Abstract
Background
Detection of breast cancer in women with high breast densities is a clinical challenge.
Purpose
To study the influence of different degrees of breast density on the sensitivity of contrast-enhanced breast magnetic resonance imaging (CE-BMRI) versus X-ray mammography (XRM).
Material and Methods
We performed an additional analysis of two large Phase III clinical trials (G1; G2) which included women with histologically proven breast cancers, called “index cancers.” Additional cancers were detected during image reading. We compared the sensitivity of CE-BMRI and XRM in women with different breast densities (ACR A→D; Version 5). For each study, six blinded readers evaluated the images. Results are given as the “Median Reader.”
Results
A total of 774 patients were included, 169 had additional cancers. While sensitivity of CE-BMRI for detecting all index cancers was independent of breast density (ACR A→D) (G1: 83%→83%; G2: 91%→91%) the sensitivity of XRM declined (ACR A→D) (G1: 79%→62%; G2: 82%→64%). Thus, the sensitivity difference between both imaging modalities in ACR A breasts of 3% (G1) and 9% (G2) increased to 21% (G1) and 26% (G2) in ACR D breasts. Sensitivity of CE-BMRI for detecting at least one additional cancer increased with increasing breast density (ACR A→D) (G1: 50%→73%, G2: 57%→81%). XRM’s sensitivity decreased (G1: 34%→20%) or remained stable (G2: 24%→25%).
Conclusion
CE-BMRI showed significantly higher sensitivity compared to XRM.
Introduction
The introduction of mammography screening was the most important preventive achievement to reduce breast cancer mortality (1). Digital X-ray mammography (XRM) is currently the standard breast imaging method worldwide (2,3). It is a high-throughput modality for cancer screening as well as for detection and characterization of suspicious clinical findings (4).
Contrast-enhanced magnetic resonance mammography (CE-MRM) has been shown to feature higher sensitivity for the detection of breast cancer compared to conventional digital XRM (5–7). However, contrast-enhanced breast magnetic resonance imaging (CE-BMRI) is also considered to be more expensive (8), to have more false positive results (9), cause increased re-excision rates (10), cause over-detection (8), or increased mastectomy rates (11). Therefore, pertinent guidelines—e.g. American College of Radiology (ACR) (12) and the European Society of Breast Cancer Specialists (EUSOMA) (13)—recommend CE-BMRI only for a limited number of specific clinical questions. These include screening of high-risk patients (13,14), determining the extent of disease (13,14), and additional evaluation of clinical or imaging findings (14). The (routine) use in women with dense breasts is still in scientific debate (2,15). However, there is increasing evidence demonstrating the usefulness of CE-BMRI for screening women with dense breasts. In an increasing number of states in the USA, a legislation has been enacted mandating women to be directly notified of the risks associated with high breast density (16).
The prevalence of women with extremely dense breasts in women aged 50+ is in the range of 3%–8% (17–19). Dense breasts constitute an increased risk for breast cancer (2,20–22), possibly because more fibro-glandular tissue bears a higher risk for development of cancerogenic mutations than fatty tissue (23). XRM has a lower sensitivity in women with extremely dense breasts (24–26) because of a masking effect of the lesions within the radio-opaque dense breast tissue (22,24,27). In addition, neovascularization is a key feature of newly developing malignant tumors. In CE-BMRI, it is the gadolinium in the vessels that triggers the MR signal. This signal is not attenuated by dense breast tissue. Thus, sensitivity of CE-BMRI remains unchanged with increasing density while sensitivity of XRM declines. As a result, CE-BMRI sensitivity is higher in women with dense breasts than XRM sensitivity (28).
Sardanelli et al. suggested that women with heterogeneously or extremely dense breasts might benefit from preoperative CE-BMRI (29,30). Later, further evidence for a higher cancer detection yield in screening women with elevated cancer risk and dense breasts by offering additional breast MRI was reported by the ACRIN 6666 study (31). Recently, in 2018 the ACR clearly recommended MRI for women with dense breast tissue (12).
Gadobutrol is a macrocyclic, extracellular gadolinium-based contrast agent (GBCA) provided in a unique 1 molar formulation (32). It is approved in 109 countries in many indications, including CNS, angiography, and breast imaging. Efficacy in lesion detection and characterizations versus other GBCAs in breast MRI has been studied by a number of other researchers (33–35). So far, 74.9 million (at 30 April 2020) gadobutrol administrations have been recorded since 1999.
The aim of the present study was to compare sensitivities of CE-BMRI versus XRM for the detection of breast cancer in women with different degrees of breast density.
Material and Methods
Data sources
We performed an additional analysis of two large Phase III studies (GEMMA-Program - Gadobutrol-Enhanced MR Mammography) registered on ClinicalTrials.gov (GEMMA1: NCT01067976; GEMMA2: NCT01104584; referred to as “G1” and “G2” below) (6).
Study population and interventions
Study population and interventions were published by Sardanelli et al. (6). In brief, in G1, 390 women (mean age = 55.7 ± 10.4 years; 74% white, 25% Asian) were included from 28 centers in seven countries in Europe, the Americas, and the Republic of Korea. In G2, 397 women (mean age = 57.1 ± 10.7 years; 71% white, 24% Asian) were included from 39 centers in Europe, Canada, Argentina, India, Taiwan, and the USA.
The “Index cancer” was defined as the lesion that triggered the diagnosis. “Additional cancers” were detected during the preoperative examinations and/or by CE-BMRI and verified by the reference standard, i.e. malignant regions were pathologically confirmed from surgical specimen and cancer-free regions by XRM together with high-frequency ultrasound or pathological examination (when available). “All cancers” were the sum of index cancers plus all additional cancers.
Study procedures
Inclusion criterion was a recently detected and histologically proven breast cancer. All patients received an additional CE-BMRI at 1.5-T. Gadobutrol (Gadovist® 1.0 mmol/mL, Bayer AG, Leverkusen, Germany) was applied intravenously at a dose of 0.1 mmol/kg body weight.
For each study, six independent readers evaluated the images: three evaluated the XRMs and three evaluated the CE-BMRIs. Breast density was determined by one radiologist with > 12 years of experience in breast imaging. Histopathology results were the standard of reference.
Target variables
The primary target variable of this additional analysis was the sensitivities of CE-BMRI versus XRM for the detection of breast cancer at different degrees of breast density according to ACR5 (36). We analyzed the index cancer and additional cancers separately. In addition, we analyzed sensitivity of both modalities of finding all cancers.
Secondary parameters were the impact of ethnicity and tumor size on the sensitivity.
Statistics
Sensitivity was defined as the proportion of patients for whom the readers detected all lesions identified by the standard of reference. To reduce the variability associated with multiple readers, median reader sensitivity was reported. Median reader sensitivity was calculated as the median of the sensitivity values for the three readers in each study. Patients were included in the analyses if they had at least one lesion identified by the standard of reference and had data available for the subgroup of interest. Two-sided 95% confidence intervals (CI) were calculated for within-group proportions using a normal approximation to the binomial distribution. The two-sided 95% CIs for the difference between CE-BMRI and XRM were calculated using the normal approximation based on Schwenke and Busse (37). No statistical adjustments were made for multiple comparisons and no formal threshold for statistical significance was declared.
Results
Study population
A total of 774 patients were included (G1: n = 386 G2: n = 388), 169 had additional cancers (G1: n = 86; G2: n = 83). The detailed demographics of the study population have been reported by Sardanelli et al. (6).
Index cancers
While the median reader sensitivity of CE-BMRI for detecting all index cancers remained stable with increasing breast density (i.e. from ACR Density A → ACR D) (G1: 83% →83%; G2: 91% → 91%) the sensitivity of XRM declined (G1: 79% → 62%; G2: 82% → 64%). As a consequence, the difference in sensitivities between both imaging modalities increased with increasing breast density (from ACR Density A → ACR D) (G1: 3% → 21%; G2: 9% → 26%) (Fig. 1a).
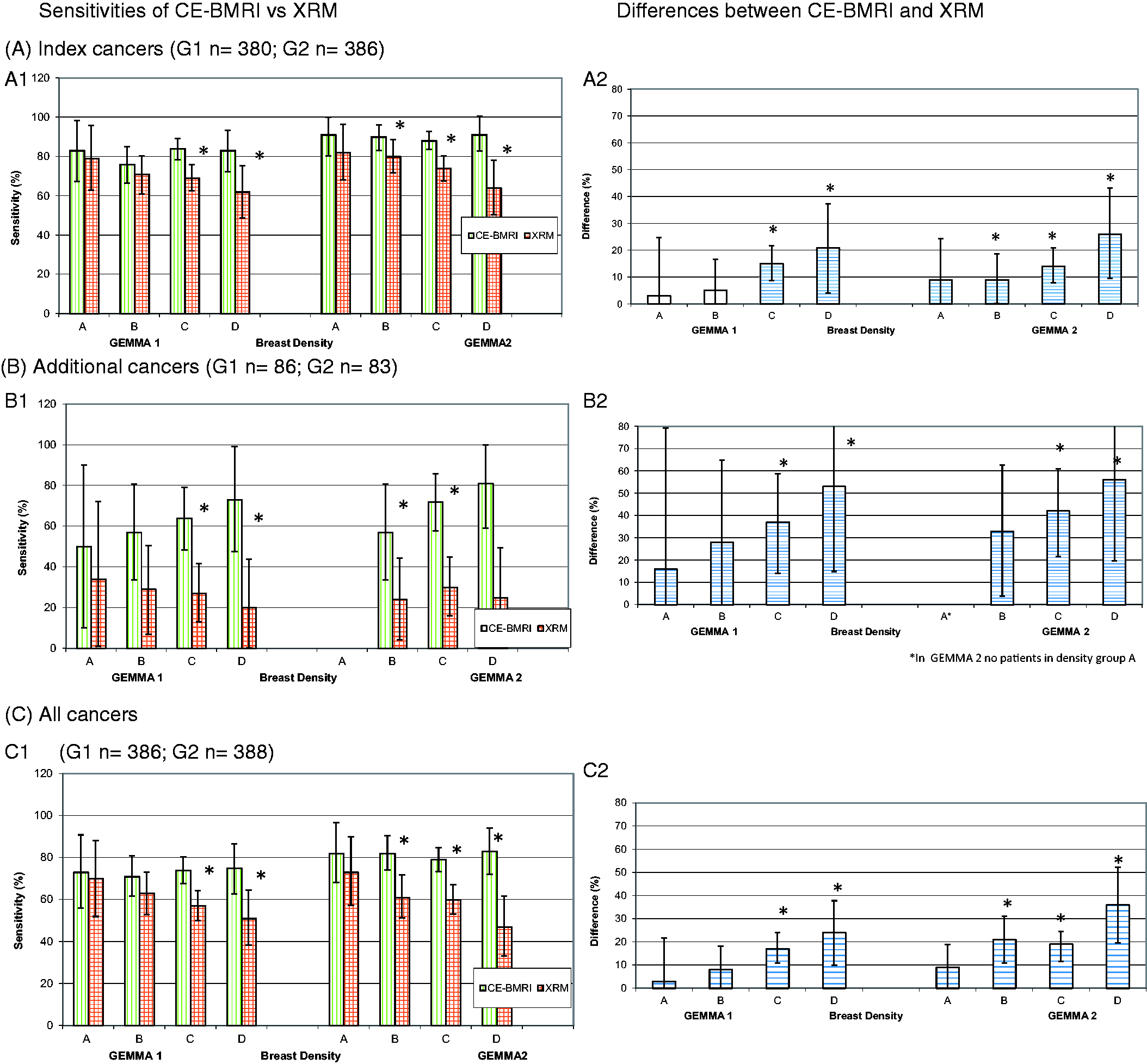
Sensitivities of CE-BMRI vs. XRM by breast density (ACR5) in Studies G1 and G2 (% (± 95% CI)).
Additional cancers
While the sensitivity of CE-BMRI for detecting at least one additional cancer increased with increasing breast density (G1: 50% →73%, G2: 57% →81%) the sensitivity of XRM decreased (G1: 34% → 20%) or remained stable (G2: 24% → 25%). These findings resulted in remarkably higher sensitivity differences for the additional tumors than for the index cancers reaching > 50% for ACR D (Fig. 1b).
All cancers
A similar picture is seen when looking at patients where all cancers have been detected. The sensitivity differences are increasing with increasing ACR grade and highest in ACR D: 24% in G1 and 36% in G2 (Fig. 1c).
Ethnicity
While the sensitivity for detecting index or additional cancers was higher in Asian patients for both imaging modalities, the difference between the two imaging modalities was similar for Caucasian and Asian patients. The difference was highest for the additional cancers (Table 1).
Sensitivities of CE-BMRI vs. XRM by ethnicity.
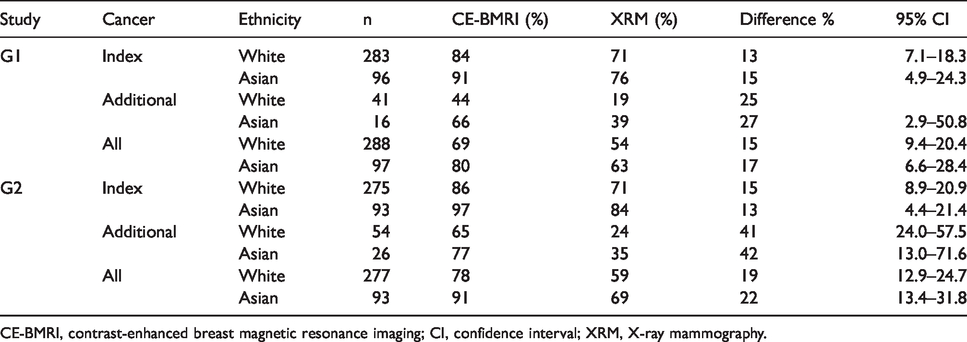
CE-BMRI, contrast-enhanced breast magnetic resonance imaging; CI, confidence interval; XRM, X-ray mammography.
Tumor size
The vast majority of all tumors were in the size categories 1.0–1.9 cm and 2.0–4.9 cm. The sensitivity of CE-BMRI was constantly ≥ 13% higher than XRM independent of tumor size, only with the exception of tumors ≥ 5 cm in G1 (4%). Thus, the 95% CIs excluded zero, consistent with a result of CE-BMRI superiority in the two major size brackets (Table 2).
Sensitivities of CE-BMRI vs. XRM by tumor size, all tumors.
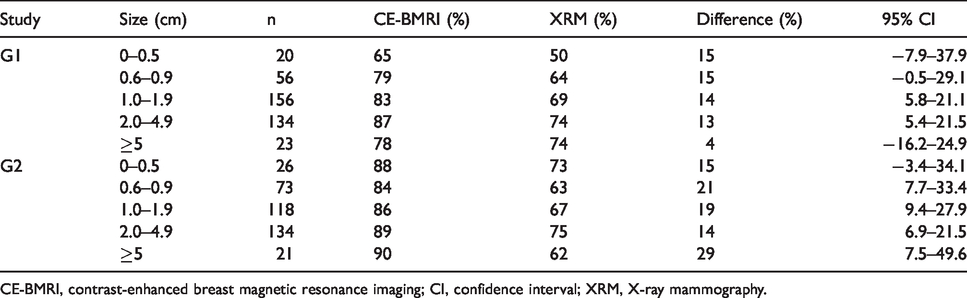
CE-BMRI, contrast-enhanced breast magnetic resonance imaging; CI, confidence interval; XRM, X-ray mammography.
Discussion
We compared sensitivities of CE-BMRI versus XRM for the detection of breast cancer in women with different degrees of ACR 5 breast density in a large dataset of two Phase III studies. While the sensitivity of CE-BMRI was almost unaffected by breast density, the sensitivity of XRM decreased remarkably with increasing breast density.
The primary inclusion criterion was a histologically proven newly diagnosed breast cancer, called index cancer, an XRM was also required for comparison (6). All blinded readers were asked to find the diagnosis solely based on the images. They had no access to the patients’ medical history or results of clinical examination and/or ultrasound, which usually provide important additional information in “normal clinical routine.” As the CE-BMRI readers were also limited to the images, both datasets, i.e. XRM and CE-BMRI, were well comparable.
While the sensitivity of CE-BMRI for detecting all index cancers was independent of the breast density, the sensitivity of XRM sensitivity declined by around 20% with increasing density (Fig. 1). The higher sensitivity of CE-BMRI was even more pronounced in detection of additional cancers. In the ACR D subgroup, the sensitivity difference between both imaging modalities increased to > 50% (Fig. 1).
This advantage of CE-BMRI over XRM in detecting malignant breast lesions is in line with findings of other groups, although the clinical trial settings are not always exactly comparable with respect to study population (patients with elevated breast cancer risk (38,39), dense breasts, proven breast cancer (40)) and imaging purpose (screening (39,41), preoperative staging (40), recurrence detection).
Berg et al. (31) added a single screening CE-BMRI to XRM in 2662 women with elevated risk of breast cancer and dense breasts. By applying supplemental CE-BMRI in 612 women with ACR density 3 and 4 plus ≥1 other risk factor, they identified nine additional cancer cases.
Kuhl et al. (5) investigated 2120 women with average risk of breast cancer. Sixty percent featured ACR breast density C or D. All had normal XRM findings. In this prospective observational study, 37/61 (61%) MR-detected additional cancers were in women with the ACR breast density category C or D.
An intriguing approach to assess the benefits of CE-BMRI in women with dense breasts has recently been reported by Bakker et al. (42) (DENSE study protocol by Emaus et al. (25)). They evaluated CE-BMRI as an additional screening modality in 40,373 women aged 50–75 years with extremely dense breasts. A total of 8061 were offered an additional CE-BMRI after a negative XRM result and 32,312 women served as controls. After two years of follow-up, they found an interval-cancer rate of 2.5 per 1000 screenings in the CE-BMRI group compared to 5.0 per 1000 screenings in the control group. The difference of 2.5 per 1000 screenings was significant (P < 0.001). While Bakker et al. (42) focus on women with extremely dense breasts, i.e. ACR D, our study confirms the higher sensitivity of CE-BMRI versus XRM by showing the “almost linear correlation” between increasing breast density—ACR A to B to C to D—and increasing sensitivity advantage of CE-BMRI.
The fact that CE-BMRI detects additional cancers besides the index cancer is of paramount clinical importance as eventually the totality of cancers determines the clinical course of the disease. Identification of additional cancers confirms a multifocal and/or multicentric cancer situation. This feature of CE-BMRI is already known independently of breast density (43). Ioacconi et al. (43) claim that these additional cancers are potentially more biologically relevant because of the presence of unsuspected invasion or a higher histological tumor grade. There is no need to say that multiple cancer lesions, either uni- or bi-lateral, heavily impact the treatment strategy.
Finally, Kuhl (44) states, that XRM is primarily detecting slow-growing, less aggressive cancers. This might explain the persistently high rates of interval cancers and high mortality rates of breast cancer despite decades of mammographic screening. CE-BMRI, instead, detects the fast-growing, more aggressive, and thus most clinically relevant cancers.
The question of whether the higher sensitivity of CE-BMRI impacts re-excision rates, recurrence rates, recurrence-free survival, or overall survival is subject to scientific debate (45).
The sensitivities of CE-BMRI and XRM were higher in Asian women compared to Caucasians but the difference between both imaging modalities was independent of ethnicity. Interestingly, the sensitivity difference between both modalities was highest for the additional cancers. A possible reason for this might be that in our study the percentage of Asian patients with additional cancers was much higher than for Caucasians, e.g. in G1, 41/96 Asians had additional cancers (43%) compared to 16/283 whites (5.7%). A potential reason for this may be the fact that the Asians have generally higher breast density than Caucasians, although paradoxically they appear to have a lower risk of breast cancer (46). A limitation of these considerations might be the smaller number of Asian women in our cohort, just about one-third the number of Caucasians (Table 1), and further studies would be needed to confirm above hypothesis.
CE-BMRI showed a higher sensitivity than XRM independent of tumor size. There was no advantage seen in a group of very small tumors, i.e. 0–0.5 cm. We did not find any research investigating this aspect.
The breast cancer risk factor “dense breast” (20,21) is prevalent in up to 8% of the female population aged 50+ years (17–19). As dense breast tissue potentially masks malignant breast lesions in XRM, CE-BMRI shows higher sensitivity with increasing degree of breast density. This analysis provides strong evidence for the usefulness of CE-BMRI, especially in women with dense breasts.
The present study has some limitations. First, this was an additional analysis, not primarily planned for this dataset. Second, as both studies were evaluated by six different blinded radiologists (three for XRM and three for CE-BMRI, each), pooling of the two studies was not possible. Third, breast density of all images was determined by only one reader (with > 12 years of experience). Fourth, the number of patients in some subgroups (e.g. additional cancers in ACR A and D) is small. Fifth, all blinded readers were asked to find the diagnosis solely based on the images. They did not have access to additional clinical information as in clinical routine, a somewhat artificial situation. Sixth, Roman et al. (47) recently reported changes of density over time. As breast density was determined at the time of cancer diagnosis and tumor growths takes time, breast density at the time of cancer initiation remains unknown. Lastly, ultrasound is frequently used as an adjunct to ambiguous XRM findings, also in dense breasts (48,49). We did not perform a comparison to this modality.
In conclusion, CE-BMRI showed significantly higher sensitivity compared to XRM in women with dense breasts.
Footnotes
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: JB received research funding and speaker honoraria from multiple companies, including Bayer; AB received funding and honoraria from various medical companies, including Bayer; MK, GB, OW, and JE are employees of Bayer AG.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
