Abstract
The need to identify therapies capable of increasing collateral blood flow after stroke has recently come into focus. This study examined the idea of repurposing an already FDA-approved drug for a new indication—inducing collaterals to remodel to larger diameters after acute stroke. We tested cilostazol, an agent whose molecular mechanism of action suggested such a capability. Importantly, cilostazol is currently used to reduce the incidence of recurrent stroke and transient ischemic attack. In mice with abundant collaterals at baseline, 7-day administration of cilostazol at the dose used clinically beginning 2 h after obstruction of the MCA, boosted collateral growth, that is, anatomic lumen diameter, such that the increase in relative collateral flow post-stroke was augmented by 31% (p = 0.004) 48 h after occlusion. The effect was stronger in mice with poor collaterals, 275% (p = 0.018), in association with a significant decrease in infarct volume and improved functional outcome. Moreover, when combined with an experimental drug that acts by a complimentary mechanism vis-a-vis cilostazol, mice with poor collaterals experienced an additive 261% (p = 0.007) additional increase 7 days post-occlusion. Cilostazol’s augmentation of remodeling extended to mice with advanced age or diabetes. This study provides a basis for further pre-clinical as well as clinical investigation.
Introduction
Stroke is the second leading cause of death and a major cause of serious long-term disability worldwide. 1 Currently, the most effective treatments for ischemic stroke have a limited therapeutic time window: generally, 4.5 h for thrombolysis and up to 24 h for thrombectomy. 2 Moreover, the recanalization rate is only 20%–30% for thrombolysis and 70%–85% for thrombectomy, and ~50% of patients with anterior large-vessel stroke who receive the latter still suffer significant disability.3–5 Blood flow mediated by pial collaterals is a major factor that lessens stroke severity by limiting early core volume, maximizing penumbra, reducing infarct progression, increasing the effectiveness of thrombolysis and thrombectomy, and decreasing the incidence of recurrent stroke. Unfortunately, collateral flow varies greatly in humans and mice.6,7 This has been shown in mice to extend primarily from genetic-dependent differences in the number and diameter of collaterals that form during development (i.e. native collateral extent) and are thus present in the adult. 6
Non-genetic factors are also capable of altering collateral extent. 7 In particular, pial collaterals undergo a remarkably rapid remodeling process that increases their anatomic lumen diameter and enhances collateral flow.7,8 Remodeling begins within hours after middle cerebral artery occlusion (MCAO) in C57BL/6 mice—the reference strain for the Mus musculus species: diameter increases ~50% by 36 h and reaches maximum by 72 h (~2-fold increase); this is three- to four-fold faster than the rate of remodeling of collaterals in non-brain tissues, for example, in mouse hindlimb after femoral artery ligation.7,8 Such rapid remodeling is especially advantageous since penumbra tissue can persist for several days after obstruction. A number of factors have been proposed to underlie the robust remodeling evidenced by pial collaterals. 7
Rapid remodeling may also occur in humans. Indirect evidence comes from the protection afforded by growth of spinal cord collaterals that is seen within 24 h on staged segmental artery occlusion, a surgical procedure used to repair thoracic aortic aneurysms that reduces the risk of subsequent paraplegia. 7 The above findings, together with the exponential fourth power relationship between vessel radius and blood flow, suggest that identifying a treatment capable of augmenting collateral remodeling could provide an important adjunctive therapy to thrombolysis and/or thrombectomy in acute ischemic stroke (AIS), as well as for the large number of patients unable to receive these treatments.
Cilostazol (CLZ) is an inhibitor of phosphodiesterase III (PDE3) that exhibits pleiotropic actions.9,10 It is approved in Japan and several other Asian countries for intermittent claudication (IC) in patients with peripheral arterial disease (PAD), reduction in risk of recurrent (secondary) stroke, transient ischemic attack (TIA), and in the United States and elsewhere for treatment of IC.9,10 It is also used off-label for Raynaud’s disease 11 and refractory variant/vasospastic angina. 12 Inhibition of PDE3 by CLZ increases intracellular cyclic adenosine monophosphate (cAMP), which activates downstream effectors leading to inhibition of platelet aggregation and vasodilation, the latter via enhanced production of endothelial cell (EC) nitric oxide (NO) and inactivation of myosin-light chain kinase.9,10,13 Cilostazol also lowers triglycerides, elevates high density lipoprotein cholesterol, and reduces coronary in-stent restenosis. 14
Previous studies reported that CLZ reduced infarct volume and improved functional recovery in rodent models of AIS.15–21 However, collaterals were not evaluated. Also, administration often preceded the incident occlusion—timing that does not simulate use of a drug during the acute phase of stroke. Herein, we hypothesized that CLZ augments the remodeling process activated by arterial occlusion. To simulate a potential clinical dose-schedule, we administered CLZ 2 h after permanent MCAO (pMCAO) and then once daily thereafter for 7 days at the dose used to treat the aforementioned indications. Mice with genetic background-dependent differences in collateral extent were studied to model patients with either “poor” or “good” collaterals. In addition, mice with advanced age or diabetes were also tested since these are common comorbidities in stroke.
Methods
Animals
C57BL/6J (B6) and B6.Rabep2 knockout (KO; C57BL/6J background 22 ) mice were obtained from laboratory colonies rejuvenated at ~2-year intervals. The Collaborative Cross (CC) strain, CC016, were from the Systems Genetics Core Facility at the University of North Carolina, Chapel Hill (UNC). B6.BKS(D)-Leprdb/J Type II diabetic mice (#000697) were from The Jackson Laboratory. Three and 24 months old C57BL/6J, Rabep2-KO, and CC016 mice, and 18 months old Rabep2-KOs were used. Diabetic mice were aged to 8 months. Mice of both sexes in approximately equal numbers were studied. All procedures were approved by the Institutional Animal Care and Use Committee (IACUC) of the University of North Carolina and adhered to the NIH Guide for the Care and Use of Laboratory Animals (IACUC #23-200).
Permanent middle cerebral artery occlusion, CLZ dosing, infarct volume
pMCAO was performed as described. 23 Briefly, mice were anesthetized with ketamine (100 mg/kg, i.p.) and xylazine (10 mg/kg, i.p.), a 1 mm diameter craniotomy was made over the right distal M1-MCA, the vessel was cauterized just-distal to the lenticulostriate branches, and buprenorphine (0.1 mg/kg) was administered every 12 h for 2 days. Rectal temperature was monitored and maintained at 37 °C with a heating pad and lamp during and after surgery until animals regained consciousness and received the first dose of CZL 2 h after pMCAO.
Cilostazol (OPC-13013, #S1294) was from Selleckchem (Houston, TX, USA). Polyethylene glycol 300 (#202371), Tween 80 (#P4780), and 2,3,5-triphenyltetrazolium chloride (TTC, #T8877) were from Millipore–Sigma (St. Louis, MO, USA). Cilostazol was dissolved in dimethyl sulfoxide to make an 18 mg/mL stock solution, which was then diluted for the final formulation: 5% CLZ stock + 40% PEG300 + 5% Tween 80 + 50% water. Cilostazol was administered at 3 or 10 mg/kg i.p. 2 h after pMCAO and then once daily. Vehicle consisted of the same formulation minus CLZ. The above information for dipyridamole (non-selective PDE inhibitor) and BI-749327 (transient receptor potential canonical 6, TRPC6, inhibitor) is in Supplemental Material. Anesthesia was not used during administration of CLZ, dipyridamole, BI-749327, or vehicle.
Infarct volume was measured on day 2 or 7 after pMCAO. Eight forebrain coronal slices (1 mm thicknesses) were stained with 1% TTC in phosphate-buffered saline (PBS) at 37 °C. Average infarct area was determined as the mean of the dorsal- and ventral-imaged lesion areas of each slice which were combined to obtain a directly measured lesion volume. Images were corrected for edema 24 : the corrected volume equals the directly measured lesion size times a factor (1 − C), where C is the proportion contributed by edema swelling; the latter is the difference in hemisphere volumes as a fraction of the contralateral side. To normalize for variation in forebrain volume among individuals, the corrected lesion volume was then divided by total forebrain volume to obtain “infarct volume, in units of percent of forebrain volume,” where total forebrain volume was determined in the same manner from the slices as described above for lesion volume.
Angiography
Heparinized mice were perfused at 100 mmHg with PBS containing freshly prepared sodium nitroprusside (10−4 M, maintained on ice in the dark) to induce maximal vasodilation. 23 Following rapid craniotomy to expose the neocortex, Evans blue dye (50 mg/mL in PBS) was perfused before infusion of Microfil® (Flowtech, Inc., Carver, MA, USA) under a stereomicroscope to ensure absence of air bubbles and complete filling of the arterial and capillary beds. Paraformaldehyde was applied to the pia, brains fixed overnight, and collaterals imaged within 24 h. Morphometry was conducted on all pial collaterals interconnecting the anterior cerebral (ACA) and MCA trees in both hemispheres. Collateral remodeling was assessed by measuring the average lumen diameter of each collateral to obtain a within-animal average.
Behavioral studies
Rotarod and grip strength tests were performed by the UNC Mouse Behavioral Core. For the rotarod test, mice were trained daily 1 week before pMCAO. The adhesive-tape-contact-and-removal test was conducted in a 30 × 30 inch 2 open field arena, with training done 1 day before pMCAO. A 3 × 4 mm² piece of tape was applied to the contralateral forepaw. The time to first contact and the time to removal were recorded. Trials were repeated in triplicate at 5-min intervals. 25
Statistical analysis
Study design and statistical analyses adhered to the ARRIVE and RIGOR guidelines.26,27 No data points were excluded as outliers. All measurements were performed by investigators blinded to treatment groups. Data are expressed as mean ± SD. Sample sizes (n) and statistical tests are given in the figure legends. Data were assessed for normality using the D’Agostino–Pearson omnibus test when n > 7, and either the Kolmogorov–Smirnov or Shapiro–Wilk test when n < 7 (GraphPad Prism). An exception was the Rabep2-KO group treated with CLZ on day 7, which missed significance for normality (p = 0.09).
Results
To evaluate whether CLZ augments collateral remodeling and whether its efficacy varies in mice with differences in collateral extent, we first studied the C57BL/6 strain. These mice have abundant pial collaterals with relatively large diameters and thus model patients with “good” collateral flow.6,8 We also studied Rabep2-KO mice (C57BL/6 background) that have fewer collaterals with smaller diameters as a model of patients with “poor” collateral flow.6,22 Additionally, we examined CC016 mice that also have “poor” collaterals. This strain has maximal genetic diversity obtained by crossing five classical laboratory strains and three wild-derived strains; it thus better models “outbred” humans with poor collateral flow.6,28 Number of collaterals per hemisphere averaged ten in C57BL/6 and four each in Rabep2-KO and CC016 mice, as previously reported. 28
Remodeling was not uniform along the length of the collateral early during remodeling, for example, on day 2. To obtain a more precise measurement of overall lumen diameter than the standard method of determining collateral diameter at mid-point or averaging two to three diameters taken along the collateral’s length, we used an area-derived method (Figure 1(a)). Cilostazol, alone, had no effect on diameter (Figure 1(b)). Remodeling, which reaches its maximum by 6–7 days after pMCAO, was similar to that reported previously (Figure 1(b)), 85% and 146% increase for C57BL/6 and Rabep2-KO mice, respectively.8,22,28,29
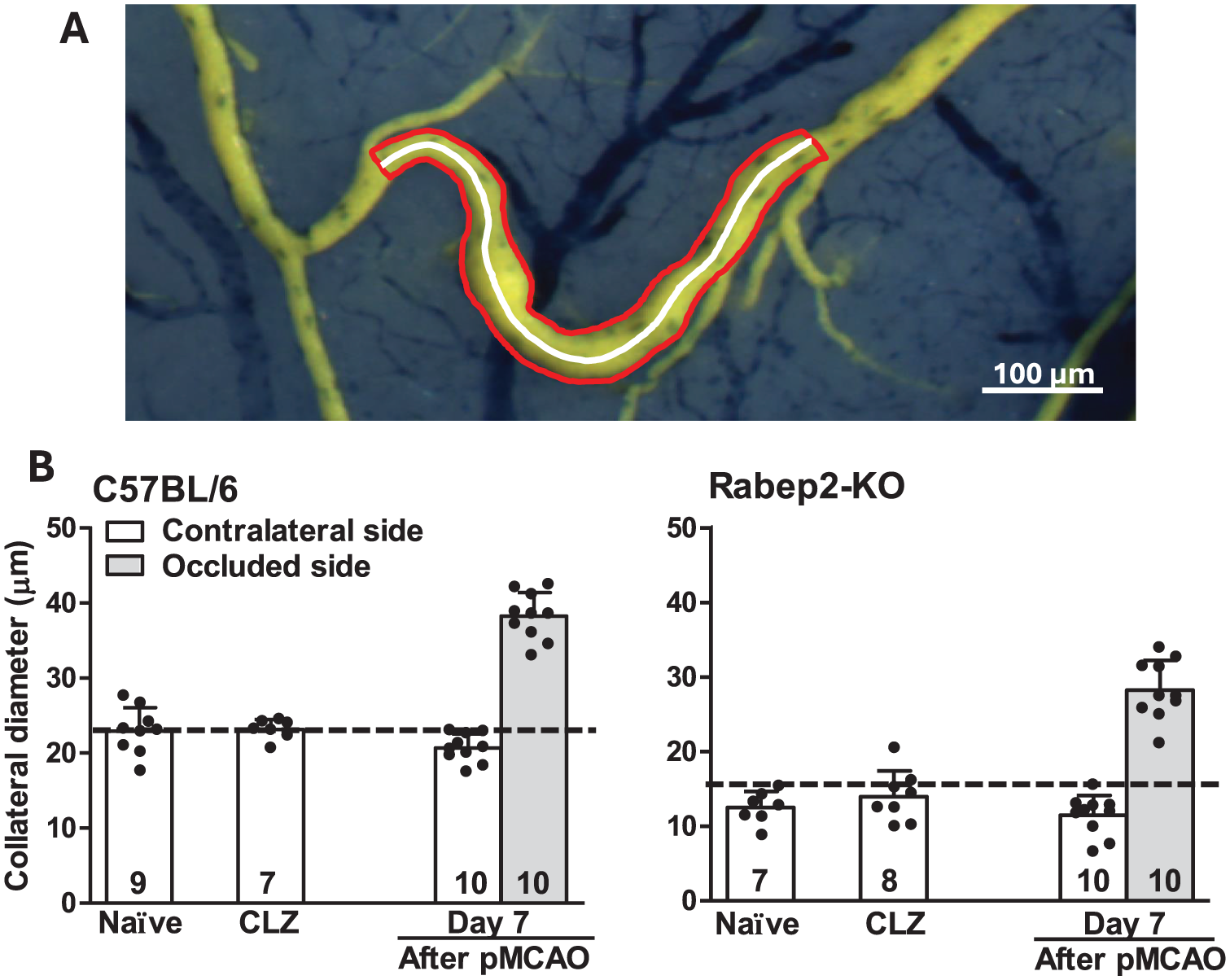
CLZ, alone, in absence of pMCAO, had no effect on diameter of ACA-to-MCA collaterals. (a) Day 2 after pMCAO. In this and subsequent figures, average lumen diameter was calculated from surface area and length (from 50× images) after the vasculature was cleared with PBS + heparin, maximally dilated at ~100 mmHg pressure with nitroprusside + Evans blue PBS, fixed, and filled with latex following distal M1-pMCAO. (b) Diameter for C57BL/6 and Rabep2 knockout mice (mice with abundant (~10) large-diameter versus sparse (~4) small-diameter collaterals, respectively). Left bar-pair in each panel: naïve mice and mice that received daily CLZ (10 mg/kg/day, i.p.) for 7 days. Right bar-pair in each panel: mice that received vehicle for 7 days after pMCAO. For this and subsequent figures: values are mean ± SD, and number of 3-month-old mice are given at the base of each bar, unless indicated otherwise; approximately equally split between male and female.
Collaterals of C57BL/6 mice enlarged by day 2 by a greater absolute amount and percent of baseline diameter than Rabep2-KO and CC016 mice: 55% versus 30% and 36%, respectively (Figure 2, vehicle bars, p < 0.01). Comparison of the rate of remodeling (baseline, days 2, and 7) indicated that it also tended to be faster in C57BL/6 than the other strains (Figure 2, vehicle bars). The smaller baseline diameters and fewer collaterals in Rabep2-KO and CC016 mice predict that their collaterals are subjected to a higher (rather than lower) shear stress after pMCAO—the primary stimulus that drives remodeling 7 —compared to C57BL/6 mice. Thus, the above differences can be attributed to genetic differences, that is, absence of Rabep2 in the Rabep2-KOs, and variation at a different locus(s) in CC016 mice since CC016 and C57BL/6 mice share the same sequence for Rabep2. 28
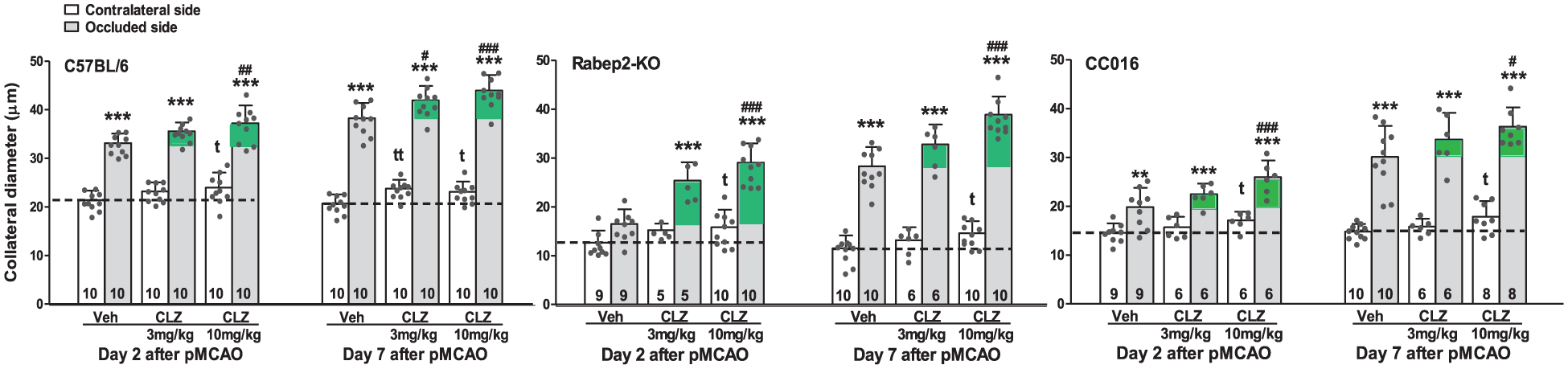
Cilostazol increased collateral remodeling after pMCAO in mice with different genetic backgrounds and collateral diameter (and number, see Results) at baseline. Cilostazol or Veh was administrated i.p. at 24 h intervals beginning 2 h after pMCAO in this and subsequent figures. Green shading, additional remodeling induced by CLZ. p < 0.0001, one-way ANOVA for each of the six-bar groupings followed by Bonferroni tests: **p < 0.01, ***p < 0.001, occluded versus contralateral side. #p < 0.05, ##p < 0.01, ###p < 0.001, CLZ versus occluded side of Veh. tp < 0.05, ttp < 0.01, t-test versus contralateral Veh group.
Cilostazol at 3 mg/kg/day tended to be less effective than 10 mg/kg/day at both time-points in C57BL/6, Rabep2-KO, and CC016 strains, respectively (Figure 2, green shading—mean ± SD microns, one-tailed t-tests; day 2: 2.5 ± 1.8 vs 4.1 ± 3.6, p = 0.11; 8.9 ± 3.8 vs 12.6 ± 3.9, p = 0.05; 2.6 ± 2.2 vs 6.1 ± 3.5, p = 0.03; day 7: 3.7 ± 3.0 vs 5.7 ± 3.2, p = 0.08; 4.5 ± 4.1 vs 10.6 ± 3.7, p = 0.004; 3.6 ± 5.5 vs 6.2 ± 3.9, p = 0.15; see Supplemental Material for additional data regarding Figure 2). C57BL/6 and Rabep2-KOs receiving 10 mg/kg/day increased their diameter by 74% ± 17% and 130% ± 31% (both p < 0.0001), respectively, on day 2, relative to contralateral collateral diameter at baseline in mice receiving vehicle (horizontal dashed lines in Figure 2). Efficacy (green shading) was greater at both time-points in Rabep2-KOs than in the other two strains (Figure 2; above “green shading” values; Supplemental Material), suggesting an influence of genetic background and other factors (see Discussion).
An unexpected finding was that, while having no effect in naïve mice (Figure 1), CLZ increased the diameter of contralateral collaterals in all three strains that had received pMCAO (Figure 2, p < 0.05 or 0.01 by t-test, p = 0.0001 by ANOVA). This was evident at both doses by day 2, significant by day 7, and confirmed in the separate control groups that were subsequently repeated contemporaneously for the aging and diabetes experiments (see below; see also Discussion regarding mechanism). This was not seen in the dipyridamole and BI-749327 experiments (see below); however n sizes were 4–6 (half the size for the aforementioned groups).
Stroke risk, morbidity, and mortality are higher in the elderly population 5 and in those with certain comorbidities including diabetes. 30 To investigate whether CLZ (10 mg/kg/day) enhances remodeling (assessed at day 7 post-pMCAO) in these conditions, we compared 3 (young-adult), 18, and 24 months old C57BL/6 and Rabep2-KO mice and 8 months old Type II db/db diabetic mice (~13, 60, 76, and 30 human-year equivalents, respectively).31,32 Remodeling was reduced in aged C57BL/6 and Rabep2-KO mice (Figure 3, vehicle bars). Cilostazol increased remodeling in aged C57BL/6 mice by 11% (Figure 3, green shading, p = 0.034 by one-tailed t-test) and by 42% in Rabep2-KOs.
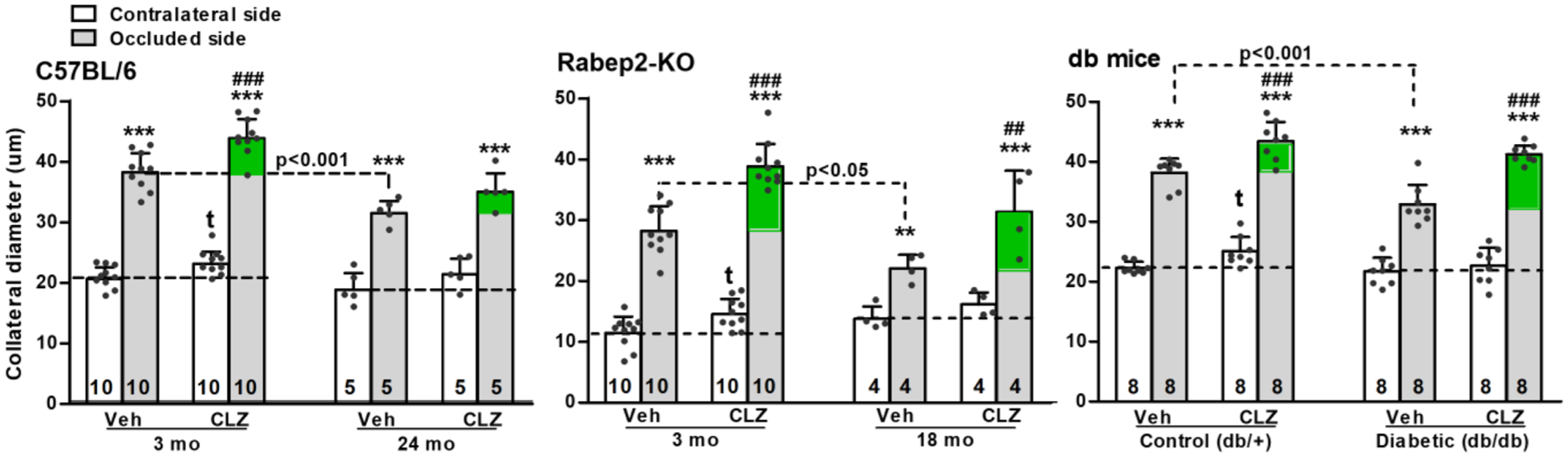
Cilostazol increased remodeling after pMCAO in aged C57BL/6, Rabep2-KO, and diabetic (db/db) mice when examined 7 days after pMCAO. Young–adult (3 months old) and aged mice (C57BL/6, Rabep2-KO), and 8 months old db/+ (normoglycemic; B6.BSK background) and db/db mice received Veh or CLZ (10 mg/kg/day). Green shading, additional remodeling induced by CLZ. p < 0.0001, one-way ANOVA followed by Bonferroni’s tests: **p < 0.01, ***p < 0.001 occluded versus contralateral side; ##p < 0.01, ###p < 0.001, CLZ versus occluded side of Veh. Remodeling was impaired in aged C57BL/6 and Rabep2-KO and in diabetic mice by 18%, 22%, and 14%, respectively (dashed-line, Bonferroni comparisons), in agreement with previous studies (see text). tp < 0.05, t-test versus contralateral Veh group.
Type II (db/db) diabetic mice have obesity, insulin resistance, moderate hyperglycemia, and dyslipidemia by 2 months age. 33 Db/db and db/+ (non-diabetic) mice had body weights of 67 ± 4 and 35 ± 3 g, and blood glucose levels of 713 ± 94 and 208 ± 23 mg/dL, respectively (mean ± SD, n = 8). Remodeling was reduced in vehicle-treated diabetic mice. Cilostazol augmented remodeling by 25% in the diabetic versus 14% in the non-diabetic mice (Figure 3).
Absolute and percentage increases in remodeling induced by CLZ were presented above. The percentage increase in calculated relative collateral flow (RCF) 34 achieved by CLZ over that seen with vehicle treatment provides a metric for the potential hemodynamic effect of remodeling, where RCF = d4 × n; d, diameter; n, average number of ACA-to-MCA collaterals for C57BL/6, Rabep2-KO and CC016 mice, that is, averaging 10, 4, and 4, respectively (see above). Accordingly, CLZ at 3 and 10 mg/kg/day on day 2 and 10 mg/kg/day on day 7, respectively, increased RCF by 31% ± 49%, 38% ± 49%, 36% ± 39%, and 76% ± 41% (p = 0.038, 0.046, 0.004, 0.0025, respectively; mean ± SD, one-tailed t-tests compared to vehicle) for C57BL/6; by 275% ± 198%, 719% ± 497%, 84% ± 112%, and 162% ± 122% (p = 0.018, 0.0008, 0.063, 0.002) for Rabep2-KO; by 60% ± 133%, 175% ± 108%, 34% ± 107%, and 48% ± 89% (p = 0.15, 0.006, 0.240, 0.085) for CC016, and by 132% ± 151%, 387% ± 295%, 55% ± 106%, and 98% ± 103% (p = 0.008, 0.0001, 0.048, 0.0006) for the mean of the latter two poor-collateral strains. The increase in RCF was greater in the poor-collateral strains, which is congruent with their greater amount of remodeling (Figure 2). However, confirmation of this conclusion regarding CLZ promoting greater remodeling in poor-collateral strains, in general, is needed in a larger cohort of strains with wide genetic-dependent differences in collateral extent (e.g. strains selected from the CC panel 6 ). Cilostazol at 10 mg/kg/day increased RCF by 46% ± 28%, 286% ± 283%, and 108% ± 61% (p = 0.01, 0.14, 0.0008) by day 7, respectively, for 24 months old C57BL/6, 18 months old Rabep2-KOs, and 8 months old diabetic mice.
Cilostazol decreased infarct volume on days 2 and 7 by 30% and 53% in C57BL/6 and by 19% and 21% in Rabep2-KO mice (Figure 4(a)). Of note, while infarct volume is known to increase in a time-dependent manner between 1.5 and 24 h after MCAO, 35 infarct volume was smaller on day 7 than 2. For example, infarct volumes in the vehicle groups were smaller by an additional 37% in C57BL/6 and 41% in Rabep2-KOs on day 7 compared to day 2. This likely reflects atrophy of infarcted tissue being evident by day 7. 36 Others have reported that infarct volume decreased by 47% from days 3 to 7 after MCAO, 37 an amount comparable to what we observed in the above vehicle-treated groups. It should also be mentioned that we corrected for edema using the widely-used method 24 wherein swelling is determined as a fraction of the total hemisphere, with the measured lesion volume being reduced by that percentage. In the distal M1-pMCAO model used herein, edema would be limited to the infarct and peri-infarct territory, which would represent a greater proportion of the uncorrected measurement. Although edema is modest with this model, compared to more proximal occlusion models, our infarct measurements may be an over-estimate at the 2-day time point. Also, since infarct progression is highly model-dependent, our days 2 and 7 findings cannot be compared to studies using other occlusion methods.
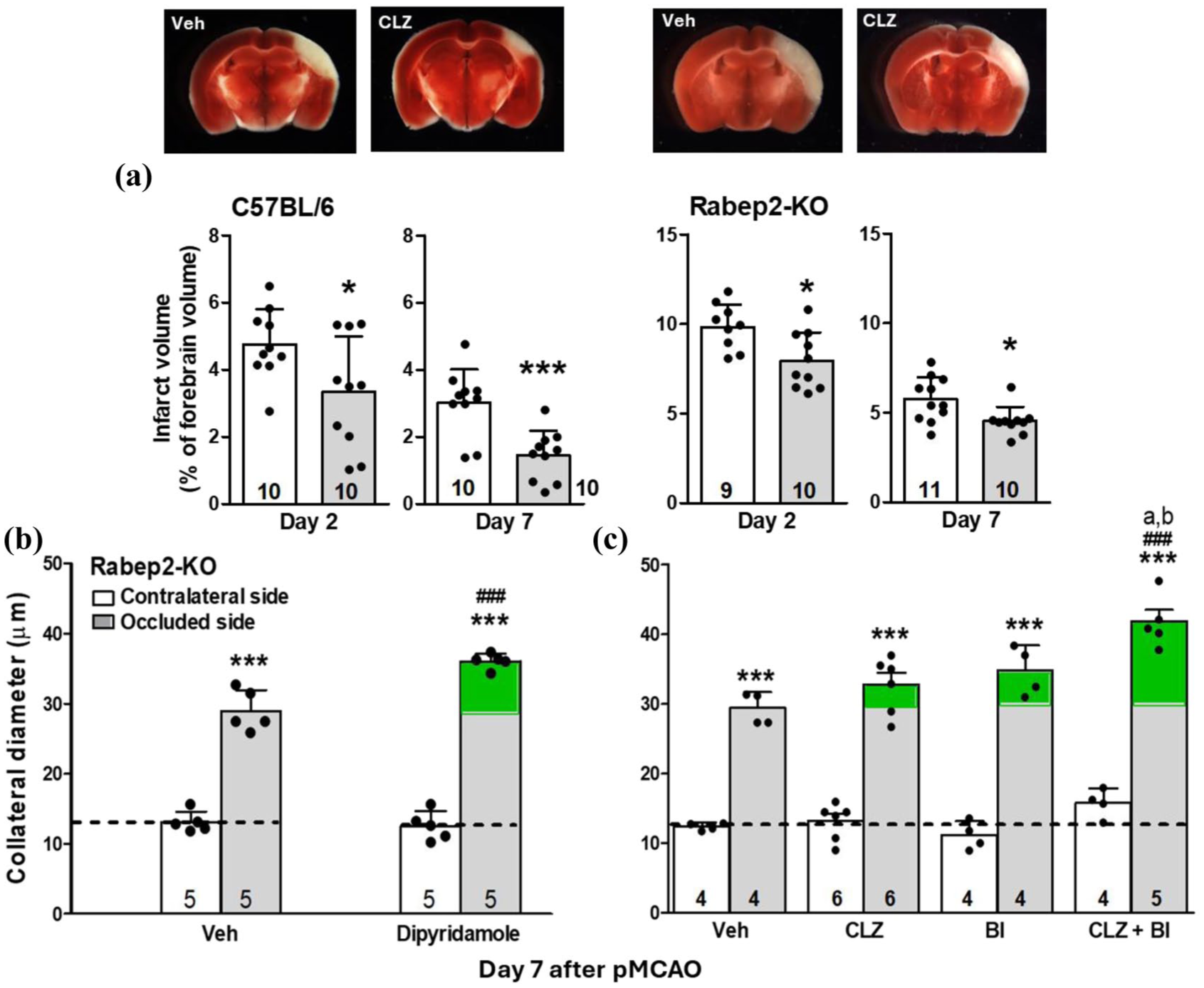
Cilostazol reduced infarct volume, dipyridamole augmented remodeling, and a TRPC6 inhibitor increased remodeling in Rabep2-KO mice and acted additively with cilostazol to enhance remodeling. (a) Cilostazol 10 mg/kg given i.p. 2 h after pMCAO and daily thereafter (gray bars); images are for day 2 after pMCAO. *p < 0.05, ***p < 0.001, t-test versus vehicle (white bars; p = 0.033, 0.0008, and 0.011, 0.015 at days 2 and 7, respectively). (b) Non-selective PDE inhibitor, dipyridamole, 200 mg/kg/day given orally 2 h after pMCAO and BID thereafter. Green shading, additional remodeling induced by dipyridamole. p < 0.0001, one-way ANOVA followed by Bonferroni test. ***p < 0.001, occluded versus contralateral side of vehicle-treated group. ###p < 0.001, dipyridamole versus ipsilateral side of vehicle. (c) TRPC6 inhibitor, BI-749327 (BI) 30 mg/kg/day given orally 2 h after pMCAO and daily thereafter, CLZ 3 mg/kg/day given i.p. BI green shading, additional remodeling induced by agents shown on abscissa. One-way ANOVA followed by Bonferroni test, ***p < 0.001, occluded versus contralateral side of vehicle-treated group. ###p < 0.001, versus ipsilateral side of vehicle. ap < 0.01 versus ipsilateral side of BI. bp < 0.001 versus ipsilateral side of CLZ.
Cilostazol reduced infarct volume less, on a percentage basis, in Rabep2-KO than C57BL/6 mice (Figure 4(a))—a finding, that is, not congruent, on first-thinking, with the greater remodeling seen in Rabep2-KOs (Figure 2). This can be attributed at least in part to absolute infarct volume being greater in the Rabep2-KO (Figure 2), which is a consequence of their fewer collaterals of smaller diameter at baseline. In addition, salvageable penumbra in poor-collateral mice is reduced, 7 favoring less at-risk tissue being available for protection from loss during CLZ’s augmentation of collateral remodeling.
Behavioral tests were conducted on days 2 and 7 in Rabep2-KO mice treated with 10 mg/kg/day CLZ, since CLZ caused the greatest relative amount of collateral remodeling in young-adult and aged mice of this strain on both days (Figures 2 and 3). Adhesive tape contact and removal times were reduced, indicating improvement in motor and sensorimotor functions (Figure 5). Although rotarod performance did not reach significance under the stringent one-way Kruskal-Wallis ANOVA test, CLZ-treated mice demonstrated longer latency to failure (p = 0.037, one-tailed t-test) on day 2, suggesting that CLZ reduced deficits in equilibrium and locomotor ability. The grip strength test detected no differences. It is worth noting that distal M1 pMCAO causes small cortical infarctions (Figure 4), in distinction to the often-used transient or permanent filament MCAO models or those that also employ carotid occlusion, which produce large, often hemispheric infarctions and large amounts of edema. Hence, behavioral deficits are also small following M1 pMCAO, making any effect of CLZ difficult to detect despite our use of relatively robust n sizes (Figure 5).
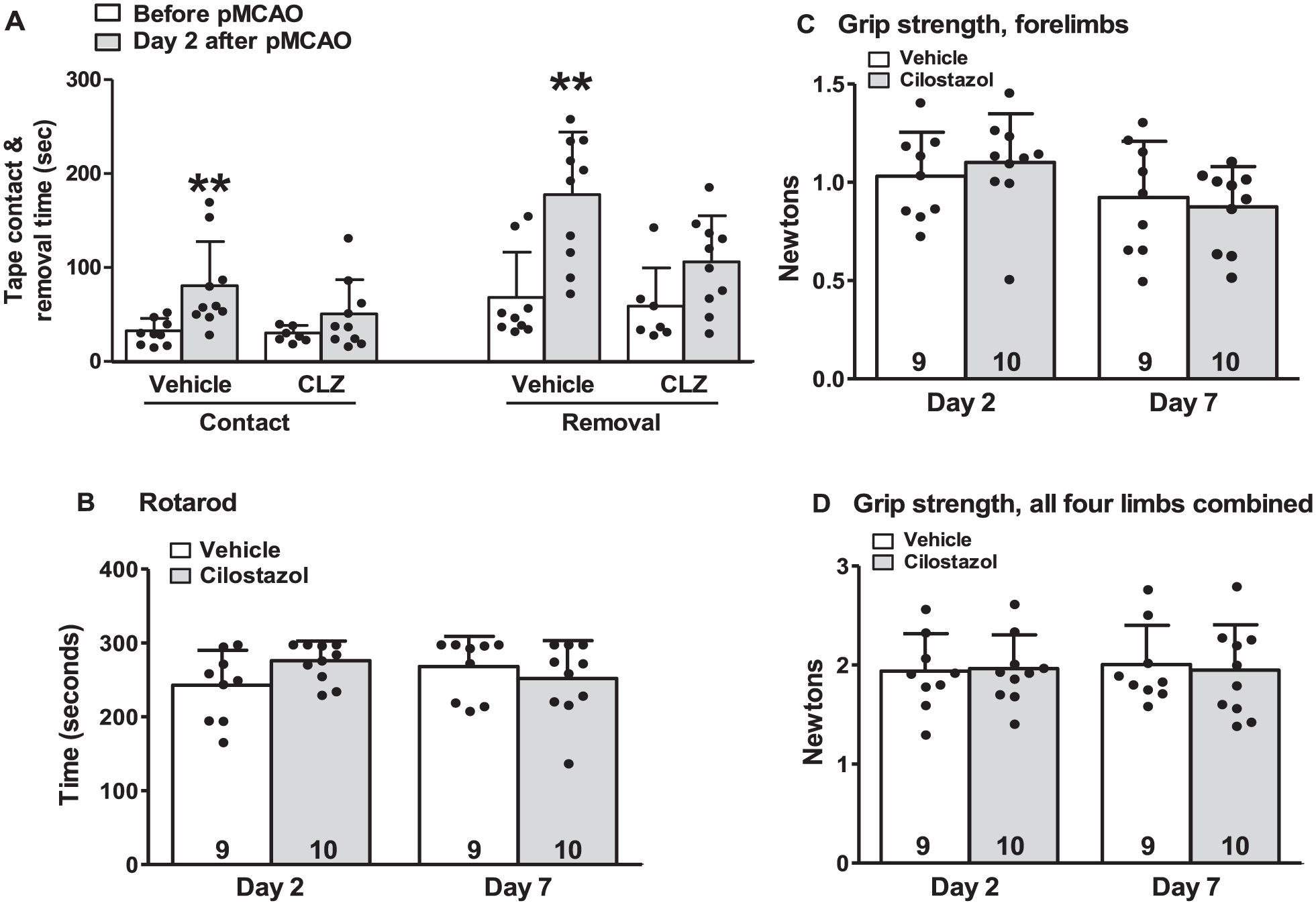
Cilostazol improved sticky tape contact and removal test scores after pMCAO (a). (b–d) Mice are examined 2 and 7 days after pMCAO. Three months old Rabep2-KO mice received CLZ at 10 mg/kg/day i.p. N sizes for bars in (a): 9, 10, 7, 10, 9, 10, 7, 10. p < 0.01, one-way Kruskal–Wallis ANOVA; **p < 0.01, post-hoc Dunn’s test.
To investigate the mechanism by which CLZ enhances collateral remodeling, we tested dipyridamole. Dipyridamole inhibits PDE3, PDE5, and other PDEs (CLZ also inhibits PDE5 but significantly less than PDE3),38,39 thereby increasing intracellular cAMP and cGMP in platelets, ECs, and SMCs 9 and enhances endothelium-dependent NO/cGMP-mediated signaling. 40 Thus, dipyridamole’s dual cAMP/cGMP actions could theoretically promote greater remodeling, compared to CLZ which only indirectly favors elevated cGMP (see Discussion). We administered 200 mg/kg 2 h after pMCAO and then twice daily, 41 which is significantly higher than the dose used for secondary stroke prevention (200 mg BID). 9 Dipyridamole increased remodeling by 174% in Rabep2-KOs (Figure 4(b)), which was <186% increase observed with CLZ at the same time-point (day 7, Figure 2). Thus, increasing cGMP, in addition to cAMP, did not augment collateral remodeling more than that achieved with CLZ.
Since CLZ has recently been shown to inhibit TRPC6 ion channel activity, 42 we investigated whether inhibition of TRPC6, alone, enhances remodeling. We administered the TRPC6 inhibitor, BI-749327, (BI, 30 mg/kg/day 43 ) to Rabep2-KOs 2 h post-pMCAO and daily thereafter. At day 7, BI augmented remodeling by an amount comparable to that achieved with CLZ at 3 mg/kg/day (179% vs 163%; Figure 4(c)). Notably, when BI and CLZ were co-administered, remodeling increased to 236% (Figure 4(c)), a level similar to that induced by CLZ at 10 mg/kg/day alone (Figure 2). These findings suggest that a portion of CLZ’s action to enhance remodeling is mediated by inhibition of TRPC6 and—importantly—demonstrate that treatment with CLZ and a drug that acts by a different mechanism to promote remodeling (see Discussion) produce an additive effect.
Discussion
Cerebro-protective effects for many agents have been reported in pre-clinical stroke studies. However none has been adopted into clinical use. Recently, the possibility of increasing collateral blood flow post-stroke has become of interest.3,7,44 Among the determinants of collateral flow, 7 a treatment that could enhance the pace and/or amount of collateral remodeling could prove especially effective. The aim of our study was to test cilostazol for such an effect. We were motivated by (discussed further below): (1) preclinical studies showing it reduced infarct volume, (2) its molecular mechanisms of action that predict it could augment remodeling, (3) its long history of use and good safety profile since approval in 1998 in Japan and by the FDA and EMA in 1999 and 2002 for treatment of IC in patients with PAD, and (4) because it is approved in many Asian countries to reduce the risk of recurrent ischemic stroke and TIA and for use in certain patients with intracranial atherosclerosis (ICAS)—effects that may derive in part from augmenting collateral remodeling induced by the initial insult or by slowly developing obstruction with ICAS. Importantly, its regulatory approvals provide a significant advantage for accelerated testing in any future clinical trial examining its use in stroke.
Cilostazol’s above-mentioned indications have been attributed to its antiplatelet and vasodilatory activities.9–19,39 Whether they also could extend from augmenting remodeling has not been investigated. We found that CLZ increased remodeling in C57BL/6 mice, a strain with abundant large-diameter collaterals that models patients with good collateral flow (i.e. “good collaterals”). 7 Cilostazol also enhanced remodeling in Rabep2-KO mice (targeted single-gene disruption on the C57BL/6 background) and the CC016 strain that has a highly admixed genetic background. Both strains have sparse, small-diameter collaterals and thus model patients with poor collateral flow (“poor” collaterals). The clinical relevance of the Rabep2-KO mouse model has recently been established: we previously identified a single nucleotide polymorphism (SNP, rs11645302) in human RABEP2 linked to increased infarct volume in patients with large-vessel anterior AIS 6 ; We then shared these findings with Zhang et al. who recently confirmed this SNP’s association with poor collateral status. 45 Possible reasons for why CLZ augmented remodeling more in Rabep2-KOs compared to C57BL/6 and CC016 mice could involve differences in collateral diameter at baseline, vigor of remodeling early-on (day 2) in the absence of CLZ, other genetic-dependent differences, or a combination of these possibilities. As expected, CLZ reduced infarct volume and improved recovery of function.
Cilostazol also increased collateral remodeling in aged (~60 and ~76 human-year-equivalence31,32) and diabetic (~30 human-year-equivalence) mice. This is noteworthy because these two common comorbidities, themselves, reduced remodeling compared to the young-adult controls (~13 human-year-equivalence), which confirmed previous reports. 7 This is consistent with reduced collateral blood flow seen 7 days after pMCAO in diabetic mice. 46 Given that impaired remodeling with aging and diabetes has been attributed, at least in part, to endothelial dysfunction,7,34 it is notable that CLZ reversed the latter in aged rats 47 and improved vascular reactivity in diabetic rats. 48 Cilostazol also enhanced endothelial function in patients with ischemic stroke. 49
The non-selective PDE inhibitor and antiplatelet agent, dipyridamole, pre-dated use of CLZ for treatment of IC and recurrent stroke. 9 When we tested it at a dose considerably higher than that used for secondary stroke prevention, dipyridamole tended to increase remodeling less than CLZ. Thus, the increase in cGMP, as well as cAMP, that this agent induces 50 did not enhance remodeling more than CLZ. This may reflect the levels of cGMP and cAMP achieved and/or that several mechanosensory mechanisms that increase cGMP in the remodeling process (Figure 6, discussed below) may already be fully engaged.
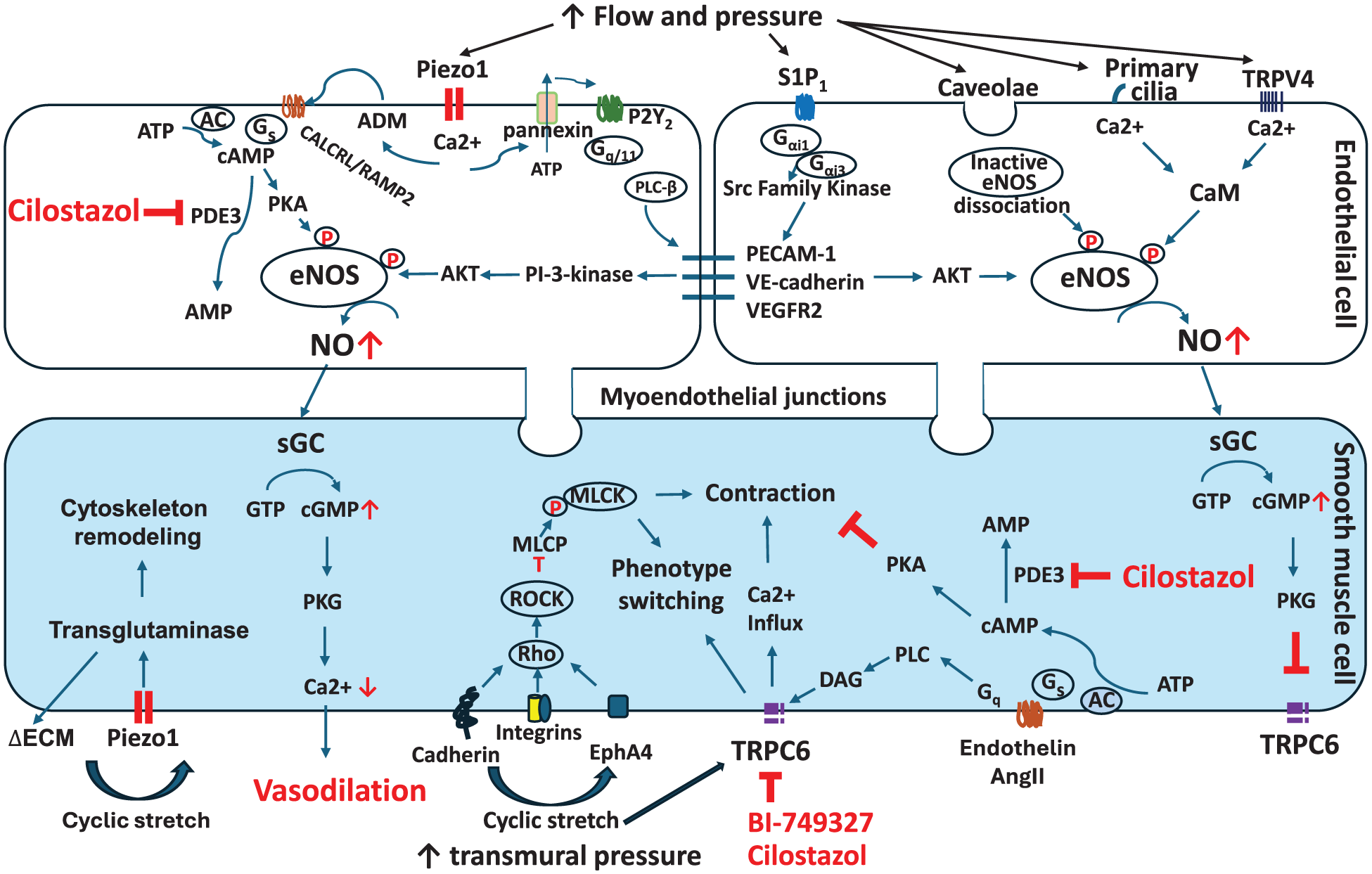
Mechanoreceptor pathways and other mechanisms potentially involved in cilostazol’s action to enhance collateral remodeling. Selected cell–surface and endothelial cell–cell junctional mechanoreceptors are shown. See Discussion for further description and Faber 7 for additional factors involved in collateral remodeling.
Collateral blood flow and shear stress increase 20-fold after occlusion, which favors flow-mediated dilation of the trees upstream from the obstructed tree.7,51 Upstream dilation increases collateral intralumenal pressure and thus circumferential (transmural) wall stress, which along with increased shear stress and the tortuous structure of collaterals, increase longitudinal wall stress. These forces are transduced by mechanoreceptors that are primarily present on ECs but also on SMCs (Figure 6; ECs have additional mechanoreceptor types that are not shown 52 ). As well, ECs and SMCs are coupled via myoendothelial junctions that mediate diffusion of ions and molecules. Several pathways are thus activated that, along with other possible mechanisms (e.g. attraction of myeloid, lymphoid and progenitor cells which contribute to remodeling in peripheral tissues), drive the remodeling process. 53 Cilostazol predictably interacts directly with two of the mechanosensory pathways, and indirectly with several others through its effects to increase cAMP and phosphorylate eNOS (Figure 6). These include inhibition of TRPC6 channels, which are activated by SMC stretch/circumferential wall stress and PLC/DAG.43,54 The resulting increase in intracellular calcium and SMC contraction opposes dilation, which is a primary driver of remodeling (see below). In support, CLZ increased blood flow recovery in the mouse hindlimb femoral artery ligation model, through phosphorylation of TRPC6 at threonine-69 which negatively regulates TRPC6 channels. 42 Activation of TRPC6 also interacts with modulation of SMCs to their non-contractile “synthetic/proliferative” phenotype, which is required for remodeling.53–55 Sustained NO release acts through multiple pathways to promote remodeling, including by suppressing TRPC6 activity 56 and promoting vasodilation and regulating SMC proliferation, apoptosis, metalloproteinases, and extracellular matrix. 57 Dilation is known to be a proximal stimulus for remodeling involving changes in extracellular matrix and rearrangement of EC and SMC cell–cell contacts, with both inward and outward remodeling evidencing a continuum of cellular changes.7,58 Remodeling has been observed within as little as 4 h. 58 The above findings suggest that CLZ could augment collateral remodeling through its direct cAMP- and indirect NO/cGMP-dependent mechanisms and also by promoting vasodilation and SMC phenotype switching through negative regulation of TRPC6.
A limitation of our study is that we did not examine remodeling and infarct volume earlier than at 48 h. However, we previously found that pial collateral diameter of C57BL/6 mice increased ~50% by 36 h, reached maximum by 72 h (~2-fold increase), 8 and that infarct volume was ~5% of forebrain volume 24 h after pMCAO 28 —findings congruent with those reported herein (Figures 2 and 4). Additional study is needed to measure diameter and infarct volume at several time points spanning the first 48 h after pMCAO. A number of studies employing distal pMCAO and TTC staining or MRI have reported that lesion volume progresses from 1.5 through as much as 72 h after occlusion.35,59–61 Since as discussed above, remodeling of arterioles can become evident within several hours 58 and flow is exponentially related to diameter, both variables may evidence similar progression. Our finding that remodeling reached 50% of maximum when examined at 36 h suggests that structural enlargement may begin before 24 h. However, determining whether a cause-and-effect relationship exists between collateral remodeling and reduction in infarct volume awaits obtaining measurements earlier than in the present study and identification of a means to prevent remodeling after pMCAO.
Given the above, we asked whether the TRPC6 inhibitor BI-749327, alone, could enhance remodeling. BI-749327 increased remodeling and RCF more than achieved with CLZ at 3 mg/kg/day (179% and 163% vs 118% and 48%, respectively). We combined BI with the lower dose of CLZ because it is equivalent (see below) to the dose used in the prevention of secondary stroke, TIA, and other indications. Dual treatment with both CLZ and BI increased remodeling and relative collateral flow 236% and 261%, respectively. This capacity for additivity is an important finding.
We also tested CLZ at a higher dose, 10 mg/kg/day, to determine whether it acts in a dose-dependent manner. This was confirmed in all three strains of mice. In humans, single-dose oral CLZ at 200 mg (2.9 mg/70 kg human; 3.5-fold less than the above dose) reached peak plasma concentration in ~3 h, with an apparent elimination half-life of ~11 h and achievement of steady state by day 4 of daily dosing.10,62 In mouse, peak plasma concentration was reached within ~1 h after intraperitoneal administration at 20 mg/kg, and half-life was estimated to be 4 h based on a one-compartment model with first-order absorption and linear elimination. 63 Therefore, we administered CLZ intraperitoneally for its faster time to peak concentration and because it avoids possible losses with intragastric administration. Cilostazol dosing was begun 2 h after pMCAO as a possible time-point for initiating administration in the clinical setting. In humans, oral CLZ at 200 mg/day had no effect on blood pressure, 64 whereas when combined with aspirin or clopidogrel, systolic blood pressure was reduced by 2–3 mmHg compared with aspirin or clopidogrel alone. 65 In mice, 10 mg/kg/day i.p. (i.p. was used herein) was accompanied by a 2–3 mmHg decrease that returned to normal within 1 h after tMCAO. 16
An unexpected finding was that remodeling occurred in the collaterals of the contralateral hemisphere of mice receiving CLZ (Figures 2 and 3). We are unaware of a previous study that tested an agent or intervention where this was reported. We hypothesize that CLZ’s actions to boost ipsilateral remodeling and collateral flow following pMCAO induces the following scenario: Cilostazol’s enhancement of ipsilateral remodeling increases flow-mediated dilation in the distal arterioles of the upstream ACA (and PCA) trees. 7 The resulting greater drop in pressure in the ipsilateral A1-ACA at the level of the anterior communicating artery causes rectification of the latter’s otherwise to-and-fro flow 66 towards the ipsilateral A1-ACA. This drops pressure in the contralateral ACA tree, which increases flow and shear stress in its collaterals, stimulating them to remodel. Why contralateral remodeling in the absence of CLZ was not observed (Figure 1) could reflect that the rate and amount of remodeling on the ipsilateral side after pMCAO at baseline is insufficient to elicit a strong enough induction of the above pressure-flow scenario to reach threshold for remodeling of the contralateral collateral network. It is also possible that changes in neuronal activity in the ipsilateral infarcted and/or peri-infarcted regions following pMCAO caused, through intracerebral connections, increased neuronal activity in the contralateral ACA territory, causing it to undergo activity-dependent vasodilation. This, in turn, would recruit collateral flow from the contralateral MCA to PCA trees, and thus stimulate remodeling. 7 New connections and activity patterns in the contralateral hemisphere after stroke have been reported in animals and humans.5,67,68 However, the several days-to-weeks that are required are not congruent with the rapid pace of contralateral collateral remodeling that we observed (i.e. significant by at least day 2). Also, why such an interhemispheric neuronal activity-based mechanism would be greater in the presence of CLZ is not evident. Interestingly, both ipsilateral and contralateral blood flow were increased in patients who received 200 mg/day CLZ for 4 weeks beginning at least 4 weeks after stroke. 69 Modified Rankin score, Brunnstrom stage, Barthel score, and Mini-Mental State Examination score were also improved.
Several previous studies using C57BL/6 mice have reported salutary effects of CLZ on stroke. However, dose and time of administration limit conclusions vis-à-vis extension to the clinical setting. Administration of CLZ at 30 mg/kg i.p. several hours before filament-occlusion of the MCA decrease infarct volume 24 h later, especially in the cortex 15 (which we note is subserved by pial collaterals, unlike subcortical regions that lack collaterals 6 ). Cilostazol given at 3 or 10 mg/kg i.p. 30 min before tMCAO or at 10 mg/kg 1–2 h after filament removal, decreased 24-h infarct volume; when administered as per the latter for 7 days, infarct volume and behavioral deficits were reduced, and peri-infarct neurogenesis and angiogenesis were increased. 16 A similar study observed increased neurogenesis in the ipsilateral subventricular area. 17 Ingestion of food containing 0.3% CLZ for 7 days before tMCAO decreased platelet-leukocyte plugging early after reperfusion, infarct volume at 24 h, and neurological deficit at 7 days. 18 Administration of CZL at 30 mg/kg i.p. 2 h before assessment did not affect CBF in the MCA territory at baseline or 1 h after filament placement for tMCAO. However, CBF was increased from 1.5-h through filament withdrawal at 2 h, in association with decreased infarct volume and neurological score obtained 22 h later. 19 Honda et al. 20 administered either 30 or 50 mg/kg CLZ at 0.5 and 4 h after pMCAO in rats. These doses are 10- and five-fold higher than the 3 and 10 mg/kg that we tested and at least 10-fold higher than the dose used clinically (see below). Thirty mg/kg decreased 24 h infarct volume by 42%; 50 mg/kg had no additional effect.
Augmentation of collateral remodeling by CLZ may also derive from downstream actions. Platelet-leukocyte adhesion and associated lumen narrowing and microclot formation are favored in the MCA tree downstream of the collaterals post-MCAO, due to ischemia, low blood flow velocity, endothelial-platelet-leukocyte activation, decreased barrier function, and other changes. 7 Cilostazol reportedly reduced ischemia-induced disruption of the blood–brain barrier and edema. 70 Studies in ischemic and hemorrhagic models of stroke support these findings in conjunction with increased eNOS/NO68,71 and inhibition of neutrophil-endothelial cell adhesion. 72 No-reflow was suppressed in the tMCAO reperfusion model when CLZ was given 7 days before occlusion. 18 Cilostazol also reportedly reduced NOX2 expression and oxidative stress. 73 Treatment of mice with 20 mg/kg CLZ at 0.5 and 24 h after pMCAO, a dose which however is seven-fold higher than used for prevention of recurrent stroke in humans, reduced infarct volume by 70% at 48 h in association with reduced NOX2 and a marker of oxidative stress. Our study did not determine whether CLZ’s actions to inhibit platelet activation and promote vasodilation, which would increase collateral flow/shear stress, contributed to its enhancement of collateral remodeling and reduction in infarct volume. Methods capable of distinguishing between a direct effect of an agent on collaterals from its downstream (or upstream) effects, which can secondarily affect collateral flow and thus remodeling, await development. 7 However, MCAO is well known to cause autoregulatory vasodilation of the MCA tree, which would diminish or eliminate additional downstream vasodilation by CLZ as a contributor to its enhancement of collateral remodeling. Nevertheless, how long after occlusion autoregulatory vasodilation remains at maximum is unclear. Also, a novel PDE3 inhibitor (“substance V”) was recently reported to reduce ischemic damage—independent of a change in platelet activity—when given before or immediately after tMCAO in mice. 74 Interestingly, platelet activation has been shown to contribute to collateral remodeling in the rodent hindlimb ischemia model by promoting leukocyte adhesion (primarily monocytes), extravasation, and subsequent recruitment of other lymphoid and myeloid cells to the peri-collateral wall where they participate in the remodeling process. 75 If this mechanism also plays a role in remodeling of pial collaterals, CLZ would predictably inhibit it—favoring reduced rather than the enhanced remodeling that we observed. However, this effect could be offset by CLZ’s potential effect, as discussed above, to inhibit platelet activation in the ischemic MCA tree post-MCAO downstream of the collaterals, and increase collateral flow and thus remodeling. The above considerations support our hypothesis that at least part of CLZ’s augmentation of remodeling and reduction of infarct volume is due to a direct effect on the collateral remodeling process (Figure 6).
This study has additional limitations besides those mentioned above. Infarct volume following ischemic stroke is known to be affected by brain temperature. Cilostazol’s vasodilatory activity could lower rectal temperature after administration. Although rectal temperature was maintained at 37 °C during and after surgery until animals regained consciousness and received the first dose of CZL 2 h after pMCAO, we did not conduct comprehensive temperature monitoring during the remainder of the study, for example, before and after each subsequent daily dose of vehicle or CZL (100–200 µl). Thus, our findings must be considered preliminary.
It is possible that prevention of secondary stroke by CLZ, in trials where it was begun in the subacute phase, derived not only from its anti-platelet action but also from increasing collateral remodeling. de Havenon et al. recently reviewed trials that evaluated CLZ for prevention of secondary stroke. 9 When administered at 200 mg/day ay for 8–180 days following stroke, recurrence was reduced, while incidence of intracerebral hemorrhage was unchanged regardless of whether patients received aspirin or clopidogrel. For example, CSPS II saw a reduced risk of ischemic or hemorrhagic stroke compared to aspirin alone, and fewer hemorrhagic events occurred. In CATHARSIS, CLZ plus aspirin was superior to aspirin alone for prevention of all vascular events and new silent infarcts (11% vs 25%); progression of ICAS was unaffected.
Cilostazol has not been approved for prevention of recurrent stroke by the FDA. However, AHA/ASA guidelines state that although supporting evidence is not sufficient to warrant a definitive recommendation, CLZ at 200 mg/day maybe be considered in combination with aspirin or clopidogrel in patients with symptomatic ICAS to reduce recurrent stroke; however, generalizability to non-Asian populations reduces the level of confidence. 76 The Japan Stroke Society guidelines 77 recommend 200 mg/day for the prevention of recurrent non-cardioembolic ischemic stroke. In China, CLZ is recognized as a second-line option for prevention of the same, particularly in patients at higher risk of bleeding or for those who are intolerant to aspirin. 78 None of the above guidelines specifies the time after stroke onset to initiate treatment. Clinical practice and trial protocols provide some insight: CLZ is typically started in the subacute-to-chronic phase once the patient is stable and the risk of hemorrhage minimized.77,78 When 200 mg/day CLZ was begun within 24 h after stroke onset, NIHSS and mRS scores did not differ between CLZ and placebo at 3 or 5 days, nor at 1 or 3 months after stroke, respectively. 79 However, NIHSS score was significantly lower in the CLZ group 2 weeks after enrollment or at discharge. In the recent CILO–CLOP trial where CLZ (vs clopidogrel) was administered to patients with moderate or moderate-to-severe ischemic stroke at 200 mg within 24 h after stroke symptoms and then 200 mg/day until 90 days, 80 CLZ did not significantly reduce the percentage of patients with mRS > 2 (52% vs 58%, p = 0.09). However, patients with hypertension experienced significant reduction in recurrent ischemic or hemorrhagic stroke. In another study, patients receiving 200 mg/day CLZ within 48 h after stroke experienced less neurological deterioration than those receiving aspirin alone and achieved more favorable functional status. 81 However, other trials beginning treatment at 48 h found no improvement in mRS or NIHSS scores, compared to aspirin alone.82,83
The efficacy of CLZ to enhance collateral remodeling and thereby contribute to improved outcome and reduction of secondary stroke could be reduced or lost when administration is begun by at least 24 h after onset of first stroke, as done in the above studies. Also, CLZ’s efficacy to increase remodeling may differ depending on the severity of stroke in the hyperacute phase, the presence of hypertension or other co-morbidities, collateral score at baseline, the efficacy of thrombolysis and/or thrombectomy, amount of no-reflow if present, or other factors that could affect collateral flow including the presence of concomitant aspirin or clopidogrel. 7 Findings from the trials discussed in the preceding paragraphs highlight the need for subgroup analysis in clinical trials and additional preclinical investigation to examine the above factors, additional dose-timing, direct measurement of collateral blood flow in animal studies, whether collateral remodeling persists after cessation of treatment, and assessment of potential adverse effects (e.g. edema, hemorrhage, reduction in blood pressure).
We observed greater remodeling at 10 compared to 3 mg/kg/day. The latter is equivalent to 200 mg/day in patients, assuming comparable bioavailability with oral administration in humans and intraperitoneal in our study. The 10 mg/kg/day dose could increase the risk of hemorrhage when given early after AIS. Additional animal studies are thus needed to identify agents like BI, especially any that are FDA-approved for another indication, that act by mechanisms that distinguish them from CLZ and augment remodeling in an additive or synergistic manner. These studies and those mentioned in the preceding paragraph would provide the foundation for future clinical investigations combining such an agent with the lower dose of CLZ, that is, a dual remodeling therapy, for reduction of brain injury and improved outcome after first-stroke as well as the incidence of recurrent stroke 84 and TIA,85,86 while not increasing the risk of hemorrhage and thrombosis.
Supplemental Material
sj-docx-1-jcb-10.1177_0271678X251409612 – Supplemental material for Cilostazol increases collateral remodeling and reduces the severity of ischemic stroke
Supplemental material, sj-docx-1-jcb-10.1177_0271678X251409612 for Cilostazol increases collateral remodeling and reduces the severity of ischemic stroke by Hua Zhang, Ming-Chieh Ding, Gabriel Gong and James E Faber in Journal of Cerebral Blood Flow & Metabolism
Footnotes
Acknowledgements
The authors thank Kathryn Conlon and Sean Thornton for collateral morphometry and assistance with animal husbandry.
Abbreviations and acronyms
ACA Anterior cerebral artery
AIS Acute ischemic stroke
BI-749327 BI, TRPC6 inhibitor
cAMP Cyclic adenosine monophosphate
CC Collaborative cross genetic reference panel of inbred strains of mice
cGMP Cyclic guanosine monophosphate
CLZ Cilostazol
DAG Diacylglycerol
EC Endothelial cell
ICAS Intracranial atherosclerosis
IC Intermittent claudication
KO Knockout (gene disruption/deletion)
MCA Middle cerebral artery
NO Nitric oxide
PAD Peripheral artery disease
PCA Posterior cerebral artery
PDE Phosphodiesterase
PKA Protein kinase A
PKG Protein kinase G
PLC Phospholipase C
pMCAO Permanent distal M1-MCA occlusion
Rabep2 Rab GTPase-effector binding protein-2
Rabep2-KO Rabep2 knock out (name of a gene is denoted in italics; its protein is not)
RCF Relative collateral blood flow
SMC Smooth muscle cell
TIA Transient ischemic attack
tMCAO Transient middle cerebral artery occlusion
TRPC6 Transient receptor potential canonical 6 (ion channel)
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: National Institutes of Health’s National Institute of Neurological Diseases and Stroke (grant NS083633).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author contributions
HZ and JEF designed the study and wrote the manuscript. HZ performed the experimental procedures and data analysis. GG assisted with morphometry and genotyping. M-CD contributed valuable discussion.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
