Abstract
Carotid artery stenting (CAS) induces hemodynamic disturbances that may trigger silent cerebral ischemia (ASILs), though the relative contributions of impaired cerebral autoregulation (dCA) versus plaque characteristics remain unclear. In this prospective cohort study, we evaluated 59 patients with severe carotid stenosis undergoing CAS, assessing dCA via transcranial Doppler (TCD) and transfer function analysis (TFA) pre- and post-procedurally, with postoperative MRI to detect ASILs. ASILs occurred in 30.5% (18/59) of patients and were associated with dyslipidemia (P = 0.010) and low-echo plaques (P = 0.022). Critically, the ASILs group exhibited significantly reduced dCA phase in the very low frequency (VLF) range post-CAS (P < 0.001), indicating impaired autoregulation. Multivariate analysis identified postoperative VLF phase (adjusted OR: 0.925, P = 0.002), dyslipidemia (OR: 11.909), and plaque morphology (OR: 10.798) as independent ASILs predictors. ROC analysis demonstrated superior predictive accuracy when combining dCA parameters with clinical/plaque features (AUC = 0.917). These findings establish dCA dysfunction as a key hemodynamic biomarker of ASILs post-CAS, surpassing plaque characteristics alone. Integration of perioperative dCA monitoring with traditional risk stratification may optimize patient selection and neuroprotective strategies during carotid revascularization.
Keywords
Introduction
Extracranial carotid artery disease accounts for approximately 20% of all stroke cases. Carotid artery stenting (CAS) has emerged as a safe and effective alternative to carotid endarterectomy (CEA) for treating carotid stenosis, demonstrating comparable post-procedural outcomes in both symptomatic and asymptomatic patients. 1 However, acute ischemic cerebral events remain a major complication following CAS, often resulting in prolonged hospitalization and increased mortality.2,3 Acute ischemic lesions (AILs) are best detected using diffusion-weighted imaging (DWI) and apparent diffusion coefficient (ADC) on MRI, characterized by hyperintense DWI and hypointense ADC signals. 4 Acute silent ischemic lesions (ASILs) are defined as AILs meeting these imaging criteria while remaining clinically asymptomatic within 10 days after procedure. Multiple studies have confirmed that the presence of acute silent ischemic lesions (ASILs) on post-procedural MRI following CAS is strongly associated with an increased risk of future ischemic stroke.5,6 A recent cohort study of 586 patients undergoing carotid revascularization demonstrated that ASILs predict significantly higher risks of serious adverse events following the procedure. 7 Given that ASILs diminish the clinical benefits of CAS and are associated with an increased risk of future adverse events, the identification of high-risk patients for ASIL development becomes critically important. 8
Impaired cerebral autoregulation plays a critical pathophysiological role in both ischemic stroke and vascular cognitive impairment. 9 Previous studies 10 have reported that dynamic cerebral autoregulation (dCA) in the middle cerebral artery (MCA) is frequently impaired in patients with severe carotid artery stenosis, particularly during ischemic stroke. This dysfunction has been further associated with compromised autoregulatory capacity under conditions of reduced perfusion pressure, as well as diminished efficiency in clearing emboli originating from carotid plaques.11,12 Current literature highlights the prognostic significance of dCA in determining functional recovery and long-term outcomes for cerebrovascular disease patients following revascularization interventions. 13 While the clinical significance of post-stenting micro-embolization and its long-term neurological consequences are well established, systematic investigations integrating quantitative dCA metrics with high-resolution DWI to predict ASIL risk are still lacking.
As a standard approach for dCA assessment, transfer function analysis (TFA) performs spectral decomposition of stationary cerebrovascular signals via fast Fourier transformation, characterizing the phase and amplitude relationships across frequency domains.14,15 TFA has been extensively applied to investigate cerebrovascular disorders including ischemic stroke, hyperperfusion syndrome, and neurodegenerative diseases. 16 However, its application in CAS remains largely unexplored.
Carotid duplex ultrasound (CDU) combined with transcranial Doppler (TCD) enables continuous blood pressure monitoring while assessing cerebral blood velocity (CBv) in the middle cerebral artery (MCA) in response to steady-state blood pressure changes.17,18 This integrated approach provides comprehensive evaluation of both cerebral hemodynamic responses and detailed characterization of carotid stenosis. This study was designed to systematically evaluate dCA parameters in patients with severe carotid stenosis, with particular focus on identifying early predictors of silent ischemic lesions following carotid artery stenting. Through comprehensive assessment of dCA characteristics and associated hemodynamic factors, we aimed to establish reliable markers that could help prevent subsequent ischemic events and other neurological complications.
Materials and methods
Study design and participants
The study protocol was approved by the Ethics Committee of Xuanwu Hospital, Capital Medical University ([2023]143) and registered at ClinicalTrials.gov (NCT06058676). In this prospective study, we consecutively recruited patients indicated for carotid artery stenting to treat extracranial internal carotid artery stenosis between September 2023 and June 2024. Written informed consent was obtained from all participants in line with the Declaration of Helsinki. In this study, the severity of internal carotid artery (ICA) stenosis was assessed by carotid duplex ultrasound (CDU) and confirmed by either computed tomography angiography (CTA) or digital subtraction angiography (DSA), following the North American Symptomatic Carotid Endarterectomy Trial (NASCET) criteria.19,20 Inclusion Criteria were as follows: (1) Male and non-pregnant, non-breastfeeding female subjects whose age is ≥18 years and <80. (2) Symptomatic patients with ICA stenosis ≥50%, or asymptomatic patients with ICA stenosis ≥70%. (3) Patients to be evaluated by CDU and TCD within 3 days before CAS and within 48 h after CAS. (4) Patient is willing and capable of complying with all study protocol requirements, including the specified follow-up visits and can be contacted by telephone. And the exclusion Criteria were patients who: (1) Severe ICA stenosis caused by non-atherosclerosis diseases, such as dissection or arteritis. (2) Carotid revascularization or MCA stent before. (3) Moderate or higher-grade stenosis of the unilateral or bilateral MCA (4) Acute ischemic stroke before CAS (5) Poor temporal sound window or closure of the temporal window. (6) Without complete clinical or imaging information. Global cognitive function was assessed using the Mini-Mental State Examination (MMSE) and Montreal Cognitive Assessment (MoCA). In accordance with established criteria for older adults, cognitive impairment was defined as a score of ≤24 on either test (maximum score = 30). All participants provided baseline data covering: age, sex, BMI; vascular risk factors (hypertension, coronary disease, diabetes, dyslipidemia, smoking); and physiological measurements (HR, BP, CBv, dCA measures).
Measurement parameters of ultrasonography
CDU and Transcranial color-coded duplex sonography(TCCS) or TCD were performed to evaluate the carotid artery and MCA by senior vascular ultrasound physicians with ≥5 years of experience. While an EPIQ7 (Philips Medical Systems, Bothell, WA, USA The Netherlands) with 3.0–12.0 MHz linear array transducer also was chosen to assess target vessels. The blood flow velocity of MCA was measured through the transtemporal acoustic window at a depth of 50–55 mm with the corrected angle adjusted to ≤60°. All ultrasound imaging and parameters were stored in PACS for subsequent analyses. The CDU pre-carotid procedure plaque imaging were also saved in PACS and the characteristics such as morphology and echogenic features (low-echo or calcified) were included.
The imaging parameters were as follows: (1) the residual diameter and original diameter at ICA stenosis; (2) the characteristics (size, echo and morphology) of palque which caused severe carotid stenosis; (3) peak systolic velocity (PSVsten) and end diastolic velocity (EDVsten) at the ICA stenosis; (4) PSV, EDV, and pulsatility index (PI) of the ipsilateral MCA before and after CAS
Assessment of dCA and data analysis
The dCA was evaluated twice by experienced neuro-sonographers with >10 years of specialized practice following the International CARNet White Paper protocol: at baseline (24 hours pre-procedure) and within 48 hours following carotid CAS. Participants were instructed to maintain strict pre-assessment conditions, including 24-hour abstinence from vasoactive substances (nicotine, caffeine, alcohol) and chocolate. All measurements were performed in a climate-controlled environment (22–24°C), with additional requirements of fasting from high-calorie meals and refraining from moderate exercise for ≥6 hours prior to evaluation.
The examination subjects were in the supine position without crossing the legs in a hospital bed for a 15-mins rest before assessing the dCA. Continuous CBv was assessed in bilateral MCA by using a TCD (EMS-9DPro; Delica Medical, Shenzhen, China). The TCD probes of 1.6-MHz were secured in the bilateral temporal windows by an adjustable headframe at a depth of 45–65 mm, respectively, a noninvasive continuous beat-to-beat BP (NIBP) by using a finger photo-plethysmograph system Finometer®, Arnhem, The Netherlands). Before each NIBP, the BP at the brachial artery was measured by using a sphygmomanometer (Omron HBP-1300; Kyoto, Japan) to correct baseline BP. A nasal catheter was connected to monitor end-tidal carbon dioxide (ET-CO2) and respiratory rate throughout the session. In addition, the precordial electrocardiography with four leads were synchronously recorded.
The dCA parameters can be evaluated between BP and CBv time series according to the standards of TFA. Therefore, absolute gain (cm·s−1 · mmHg−1) phase and coherence parameters were calculated in the traditional three frequency bands recommended for the TFA: a very low frequency range (VLF, 0.02–0.07 Hz), low frequency (LF, 0.07–0.20 Hz), and high frequency (HF, 0.20–0.50 Hz). For interpretation, phase reflects the time delay of cerebrovascular adaptation between BP and CBv, whereas the gain quantifies the damping effect of dCA on amplitude. 21 That is, a lower phase and higher gain reflected that the dCA was impaired. Coherence represented a linear relationship between BP oscillations and CBv. Only dCA data with a coherence ≥0.50 (number of windows: 3; critical values of coherence:5%) were included in the subsequent statistical analysis.14,15
Transfer function analysis
Signal processing and TFA were performed according to the 2022 CARNet White Paper guidelines. 14 Temporal delays between flow velocity (FV) and arterial blood pressure (ABP) signals were corrected using cross-correlation. Signals were preprocessed with a 3rd-order Butterworth low-pass filter (0.5 Hz cutoff) to prevent aliasing before down sampling to 1 Hz, followed by linear detrending to minimize spectral leakage. Power and cross-spectra were estimated using Welch’s method with 102.4 s Hanning windows (50% overlap) and triangular spectral smoothing.14,22
Surgical procedure and medical management
The CAS was selected by the experience of neurosurgeons with >10 years of carotid revascularization experience 23 according to the contemporary guideline promulgated by the American Heart Association/American Stroke Association (AHA/ASA). 24 For the CAS procedure, approved devices including FilterWire EX (Boston Scientific, Natick MA, USA) and Spider (Covidien, Plymouth MN, USA) were used based on the stenting procedure. The distal protection devices were used in all cases. Approved stents included Carotid Wallstent (Boston Scientific, Natick MA, USA) and Precise (Cordis, Miami FL, USA). A suitable devices, stents, balloon size and dilation strategy selected based on the ICA diameter, as shown on the DSA image was also left to the discretionary judgment of the physician. The medical treatment strategies of drugs was consistently implemented during perioperative period in accordance with AHA/ASA guidelines. 24
Magnetic resonance imaging
All participants underwent MRI assessments at two time points: within seven days preceding CAS and three days following the procedure. MRI was acquired on a clinical 1.5-T Philips scanner using optimized sequences covering the range from the cranial vertex to the foramen magnum. Multiple MRI sequences were acquired for comprehensive evaluation, including: T1-weighted (T1W) and T2-weighted (T2W) imaging for anatomical assessment. DWI with ADC mapping for detection of acute ischemic changes. These MRI sequences permitted comprehensive evaluation of ASILs. The presence of lesions on MRI (consisting of areas of focal hyperintensity on T2-weighte, hypointensity on T1-weighted images and a heightened DWI signal coupled with a reduced ADC signal). Comprehensive neurological assessments were conducted, with detailed documentation of clinical manifestations including headache, ipsilateral weakness, focal deficits, and other focal neurological signs. DWI sequence parameters: repetition time (TR)/echo time (TE) = 3500/80 ms, FOV(mm) = 220 × 220 mm, Acquisition Matrix = 128 × 128, Slice thickness = 5 mm with 1 mm gap, Flip angle(°) = 90, with b = 0, 1000 s/mm2. All DWIs were reviewed by two neuroradiologists to ensure inter-rater reliability.
Statistical analysis
All statistical analyses were executed employing IBM SPSS software (version 27.0; Armonk, NY, USA).The Shapiro–Wilk test was used to assess whether continuous variables conformed to a normal distribution (p > 0.05, normal). Continuous data with normal distributions were expressed as the means ± standard deviation (SD), such as age, BMI, residual diameter and original diameter. Two-sided t-test was used to compare these continuous variables between ASILs group and non-ASILs group. Demographic and clinical baseline characteristics were compared between two groups before procedure. In contrast, the interquartile range [M (P25, P75)] was used to represent the variables data of non-normally distributed and were compared using Wilcoxon rank-sum test. Categorical variables are expressed as percentages and were analyzed using the chi-square test or Fisher’s exact test as appropriate. Multivariate analysis was performed using a logistic regression analysis to investigate, including the indicators with P < 0.1 in the univariate analysis. We constructed ROC curves and calculated the area under the ROC curve (AUC) to investigate the value of early postoperative dCA for predicting ASILs. All tests were two-tailed, and p < 0.05 was considered statistically significant.
Results
Demographical data
A total of 128 consecutive patients with hemodynamically significant carotid artery stenosis were initially evaluated for eligibility for carotid artery stenting (CAS). After applying stringent exclusion criteria, 59 patients (mean age 65.98 ± [6.95]; range 52–79 years) were prospectively enrolled in this study. The cohort exhibited a pronounced male predominance (n = 54, 91.5%), with female participants constituting a smaller subset (n = 5, 8.5%). All 59 patients underwent successful CAS with complete perioperative dCA assessment. Postprocedural DWI detected ASILs in 18 patients (30.5%). The enrollment pathway is detailed in Figure 1. Figure 2 presents comprehensive perioperative multimodal neuroimaging findings – including ultrasonography, DSA, and MRI – in a representative case with radiologically confirmed ASILs. The detailed information among the baseline demographic characteristics of the ASILs group (n = 18) and non-ASILs group (n = 41) is summarized in Table 1. Patients in the ASILs group demonstrated significantly higher prevalence of dyslipidemia compared to the non-ASILs group (72.2% vs. 31.7%; χ2 = 8.33, p = 0.004) and elevated body mass index (27.1 ± 3.6 vs. 24.9 ± 2.4; p = 0.028). While hypertension, diabetes mellitus, and coronary artery disease showed numerically higher incidence rates in the ASILs group, these differences did not reach statistical significance (all p > 0.05). No significant intergroup differences were observed in age, sex distribution or smoking history.
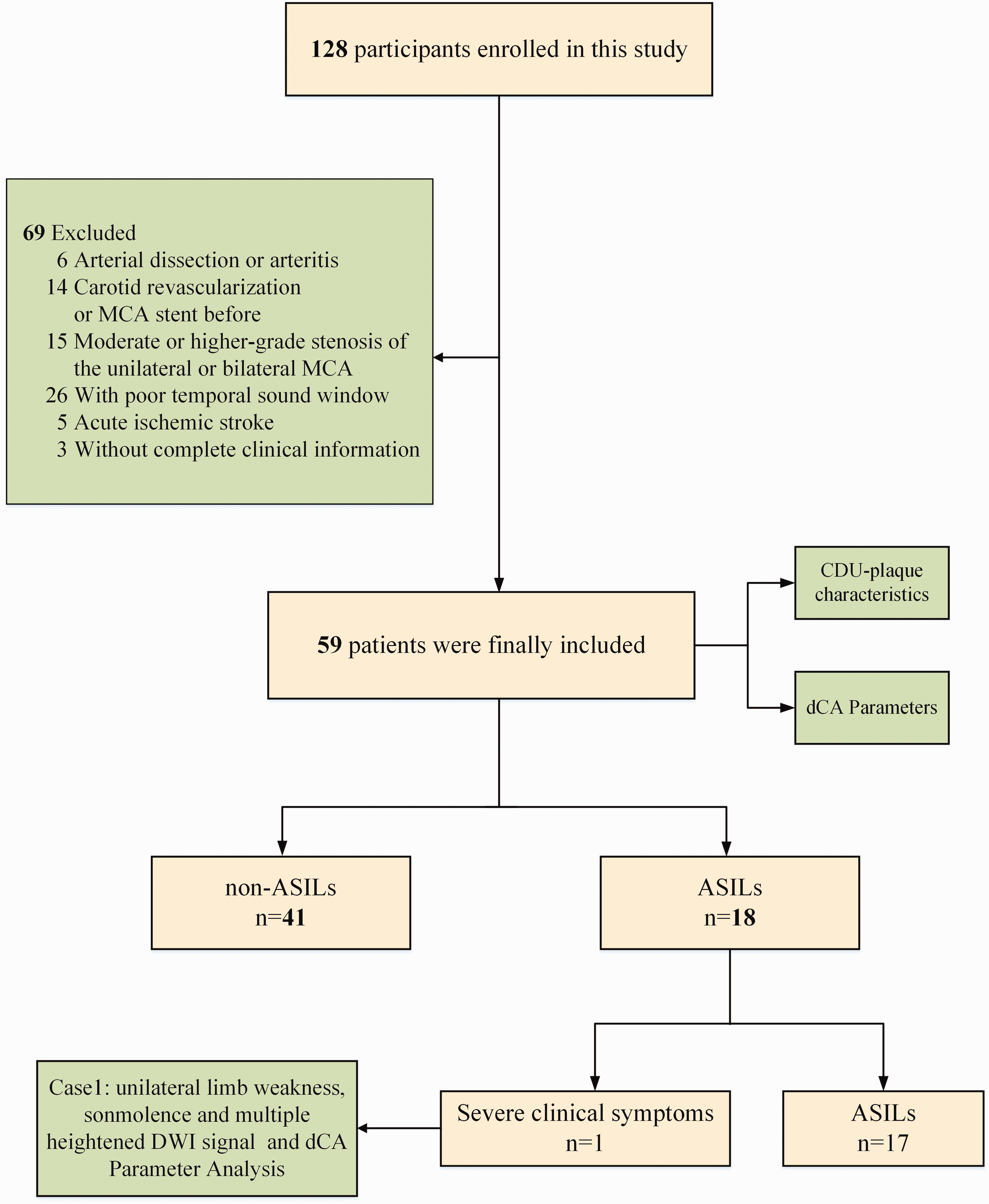
Flow diagram of included studies. N = number.
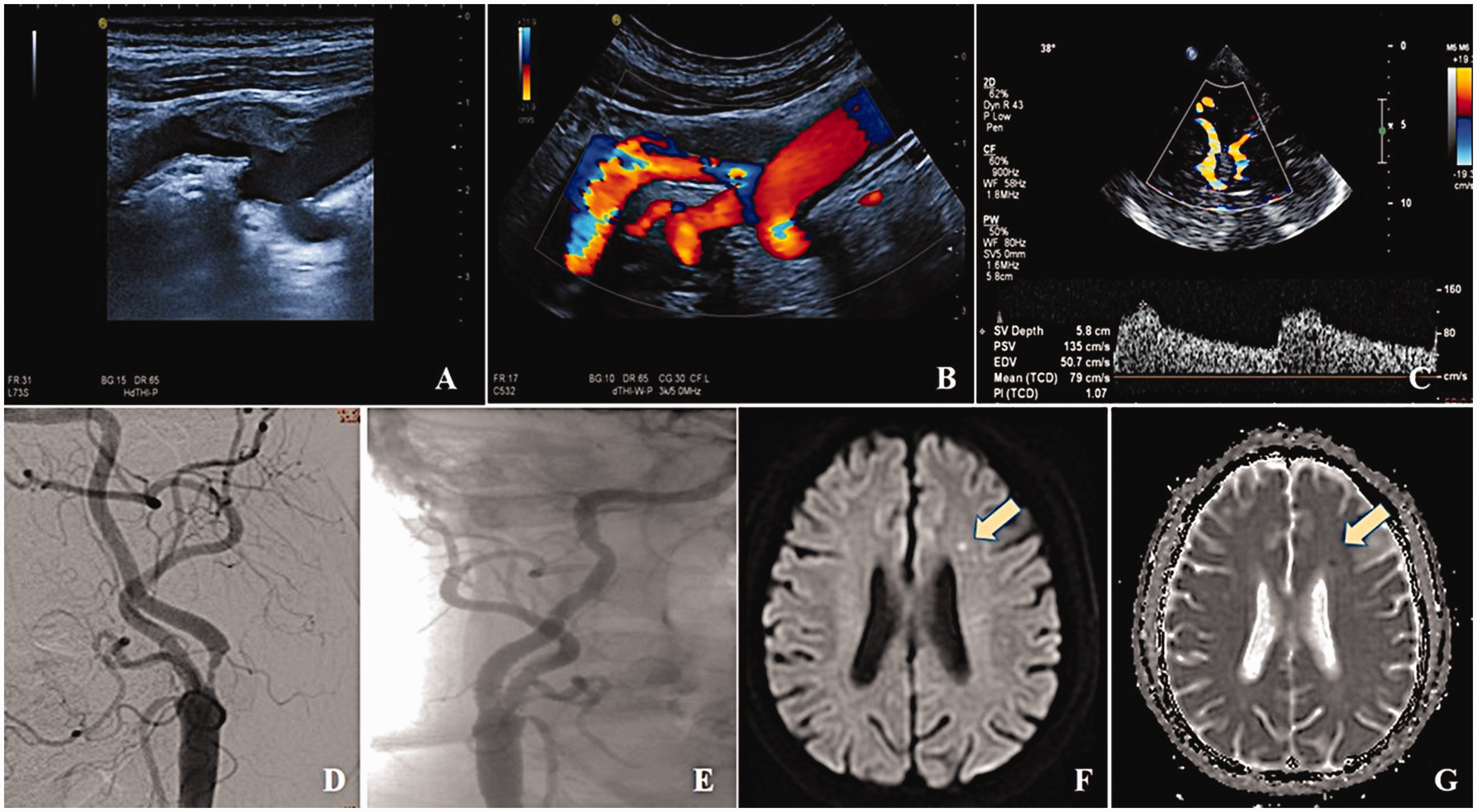
The perioperative evaluation of a patient with carotid artery stenosis. (a,b) Two-dimensional ultrasound imaging and color Doppler flow imaging (CDFI) before CAS showed carotid artery stenosis at the initial segment. (c) TCCD showed left MCA hemodynamics after CAS. (d) Preoperative digital subtraction angiography (DSA) showed severe stenosis at the proximal of the internal carotid artery. (e) DSA showed blood flow patency recovery after CAS and (as arrow showed) in (f,g) A heightened diffusion-weighted imaging (DWI) signal coupled with a reduced apparent diffusion coefficient (ADC) signal in patients with ASILs that transpired after CAS.
Characteristics of patients in ASILs and non-ASILs group.
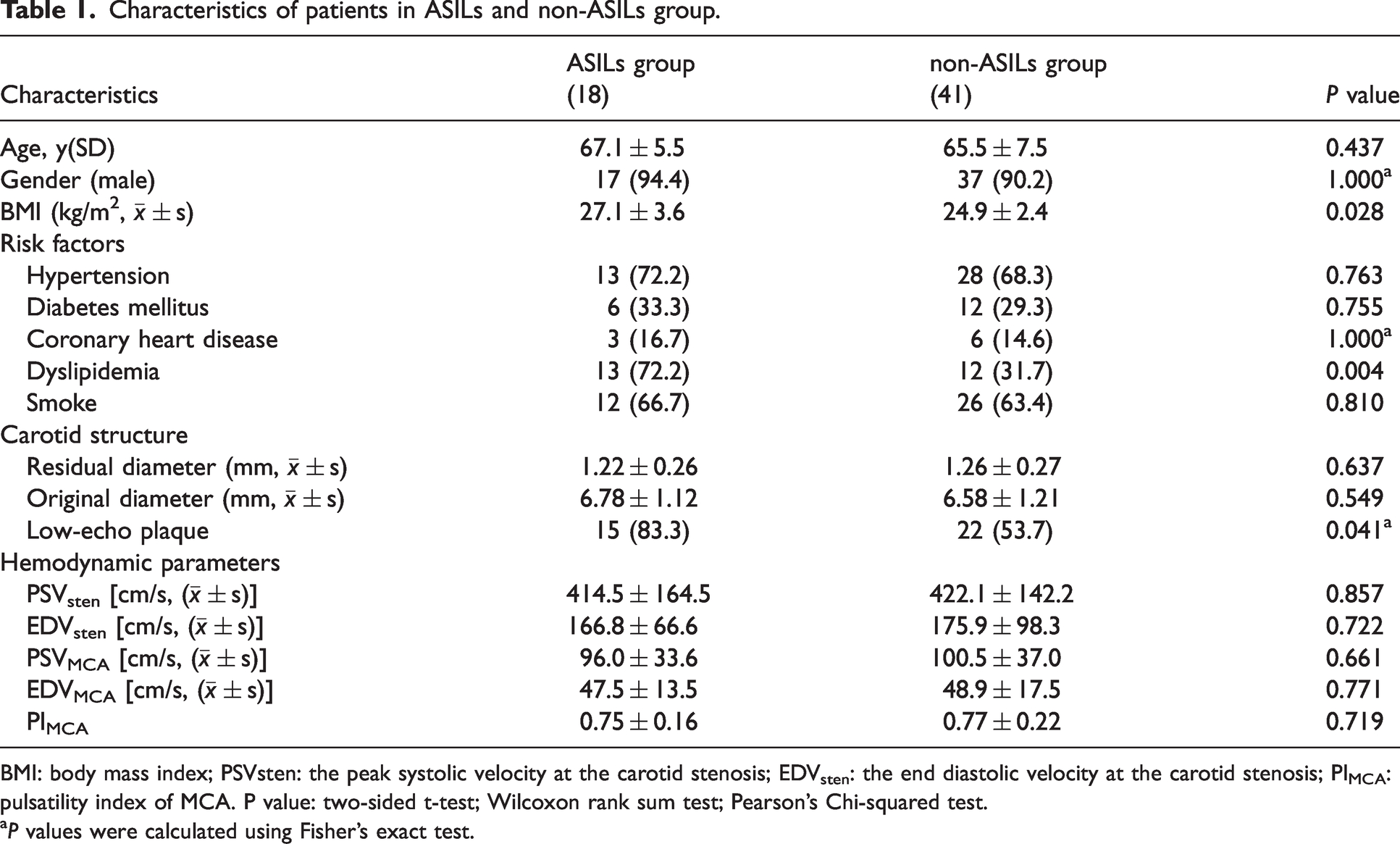
BMI: body mass index; PSVsten: the peak systolic velocity at the carotid stenosis; EDVsten: the end diastolic velocity at the carotid stenosis; PIMCA: pulsatility index of MCA. P value: two-sided t-test; Wilcoxon rank sum test; Pearson’s Chi-squared test.
P values were calculated using Fisher’s exact test.
Carotid characteristics and ASILs
The cohort with ASILs exhibited a significantly greater prevalence of low-echoic plaques compared to non-ASILs (83.3% vs 53.7%, P = 0.041). While the ASILs group demonstrated a trend toward greater luminal narrowing at the stenosis site (residual diameter: 1.22 ± 0.26 mm vs 1.26 ± 0.27 mm), this difference did not achieve statistical significance (p = 0.065). Hemodynamic parameters, including PSVsten (414.5 ± 164.5 vs 422.1 ± 142.2) and EDVsten (166.8 ± 66.6 vs 175.9 ± 98.3), PSVMCA (96.0 ± 33.6 vs 100.5 ± 37.0) and EDVMCA (47.5 ± 13.5 vs 48.9 ± 17.5), and MCA pulsatility index (0.75 ± 0.16 vs 0.77 ± 0.22), showed no significant differences between ASILs and non-ASILs groups (all p > 0.05), despite numerically lower flow velocities in the ASILs cohort (Table 1).
Comparison of dCA parameters between the two groups
The dCA parameters were quantitatively assessed using TFA method in accordance with the international CARNet guidelines,14,15 including gain, phase, and coherence at very low frequency (VLF, 0.02–0.07 Hz), low frequency (LF, 0.07–0.20 Hz), and high frequency (HF, 0.2–0.50 Hz). The phase degree was found lower before and after CAS in ASILs group than in the non-ASILs group, but there was not statistically significantly different (P > 0.05). Preoperative phase degree was consistently lower in the ASILs group and non-ASILs group (32.18° vs 33.85°, p = 0.59), though this difference did not reach statistical significance. Postoperative phase degree remained significantly lower in the ASILs group compared to the non-ASILs group (39.3° [26.7–45.8] vs 58.9° [46.9–75.4], p < 0.001), failing to normalize within 2 days post-CAS. Coherence values showed no significant intergroup differences across all frequency bands (VLF/LF/HF) either pre- or post-CAS (all p > 0.05). In addition, gain values showed no significant differences at either time point (both p > 0.05). Tables 2 and 3 summarize pre- versus post-CAS dCA characteristics (gain/phase/coherence) for ipsilateral MCA hemodynamics. Figure 3 presents the comparative analysis of pre- versus postoperative dCA parameters in both group.
Comparison of dCA parameters of MCA before CAS in ASILs and non-ASILs group.
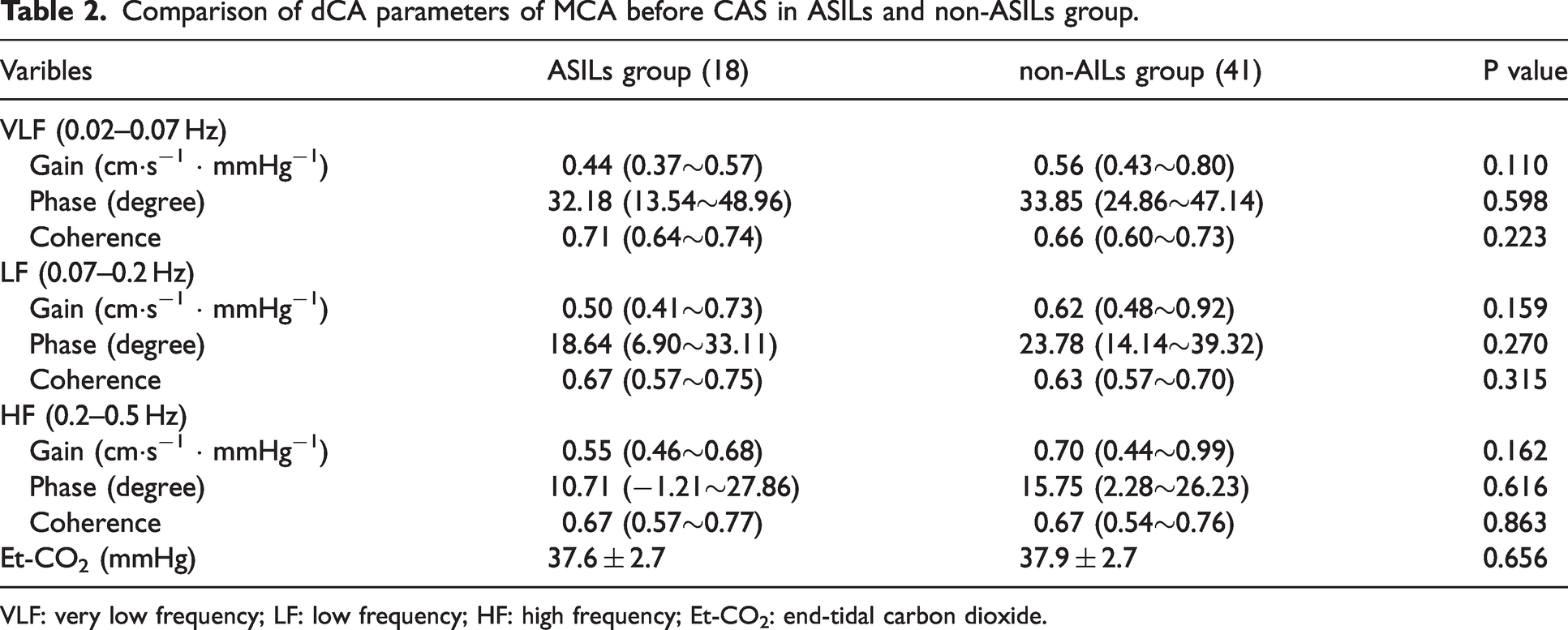
VLF: very low frequency; LF: low frequency; HF: high frequency; Et-CO2: end-tidal carbon dioxide.
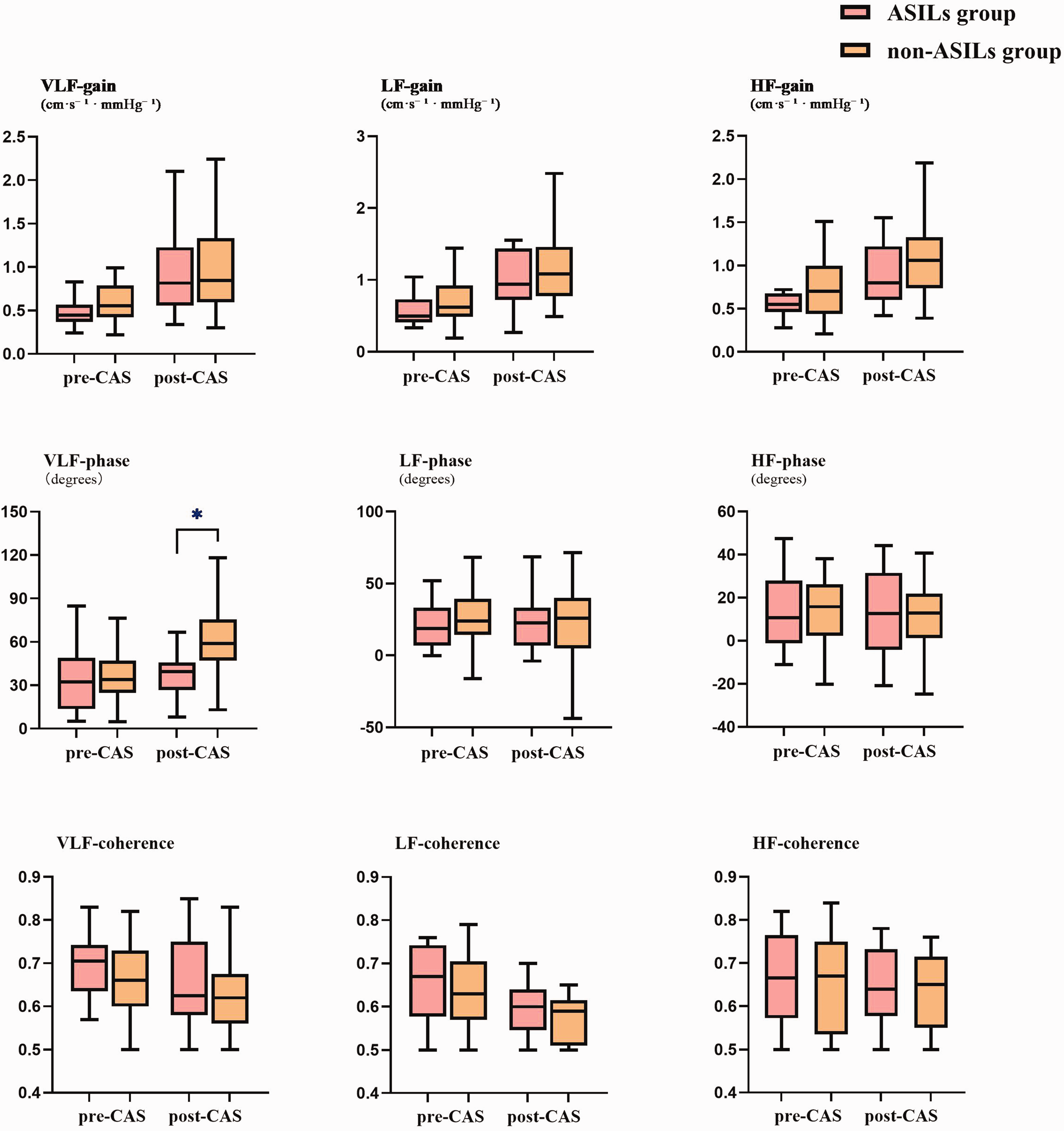
Group comparisons of transfer function analysis (TFA) metrics (gain, phase, coherence) across very low frequency (VLF), low frequency (LF), and high frequency (HF) bands in ASILs and non-ASILs patients before and after CAS. Bars denote medians, boxes denote interquartile ranges. For significances see Table 2. Postoperative VLF phase was significantly reduced in the ASILs group compared to non-ASILs (p < 0.05).
Comparison of dCA parameters of MCA in early post-CAS in ASILs and non-ASILs group.
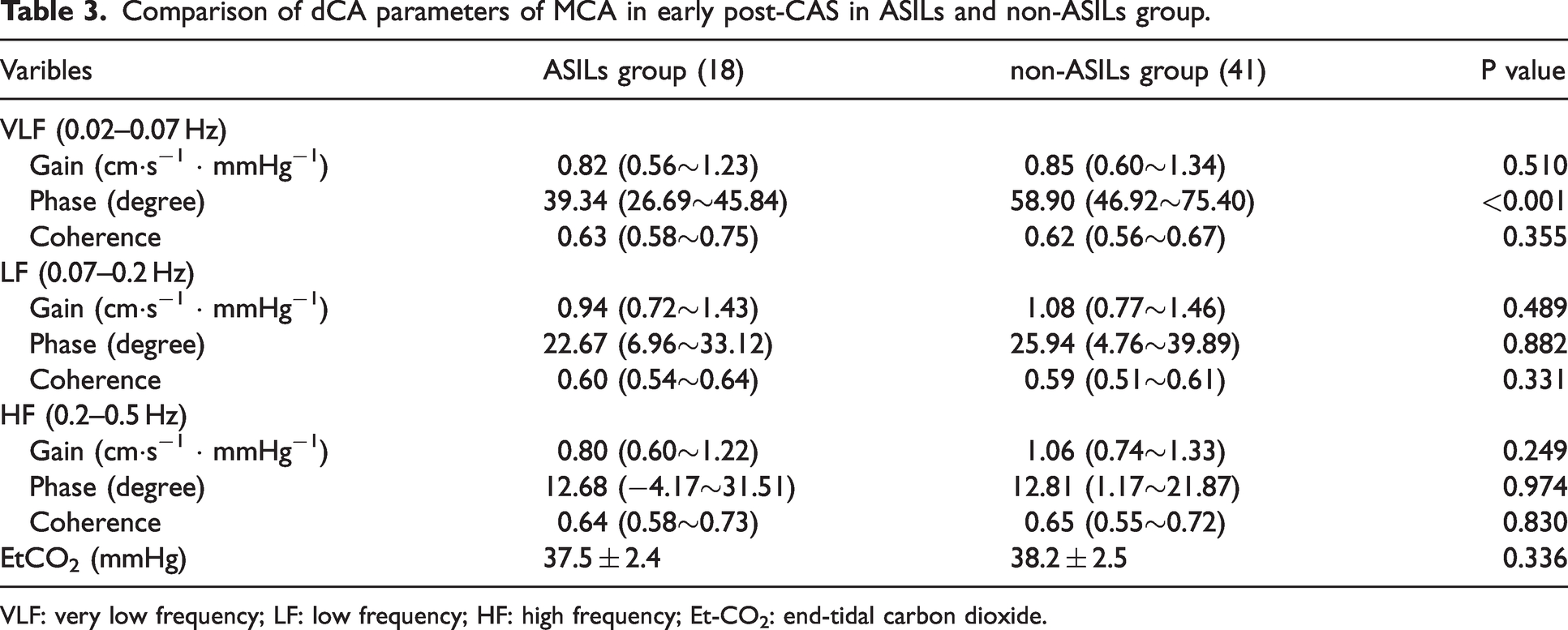
VLF: very low frequency; LF: low frequency; HF: high frequency; Et-CO2: end-tidal carbon dioxide.
Stratified analysis of dCA improvement in ASILs subgroups
Among the 18 patients with ASILs, 7 cases (38.9%) failed to demonstrate improvement in postoperative VLF-dCA (defined as <10% improvement in phase shift from baseline). Subgroup analysis of ASILs patients revealed that 85.7% (6/7) of those without postoperative dCA improvement exhibited low-echo plaques, compared to 81.8% (9/11) of patients with dCA improvement (P > 0.05, Fisher’s exact test). Preoperative hemodynamic evaluation revealed no statistically significant differences in either peak systolic or end-diastolic velocities in the middle cerebral artery between the two groups (P > 0.05).
Multivariate analysis and comparison of ROC curves
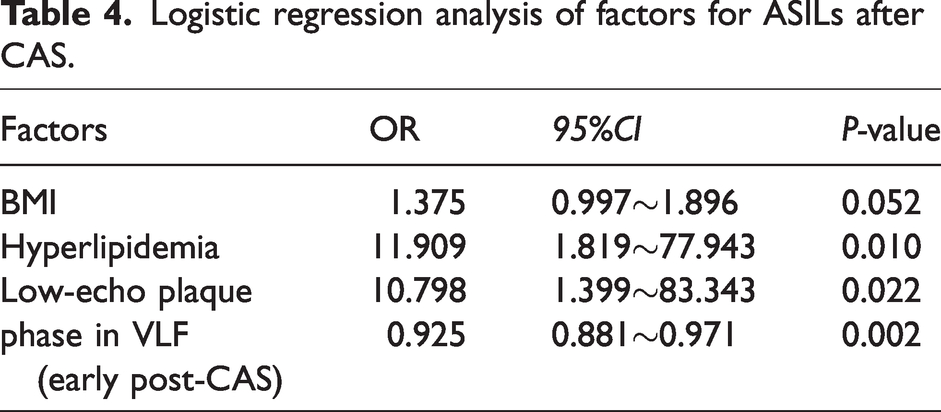
Multivariable logistic regression analysis incorporating BMI, dyslipidemia, low-echo plaque, and early postoperative ipsilateral phase degree at very low frequency (VLF) following CAS identified three independent predictors of ASILs: early postoperative ipsilateral phase at VLF (adjusted odds ratio: 0.925, 95%CI: 0.881∼0.971, P = 0.002), dyslipidemia (adjusted odds ratio: 11.909, 95%CI: 1.819∼77.943, P = 0.010) and low-echo plaque (adjusted odds ratio: 10.798, 95%CI: 1.399∼83.343, P = 0.022). The factors associated with ASIL are summarized in Table 4.
Logistic regression analysis of factors for ASILs after CAS.
The overall ROC curve analysis was performed to evaluate and compare the predictive performance of four distinct models for ASILs after CAS. Model 1: Early postoperative ipsilateral phase at VLF alone; Model 2: comprised a combination of Model 1 and dyslipidemia; Model 3: comprised a combination of Model 1 and low-echo plaque; Model 4: comprised a combination of Model 2 and low-echo plaque. The area under the curve (AUC) values for the five prediction models were as follows (Figure 4): Model 1: 0.820 (95% CI: 0.702–0.937); Model 2: 0.890 (95% CI: 0.810–0.971); Model 3: 0.856 (95% CI: 0.759–0.954) and Model 4: 0.917 (95% CI: 0.837–0.998) (shown in Figure 4). Model 4 demonstrated the highest discriminative performance among all models; Specifically, no significant differences were observed between Model 1, Model 2, Model 3, and Model 4 in any pairwise comparisons (all P > 0.05). Notably, the standalone predictive performance of low-echo plaque for ASILs was suboptimal, with an AUC of 0.648 (95% CI: 0.502–0.795, p = 0.071), failing to reach statistical significance. The optimal discriminatory threshold of the postoperative ipsilateral phase for distinguishing patients with ASILs was derived through Youden index maximization in Model 1. The optimal cutoff value for the early postoperative ipsilateral phase was determined to be 45.48°. Patients with ipsilateral phase values ≤45.48° demonstrated significantly higher prevalence of ASILs compared to those with values >45.48° (60.9% vs. 11.1%, respectively; P < 0.001).
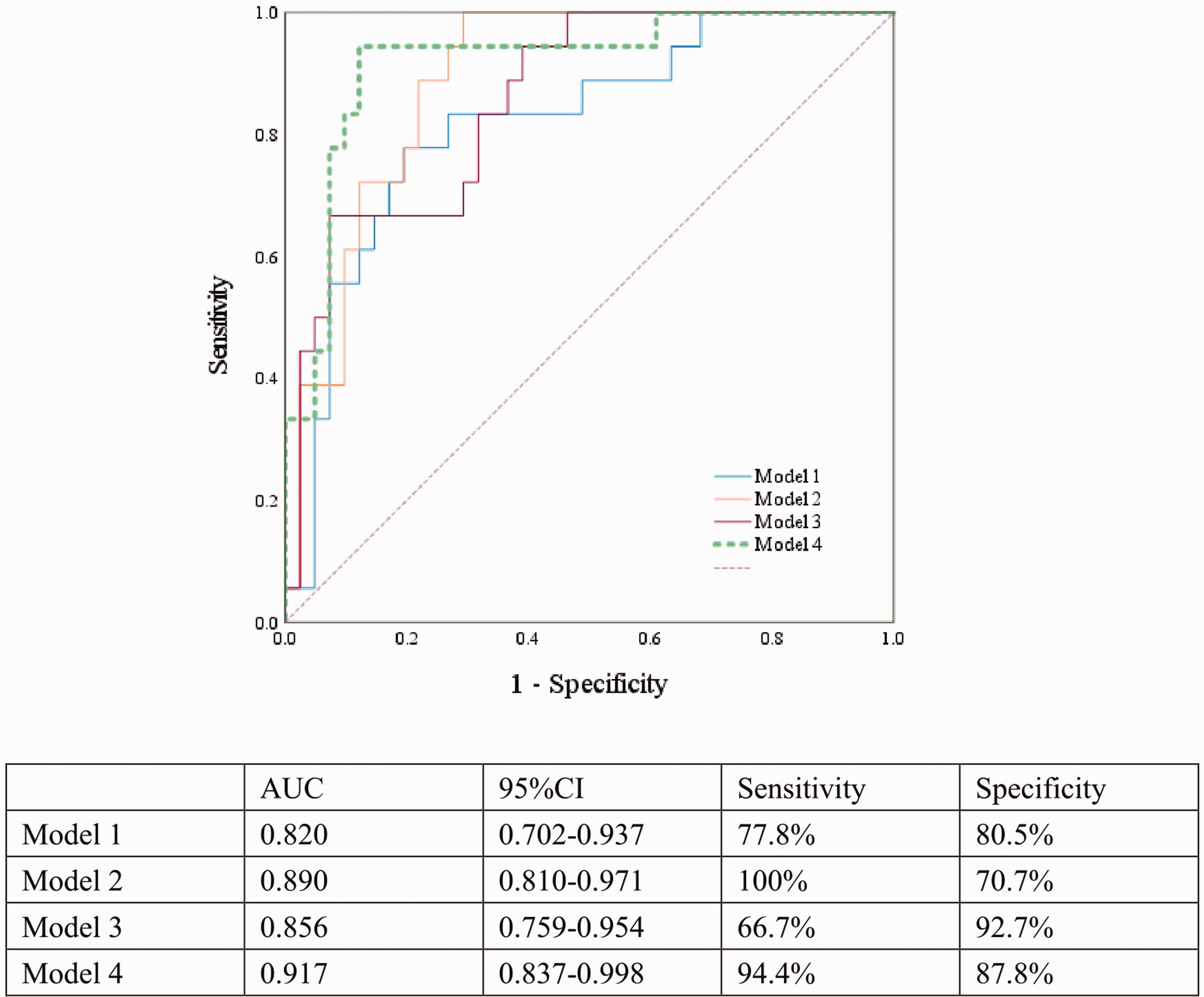
ROC curves for predicting the outcome. Model 1: Early postoperative ipsilateral phase at VLF alone; Model 2: comprised a combination of Model 1 and dyslipidemia; Model 3: comprised a combination of Model 1 and low-echo plaque; Model 4: comprised a combination of Model 2 and low-echo plaque.
Discussion
Our analysis identified three notable associations. First, the presence of dyslipidemia and low-echo plaques was more common among patients who developed new ischemic brain lesions after CAS. Second, analysis of dCA before and after CAS demonstrated that the phase difference remained reduced in the ASILs group and did not return to normal levels within two days post-procedure. Third, dCA monitoring was identified as an independent predictor of new ischemic lesions during the perioperative period, and its predictive accuracy was further strengthened when integrated with other risk factors.
Carotid artery revascularization procedures, such as CEA or CAS, are commonly performed to restore blood flow in patients with significant carotid stenosis, thereby reducing the risk of stroke. Among this total cohort of 59 patients, ASILs were detected in 18 cases (30.5%) by DWI examination. Our 72-hour post-CAS MRI surveillance captured both acute (0–24 h) and delayed microembolic events, including delayed embolization accounting for the moderately higher ASIL incidence observed in our study compared to those with shorter observation periods. 25 This single-center prospective cohort study investigated the association between ASILs detected by DWI and potential risk factors. A study with a mean follow-up period of 6 months has demonstrated that both the presence and number of DWI lesions are significantly associated with an increased risk of recurrent stroke or TIA following treatment. 26 Notably, dyslipidemia was identified as an independent risk factor for ASILs following CAS. The long-term follow-up study 27 of 921 patients evaluated the clinical significance of ischemic lesions detected by DWI after carotid artery stenting in patients with dyslipidemia and found that post-procedural ischemic lesions were significantly associated with an increased risk of long-term stroke. Furthermore, additional studies have investigated that statin pretreatment can significantly reduce the incidence of ischemic lesions following CAS. 28 The future cohort studies are warranted to validate whether the impact of dyslipidemia management measures on DWI-detected lesions correlates with procedural safety outcomes.
Extensive research29,30 has established that hypoechoic plaques constitute a specific subtype of carotid atherosclerotic lesions, characterized by high-risk vulnerability features including an expansive lipid-rich necrotic core, attenuated fibrous cap, and intra-plaque hemorrhage. An article has previously reported that the carotid plaque with a lipid-rich necrotic core or intraplaque hemorrhage, leading to the dislodgment of plaque fragments, thereby triggering embolic events are significantly associated with new-onset infarction during or following CAS. 31 Therefore, preoperative assessment of plaque morphology using ultrasonography is of great significance for predicting the risk of post-procedural stroke. A total of 1,013 articles were screened and 23 studies with 6,706 carotid plaques demonstrated that plaques with complex features, particularly those with echo-lucent, neovascularization, ulceration and intraplaque motion are associated with ischemic symptoms. 32 This study provides evidence supporting the hypothesis that new ischemic lesions detected on DWI following CAS result from thromboembolic material or atherosclerotic plaque debris dislodgement during or after the intervention. However, several important limitations should be acknowledged. First, while ultrasonography is clinically practical for plaque characteristics assessment, it lacks the capability of high-resolution MRI to quantitatively evaluate lipid cores or fibrous cap integrity. 33 The fibrous cap, especially when thin and prone to rupture, cannot be detected precisely because of the limited spatial resolution of current ultrasound machines. 34 Second, this study did not implement routine intraoperative TCD monitoring for cases with low-echo plaques, which limits our ability to correlate real-time microembolic signals with postoperative ischemic lesions. Future studies should include the need for incorporating high-resolution MRI based on the limitations of single-modality assessment in our study and standardized intraprocedural monitoring to better characterize which plaque morphologies most affect clinical outcomes.
The success of carotid revascularization in improving cerebral hemodynamics depends on the integrity of the cerebral autoregulatory system. 35 In patients with preserved autoregulation, the brain can effectively modulate vascular resistance to maintain stable CBF despite changes in perfusion pressure. Moreover, Et-CO2 influences dCA through cerebrovascular diameter changes, 36 with 1 mmHg alterations inducing 3–6% CBF variation. To account for this confounder, Et-CO2 was measured and corrected for in all dCA analyses. With the development of carotid revascularization in recent years, increasing attention has arisen toward dCA, which have been used as periprocedural outcome marker, for example, hyper-perfusion syndrome studies. 37 In a retrospective analysis, dCA was found to significantly improve one month after carotid revascularization. The study highlighted the utility of dCA parameters in identifying the likelihood of stroke occurrence during the acute postoperative period. 38 However, studies between dCA and DWI lesions after carotid revascularization and long-term follow-up data are still lacking. Our study revealed that dCA in 12 patients failed to demonstrate improvement within 2 days post-procedure compared to pre-procedure levels, with 7 of these patients presenting ASILs. In our subgroup analysis of ASILs patients, we observed a non-significant trend toward higher prevalence of low-echo plaques in those without dCA improvement compared to those with improvement. However, low-echo plaque as an independent predictor for ASILs demonstrated only modest discriminative ability (AUC = 0.648, 95% CI: 0.502–0.795; p = 0.071), significantly underperforming compared to multimodal models (all AUC > 0.820). These findings suggest that low-echo plaques may exert a synergistic effect on postprocedural cerebral autoregulation (dCA) impairment through multiple interacting mechanisms: artery-to-artery embolism from plaque components; microvascular obstruction in perforating arteries due to enhanced embolization of lipid-rich debris; and pre-existing autoregulatory dysfunction. Additionally, the relatively small sample size of patients may have affected the statistical power to detect smaller but potentially clinically relevant effects.
Although carotid artery recanalization has the potential to enhance dCA, 39 the observed divergence in dCA outcomes between our CAS cohort and historical CEA data likely stems from differences in procedural mechanisms: CEA achieves physiological restoration through direct atheroma removal and adventitial nerve preservation, CAS induces complex vessel wall interactions and the concurrent endothelial injury may progressively exacerbate dCA impairment. Furthermore, intraoperative micro-emboli could potentially induce cerebral cortex ischemia, potentially leading to cognitive dysfunction, which necessitates long-term follow-up and further comprehensive investigation to elucidate the underlying mechanisms and clinical implications. In patients with impaired cerebral autoregulation, particularly those with chronic conditions such as hyperlipidemia or diabetes, the risk of postoperative complications—including hyperperfusion and hypoperfusion—is significantly elevated. 40 Early postoperative monitoring of dCA not only facilitates the timely evaluation of cerebral blood flow regulatory capacity but also offers critical predictive insights into the potential occurrence of acute cerebral ischemic events.
Despite its contributions, this study has notable limitations. First, a significant limitation of this study is the notable gender disparity in our patient cohort, with a substantially higher representation of male patients compared to their female counterparts. This observed imbalance may be primarily explained by the well-established epidemiological evidence indicating a lower prevalence of carotid artery stenosis in female populations.41,42 Second, Studies in severe carotid disease showed significantly improved dCA after revascularization. 39 Similar to our patient, carotid stenting immediately normalized dCA parameters, though some patients in ASILs group remained near the lower normal limit. The 45.48° threshold derived from Model 1 (early postoperative ipsilateral VLF phase shift alone). Although this value falls near the lower limit of the normal range defined in healthy populations, 14 it may reflect significant correlation with postoperative new infarcts, suggesting that optimal risk stratification thresholds likely vary across populations with different hemodynamic features. A key limitation is the lack of long-term follow-up data to validate this cutoff and explore optimal thresholds across disease stages. Future studies should expand the ASIL cohort with impaired dCA across distinct frequency domains, incorporating longitudinal multimodal imaging and extended follow-up to delineate frequency-specific hemodynamic contributions to lesion progression and clinical outcomes post-revascularization.
In conclusion, this single-center prospective cohort study explored the relationship between ASILs identified on DWI and factors such as dyslipidemia, low-echo plaque, and the early postoperative impaired dCA. Advanced imaging techniques can provide valuable insights into cerebral hemodynamic changes and help identify patients at risk after CAS. Furthermore, standardized dCA assessment protocols and biomarker-driven trials are needed to validate its clinical utility and establish dCA optimization as a precision therapy in carotid revascularization.
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was supported by the Noncommunicable Chronic Diseases - National Science and Technology Major Project (NO.2024ZD0521601) and the National Natural Science Foundation of China (U24A20686).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Authors’ contributions
Yunlu Tao: conceptualization, data curation, formal analysis, methodology, writing – original draft, critical revision of manuscript; Songwei Chen: data curation, methodology, critical revision of manuscript; Yifan Yang: data curation, methodology, writing – review & editing; Fubo Zhou: formal analysis, writing – review & editing; Hongxiu Chen: data curation, methodology, critical revision of manuscript; Liuping Cui: data curation, methodology, formal analysis; Zihao Ni: data curation, methodology; Xia Lu: data curation, writing – review & editing; Shengnan Li: data curation, writing – review & editing; Yan Ma: Investigation, methodology; Liqun Jiao: investigation, methodology; Yingqi Xing: project administration, investigation, methodology, critical revision of manuscript. All authors contributed to the article and approved the submitted version.
