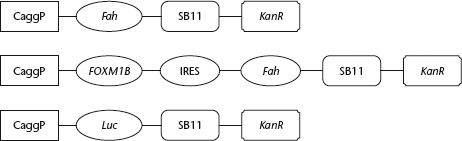
Injection of Constructs
The Fah–/– mice were put under general anaesthesia with 2,2,2-tribromoethanol (Avertin® Sigma-Aldrich, St Louis, MO, USA). There were equal copy numbers (copies/ml = 6.02 × 1023 × concentration, g/ml] / DNA length × 660) of each construct. The Fah-SBTS, FOXM1B-Fah-SBTS and luciferase-SBTS constructs (2 × 1015) were individually mixed with 2 ml saline. Solutions were administered by hydro -dynamic tail vein injection within 10 s into the mice. Each mouse was randomly chosen for specific injection of one of the different constructs by a technician blinded to the construct being used. Each mouse was then kept on a warm pad until it woke up. Immediately following the injection, NTBC was withdrawn from the diets of treated mice.
Mice were then weighed daily and their drinking water was supplemented with NTBC as required, if there was a reduction in body weight > 40% of the baseline level. One mouse from each study group was sacrificed (as described below) at 1 week and 1 month after construct injection, for reverse transcription–polymerase chain reaction (RT–PCR), Western blot and immuno -cytochemical analyses. After 6 months, the remaining mice (n = 13 per group) were sacrificed for collection of liver tissue and isolation of hepatocytes.
In VIVO Bioluminescence Imaging
To monitor incorporation of the gene constructs, bioluminescence imaging was conducted using luciferase-SBTS on days 1, 7, 14 and 21. All mice were scheduled to undergo this experiment. Mice were anaesthetized with 2,2,2-tribromoethanol, then injected with 100 μl of 28.5 mg/ml D-luciferin (Sigma-Aldrich) intraperitoneally as a trace marker. After 10 min, mice were exposed under 560 nm light for 1 min using the IVIS® 50 imaging system (Caliper Life Sciences, Hopkinton, MA, USA) to measure the luciferase optical density.
Liver Tissue Collection and Isolation of Hepatocytes
After 1 week, 1 month or 6 months, mice were put under general anaesthesia with 2,2,2-tribromoethanol and sacrificed. Midline and abdominal incisions were made to expose the liver. One liver lobe was collected and stored at –80 °C in a freezer; the remaining liver tissue was perfused as described below, to collect hepatocytes. An intravenous catheter was filled with Earle's Balanced Salt Solution (EBSS; Invitrogen, Carlsbad, CA, USA) with 0.5 mM ethylene glycol tetra-acetic acid and 10 mM Hepes (pH 7.4), without calcium and magnesium. A 27-gauge needle was used to annulate the portal vein from its lower part and the perfusion pump was then started at a speed of 5 ml/min. The inferior vena cava was cut to allow the blood to wash out. After 3 min, the solution was switched to EBSS with calcium, magnesium and 10 mM Hepes (pH 7.4) for a further 3 min, then switched to EBSS with calcium, magnesium, 10 mM Hepes (pH 7.4) and 0.5 mg/ml collagenase D (Sigma-Aldrich) for 8 – 15 min until the liver became tender. When the liver was completely digested, the catheter was removed and the gallbladder was excised. Tissue surrounding the liver was also excised and the entire liver was transferred to a petri dish with 5 ml of EBSS with calcium, magnesium, 10 mM Hepes (pH 7.4) and 0.5 mg/ml collagenase D. Under a cell culture hood, liver tissue was disrupted with forceps. The hepatocytes were collected by filtering through a nylon mesh into a sterile 50-ml tube containing 10 ml high-glucose Dulbecco's modified Eagle's medium (DMEM; Invitrogen) with 10% fetal bovine serum (FBS; Hyclone Laboratories, Logan, UT, USA). Cells were collected by centrifuging for 2 min at 250
In VITRO Bioluminescence Imaging
Hepatocytes were thawed and cultured with DMEM plus 10% FBS overnight at 37°C. After washing with 5 ml 1x phosphate-buffered saline (PBS; pH 7.4, Hyclone Laboratories), 2 ml of 28.5 mg/ml D-luciferin was added and cells were incubated for 10 min at room temperature. The luciferase content of the cells was measured using the IVIS® 50 imaging system under 560 nm light for 1 min, as described above.
RT–PCR Analysis of Foxm1b MRNA
Frozen liver tissues (1 g) from mice sacrificed at 1 week and 1 month were ground into a fine powder with liquid nitrogen and homogenized using a QIAshredder® kit (Qiagen, Valencia, CA, USA) for total RNA and protein extraction. Total RNA was extracted using a RNeasy® Micro Kit (Qiagen); RT was performed using a M-MLV RT Kit (Clontech Laboratories, Mountain View, CA, USA), both according to the manufacturers' instructions. PCR was carried out using a PCR master mix purchased from Promega (Madison, WI, USA) and the S1000 thermal cycler (Bio-Rad, Hercules, CA, USA) under the following conditions: preliminary denaturation at 95°C for 5 min, then 35 cycles of denaturation at 95 °C for 30 s, annealing at 58 °C for 30 s and elongation at 72 °C for 30 s, followed by a final elongation step at 72°C for 10 min and 4 °C for 10 min. The primer sequences were: human FOXM1B (forward) TGATTCAACA CAGGGA GAGG and (reverse) GACGTCTATATCTGAGGG AGAAGA; mouse albumin (forward) GACGTGTGTTGCCGATGAGTC and (reverse) CAGCCTCTGGCCTTTCAAATG. The PCR products were separated on 1.5% agarose gel and visualized using ethidium bromide under ultraviolet light. Each RT–PCR analysis was repeated at least three times.
Western Blotting Analysis of Foxm1b and FAH Protein
Homogenized liver tissue, prepared as described in the RT–PCR section above, was lysed in radioimmunoprecipitation assay lysis buffer (Millipore, Billerica, MA, USA) for 15 min at 4 °C. Samples were centrifuged at maximum speed for 15 min (10000
Immunocytochemical Analysis of Foxm1b and FAH Protein
Mouse hepatocytes, collected at 6 months after injection and stored in liquid nitrogen, were thawed and cultured on cell-culture chamber slides (with high-glucose DMEM with 10% FBS), overnight at 37 °C. The hepatocytes were fixed with ice-cold acetone for 30 min on ice, washed three times with cold 1x PBS (described previously) and incubated with 0.5% normal donkey serum (Santa Cruz Biotechnology) for 1 h at room temperature. The hepatocytes were then washed three times with 1x PBS and incubated with rabbit anti-FOXM1B antibody (1:200 dilution) or rabbit anti-Fah antibody (1:5000) for 3 h at room temperature, followed by incubation at room temperature with either cyanine 5 or fluorescein isothiocyanate-conjugated donkey antirabbit secondary antibody for 1 h at room temperature (1:250; Santa Cruz Biotechnology). After washing three times with 1x PBS, cells were fixed with fluorescent mounting medium (DAKO, Glostrup, Denmark). Samples treated in the same way but without primary antibody were used as negative controls. Cells were visualized using a Leica TCS SPE confocal microscopy device (Leica, Buffalo Grove, IL, US).
Statistical Analyses
Data were collected onto an Excel® spreadsheet. Differences between groups for all data were compared using an unpaired Student's t-test. A P-value of < 0.05 was considered to be statistically significant.
Results
The Fah–/– mice selected for the study were of similar age and body weight. Only mice that received hydrodynamic tail vein injections successfully without repeat injection were included: 10 mice each successfully received injection with the Fah-SBTS or FOXM1B-Fah-SBTS constructs. Five wild-type positive controls and five Fah–/– negative controls were also included. All mice successfully received the luciferase-SB TS construct.
Immediately after injection with Fah-SBTS, the body weight of the mice remained stable without additional NTBC feeding. These mice behaved normally and exhibited no adverse immune-response symptoms such as fever, itching or skin rash. The body weight of all Fah–/– mice injected with FOXM1B-Fah-SBTS dropped significantly (i.e. > 40% reduction) without NTBC feeding within the first week, therefore their diets were supplemented with NTBC to prevent their body weight from dropping further.
At 1 week after injection, Fah protein was detected in the sacrificed Fah-SBTS injected mouse but not in the sacrificed FOXM1B-Fah-SBTS injected mouse (Fig. 2A). The FOXM1B gene was detected in the FOXM1B-Fah-SBTS-injected mouse sacrificed after 1 month (Fig. 2B), and the remaining mice in this group ultimately became NTBC independent, suggesting that they began to express the exogenous Fah gene in their liver 1 month after injection of the construct.
Differences in timing of foreign fumarylacetoacetate hydrolase (Fah) and forkhead box M1b (FOXM1B) gene integration into the host chromosome. (A) Western blots of hepatocytes from: wild-type control mouse (wt); Fah-deficient mouse (Fah–/–); Fah–/– mouse injected with Fah-Sleeping Beauty transposon system (SBTS; Fah); Fah–/– mouse injected with FOXM1B-Fah-SBTS (FOXM1B). Hepatocytes were collected 1 week after injection. Images show that Fah protein was detected in the Fah-SBTS-injected mouse but not in the FOXM1B-Fah-SBTS mouse. β-actin protein was used as the loading control. (B) Reverse transcription–polymerase chain reaction in hepatocytes from wt, Fah–/– and FOXM1B groups, collected 1 month after injection. The human FOXM1B gene was detected in the FOXM1B-Fah-SBTS injected mouse; there was no human FOXM1B expression in the wt or Fah–/– mice. Mouse albumin (mAlbumin) was used as the positive control
The density of the luciferase signal reached its peak 1 week after construct injection, and decreased dramatically and significantly within 1 month (P < 0.05) (Fig. 3). Immunocytochemistry showed normal hepatocyte morphology 6 months after construct injection. Fah was present in hepatocyte cytoplasm (Fig. 4A), and FOXM1B was present in both the cell cytoplasm and nucleus, with the majority of staining observed in the nucleus (Fig. 4B). Using in vitro bioluminescence imaging, luciferase signals remained detectable in hepatocytes (Fig. 4C).
Firefly luciferase expression levels were analysed after injection of the luciferase-Sleeping Beauty transposon system construct into Fah–/– mice. (A) Representative results of in vivo bioluminescence imaging in a single mouse, after injection of 100 μl of 28.5 mg/ml D-luciferin, at 24 h, 1, 2 and 3 weeks after injection of the construct; the signal reached its peak level after 1 week and decreased to just traceable after 3 weeks. (B) Quantification of the luciferase signal over 3 weeks: data presened as mean ± SD; *P < 0.05 and **P < 0.005 versus 7 days; unpaired Student's t-test Localization of fumarylacetoacetate hydrolase (Fah), forkhead box M1b (FOXM1B), and luciferase in hepatocytes collected from Fah–/– mice 6 months after injection with Fah-Sleeping Beauty transposon system (SBTS), FOXM1B-Fah-SBTS or luciferase-SBTS. Immunocytochemisty showing: (A) Fah protein in hepatocytes from Fah-SBTS-injected mice liver; (B) FOXM1B protein in hepatocytes from FOXM1B-Fah-SBTS-injected mice liver (scale bars, 10 μm). Fah protein was localized in the cytoplasm and the FOXM1B protein in the cytoplasm and nucleus. (C) In vitro bioluminescence imaging showing the luciferase signal in the hepatocytes after incubating with the luciferase substrate 28.5 mg/ml D-luciferin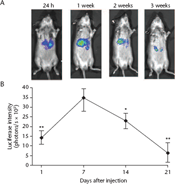

Discussion
A restricted phenylalanine and tyrosine diet helps to ameliorate the symptoms of HT-1, and lifelong daily single doses of NTBC are necessary to improve metabolic control. 3 Irrespective of dietary intervention and pharmaceutical treatment, hepatocellular carcinoma is often the outcome of HT-1.2,3 Gene therapy has the potential to compensate for the loss of the FAH gene in HT-1, thereby improving the patient's health-related quality of life.
The major barrier in gene therapy is finding an appropriate gene delivery system. The SBTS is a nonviral gene carrier that can deliver foreign genes into vertebrate chromosomes. 9 The SBTS was used in the present study to deliver the Fah gene successfully into Fah–/– mice (an animal model for HT-1). Genetically corrected mice were able to live on normal diets without NTBC, and behaved like normal mice without adverse immune-response symptoms such as fever, itching or skin rash. Additionally, the morphology of the hepatocytes collected at 6 months after injection of the constructs remained normal. This suggests that the SBTS is not only nontoxic for liver gene therapy but may also produce a permanent cure for HT-1. The toxicity of the SBTS may vary in larger animals such as cats, dogs or humans, therefore further experiments using large-animal models are needed. This treatment method might be applicable in future as gene therapy for HT-1, and for other genetically deficient diseases. 24
The FOXM1B-IRES-Fah-SBTS construct was designed to evaluate whether overexpression of FOXM1B and Fah improved hepatocyte repopulation in the same Fah–/– mouse hepatocytes. Fah–/– mice that received the FOXM1B-IRES-Fah-SBTS construct needed NTBC treatment for 3 weeks longer than those that received the Fah-SBTS construct. The length of the FOXM1B-IRES-Fah gene is 4.5 kb, which should be delivered into the host chromosomes successfully. The luciferase signal decreased significantly 1 week after injection, suggesting that temporarily integrated genes were excluded from the host chromosomes. These two foreign genes were both driven by the CAGGS promoter. This construct design excludes the possibility of different timings of foreign gene insertion related to varied promoter potency. Thus, it was concluded that the shorter gene had an advantage over the longer gene in achieving stable integration into the host chromosomes by the SBTS.
The FOXM1B gene is expressed in actively dividing cells and is critical for cell-cycle progression; 25 it is dramatically down -regulated in adult tissue, but can be reactivated in response to organ injury and in a variety of cancers. Overexpression of FOXM1B contributes to the hepatocyte proliferation in regenerating liver of ageing mice and chronic injured liver.21,22 NTBC was withdrawn from the diets of Fah–/– mice after SBTS injection in the present study. Under this challenge, it was thought that hepatocytes with FOXM1B-IRES-Fah gene expression would proliferate faster than those with only Fah gene expression, and that Fah–/– mice with FOXM1B-IRES-Fah gene expression would recover faster than those with Fah gene expression. This phenomenon was not observed, however. The Fah–/– mice used in the present study were 6 months old; future studies on ageing mice are planned.
In conclusion, the present study demonstrated that the SBTS is a well-tolerated and efficient gene carrier for Fah, suitable for gene therapy of the Fah–/– mouse model for HT-1. The shorter gene had an advantage over the longer gene in terms of stable chromosome integration.
Footnotes
Acknowledgements
Dr Xiaojing Pan thanks Ms Genya Gekker and Mr Paul Marker at the University of Minnesota for helping with confocal microscopy.
The authors had no conflicts of interest to declare in relation to this article.
