Abstract
Background
Neuroprotective agents are needed to reduce cerebral damage during surgical or neurointerventional procedures including stroke patients.
Purpose
To evaluate if thiopental can be used as a neuroprotective agent when injected intra-arterially in a transient ischemia model.
Material and Methods
In total, 24 rabbits were studied as four groups of six animals. Group 1 served as the control group. In group 2, transient ischemia was obtained by intracarotid administration of degradable starch microspheres (DSM). Group 3 was administered thiopental intra-arterially via the carotid artery. Group 4 (experimental group) received both thiopental and DSM intra-arterially. DSM and thiopental were administered through a microcatheter placed into the common carotid artery via the central ear artery access. After sacrifice, apoptotic cells in the cerebral tissues of the animals were evaluated in H&E and TUNEL stained slides.
Results
There was a significant increase in the number of apoptotic glial or neuronal cells in group 2 compared to the control group and group 3. The mean number of both the apoptotic neuronal cells (6.8 ± 2.1 vs. 2.5 ± 1.3, P < 0.001) and the apoptotic glial cells (9.4 ± 3.1 vs. 4.6 ± 1.6, P < 0.001) were higher in group 2 compared to group 4. In addition, a higher level of neurological improvement was observed in group 4 compared to group 2 based on neurological assessment score.
Conclusion
The intra-arterial administration of thiopental has a protective effect on both glial and neuronal cells during temporary cerebral ischemia in low doses.
Introduction
Neuroprotection is desirable for certain neurosurgical operations in which temporary arterial clipping is employed and also for neuroendovascular procedures in which thrombectomy or balloon occlusion is applied, such as endovascular procedures for acute stroke. Among the drugs used as neuroprotective agents, systemically administered barbiturates have been utilized in cerebral ischemia models due to their brain-protective effects in cerebral ischemia (1). The effects of thiopental, a barbiturate derivative, in cerebral ischemia have been well-demonstrated (2,3) and include slowing of brain metabolism, reduction of blood flow to the brain, and reduction in intracranial pressure (3). However, when used in systemic doses, thiopental-related complications such as arterial hypotension, hypokalemia, respiratory complications, infections, and hepatic or renal dysfunctions may lead to morbidity and mortality (4).
Comprising amylopectin and amylose, degradable starch microspheres (DSM) are hydrophilic granular molecules that are used as embolic agents for the locoregional transarterial chemoembolization of tumors of the head and neck, the lung, and more commonly the liver (5,6). The mean half-life of DSM in plasma is about 35–40 min (7). Recently, its use as a titratable agent for inducing reversible cerebral ischemia was described in an animal model (8). The aims of the present study were to demonstrate if an intra-arterial injection of barbiturates, namely thiopental, has the potential to delay cerebral ischemic changes in reversible cerebral ischemia and to evaluate its potential for utilization in surgical and neuroendovascular procedures.
Material and Methods
The study was approved by the interventional ethical board of Hacettepe University.
A total of 24 male New Zealand rabbits weighing 2500–3500 g were randomly divided into four groups. The animals underwent a carotid angiogram via the central ear artery, and DSM was used with or without thiopental for inducing transient ischemia. The subjects were kept alive for 24 h after the procedure and then sacrificed to collect samples for histopathological analysis. The subjects were divided into the following groups:
Group 1 (control group, n = 6): a microcatheter was inserted through the central ear artery and then into the common carotid artery. Normal saline and non-ionic contrast agent were injected into the right common carotid artery (CCA);
Group 2 (ischemia with DSM group, n = 6): a microcatheter was inserted into the right common carotid artery as in the previous group and a non-ionic contrast agent diluted with saline was injected together with DSM. For this procedure, starch particles were injected in a pulsatile fashion under fluoroscopic control until flow stagnation was achieved;
Group 3 (intracarotid thiopental group, n = 6): thiopental was mixed with diluted non-ionic contrast agent and using the same method of catheterization, it was injected into the right CCA. A total of 12.3 mg thiopental was administered to the subjects, which is the known required dose to achieve burst suppression on the electroencephalogram (EEG) (9);
Group 4 (experimental group, ischemia with DSM + intracarotid thiopental group, n = 6): the right CCA was catheterized as described above. Intracarotid thiopental was administered as in group 3 and then DSM was given in a similar fashion as used in group 2.
Transarterial procedure
To provide anesthesia and analgesia, 50 mg/kg ketamine hydrochloride and 5 mg/kg xylazine were intramuscularly injected into the rabbits that were fasted overnight. After anesthesia, the subjects were left to breathe on their own. In case of insufficient analgesia, additional doses of ketamine and xylazine were applied. The right ear was wiped externally with 70% alcohol to induce vasodilation. The central ear artery was punctured with a 24-gauge needle; a 0.014-inch micro-guidewire (Synchro; Stryker, Kalamazoo, MI, USA) was navigated into the central ear artery retrogradely and then an SL-10 microcatheter (Synchro; Stryker, Kalamazoo, MI, USA) was exchanged with the IV catheter. Then 0.2 mg of nimodipine diluted in normal saline was injected through the microcatheter. The microcatheter was navigated retrogradely into the right common carotid artery over the guidewire. After entering the right common carotid artery, 50% non-ionic iodinated contrast agent diluted in normal saline was administered with 2.5-mL luer-lock syringes for cerebral angiography. Thiopental was administered in groups 3 and 4 after the initial angiogram. If DSM were used, they were injected in a 25% contrast medium, under fluoroscopy with intermittent, pulsed, gentle injections over 3–4 min. Then an angiogram was obtained to determine diminished distal perfusion and the catheter was removed, hemostasis was maintained by manual compression over the ear, and the animals were placed in cages to be monitored for 24 h (Fig. 1).
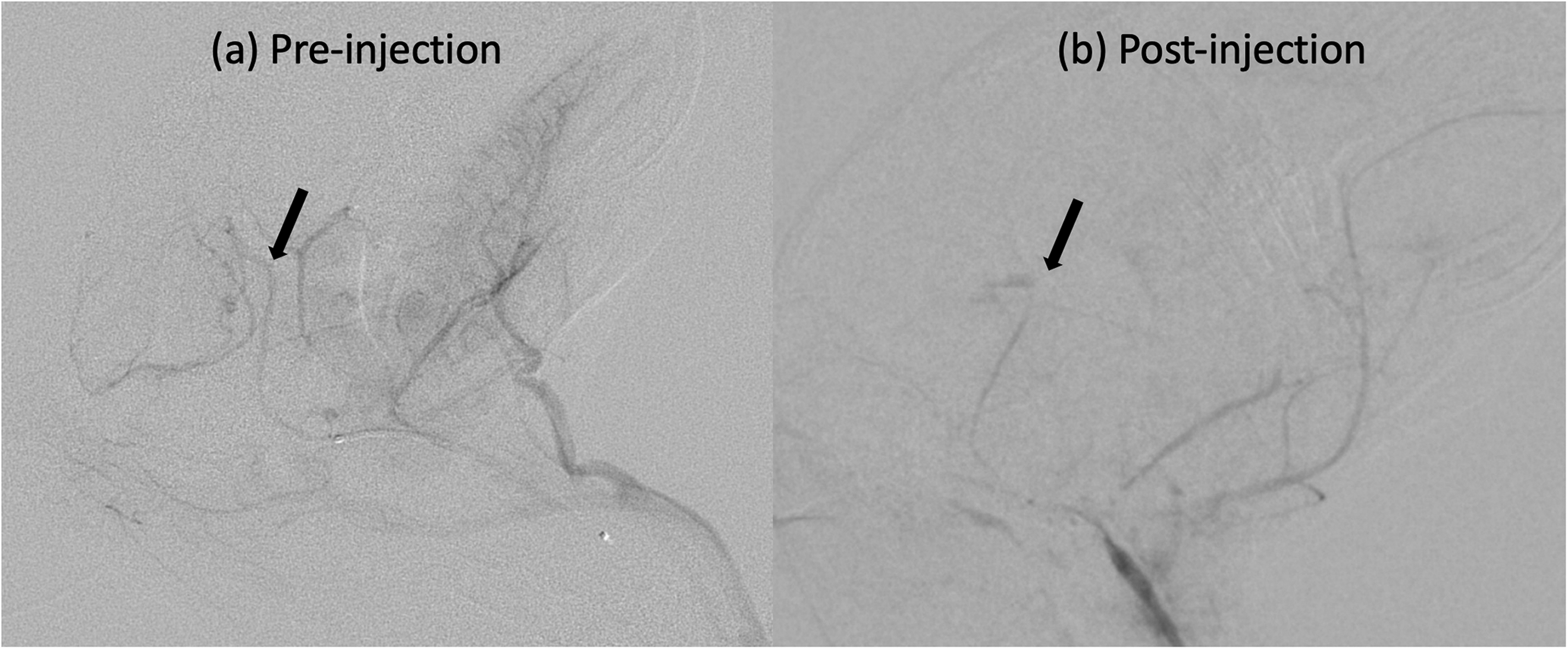
Angiographic images (a) before and (b) after embolization with starch particles. The arrow indicates the intracranial part of internal carotid artery, and pruned appearance after embolization with starch particles.
The sacrifice of the subjects and removal of the tissues
Twenty-four hours after the transarterial embolization procedure, the neurological assessment score was calculated as described previously (Table 1) (10), and an overdose of xylazine (10 mg/kg) was given. After that, perfusion fixation was performed by administering 4% paraformaldehyde (in 0.1 mol/L phosphate buffer, pH: 7.4) solution at a constant rate into the left ventricle of the subjects until their neck and extremities were stiff, and the subjects were sacrificed. The brains of the sacrificed subjects were removed after craniectomy and postfixed by immersion in 10% paraformaldehyde at room temperature for 48 h.
Neurological assessment score.
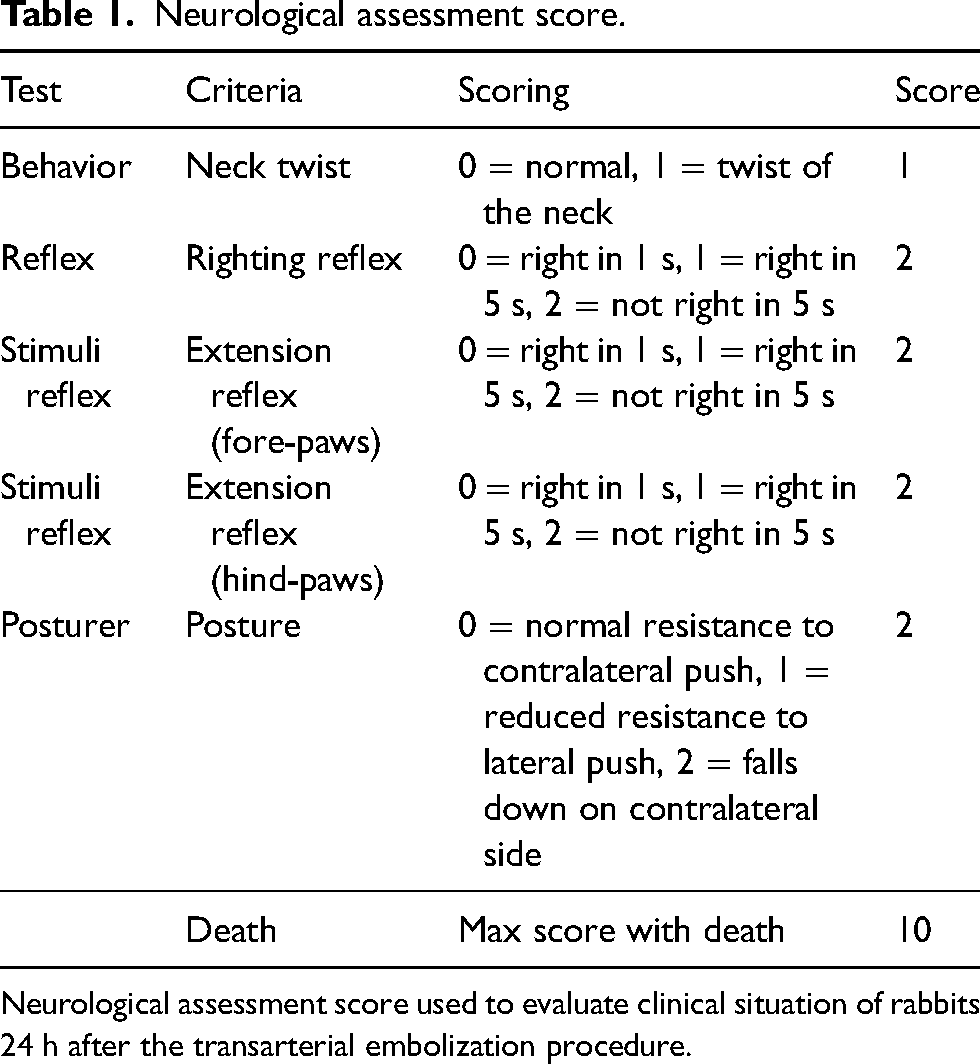
Neurological assessment score used to evaluate clinical situation of rabbits 24 h after the transarterial embolization procedure.
Histopathologic evaluation
The histopathological evaluation was carried out in a blinded fashion from the group of animals. The samples obtained by 5-µm serial sections were stained with hematoxylin and eosin (H&E) and a terminal deoxynucleotidyl transferase-mediated deoxyuridine triphosphate (TUNEL) method and evaluated under a DM6000B light microscope. The detection of apoptosis in the cerebral cortex region of the brain was performed using the Cell Death Detection kit by enzymatically labeling DNA strand breaks with TUNEL. All cells were randomly counted in five pre-described sections oriented anteroposteriorly within the brain, with a separate histopathologic slide obtained from each of these sections. Each of these five slides were used for evaluating cells at three different microscopic fields at 40× magnification. The numbers of glia and neurons were determined from the total number counted. The ones with a TUNEL-positive stain were noted. Pyknotic neurons with basophilic cytoplasm were counted in three areas at 40× microscopic magnification by two histologists blinded to the groups, and the average number of apoptotic cells in three evaluation fields were calculated for each area. Images were taken using a microscope.
Single- and double-chain immunoreactivity associated with apoptosis was evaluated in the neurons and glia in the cerebral cortices of all subjects. Apoptotic cell ratios were calculated as the mean percentage of apoptotic cells (number of positively labeled apoptotic cells/total number of cells), and the extent of apoptosis was used for the calculation.
Statistical analysis
All statistical analyses were performed using R statistical and computing software, version 3.5.0 (http://www.r-project.org). Results were presented as the median (interquartile range) or percentage. Normality distribution of scale variables was checked using the Shapiro–Wilk test, and the independent samples were compared with the Kruskal–Wallis H test. The Mann–Whitney U-test was used as a post-hoc test in pairwise comparisons of variables with significant results. Two-sided P values <0.05 were considered statistically significant.
Results
There was no significant difference between the groups in the number of observed normal glial or neuronal cells in any area (Supplemental Figs. 1 and 2). The histopathologic and immunohistochemical evaluations identified apoptotic neurons and glial cells close to the blood vessels, which was consistent with ischemic damage. The chromatin distribution in their nuclei was marginally located.
The mean number of apoptotic glial cells in all areas was 9.4 ± 3.1 in the ischemia group, 4.6 ± 1.6 in the experiment group, 2.6 ± 1.7 in the control group, and 1.9 ± 0.9 in the thiopental group. Except for the control and thiopental groups, comparisons of the total number of apoptotic glial cells in all areas showed a significant difference between all groups compared (Fig. 2). When each area was evaluated separately, there was always a significant increase in the number of apoptotic glial cells in the ischemia group compared to the control and thiopental groups (Fig. 3). Injection of thiopental in the experimental group resulted in a significant decrease in apoptotic glial cells in areas 2, 3, and 4, while the difference was marginally non-significant in areas 1 (P = 0.060) and 5 (P = 0.089).
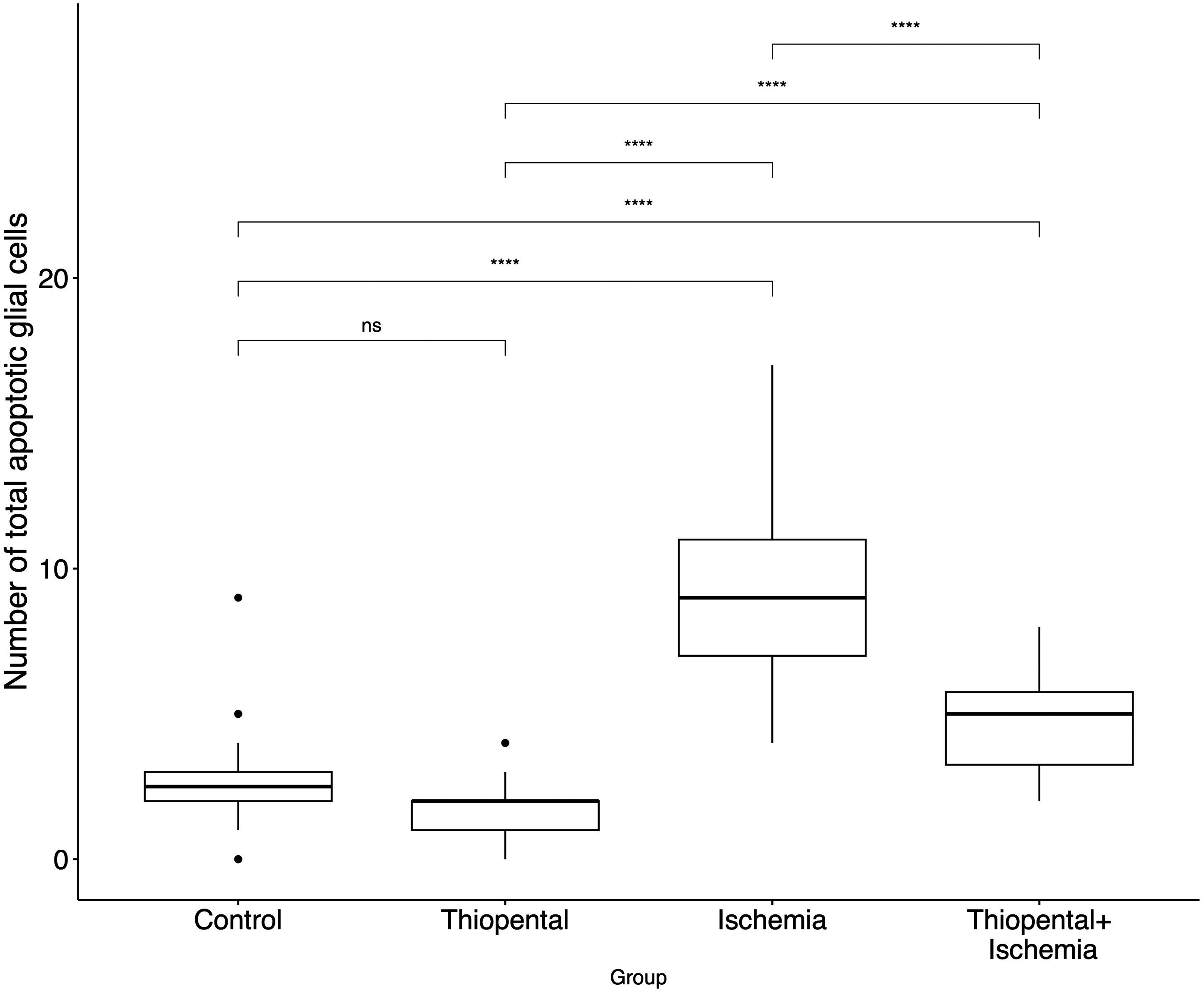
Comparison of total number of apoptotic glial cells in each group. *P ≤ 0.05; **P ≤ 0.01; ***P ≤ 0.001; P ≤ 0.0001. ns, non-significant.
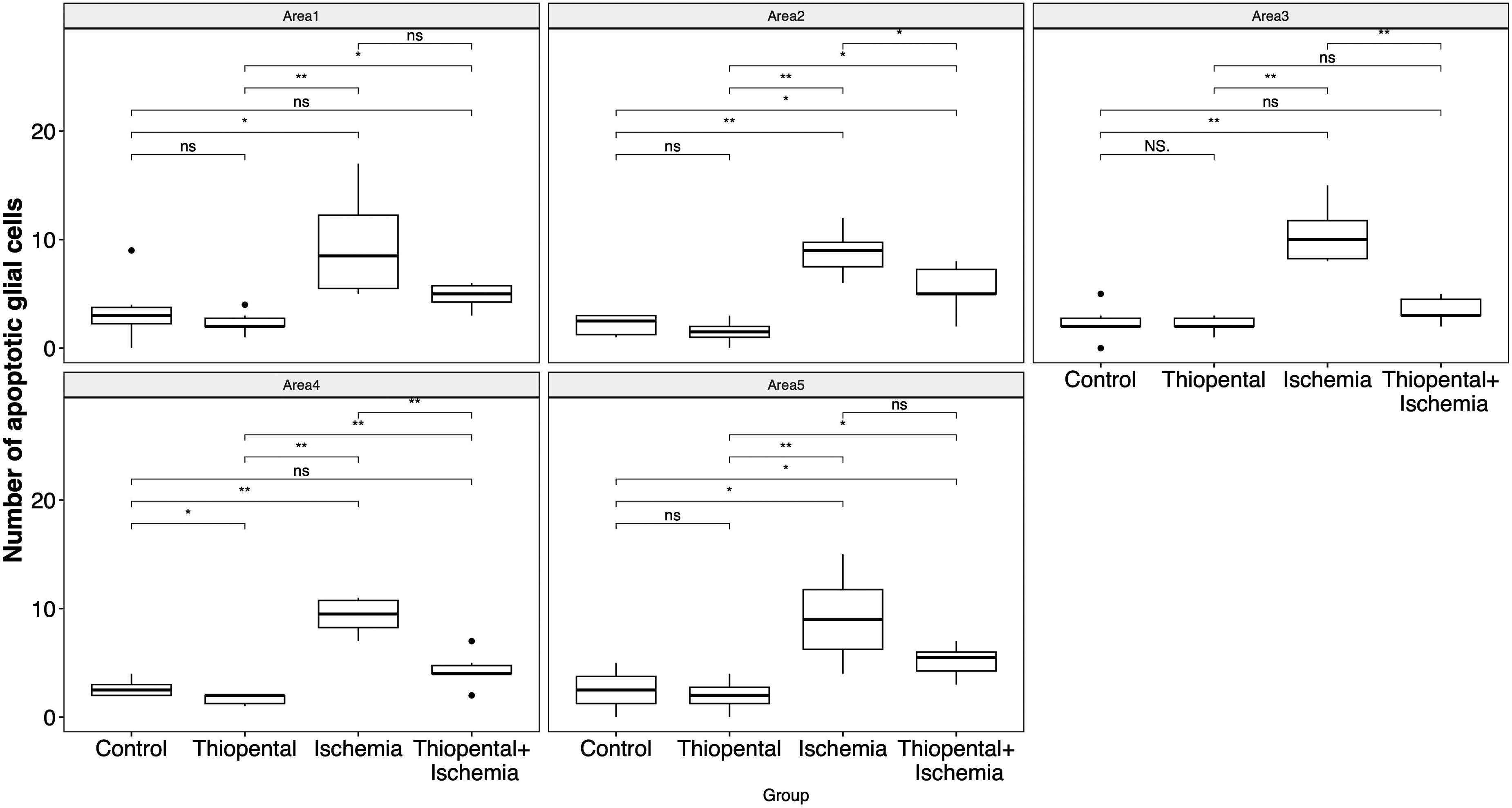
Comparison of apoptotic glial cells in each area between groups. *P ≤ 0.05; **P ≤ 0.01; ***P ≤ 0.001; P ≤ 0.0001. ns, non-significant.
Similarly, the ratio of apoptotic glial cells to all glial cells was significantly higher in the ischemia group compared to the three remaining groups (Fig. 4). Evaluation of each area separately showed that the experimental group had a lower ratio of apoptosis compared to the ischemia group in all areas (Supplemental Fig. 3).
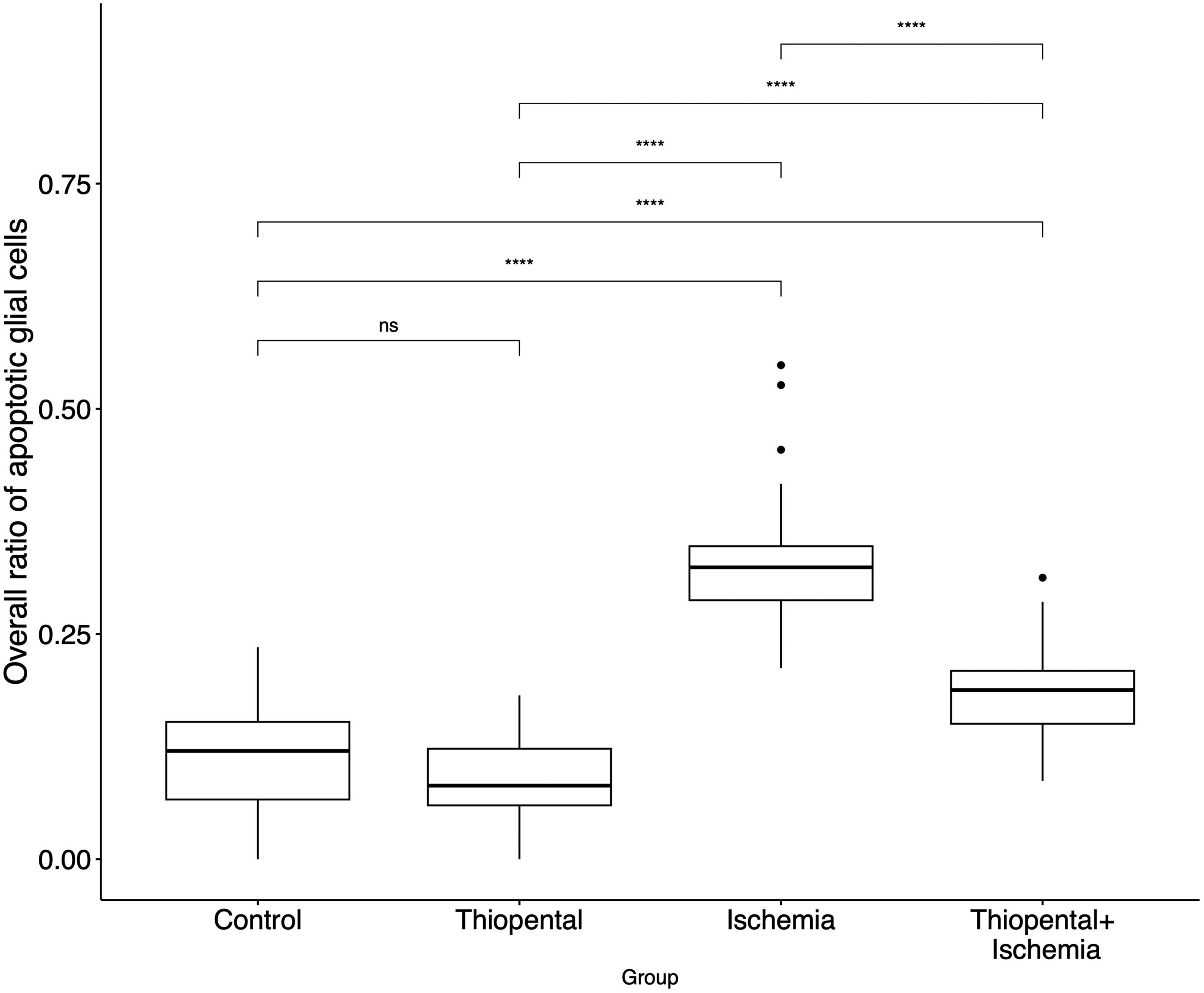
Comparison of ratio of apoptotic glial cells in each group. ns: *P ≤ 0.05; **P ≤ 0.01; ***P ≤ 0.001; P ≤ 0.0001. ns, non-significant.
The mean number of apoptotic neuronal cells was 6.8 ± 2.1 in the ischemia group, 2.5 ± 1.3 in the experiment group, 0.4 ± 0.6 in the control group, and 0.7 ± 0.7 in the thiopental group. The total number of apoptotic neuronal cells and that of apoptotic glial cells were significantly higher in the ischemia group among all groups, and in the experimental group compared to the control and thiopental groups (Fig. 5, Supplemental Fig. 4). The ratio of apoptotic neuronal cells to all neuronal cells was also significantly higher in the ischemia group in all assessed areas (Fig. 6, Supplemental Fig. 5).
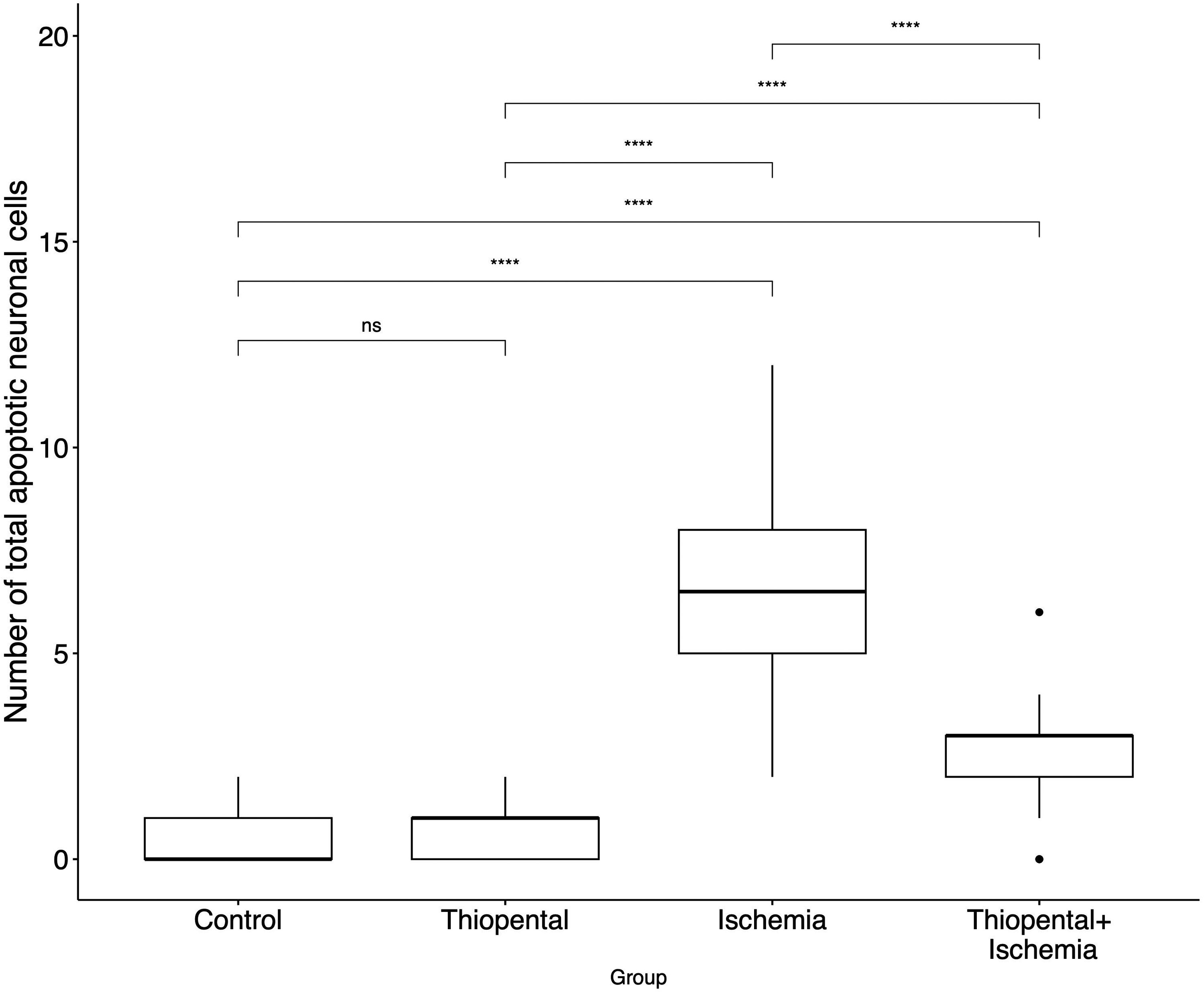
Comparison of total number of apoptotic neuronal cells in each group. *P ≤ 0.05; **P ≤ 0.01; ***P ≤ 0.001; P ≤ 0.0001. ns, non-significant.
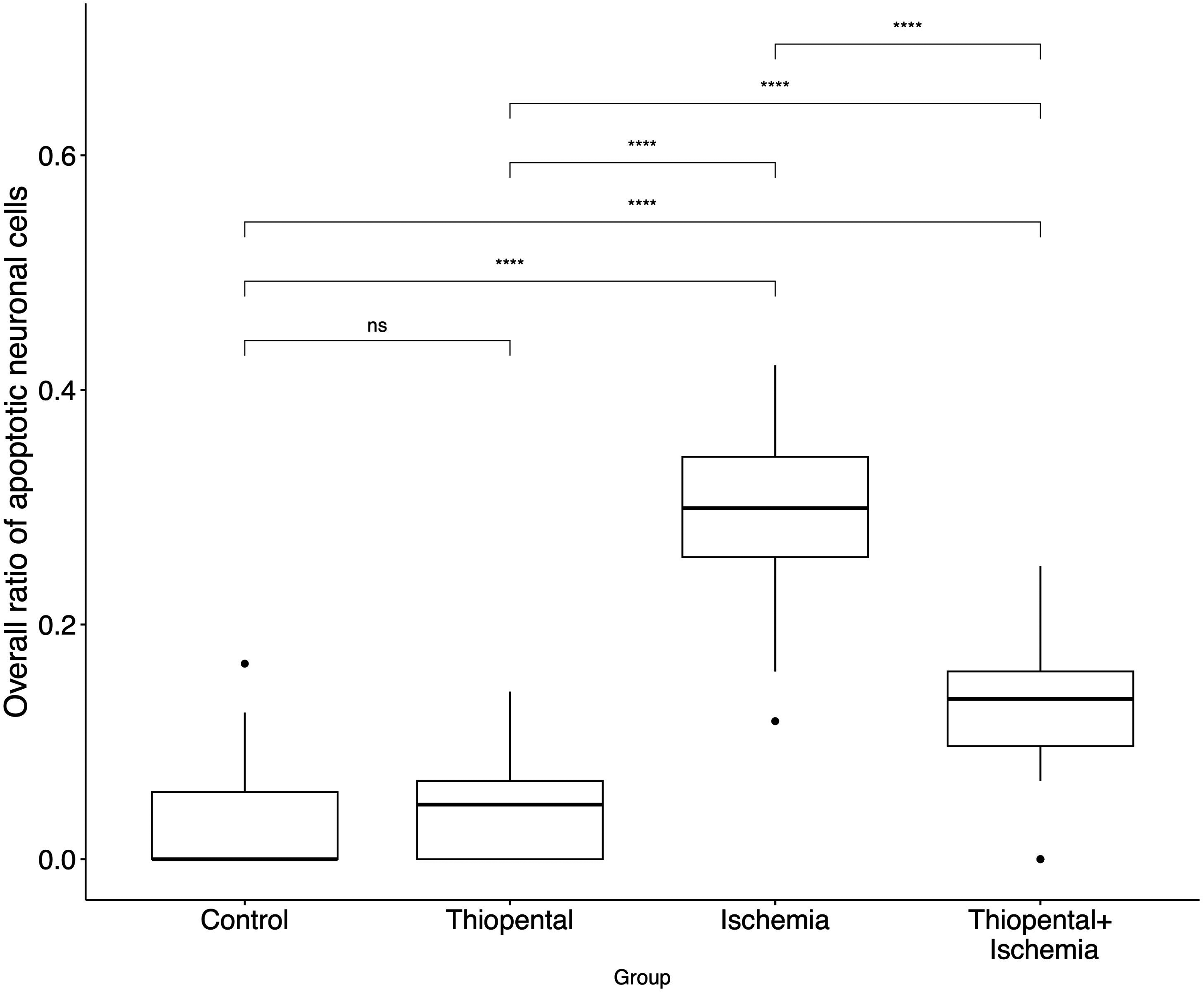
Comparison of ratio of apoptotic neuronal cells in each group. *P ≤ 0.05; **P ≤ 0.01; ***P ≤ 0.001; P ≤ 0.0001. ns, non-significant.
In terms of clinical evaluation with the neurological scoring system, complete recovery without any neurological deficit was achieved in only the thiopental and control groups. In the ischemia group, three animals were completely hemiplegic, two were severely hemiparetic, and one recovered without sequelae. The mean neurological assessment score (NAS) in this group was 6.8. In the experimental group, one animal was hemiplegic, four were hemiparetic, and two showed full recovery. The mean NAS of the animals in this group was 5.3.
Discussion
In this study, we verified that transient cerebral ischemia could be induced by using DSM with the selective transarterial microcatheterization method. In addition, we showed the favorable effect of thiopental during reversible cerebral ischemia.
Several studies have described the neuroprotective effect of thiopental in a variety of clinical scenarios (11,12); however, the practice of intravenous thiopental administration has been abandoned because the total dose that would cause neuronal suppression is too high (13). On the other hand, intracarotid injection of thiopental resulted in similar results with about 18% of the intravenous dose (9). In the literature, intracarotid thiopental injection has been reported (9,14), yet the results of intra-arterial thiopental administration in the setting of reversible perfusion deficit still remains unknown. Currently, none of the agents that have been investigated in patients with acute stroke demonstrated an improved outcome (15,16). In a similar fashion, no drug has been shown to be effective as a neuroprotective medication for the salvageable tissue in the endovascular treatment of acute stroke. In the present study using DSM to induce reversible ischemia, both the control group (no particle embolization) and the thiopental group (animals with thiopental administration only) recovered completely without sequelae after anesthesia. Complete obstruction and hemiplegia were observed in 50% of the subjects with induced ischemia. On the other hand, only one rabbit in the experiment groups (embolization and thiopental) had total hemiplegia. These findings suggest the protective effect of intracarotid administration of thiopental on the neurons and glia when the ischemic insult is temporary. These findings were also confirmed by histopathologic evaluation. A higher number of apoptotic cells were observed in the ischemia group as compared to the control or thiopental groups. The areas affected by ischemia were fewer in the experimental group than in the ischemia group.
As thiopental was administered intra-arterially, sufficient drug effect could be achieved at low doses (around 12.3 mg). In contrast to the systemic administration of thiopental, intra-arterial administration resulted in no major adverse events. After the administration of thiopental, subjects were left to breathe on their own. Although there was a decrease in respiratory rate and shallower breathing when thiopental was injected, the oxygen saturation of the subjects was stable. This situation underlines the potential of intra-arterial usage to overcome dose-limiting toxicities after systemic application.
To date, a few experimental studies utilized DSM or similar starch-based embolic agents to induce ischemia in the brain. The increased number of apoptotic cells and worse neurological outcomes in the ischemia group compared to the control group in our study confirmed the applicability of this model. Compared with the other experimental ischemia models, such as arterial ligation or insertion of a foreign body into the arterial lumen, the DSM model does not cause arterial wall injury. In the other cerebral ischemia models in which subjects are exposed to ischemia by surgically opening a window from the cranium, additional parenchymal damage and increased intracranial pressure may occur during the procedure (17), overestimating the effect of ischemia. Finally, in the ischemic model utilizing non-resuscitated cardiac arrest, the major disadvantage is the inability to provide circulation after ischemia (18). In the ischemic model created using DSM, vascular access is obtained either at the inguinal region or the ear, and ischemia is demonstrated angiographically by the induction of stasis of the blood. In addition, precise embolization of the microvascular bed is induced, hence the pathophysiology of ischemia can be directly studied. On top of that, the subjects are not exposed to major surgical stress.
The present study has some limitations. The most important one is the presence of collateral blood flow via the circle of Willis. The topography of the infarct area and therefore on the histopathologic results may potentially be affected by this flow, leading to underestimation of cellular damage. Due to the tiny sizes of both the posterior and anterior communicating arteries, it is not technically possible to visualize the exact distribution of DSM or thiopental to non-target territories or detect the washout of DSM or thiopental from the target territory secondary to collateral flow. However, our results suggest that intra-arterial administration of thiopental can prevent, or at least reduce, permanent brain damage in reversible ischemia. This was an initial study in line with STAIR guidelines (19,20). Further human studies need to be conducted to determine whether selective intra-arterial thiopental injection increases the tolerance of cerebral tissue to ischemia when incorporated into mechanical thrombectomy for acute stroke or into hybrid open/endovascular procedures when temporary arterial clipping is used.
In conclusion, when administered intra-arterially, thiopental had a protective effect on both glial cells and neurons during a brief episode of cerebral ischemia as evidenced by a lower rate of apoptosis. The reduction in cerebral edema in the animals receiving intra-arterial thiopental lends further support to our histopathologic data. In this first in vivo study that investigated the effects of the administration of intra-arterial thiopental on cerebral tissue during reversible ischemia, it shows promise as a neuroprotective strategy during endovascular and cerebrovascular surgery. The safety parameters regarding the amount, concentration, and rate of intra-arterial thiopental administration need to be tested in further studies for each of these clinical conditions.
Supplemental Material
sj-jpg-1-acr-10.1177_02841851231206503 - Supplemental material for In vivo study of the utility of selective intra-arterial injection of thiopental for neuroprotection in reversible cerebral ischemia
Supplemental material, sj-jpg-1-acr-10.1177_02841851231206503 for In vivo study of the utility of selective intra-arterial injection of thiopental for neuroprotection in reversible cerebral ischemia by Murat Gökten, Osman öcal, Can Sezer, Selim Zırh, Sevda Muftuoglu, Elif öcal, Burcak Bilginer and Anıl Arat in Acta Radiologica
Supplemental Material
sj-jpg-2-acr-10.1177_02841851231206503 - Supplemental material for In vivo study of the utility of selective intra-arterial injection of thiopental for neuroprotection in reversible cerebral ischemia
Supplemental material, sj-jpg-2-acr-10.1177_02841851231206503 for In vivo study of the utility of selective intra-arterial injection of thiopental for neuroprotection in reversible cerebral ischemia by Murat Gökten, Osman öcal, Can Sezer, Selim Zırh, Sevda Muftuoglu, Elif öcal, Burcak Bilginer and Anıl Arat in Acta Radiologica
Supplemental Material
sj-jpg-3-acr-10.1177_02841851231206503 - Supplemental material for In vivo study of the utility of selective intra-arterial injection of thiopental for neuroprotection in reversible cerebral ischemia
Supplemental material, sj-jpg-3-acr-10.1177_02841851231206503 for In vivo study of the utility of selective intra-arterial injection of thiopental for neuroprotection in reversible cerebral ischemia by Murat Gökten, Osman öcal, Can Sezer, Selim Zırh, Sevda Muftuoglu, Elif öcal, Burcak Bilginer and Anıl Arat in Acta Radiologica
Supplemental Material
sj-jpg-4-acr-10.1177_02841851231206503 - Supplemental material for In vivo study of the utility of selective intra-arterial injection of thiopental for neuroprotection in reversible cerebral ischemia
Supplemental material, sj-jpg-4-acr-10.1177_02841851231206503 for In vivo study of the utility of selective intra-arterial injection of thiopental for neuroprotection in reversible cerebral ischemia by Murat Gökten, Osman öcal, Can Sezer, Selim Zırh, Sevda Muftuoglu, Elif öcal, Burcak Bilginer and Anıl Arat in Acta Radiologica
Supplemental Material
sj-jpg-5-acr-10.1177_02841851231206503 - Supplemental material for In vivo study of the utility of selective intra-arterial injection of thiopental for neuroprotection in reversible cerebral ischemia
Supplemental material, sj-jpg-5-acr-10.1177_02841851231206503 for In vivo study of the utility of selective intra-arterial injection of thiopental for neuroprotection in reversible cerebral ischemia by Murat Gökten, Osman öcal, Can Sezer, Selim Zırh, Sevda Muftuoglu, Elif öcal, Burcak Bilginer and Anıl Arat in Acta Radiologica
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by Hacettepe University.
Data availability
The data that support the findings of this study are not publicly available but are available from the corresponding author on reasonable request.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
