Abstract
Background
Endothelial TGF-β1 signaling is a primary driver of atherosclerosis-associated vascular inflammation. Targeted imaging and inhibition of the expression of TGF-β1 may reduce the atherosclerotic vessel wall inflammation and stop the progression of atherosclerotic plaque.
Purpose
To investigate the possibility of the anti-TGF-β1-ultrasmall superparamagnetic iron oxide (USPIO) specific probe as an imaging marker for the expression of TGF-β1 in ApoE–/– mice atherosclerosis detected with 7.0-T magnetic resonance imaging (MRI).
Material and Methods
Here, 70 ApoE–/– mice on a high-fat diet served as the experimental group and 30 C57BL/6 mice on a normal diet served as the control group. The morphology of plaques was viewed by H&E staining, and the expression and distribution of TNC and TGF-β1 were detected by immunohistochemical staining. Another 40 mice in the experimental group were classified into a targeted group, which was administrated an anti-TGF-β1-USPIO probe, and the pure group, which was injected with pure USPIO.
Results
The 7.0-T MRI showed that the relative signal intensity (rSI) changes of the targeted group decreased more than those of the pure group (−19.34 ± 0.68% vs. −5.61 ± 0.57%; P < 0.05). Histopathological analyses demonstrated expression of TGF-β1 in atherosclerotic plaque formation progression from 10 to 28 weeks. The MR images of the expression of TGF-β1 in atherosclerosis correlated well with the pathological progression of atherosclerotic plaque formation.
Conclusions
Anti-TGF-β1-USPIO could provide a useful molecular imaging tool for detecting and monitoring the expression of TGF-β1 in atherosclerotic plaques by MRI.
Keywords
Introduction
Cardiovascular and cerebrovascular diseases are a major health concern worldwide and atherosclerosis is a common pathological foundation (1). Most major cardiovascular diseases may be ascribed to rupture of vulnerable atherosclerotic plaque and secondary thrombosis formation. The occurrence of vulnerable plaque is a complicated biological process, including endothelial dysfunction, macrophage infiltration, inflammatory factors expression, etc. (2). How to dynamically monitor changes of biomarkers for vulnerable plaque remains a crucial problem.
TGF-β are a large family of signaling molecules with common biological characteristics. They can be generated by a variety of cells in the body and are involved in many biological processes including embryo development, cell growth and differentiation, extracellular matrix (ECM) synthesis, inflammation, wound repair, and immunity (3,4). Transforming growth factor-β1 (TGF-β1) is a multifunctional peptide that stimulates cell proliferation, migration, and ECM deposition, thus contributing to the vascular remodeling of atherosclerosis (5,6). Several studies have found that the imbalance of the signaling pathway of TGF-β1 can accelerate the process of atherosclerosis in mice and induce unstable plaque formation (7). In addition, TGF-β1 is a protective factor for the onset of atherosclerosis and is a key mediator of fibrosis by regulating the remodeling of ECM. Upregulation of TGF-β1 activity can promote the formation of atherosclerotic plaques and is more likely to promote the transition of unstable plaques to stable plaques or fibrous plaques (3,8). Therefore, TGF-β1 is an important factor in the maintenance of atherosclerotic plaque stability.
Acquiring more specific biomarkers aimed at different composition of plaque is critical to the construction of multimodal molecular imaging platform. Molecular imaging, which can non-invasively monitor the development of lesions on cellular and protein levels, enables more accurate diagnosis of atherosclerotic plaque, magnetic resonance imaging (MRI) is a promising examination method providing high-resolution, as well as functional and physiological, information about tissues in vivo, and to identify the biological procession of atherosclerosis due to its high sensitivity and specificity (9,10). Targeted MRI can provide atherosclerotic information about the stenosis process and plaque composition, and can help to evaluate the biological and pathological progression of atherosclerosis (11–13).
We dynamically observed the expression changes of TGF-β1 in mouse AS plaque, and studied their regulatory effects on AS plaque and vascular fibrosis. The aim of the present study was to evaluate the feasibility of targeting TGF-β1 molecule expression in AS plaque through synthetic probe MR, so as to explore whether TGF-β1 can become one of the molecular targets in atherosclerosis, to provide a new entry point for diagnosis of AS-related molecular imaging research and clinical prevention and treatment of cardiovascular diseases. The distribution and expression of TGF-β1 in AS plaque of mice were measured by synthesizing a new anti TGF-β1 nano probe and using 7.0-T MR in vivo examination .
Material and Methods
Induction of atherosclerosis in ApoE-knockout mice
All experimental procedures involving animals were approved by the local ethical animal care committee. The six-week-old male ApoE–/– mice (n = 70) were purchased from Vital River (Beijing, PR China) as an experimental group, and fed a high-fat diet (containing 2.5% cholesterol) to induce atherosclerosis. The six-week-old male wild-type C57BL/6 mice (n = 30) were purchased from the animal core facility, and fed a normal diet as a control. Mice were bred and maintained in the animal research center.
Atherosclerotic plaque formation was dynamically viewed at different times (10, 16, 22, and 28 weeks). The morphology of aorta plaques was observed by optical microscope (Nikon, Japan) on hematoxylin and eosin (H&E) staining and immunohistochemical staining.
After the successful establishment of atherosclerosis mice models, another 40 mice from the experimental group were classified into the targeted probe group, which was administrated the anti-TGF-β1-ultrasmall superparamagnetic iron oxide (USPIO) probe, and the pure group, which was injected on pure USPIO. A 7.0-T MRI scan was performed before the targeted probe and administration of the USPIO agent as the baseline imaging, and a second MRI scan was obtained 24 h later.
Preparation and characteristics of anti-TGF-β1-USPIO probe
The morphology of the USPIO was observed by a transmission electron microscope (TEM) (JEOL-100CX, Tokyo, Japan). The size and distribution of the particles were measured by Image-Pro Plus 6.0. Zeta potential and hydrated particle sizes were measured by dynamic light scattering (DLS) (Nano ZS, Malvern, UK). The T2 relaxation rate was measured using a 7.0-T MRI scanner (Verio Tim; Siemens, Erlangen, Germany); anti-Tenascin C-USPIO was diluted to the following concentration gradients: 0.1 mmol/L; 0.2 mmol/L; 0.3 mmol/L; 0.4 mmol/L; 0.5 mmol/L; and 0.6 mmol/L. The T2 relaxation rate was calculated as 1/T2. The targeted USPIO specificity was evaluated by a TGF-β1 mouse enzyme-linked immunosorbent assay (ELISA) kit (R&D System). Pure USPIO particles were used as controls in ELISA analysis.
Acquisition of MR images
In vivo MRI was performed using a 7.0-T small animal MRI system (Bruker, Germany) with a small animal coil and mouse cradle. Double cardiac and respiratory-gated devices were connected. Three patch electrodes were placed on the mice's paws to collect the electrocardiogram (ECG) signal, and a pressure sensor was placed under the abdomen for the respiratory signal. The mice were initially anesthetized by 4% isoflurane /gas mixture and were maintained on anesthesia with 1.5% isoflurane /gas mixture during scanning. Mice in the targeted probe group were injected with anti-TGF-β1-USPIO via tail vein at a dose of 10 mg Fe/kg body weight, mice in the control group were injected with the same dose of pure USPIO.
A bright-blood cine-mode FLASH sequence was used for localization, in order to ensure a similar observation range, the lower pole of the left kidney was taken as the lowest point, and 18 layers were scanned upward to observe the abdominal aorta. Based on these overview scans, a proton density weighted (PDW) dual-echo multi-slice multi-echo (MSME) sequence can be planned and then obtained in the transverse plane. The parameters of this sequence were as follows: TR = 1891.5 ms; TE = 65/13 ms; field of view (FOV) = 2.0 × 2.0 cm; matrix = 256 × 256; slice thickness = 0.7 mm; number of averages (NA) = 2; voxel size = 0.8 × 0.8 × 0.4mm.
Immunohistochemical analysis
The primary antibody was rabbit anti-rat anti-TGF-β1. The tissue was covered with 5% bovine serum albumin and diaminobenzidine staining for 10 min. Prussian blue staining used Perls solution prepared with 2% sodium cyanide ferrous mixes with 2% hydrochloric acid. After 30 min of staining, the tissue was kept in 0.5% neutral red dye water for 2 min.
Statistical analysis
All analyses were performed using SPSS version 25.0 (IBM Corp., Armonk, NY, USA). All data are reported as mean ± SD. The expression of TGF-β1 was compared, and signal changes on MRI were investigated; one-way analysis of variance and pairwise comparison were conducted if the difference was statistically significant. Statistical significance was defined as P < 0.05.
Results
Establishment of atherosclerosis mice model
There was no plaque in the control mice at any time (Fig. 1a–d). In the experimental group, several foam cells and inflammatory cells gathered in the aortic wall of 10-week-old mice (Fig. 1e). With longer feeding times, plaque started to form in 16-week-old mice, and the plaque area increased gradually. The aorta of 16-week-old mice had small plaques with small fibrous caps (Fig. 1f). The plaque area of 22-week-old mice increased with slight deposition of cholesterol crystals (Fig. 1g). The plaque of 28-week-old mice was obvious with a large lipid core that is rich in cholesterol crystals, leading to lumen stenosis (Fig. 1h). The results show that the atherosclerosis model was successfully established in 28-week-old mice.
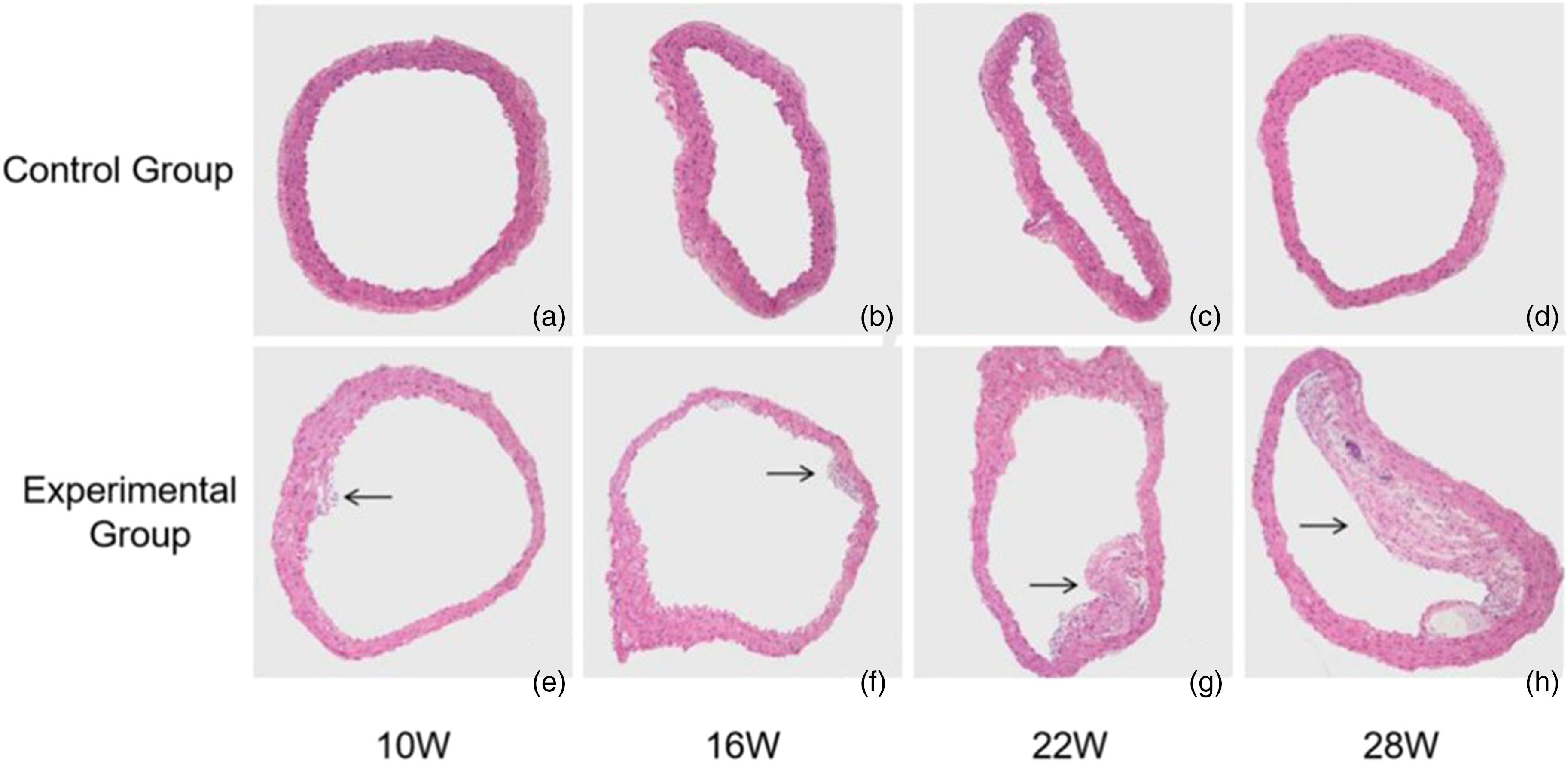
Dynamic observation process in atherosclerosis both in the experimental group and control group (H&E staining, ×100). (a–d) There was no plaque formation and the intima was uniform in the control group. (e) In the experimental group, several foam cells and inflammatory cells gathered and (f–h) plaque formed with lipid core and cholesterol crystal. The plaque is shown above (black arrow).
Expression and distribution of TGF-β1 in atherosclerotic plaque
Immunohistochemical staining showed that the expression of TGF-β1 in plaques at different times (Fig. 2): compared with 16-week-old and 22-week-old mice, the expression of TGF-β1 in the arterial plaque of 28-week-old mice was significantly higher, and the positive expression area was mainly located around the plaque. The expression trend increased gradually with longer feeding times.
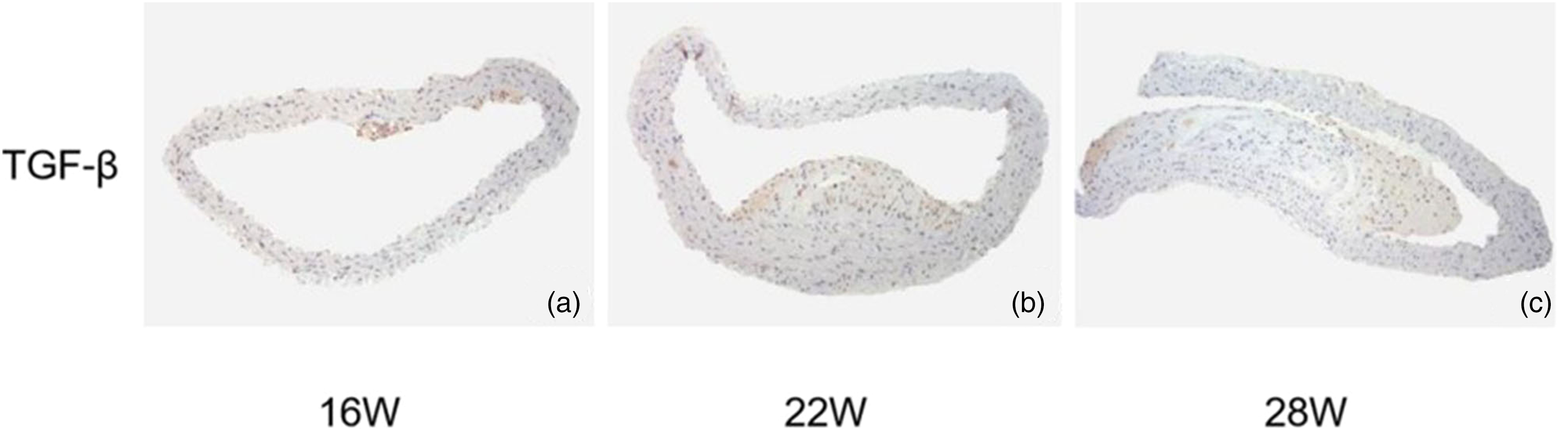
The expression and distribution of TGF-β1 in atherosclerotic plaques (immunohistochemical staining, ×100). (a–c) The expression and distribution of TGF-β1 in atherosclerotic plaques.
Characteristics of anti-TGF-β1-USPIO probe
The anti-TGF-β1-USPIO solution was black-brown in color and was drawn to a permanent magnet. TEM imaging demonstrated the probe was spherical or spheroidal in shape and well distributed in phosphate buffered solution (PBS) solution (Fig. 3a). The hydrated diameter of the probe was greater than untargeted USPIO, indicating that the conjunction was successful. The T2 relaxation rate was r2 = 168.24 mM−1 s−1 (Fig. 3b and c). The ELISA results showed that the anti-TGF-β1-USPIO maintained high biological activity, whereas the pure USPIO showed negative activity (Fig. 3d).
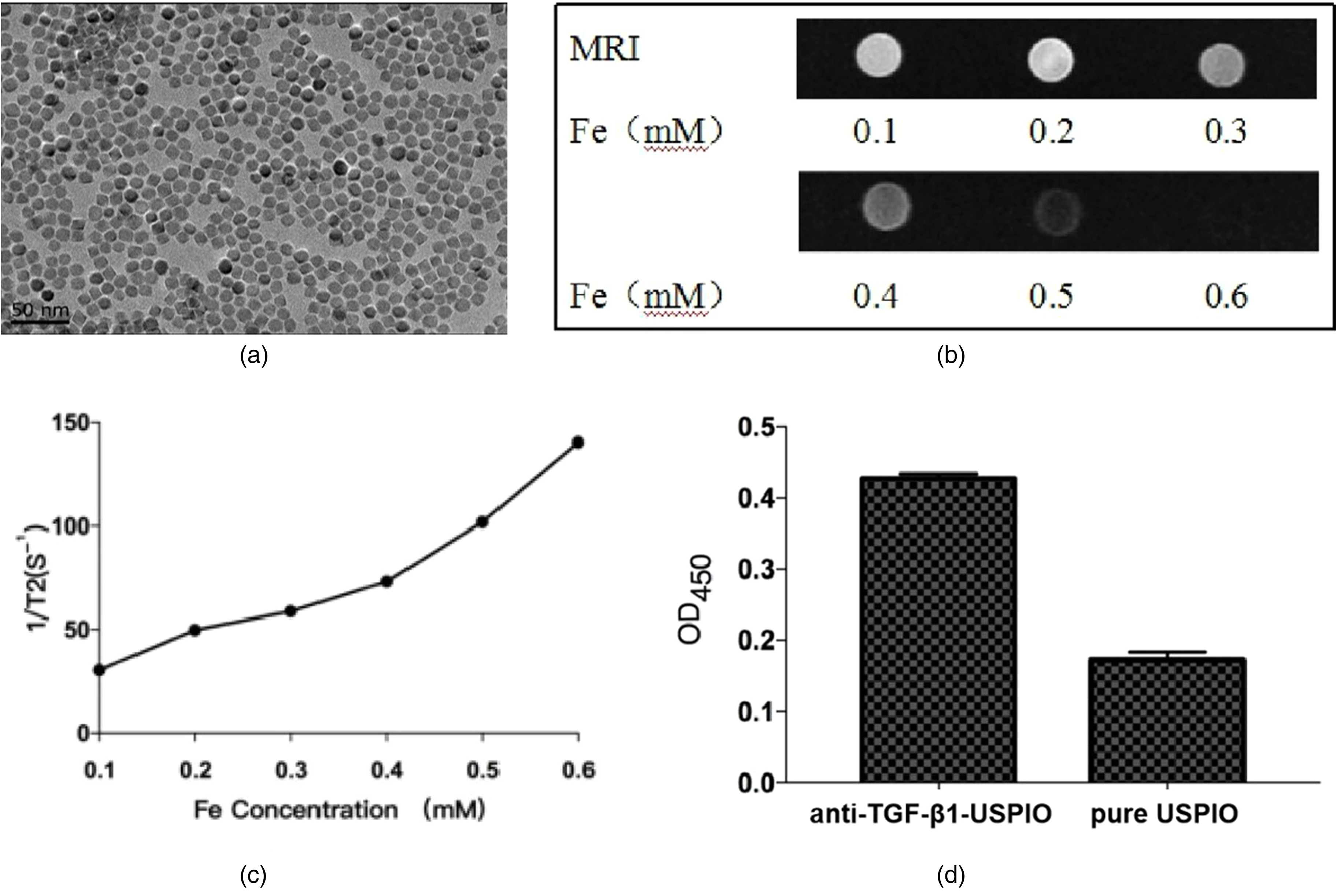
The characteristics of anti-TGF-β1-USPIO probe. (a) TEM imaging shows that the targeted probe is well dispersed in PBS solution. Top right insert shows apparent magnetism of the probe liquid. (b, c) T2 relaxation rate of the probe under different concentrations of Fe and T2 relaxivity. (d) ELISA results of different nanoparticles. Data are expressed as the mean ± SEM (n = 5). *P < 0.05. PBS, phosphate buffered solution; TEM, transmission electron microscope; USPIO, ultrasmall superparamagnetic iron oxide.
In vivo MR imaging of TGF-β1 and statistical analysis
Fig. 4 shows representative MR images of atherosclerotic plaques between the targeted group (Fig. 4a and b) and the pure group (Fig. 4c and d). The MR images show the targeted group exhibited an apparent signal decrease in the plaque and wall of the aorta relative to the pure group. The relative signal intensity (rSI) changes showed a distinct difference compared with the pure group (−19.34% ± 0.68% vs. −5.61% ± 0.57%; P < 0.05) (Fig. 5). The presence of USPIO was verified by Prussian blue staining. More USPIO deposition was found in the targeted group than that in the pure group (Fig. 6).
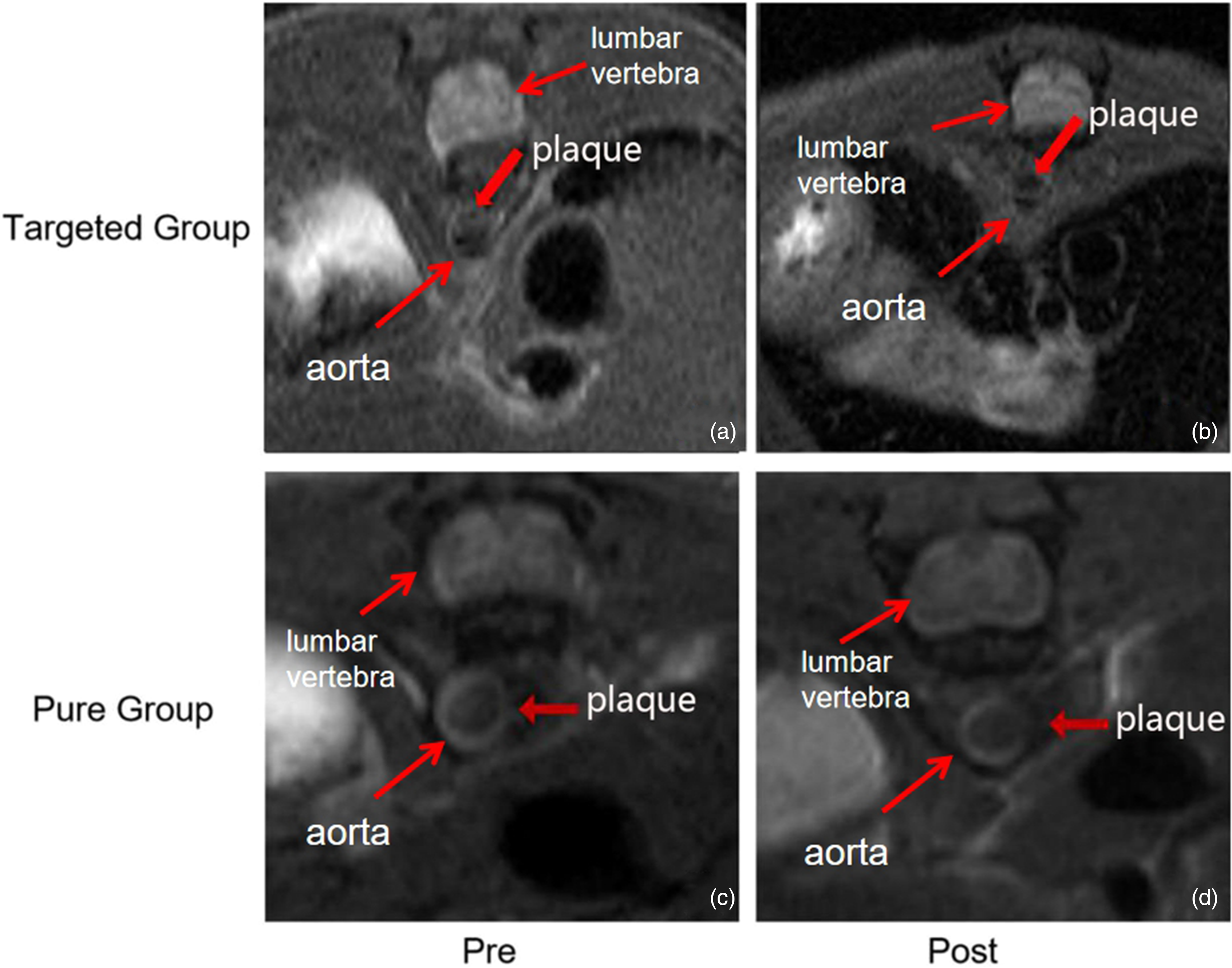
MR images of aorta atherosclerotic plaques before and 24 h after administration of targeted USPIO and pure USPIO. The MR images show (a, b) the targeted group exhibit an apparent signal decrease in the plaque and the aorta wall relative to (c, d) the pure group. The red arrows indicate atherosclerotic plaques. MR, magnetic resonance; USPIO, ultrasmall superparamagnetic iron oxide.
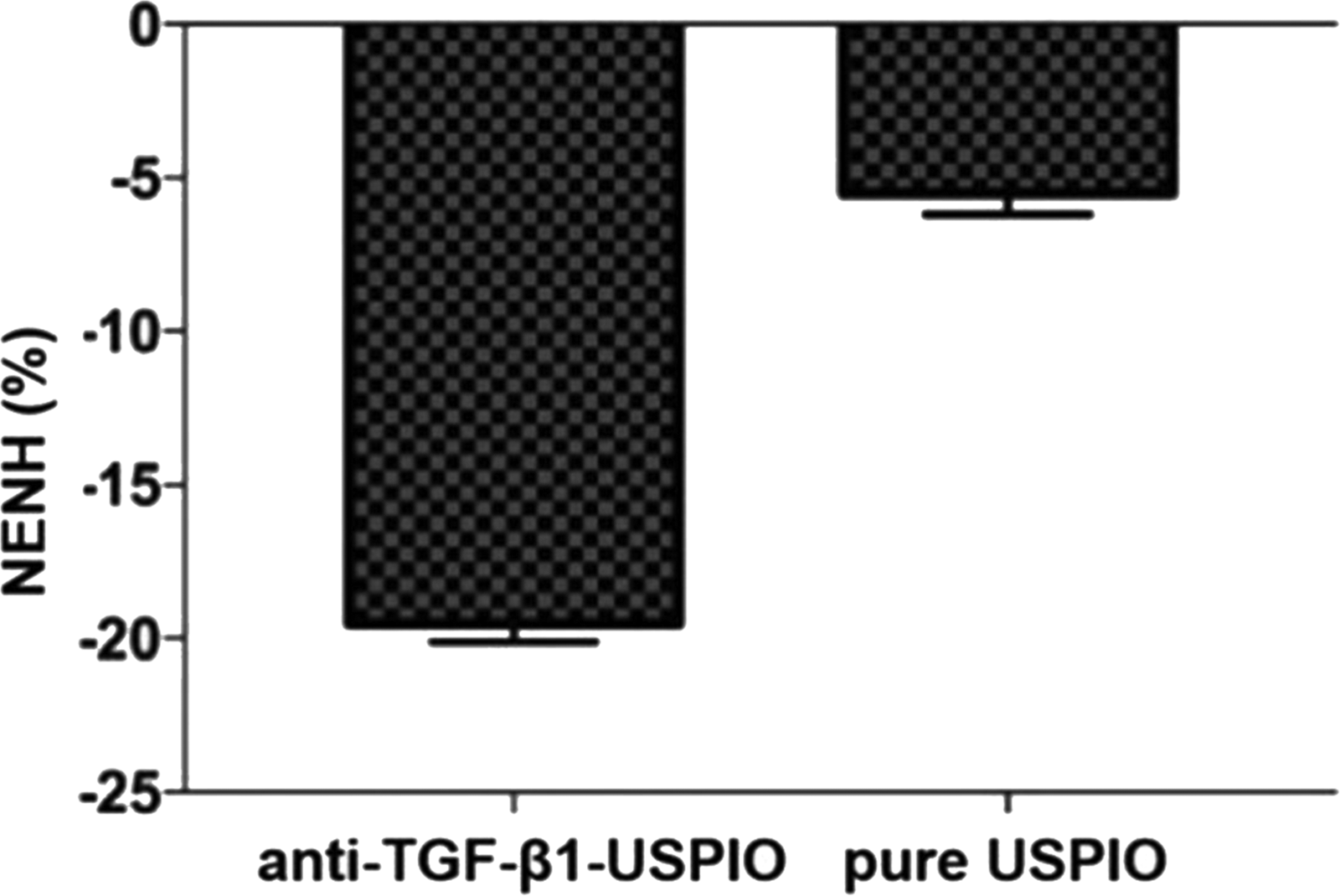
Comparisons of NENH% between the targeted probe group and pure group are shown. *P < 0.05.
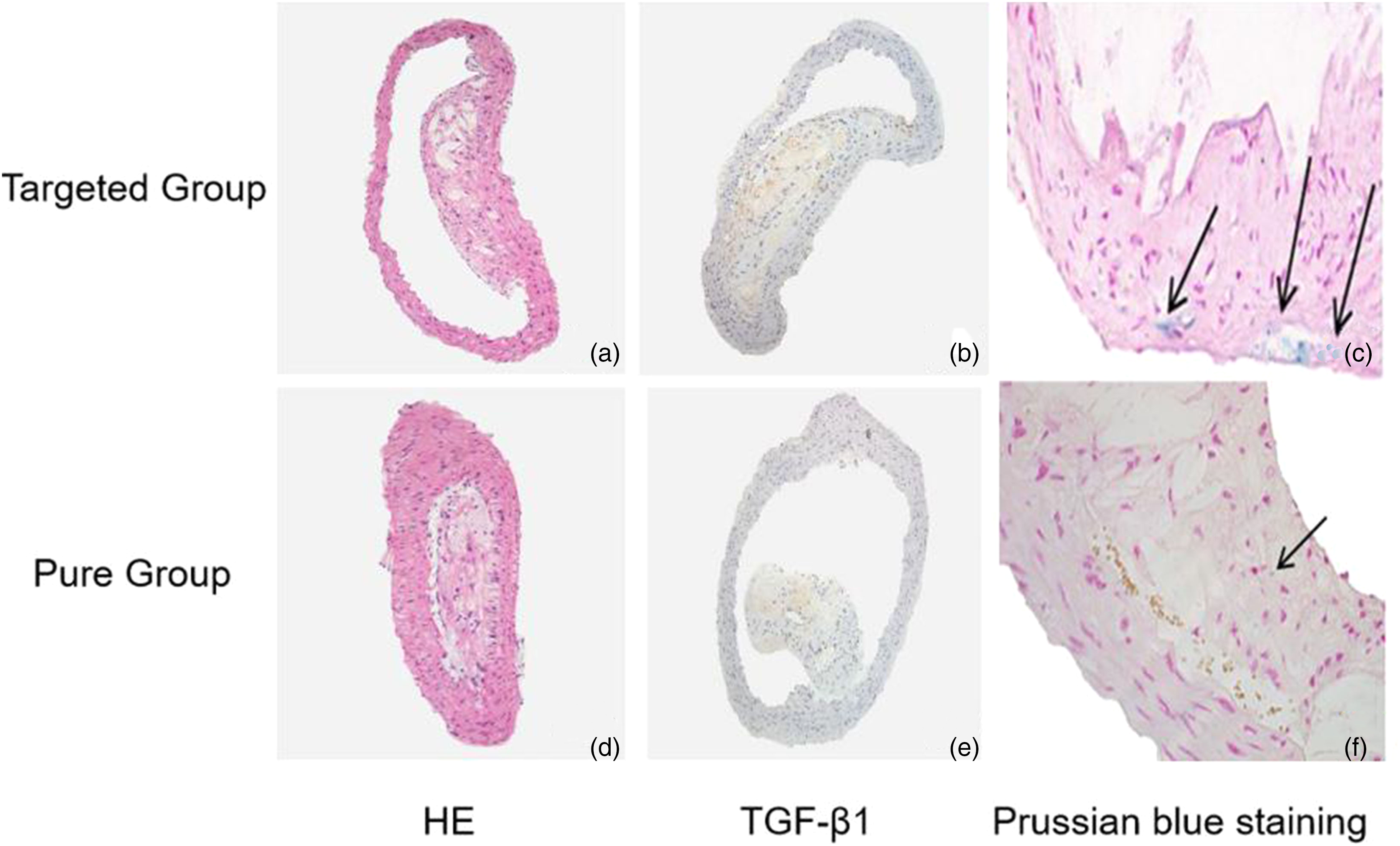
Histopathological and immunohistochemical analyses of the atherosclerotic lesions in ApoE–/– mice. The plaques of aorta were stained by (a, d) H&E staining (× 100) and (b, e) immunohistochemical staining for TGF-β1 (×100). (c, f) Prussian blue staining verified the existence of USPIO in the plaques (×400). USPIO, ultrasmall superparamagnetic iron oxide.
Discussion
The development of atherosclerotic vessel wall disease is the result of complex interactions between different molecular components of the atherosclerotic plaque (14,15). Molecular imaging is a promising technique for non-invasively visualizing and characterizing atherosclerotic development. However, its clinical application is still limited, mainly due to the lack of proper contrast agents with fine biocompatibility and specificity. TGF-β1 plays a fundamental role in the regulation of vascular function by affecting SMC proliferation, migration, differentiation, and fibrosis in atherosclerosis progression (16). In the present study, we observed that TGF-β1 was highly expressed in atherosclerotic plaque. Then, we constructed anti-TGF-β1-USPIO targeting probe as molecular imaging probe and used it to in vivo visualize atherosclerotic plaque in apoE–/– mice through MRI. It is likely that increased accumulation of anti-TGF-β1-USPIO in atherosclerotic lesions led to significant signal changes in T2-weighted (T2W) imaging of plaque compared to other non-targeted probes.
Previous studies observed an increased expression of TGF-β1 in atherosclerotic lesions and an especially strong over-expression and higher expression rate in the advanced lesions. TGF-β1 is a key mediator of fibrosis in atherosclerosis by regulating the remodeling of ECM by inducing fibroblasts to differentiate into collagen-secreting myofibroblasts and adjusting the balance between matrix metalloproteinases (MMPs) and tissue inhibitors of metalloproteinase (TIMP) (16–18). Here, thoracic aortic atherosclerotic plaques of ApoE–/– mice were assessed by targeting this protein. The expression level of TGF-β1 is closely related to the stability of the plaques, and the stability of the plaques can be predicted based on the extent of signal decline.
ApoE–/– mice were raised on a high-fat diet for 28 weeks until advanced atherosclerotic plaques formed. Before injecting the targeted probe, baseline MRI showed thoracic aortic plaques in mice, and the thickened vessel walls showed slightly higher signal on T2W and PDW imaging. This was the result of a large amount of lipid precipitation in the intima, of which T2W imaging was the best. MRI was repeated after the anti-TGF-β1-USPIO injection or pure USPIO injection for 24 h. The dose was 10 mg Fe/kg in each group. At 24 h after the anti-TGF-β1-USPIO injection, the signal of atherosclerotic plaques of ApoE–/– mice was significantly reduced, with a signal reduction rate of −19.34% ± 0.68%. Prussian blue staining confirmed iron granule deposition in the signal reduction area consistent with the immunohistochemical-positive area of TGF-β1. At 24 h after injection of pure USPIO, the signal of atherosclerotic plaque was slightly decreased, and the signal reduction rate was −5.61% ± 0.57%. The difference was statistically significant versus the probe group (P < 0.05). Prussian blue staining confirmed that only a few iron particles were deposited in the signal reduction area. The results of MR studies and immunohistochemistry in mice confirmed that anti-TGF-β1-USPIO can be used as a specific targeted molecular imaging vector for TGF-β1, and the distribution of TGF-β1 in thoracic aortic plaques can be detected in vivo and non-invasively.
USPIOs are superparamagnetic and can shorten the relaxation time of hydrogen protons in tissues, thus reducing the signal intensity of T2W imaging. The degree of decline is related to the local iron accumulation (19–21). Here, 24 h after administration was selected as the optimal imaging time, and signal decline is usually seen within 24 h after administration (22,23). The signal decrease was detected at the lesion site. Prussian blue staining confirmed that the iron deposition in the plaque was mainly in the ECM region. Only a small amount of iron deposition was found in the pure group, suggesting that a decrease in T2W imaging signal was caused by the target probe entering the plaque and binding to TGF-β1.
The present study has some limitations. First, the method of synthesizing probes had some deficiencies that may affect the binding rate of antibody and probe. Second, the imaging time selected was only 24 h after injection and it was a single injection. Tail vein injection might have led to iron particle ingestion by the phagocytic system to affect the number of probes reaching the target site. How to reduce this non-specific uptake will be the focus of future research.
In conclusion, we reported a specific anti-TGF-β1-USPIO probe. MRI can be a non-invasive method of monitoring the expression of TGF-β1 in atherosclerotic plaques. The signal changes on the images can depict the stability of atherosclerotic plaques. These findings, which suggested that intervention in the early stage of the disease is critical to preventing acute cardio-cerebrovascular events, could offer a new direction for the early diagnosis of AS.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received the following financial support for the research, authorship, and/or publication of this article: The research was supported by the National Natural Science Foundation of China (grant no. 81971669).
