Abstract
Background
Patient decision aids should help people make evidence-informed decisions aligned with their values. There is limited guidance about how to achieve such alignment.
Purpose
To describe the range of values clarification methods available to patient decision aid developers, synthesize evidence regarding their relative merits, and foster collection of evidence by offering researchers a proposed set of outcomes to report when evaluating the effects of values clarification methods.
Data Sources
MEDLINE, EMBASE, PubMed, Web of Science, the Cochrane Library, and CINAHL.
Study Selection
We included articles that described randomized trials of 1 or more explicit values clarification methods. From 30,648 records screened, we identified 33 articles describing trials of 43 values clarification methods.
Data Extraction
Two independent reviewers extracted details about each values clarification method and its evaluation.
Data Synthesis
Compared to control conditions or to implicit values clarification methods, explicit values clarification methods decreased the frequency of values-incongruent choices (risk difference, –0.04; 95% confidence interval [CI], –0.06 to –0.02;
Limitations
Some meta-analyses had high heterogeneity. We grouped values clarification methods into broad categories.
Conclusions
Current evidence suggests patient decision aids should include an explicit values clarification method. Developers may wish to specifically consider multicriteria decision analysis. Future evaluations of values clarification methods should report their effects on decisional conflict, decisions made, values congruence, and decisional regret.
Highlights
Current evidence suggests patient decision aids should include an explicit values clarification method.
To support health decisions that align with values, patient decision aid developers may wish to specifically consider multicriteria decision analysis.
Introduction
Shared decision making is appropriate in many situations and is particularly indicated in clinical situations where the “best” option may differ between people, depending on what matters to them.1–3 What is important to one person might be different from what is important to others, and determining what is important can be difficult even with the appropriate information and evidence at hand. The process of shared decision making therefore aims to help people make health-related decisions that are informed by high-quality, well-presented, and well-understood evidence,4–8 aligned with what matters to the person or people affected by the decision and acted upon.9–12 It follows that the process of clarifying and expressing values is an important aspect of shared decision making and thus of patient decision aids. Within patient decision aids, this process is supported by explicit values clarification methods.
Explicit values clarification methods require users to interact with something such as a worksheet or an interactive website to clarify what matters to them relevant to a health decision. Such methods have been shown to encourage desirable outcomes such as better alignment with patients’ values13,14 and reduced decisional regret, the latter particularly among people with lower health literacy. 15 However, explicit values clarification methods are extremely diverse, 16 and there has been little guidance regarding their comparative effects on users’ decision making processes or outcomes, 17 making it difficult for patient decision aid developers to know which explicit method to use. Patient decision aid developers might look toward the preference elicitation literature for guidance, but the guidance available 18 is often tailored toward aggregate-level decision making, such as regulatory decisions 19 or health technology assessment, 20 not for supporting individual-level decision making.
This updated review sought to build upon previous versions of the International Patient Decision Aids Standards’ chapter on values clarification21,22 as well as previous evidence syntheses that have established the advantages of explicit values clarification methods over implicit methods or no values clarification.13,14 We sought to advance the science and practice of values clarification methods in 3 ways. First, we aimed to offer clear definitions and an annotated summary of existing approaches that have been or could be used as values clarification methods. Second, we aimed to synthesize evidence of different techniques’ effects on health decision outcomes. Third, we aimed to foster future evidence by offering researchers a proposed set of outcomes to consider when evaluating the effects of values clarification methods.
Definitions
Part of the challenge in studying or using values clarification methods is that definitions vary and terms like
Definitions of Terms

As noted above, we continue to use the term
Theoretical Rationale
Our interdisciplinary team determined that the theoretical rationale for values clarification required only a small edit, shown in square brackets, to reflect the focus on explicit methods. Like Fagerlin et al., 22 we assert the theoretical rationale for explicit values clarification methods as being that they “should aim to [explicitly] facilitate at least one or more of the following six decision making processes: 1) Identifying options, which can include either the narrowing down of options, or the generation of options that were not offered at the outset, 2) Identifying attributes of the situation and/or the options which ultimately affect the patient’s preference in a specific decision context, 3) Reasoning about options or attributes of options, 4) Integrating attributes of options using either compensatory or both compensatory and noncompensatory decision rules, 5) Making holistic comparisons, and 6) Helping decision makers retrieve relevant values from long-term memory.” Pieterse et al. 27 provided theory-based recommendations on processes that values clarification methods could aim to facilitate.
Although reasoning is one of the potential processes supported by values clarification, neither the definition nor the theoretical rationale of values clarification methods requires that people who are being supported in making a personal health decision must rationally deliberate about each option, or that the goal must always be a fully rational choice. In some decision making situations, rational deliberation and rational choice may be desired, while in others, they may not.28,29
Explicit Values Clarification Methods
Table 2 organizes strategies that can be used as explicit values clarification methods in patient decision aids, building upon previously developed lists of types of values clarification methods3,16 and reviews of preference elicitation methods.30,31 Methods range from highly structured strategies that can also be used for preference elicitation in the context of health policy decision making to substantially less structured strategies. While not every use of a given method will be exactly the same, we deemed them functionally similar in terms of how they might be used and what the user experience might be in a patient decision aid. Patient decision aids may use multiple strategies. For example, a user may be asked to use a rating scale or visual analog scale whose values are then used in a decision-analytic model.
Explicit Values Clarification Methods

Methods
Our overall methods were guided by the Cochrane handbook. We report according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) 45 guidelines.
Eligibility Criteria
We included published reports of comparative evaluations of explicit values clarification methods, whether they were called “values clarification methods” in the publications or not. This meant that we included trials of preference elicitation methods that had been trialed as values clarification methods (e.g., multicriteria decision analysis or discrete-choice experiments). We included evaluations using comparative methods (i.e., randomized controlled trials or randomized experiments of 1 or more values clarification methods). The comparisons could be 1 or more values clarification methods compared to a control method or compared to each other. Because we sought to understand the effects of values clarification methods, we excluded evaluations using descriptive study designs (e.g., acceptability and feasibility study, development study), observational study designs (e.g., reporting outcomes before and after use of a values clarification method), and reports of values clarification methods that did not evaluate the method independently of the patient decision aid in which it was used. Randomized experiments comparing 1 or more values clarification methods had to use distinctly different methods, meaning that more than the content or presentation of information in the values clarification method varied.
We did not apply language restrictions. We applied date restrictions to the portion of the review for which we had already conducted a systematic review (i.e., evaluations of values clarification methods that used the term
Information Sources
We performed a systematic literature search in MEDLINE, EMBASE, Web of Science, the Cochrane Library, and CINAHL.
Search Strategy
We developed a draft search strategy in collaboration with an information specialist (FB; see Acknowledgments). Search strategies for each database are shown in online Appendix 1. We reviewed search strategies with all authors to ensure they were inclusive of relevant preference elicitation methods that might be used for values clarification. We conducted hand searches by reviewing articles that cited the previous version of these standards (values clarification chapter) or a previous systematic review of values clarification methods.
Study Records: Data Management
We managed data with Covidence (Melbourne, Australia), reviewing data records at regular team meetings.
Study Records: Selection Process
Two independent reviewers (SC, MM, TP, CR, CR-B) screened titles and abstracts to assess potential relevance, with a third reviewer adjudicating discrepancies and discussions of questions and points of disagreement at regular team meetings. Two independent reviewers then reviewed the full text of all articles deemed potentially relevant based on their title and abstract. Discrepancies in inclusion and exclusion at full text were adjudicated through team discussions at regular meetings until we reached consensus.
Study Records: Data Collection Process
Two independent, trained research team members (SC, MM, TP, CR, CR-B) extracted data from each article using a standardized and pilot-tested data extraction form based on a previous form 17 and adapted to this review. We resolved disagreements through discussion until consensus was reached. We contacted authors to collect any needed data that they did not report or were unable to report in their publication.
Data Items
Regarding study participants, we recorded the sample size for control and intervention groups along with basic inclusion and exclusion criteria and whether or not they were making the actual decision or if the study was hypothetical. We defined a hypothetical scenario as one in which people are asked (explicitly or implicitly) to imagine that they are in a certain situation or facing a certain decision. We defined a real scenario as one in which people are facing a decision (e.g., because they have received a diagnosis) or are members of a population likely to face the decision in the near term (e.g., parents of children eligible to receive vaccines within the coming months).
Regarding interventions, we recorded the type of explicit values clarification method as listed in Table 2. We also recorded specific characteristics of each values clarification method, namely, whether it explicitly requires the user to engage with tradeoffs, whether it explicitly shows the user the correspondence between their options and what they value, and which, if any, theoretical or conceptual framework underpins it. Where relevant, we recorded whether a variable was collected via self-report, meaning whether responses were completed by participants themselves or by independent researchers based on direct observation, including coded qualitative data.
For comparators (controls), we recorded whether the comparator was no values clarification method or an implicit method and treated both as equivalent controls. The Cochrane review of patient decision aids specifies that all patient decision aids must contain implicit values clarification methods at minimum, 14 and it is accordingly rare to have patient decision aids that do not present potential benefits and harms of options in organized ways. In other words, in the context of patient decision aids, there is no meaningful distinction between implicit methods and no values clarification. The different terminology is simply a function of how authors choose to name their control. We also recorded studies that compared different types of explicit values clarification methods to each other.
Outcomes
Whenever such data were available, we extracted data regarding values congruence (i.e., the extent to which choices aligned with stated values) as our primary outcome, as well as secondary outcomes: decision readiness (worry, decision uncertainty, decision making preparation, knowledge), decisional conflict (measured with a version of the Decisional Conflict Scale and/or its subscales 46 ), decision made, and postdecision and postimplementation health and well-being (decisional regret, longer-term health outcomes). Following data extraction by pairs of trained reviewers (SC, MM, TP, CR, CR-B), 3 authors (HOW, SCD, JJ) mapped all outcomes into broad outcome groups: worry (including perceived risk), decision uncertainty (not including decisional conflict), decisional conflict (decisional conflict scale or any subscales), decision making preparation (including self-efficacy for decision making), beliefs (including beliefs about the condition or underlying decision structure), knowledge, values (including reported utilities), shared decision making (i.e., the extent to which shared decision making occurred or not), effects on communication (including quality, length, or existence of communication), satisfaction with care, preferences (i.e., preferences expressed), decision (choice made and implemented) or decisional intent (choice intended or made and not yet implemented), values congruence, informed decision making (i.e., the extent to which someone made an evidence-informed, values-congruent, behaviorally implemented decision9,47), postdecision feelings (including satisfaction, regret), postdecision health, and user assessment of the intervention (including acceptability, satisfaction, perceived balance). We conducted meta-analyses on primary outcome values congruence and secondary outcome decisional conflict, as these outcomes had sufficient studies to do so.
Risk of Bias in Individual Studies
Independent, trained research team members assessed risk of bias for each study using methods as defined in the Cochrane handbook, section 8.5. 48 We conducted quantitative data syntheses with and without studies identified as being at high risk of bias to determine the sensitivity of overall findings to these studies.
Data Synthesis
We synthesized frequency-based results (e.g., how many values clarification methods reflect a given design) descriptively. To synthesize effects on outcomes, we pooled all experiments that evaluated a values clarification method against no values clarification method or an implicit method. For multiarmed studies in which the comparison of a decision aid with and without a values clarification method included an arm that was not relevant to our comparison of interest (e.g., an information booklet serving as a control condition in an evaluation of the decision aid), we ignored the third arm. For multiarmed studies containing 2 or more different values clarification methods and 1 arm of implicit values clarification or control, we considered each comparison of a values clarification method against implicit values clarification, meaning that each of the multiarmed studies included in this review contributed multiple comparisons to the pooled set.
To meta-analyze results for values congruence, we pooled results from 11 studies using risk differences and applying a random-effects model. Here,
Results
Articles Identified
Out of 30,648 records screened at the title and abstract stage and 279 screened at the full-text stage, we identified 33 articles that met our inclusion criteria describing trials of 43 values clarification methods. Twenty-four of the articles were new articles identified in this update of the International Patient Decision Aids Standards (IPDAS). We excluded 2 of the articles previously included in the IPDAS values clarification chapter because they did not meet our revised inclusion criteria requiring randomized controlled trials and instead reported, for example, pre–post study designs. The PRISMA diagram of included articles is shown in Figure 1.
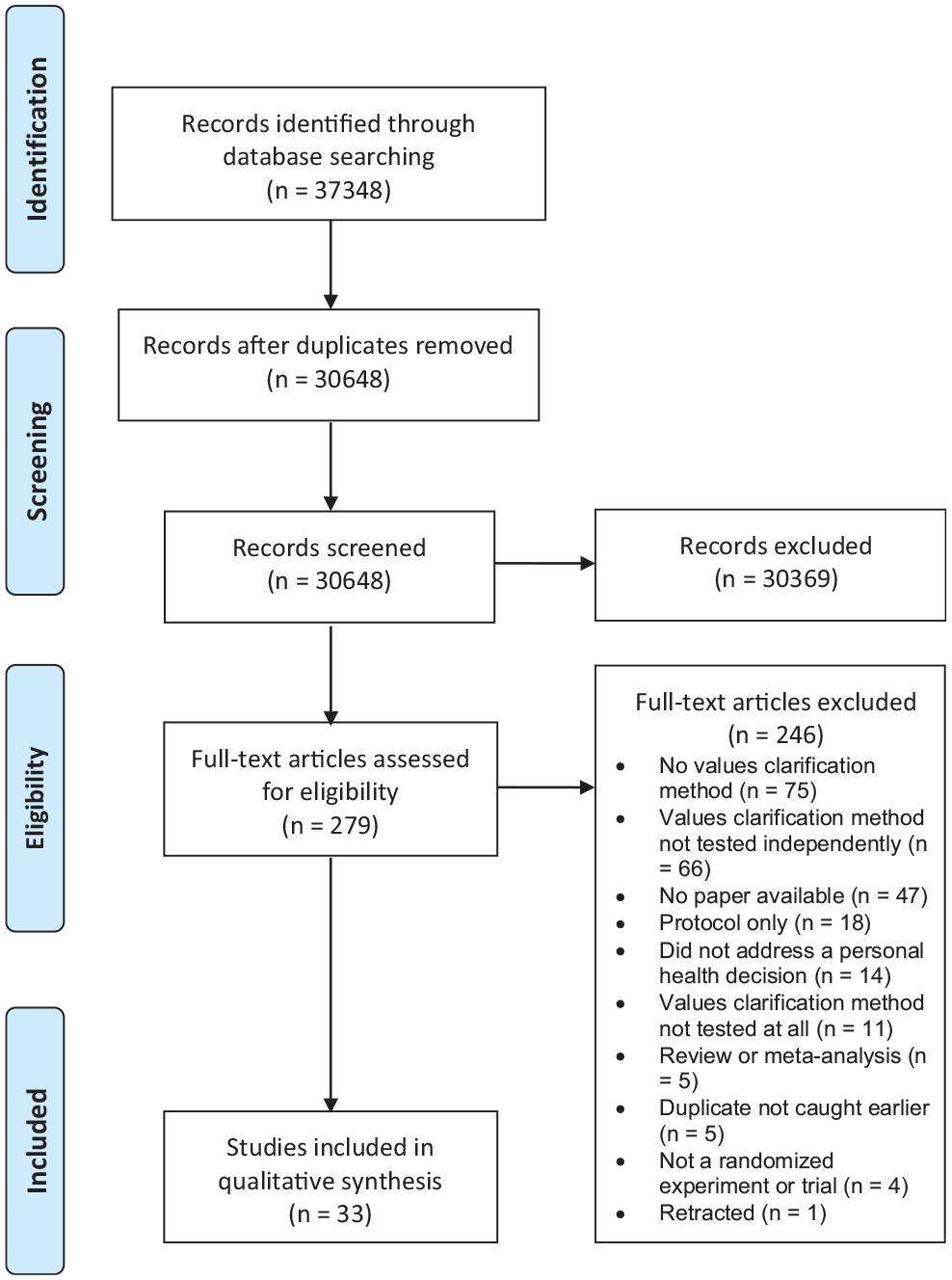
Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) diagram.
The decision context varied across studies. Out of the 43 included trials, 25 (58%) addressed treatment decisions, 9 (21%) screening decisions, 4 (9%) prevention, 3 (7%) genetic testing, and 2 (5%) diagnostic testing. Thirteen of the 43 trials (30%) centered on a yes/no decision to take an option or not, 18 (42%) a choice between 2 or more options, and 12 (28%) both a yes/no and a choice between 2 or more options. Most decisions (22/43, 51%) were real decisions, meaning that the person was making this decision in their actual life. The rest were hypothetical (18/43, 42%), or it was not entirely clear whether the decision was real or hypothetical (3/43, 3%). The most commonly reported outcomes were decisional conflict and/or its subscales (29/43, 67%), decision and/or decisional intentions (22/43, 51%), knowledge (13/43, 30%), and values congruence (12/43, 28%).
As shown in the overview of included studies in Table 3, there was substantial diversity in the types of values clarification methods used. Decision analysis or multicriteria decision analysis was the most commonly trialed method. Full study details are available in online Appendix 2.
Study Details
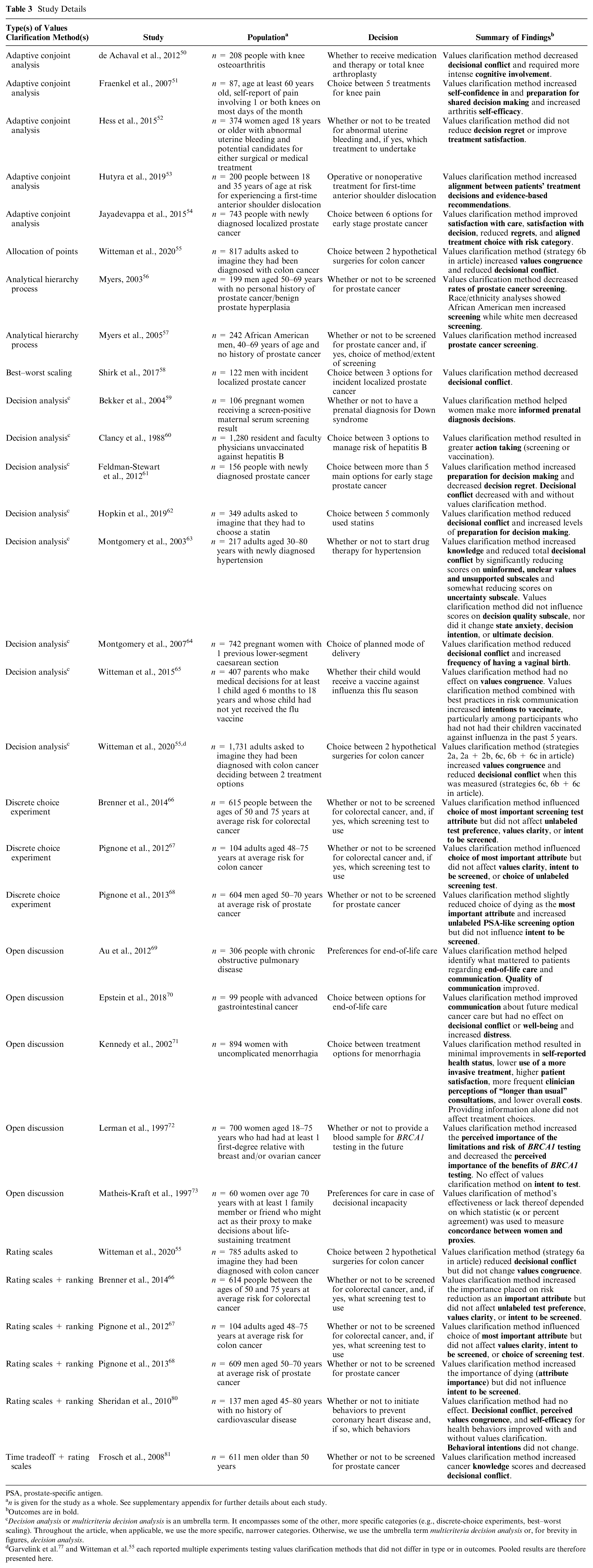
PSA, prostate-specific antigen.
Outcomes are in bold.
Garvelink et al. 77 and Witteman et al. 55 each reported multiple experiments testing values clarification methods that did not differ in type or in outcomes. Pooled results are therefore presented here.
Quality Assessment
Overall study quality was acceptable, with the majority of studies at low risk of bias on most elements. Eight studies were deemed to be at high risk of bias on 1 element, with the majority in Blinding of Participants and Personnel (Performance Bias). Eighteen additional studies were deemed unclear on this element. Blinding of Outcome Assessment (Detection Bias) was the next most common source of potential bias, with 1 study at high risk of bias and 20 more unclear. Full details of risk of bias assessments are available in online Appendix 3.
Values Congruence
As shown in Figure 2a, included explicit values clarification methods, as a group, increased values congruence, meaning people making decisions that aligned with their stated values. Eleven out of 43 trials (26%) reported the number of people who made values-congruent or values-incongruent decisions. The pooled risk difference of making a values-incongruent decision when using one of the trialed values clarification methods was –0.04 (95% confidence interval [CI], –0.06 to –0.02;
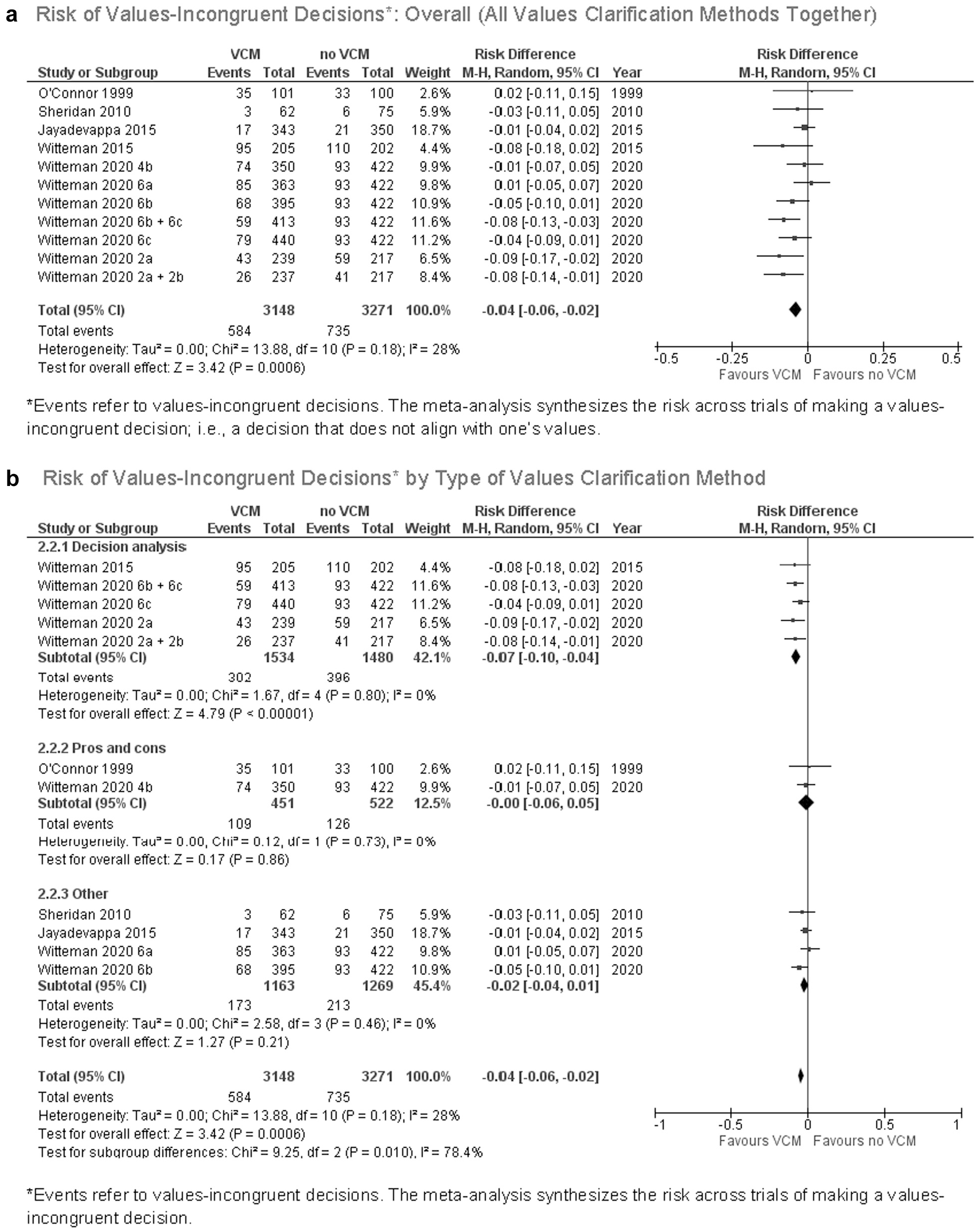
(a) Risk of values-incongruent decisions: overall (all values clarification methods together). (b) Risk of values-incongruent decisions by type of values clarification method.
Figure 2b shows a statistically significant subgroup difference by type of values clarification method. The results suggest that decision analysis is more likely to encourage values-congruent decisions compared to other explicit values clarification methods within this set of trials (χ2 = 9.25,
Decisional Conflict
As shown in Figure 3a, explicit values clarification methods decrease decisional conflict. For the 14 of 43 (33%) trials for which we had complete data, the pooled standardized mean difference for decisional conflict was –0.20 (95% CI, –0.29 to –0.11;
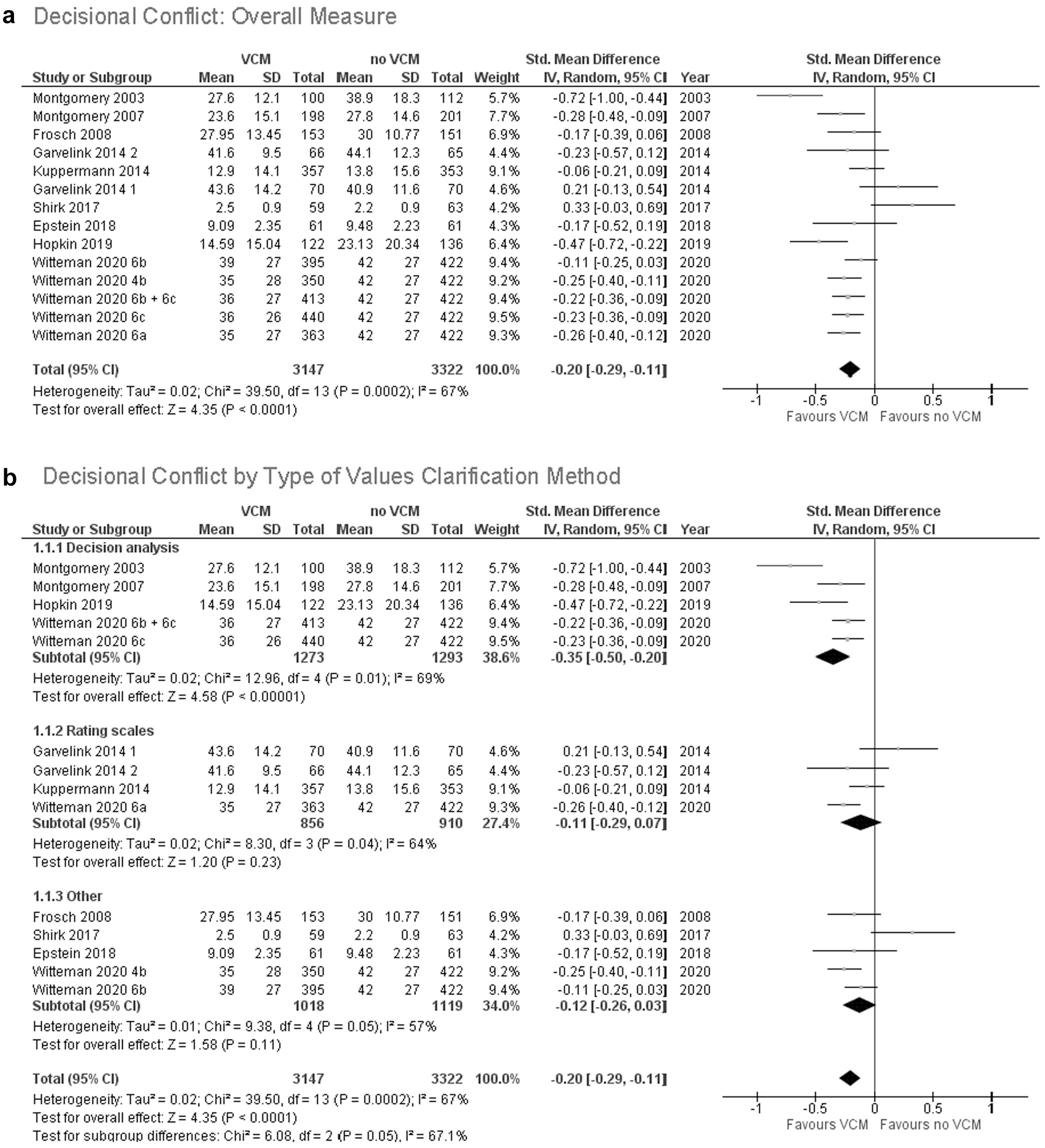
(a) Decisional conflict: overall measure. (b) Decisional conflict by type of values clarification method.
Head-to-Head Evaluations of Values Clarification Methods
The 5 studies that compared values clarification methods to each other reported findings that align with the findings of our meta-analyses. Methods that provided users with explicit feedback regarding how the decision options align with their stated values led to somewhat better outcomes, including greater values congruence. 55 When asked to compare methods to each other, study participants also preferred a values clarification method that explicitly showed them how the decision options align with their stated values. 79 Different values clarification methods yielded different patterns of attribute importance.66–68 Brief summaries of each study are available in online Appendix 3.
Discussion
Overall, our systematic review and meta-analyses confirm that explicit values clarification methods improve decision outcomes, notably by increasing values congruence and decreasing decisional conflict. Patient decision aids should include an explicit values clarification method.
While the best explicit values clarification method may depend on context—for example, urgent v. routine care or the extent to which a decision has a clear set of decision attributes—our analyses suggest that patient decision aid developers may wish to consider methods that draw on multicriteria decision analysis. The apparent advantages of such methods shown in our analyses may reflect similarities between the process and the outcome. In other words, increased values congruence yielded by decision-analytic methods may be a function of the ways in which such methods transparently show people how their options align with their stated values. We also caution that when these methods use prespecified attributes, there might not be the flexibility for users to add new attributes, highlighting the importance of research to inform attribute selection. We acknowledge that some researchers have argued that health professionals having an unhurried, high-quality conversation with patients may be a preferred approach for at least some patients, especially when decision attributes are many and varied. However, in this systematic review, trials of Open Discussion values clarification methods did not demonstrate strong results, suggesting that such an ideal may be difficult to achieve.
To advance further knowledge on the merits and pitfalls of different values clarification methods, we recommend that authors of future trials of values clarification methods report 4 outcomes: decisional conflict, decision or decision intention, values congruence, and decisional regret. When possible, authors should make use of validated scales that have good psychometric properties and are commonly reported, as this facilitates evidence synthesis.
Decisional conflict should be assessed before people make the decision, using a version of the Decisional Conflict Scale.46,83 Decisions or decision intentions should be assessed when the decision is made.
Values congruence should be assessed once the decision is made. We acknowledge that including values congruence as an outcome brings both measurement and conceptual issues. Measurement issues exist because there are disagreements about how to measure what matters to people (or, indeed, whether it is conceptually possible to do so) and compare such measures to what people choose. 84 Values congruence should not be measured using the values clarity subscale of the Decisional Conflict Scale, as this subscale measures perceived values clarity, not values congruence. 55 Further research is required to determine whether measuring values congruence might introduce bias or otherwise negatively influence decision making.
Decisional regret should be assessed with a version of the Decisional Regret Scale85,86 after people make the decision, ideally with a sufficiently long delay so that longer-term effects can be captured. This scale, whose items include, “I would go for the same choice if I had to do it over again,” assesses how people feel about the decision itself, not the decision process. An included study in this review showed that a values clarification method reduced decisional regret but only after a year had passed following implementation of the decision. 61
For all 4 measures, authors should clearly report sample mean and sample standard deviation for continuous measures, numbers in each category for categorical measures, and sample size per study arm in all cases. Finally, we recommend that patient decision aid developers explain the rationale for their choice of values clarification method.
Our study has 4 main limitations. First, the included data were of moderate quality. Although this review includes many robust trials, the included studies often measured different outcomes or the same outcomes in different ways, there were missing data in some studies, some studies had high risk of bias (often because it was not possible to prevent study participants from ascertaining the study arm to which they were assigned), and some of our meta-analyses had high heterogeneity. Together, these issues suggest a degree of caution in our conclusions. Second, we did not distinguish between subtypes of values clarification methods. For example, different adaptive conjoint analysis exercises may be very different from each other, as might open discussions or many other values clarification methods we grouped together, particularly those we grouped under the broad umbrella term of
Our study also has 3 main strengths. First, we catalog definitions and resources regarding values clarification methods, as well as recommended outcomes to report in studies. In doing so, we hope to offer more clarity and structure to a literature that can be confusing to navigate, particularly for those who are newer to developing patient decision aids. Second, we begin to answer a core question that commonly arises when developing a patient decision aid: when including a values clarification method, which type of method should one use? Third and finally, we used rigorous methods and an expansive, systematic search. By conducting a systematic review, we reduced our likelihood of missing relevant studies. By including meta-analyses, we offer stronger findings and recommendations than would be possible without pooling data across multiple studies.
In conclusion, particularly in contexts in which people may make health decisions unaligned with what matters to them, patient decision aids should include an explicit values clarification method. Patient decision aid developers may wish to consider the potential advantages of multicriteria decision analysis. Future research should further investigate which methods lead to the best outcomes across or within particular decisions, populations, and settings. Authors of randomized controlled trials of explicit values clarification methods should report decisional conflict, decision made, values congruence, and decisional regret.
Supplemental Material
sj-doc-1-mdm-10.1177_0272989X211037946 – Supplemental material for Clarifying Values: An Updated and Expanded Systematic Review and Meta-Analysis
Supplemental material, sj-doc-1-mdm-10.1177_0272989X211037946 for Clarifying Values: An Updated and Expanded Systematic Review and Meta-Analysis by Holly O. Witteman, Ruth Ndjaboue, Gratianne Vaisson, Selma Chipenda Dansokho, Bob Arnold, John F. P. Bridges, Sandrine Comeau, Angela Fagerlin, Teresa Gavaruzzi, Melina Marcoux, Arwen Pieterse, Michael Pignone, Thierry Provencher, Charles Racine, Dean Regier, Charlotte Rochefort-Brihay, Praveen Thokala, Marieke Weernink, Douglas B. White, Celia E. Wills and Jesse Jansen in Medical Decision Making
Supplemental Material
sj-pdf-1-mdm-10.1177_0272989X211037946 – Supplemental material for Clarifying Values: An Updated and Expanded Systematic Review and Meta-Analysis
Supplemental material, sj-pdf-1-mdm-10.1177_0272989X211037946 for Clarifying Values: An Updated and Expanded Systematic Review and Meta-Analysis by Holly O. Witteman, Ruth Ndjaboue, Gratianne Vaisson, Selma Chipenda Dansokho, Bob Arnold, John F. P. Bridges, Sandrine Comeau, Angela Fagerlin, Teresa Gavaruzzi, Melina Marcoux, Arwen Pieterse, Michael Pignone, Thierry Provencher, Charles Racine, Dean Regier, Charlotte Rochefort-Brihay, Praveen Thokala, Marieke Weernink, Douglas B. White, Celia E. Wills and Jesse Jansen in Medical Decision Making
Supplemental Material
sj-pdf-2-mdm-10.1177_0272989X211037946 – Supplemental material for Clarifying Values: An Updated and Expanded Systematic Review and Meta-Analysis
Supplemental material, sj-pdf-2-mdm-10.1177_0272989X211037946 for Clarifying Values: An Updated and Expanded Systematic Review and Meta-Analysis by Holly O. Witteman, Ruth Ndjaboue, Gratianne Vaisson, Selma Chipenda Dansokho, Bob Arnold, John F. P. Bridges, Sandrine Comeau, Angela Fagerlin, Teresa Gavaruzzi, Melina Marcoux, Arwen Pieterse, Michael Pignone, Thierry Provencher, Charles Racine, Dean Regier, Charlotte Rochefort-Brihay, Praveen Thokala, Marieke Weernink, Douglas B. White, Celia E. Wills and Jesse Jansen in Medical Decision Making
Supplemental Material
sj-xlsx-1-mdm-10.1177_0272989X211037946 – Supplemental material for Clarifying Values: An Updated and Expanded Systematic Review and Meta-Analysis
Supplemental material, sj-xlsx-1-mdm-10.1177_0272989X211037946 for Clarifying Values: An Updated and Expanded Systematic Review and Meta-Analysis by Holly O. Witteman, Ruth Ndjaboue, Gratianne Vaisson, Selma Chipenda Dansokho, Bob Arnold, John F. P. Bridges, Sandrine Comeau, Angela Fagerlin, Teresa Gavaruzzi, Melina Marcoux, Arwen Pieterse, Michael Pignone, Thierry Provencher, Charles Racine, Dean Regier, Charlotte Rochefort-Brihay, Praveen Thokala, Marieke Weernink, Douglas B. White, Celia E. Wills and Jesse Jansen in Medical Decision Making
Footnotes
Acknowledgements
We thank Frédéric Bergeron, MLIS, for assistance with search strategy, Caroline Beaudoin for assistance in resolving article counts, and all authors of the original articles who generously gave their time to provide missing data when we were unable to extract the data needed from their papers.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Financial support for this study was provided in part by the Canadian Institutes of Health Research (CIHR) FDN-148426 (principal investigator: HOW). HOW receives salary support from Tier 2 Canada Research Chair in Human-Centred Digital Health and received salary support during this study from a Fonds de Recherche du Québec-Santé (FRQS) Research Scholar Junior 2 Career Award. The funding agreements ensured the authors’ independence in designing the study, interpreting the data, writing, and publishing the article.
