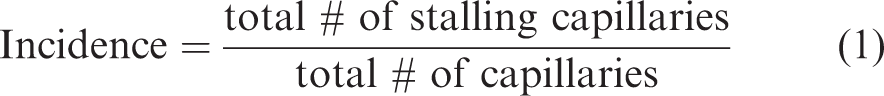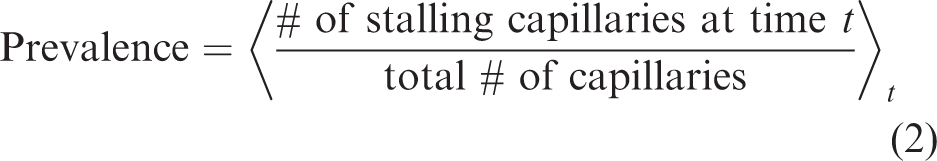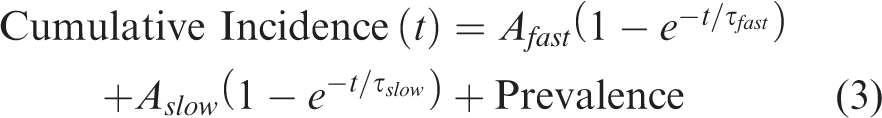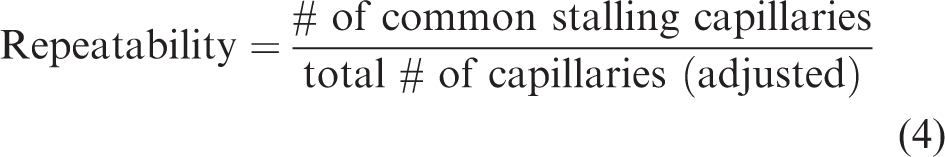Abstract
Transient stoppages of red blood cell (RBC) flow through capillaries—termed capillary stalls—occur persistently in neurological disorders such as Alzheimer’s disease and ischemic stroke and can interrupt oxygen delivery and exacerbate neurological damage. Effective imaging tools and analyses are necessary to understand the nature, role, and prevention of stalls. In this study, we dissect differences in stalls measured by two-photon Bessel beam microscopy (Bessel-2PM) and optical coherence tomography (OCT) to gain insight into the temporal dynamics of stalls. Twenty-minute series of volumetric angiograms were obtained separately with Bessel-2PM and OCT on the same day in awake, head-fixed mice. The temporal dynamics of stalling in both methods revealed a minority population of susceptible capillaries that exhibited frequent stalls and a large majority of capillaries with infrequent stalls. Differences between OCT and Bessel-2PM in the repeatability and dynamics of stalls are explained by differences in their sensitivity to short or infrequent stalls based on scanning speed and detection off-time. Finally, stroke caused a shift toward the frequently stalling capillary subpopulation, lasting 1 week post-stroke. Dynamic stall analysis therefore enables examination of physiological and methodological contributions to the stalls measured in disease models and across studies.
Introduction
In an organ as active as the brain with very little metabolic reserve, the efficient transport and delivery of nutrients through its capillary networks is a fundamental requirement to sustain healthy function.1,2 Each capillary constantly perfuses its surrounding tissues with adequate oxygen and other nutrients, as long as there are red blood cells (RBCs) continually passing through.2,3 However, a steady passage of RBCs is not always guaranteed: “capillary stalls,” or transient periods of no flow in individual capillaries lasting seconds to minutes, disrupt perfusion and risk inducing hypoxia in the surrounding brain tissue.4,5 Current research on stalls relies on in-vivo imaging techniques aiming to understand their physiological causes and identify potential therapeutic targets in disease models as mounting evidence has implicated stalls as a contributor to negative outcomes in a multitude of neurological disorders including Alzheimer’s disease, ischemic stroke, and epilepsy.6–10 These pathologies are associated with elevated numbers of stalling events, which may lead to hypoxic cell damage or death in vulnerable tissue patches. Previous work has linked changes in stalling to dynamic physiology such as pericyte constriction and neurovascular coupling in the cortex of both healthy and diseased animal models.5,9–13 However, the temporal dynamics of stalls have not yet been extensively quantified, despite being critical to understanding the causes of stalls and their significance in pathology.5,8
To this end, recent methodological advancements have improved the temporal resolution and throughput of stall quantifications, which has enabled capturing stalls as brief as ∼1 s.14–16 We have utilized Optical Coherence Tomography (OCT) angiography, which relies on endogenous backscatter from moving RBCs to identify blood vessels and measure stalling dynamics. Its volumetric scanning capability enables imaging 100 to 200 capillaries every 8 to 10 s in the mouse brain.5,8,16 This is complemented by two-photon (2 P) microscopy, which utilizes fluorescent labeling of the blood plasma or blood cells to identify when blood cells are stationary within the blood vessels during a stall. 2 P microscopy has the advantage of fluorescent markers that can distinguish cell types responsible for stalling events.6,7,17–20 However, traditional 2P microscopy with a Gaussian focus is inefficient for measuring stalling dynamics among large capillary populations because it can only image one plane in depth at a time. As such, 2P microscopy with a Gaussian focus has not been used for measuring stalling dynamics, but instead has been used for surveying a volume of capillaries to identify the percentage that are stalled.7,18 2P microscopy with a Bessel focus (Bessel-2PM)21,22 extended over 60 to 100 µm in depth permits volumetric imaging of more than 100 capillaries with an ∼2 s image frame period, as recently demonstrated in our home-built system.14,15 Due to their faster volume rate, Bessel-2PM14,15 and OCT5,8,9,23–25 have been adopted as the methods of choice to capture dynamic stalls.
Differences in the scanning and analysis strategies of each method can produce differences in the stalling phenomena they record. OCT relies on backscatter from moving RBCs to see a capillary. When there are no moving RBCs, the capillary disappears in that given image frame, when using the difference of two repeated B-scans (images in xz-plane). On the other hand, for 2P microscopy with fluorescently labeled blood plasma, blood cell shadows that are stationary for 2 or more frames will indicate a stall. Despite the shorter frame period (1.8 s) of Bessel-2PM over OCT (7.5 s), OCT may still be capable of detecting shorter stalls as only one frame is needed, leading to a lower limit of the duration for which a capillary is scanned over (∼1 s), although the exact duration of a stall under 8 s cannot be determined.5,8,14,15 These differences can lead to inconsistencies between quantification of stalls depending on the method used, and an incomplete understanding of these effects will bias quantification of the dynamic behavior of stalls and impact interpretations of stalls in different pathologies. Therefore, there is a need to investigate the nuances of capillary stall dynamics and how their apparent behavior is affected when captured with each method.
To address these challenges, we investigated the spatiotemporal differences between stalls recorded by Bessel-2PM and OCT angiography. Cumulative incidence dynamics averaged from n = 12 datasets in 7 mice departed from previously reported models, revealing that a minority of capillaries (<10%) experience frequent stalls while the majority of capillaries only experience infrequent stalls. The distributions of stall rates and durations further supported that the majority of stalling capillaries experience infrequent and short stalls. Differences were observed in the details of the proportion of frequent and infrequent stalling capillaries when comparing OCT and Bessel-2PM, and we show that these differences likely arise from the different imaging frame rate and duty cycle of each imaging method. We repeated the same dynamic analysis of frequent versus infrequent stalling capillaries in a mouse model of ischemic stroke, where the stall rate distribution shifted toward frequently stalling capillaries at Day 2 and Week 1 post-stroke before recovering to baseline. The present study shows the patho-physiological relevance of the temporal dynamics of stalls and the importance of considering methodological differences between Bessel-2PM and OCT when interpreting details of the observed stalling dynamics with results acquired with different image scanning parameters.
Materials and methods
Animal preparation
All experiments and animal procedures were conducted according to the Guide for the Care and Use of Laboratory Animals and protocols approved by the Boston University Institutional Animal Care and Use Committee. Animals were housed in normal light-dark cycles with nestlet enrichment and unrestricted food and water. For in-vivo imaging, a custom aluminum head bar and half crystal skull were implanted in the crania of C57BL/6 mice (Jackson Laboratory) over the intact dura.26–28 Animals recovered for 2–3 weeks post-surgery and then underwent head fixation training (daily, increasing duration until 90 minutes achieved) to minimize animal motion during awake imaging. Once acclimated, the mice were imaged under Bessel-2PM and/or OCT. During both training and imaging, the experiment was terminated if the mouse exhibited signs of distress. Measurements were completed in 7 healthy mice (3 F and 4 M) and 8 stroked mice (3 F and 5 M; see below) at 12–16 weeks of age. Group sizes were selected based on Lamorte statistical power calculations (Supp. Table S1). One other mouse was excluded from the study due to poor post-surgical recovery, totaling 16 mice. We used the ARRIVE checklist when writing our report. 29
Optical coherence tomography
A spectral-domain OCT system (Thorlabs Telesto II; 1310 nm center wavelength and 170 nm bandwidth) with an axial and transverse resolution of 3.5 µm was used to image the cerebral cortex through the cranial window. Images were acquired at ∼0.125 Hz volume rate with a 10X objective (Mitutoyo Corporation) and an A-line speed of 50 kHz, controlled by a custom LabVIEW program (National Instruments Corporation). OCT angiograms were acquired using a decorrelation-based approach using two repetitive B-scans. 16 The k-space intensity difference between repeated B-scans results in static tissue appearing as dark and dynamic tissue (e.g. moving RBCs in vessels) appearing as bright in the angiogram. Each dataset acquired was a 20-minute-long time series (160 total frames) and consisted of a 600 × 600 µm (400 × 400 pixels) region of interest (ROI). The angiogram was cropped in depth to a ∼100 µm (31 pixels) section starting ∼200 µm below the cranial window to isolate the capillary bed. The maximum intensity projection (MIP) along the depth axis of each angiogram in the time series was used for classification of stalling. Stalling capillary segments were manually annotated and identified by a sudden drop in intensity, seen as a temporary disappearance of the entire capillary from one or more angiograms in the time series. 5
Two-photon bessel beam microscopy
For two-photon imaging, a custom-built Bessel-2PM microscope with a 100 µm axial PSF was used. 14 Animals were anesthetized with isoflurane (3% induction, 1% maintenance) before being retro-orbitally injected with 50 µL of Texas Red dextran (5% in PBS; 150 kDa). 15 The dye was excited at 920 nm using a tunable Ti: Sapphire laser (Insight X3, Spectra Physics) to acquire fluorescence images of vasculature at ∼0.57 Hz for 20 minutes (700 total frames). Each dataset had an ROI of 713 × 713 µm (512 × 512 pixels). Stalls were annotated using a semi-automated approach developed previously. 15 This analysis identified stationary blood cells between two or more frames using an intensity correlation-based classification, which was manually corrected after analysis. Capillaries which did not have RBCs for two or more frames were classified separately as “plasma-only stalls” and not included in further analyses (see Discussion). A 3 D Z-stack was acquired for the same ROIs using a laser with a Gaussian focus. Capillary branch order was found for Bessel-2PM data manually by counting branches to the nearest arteriole or venule.
In-vivo imaging
Initial imaging was performed using both modalities on seven mice with a single ROI, with one-week repeat measurements performed on four of these animals. One mouse was subject to measurement in a second ROI without repeat. This resulted in 12 datasets from 7 mice in 8 different ROIs. One mouse was also used for a 1-hour OCT measurement in a new ROI.
Each OCT and Bessel-2PM dataset pair was captured on the same day using separate systems with an ∼1–3-hour break in between imaging sessions. OCT imaging was performed first to be able to easily capture the same ROI with Bessel-2PM afterward (measurements were not randomized). OCT angiograms were processed before starting the subsequent Bessel-2PM measurement so that the ROI and focus could be matched. Matching was done by adjusting the x-y-z imaging position of Bessel-2PM to maximize the number of Bessel-2PM visible capillaries that were also visible in the prior OCT. For Bessel-2PM, mice were briefly anesthetized to perform retro-orbital injections of Texas Red dextran, and imaging began after the mouse recovered from effects of isoflurane (∼30 min). We did not randomize OCT and Bessel-2PM because our OCT did not provide real-time viewing of the capillary network necessary to enable acquisition of the same capillaries. In order to rule out any potential bias of OCT always occurring before Bessel-2PM, we performed a separate set of experiments in n = 3 different mice under an identical experimental schedule, except a second OCT dataset was taken after injection of Texas Red dextran instead of taking Bessel-2PM, to verify that this sequencing had no observable impact on the stalling parameters.
Stroke measurements
A photothrombotic model 26 was used to induce pial artery-targeted ischemic stroke in the somatosensory cortex of mice (n = 8) by performing retro-orbital injection of 100 µL of 0.015 M Rose Bengal dye in saline, followed by 520 nm laser irradiation on distal branches of the middle cerebral artery and collaterals. OCT stalling measurements of a single matched ROI were taken pre-stroke and 2 days, 1 week, 2 weeks, and 4 weeks post-stroke. These datasets were blinded and manually annotated, and statistics were calculated in the same way as for other OCT measurements (see below).
Statistical analyses
After performing manual (OCT) and semi-automated (Bessel-2PM) stall annotations, the results of each dataset were summarized in a stall-o-gram, which is a binary representation of when each capillary stalled in time (Figure 1(c) and (d)). From these stall-o-grams, stall incidence (equation (1)) and point prevalence (equation (2)) were calculated. The total number of capillaries was counted in 2 D images of either Bessel-2PM or the OCT MIPs, averaged over the recording duration, in the same volume of cortex.
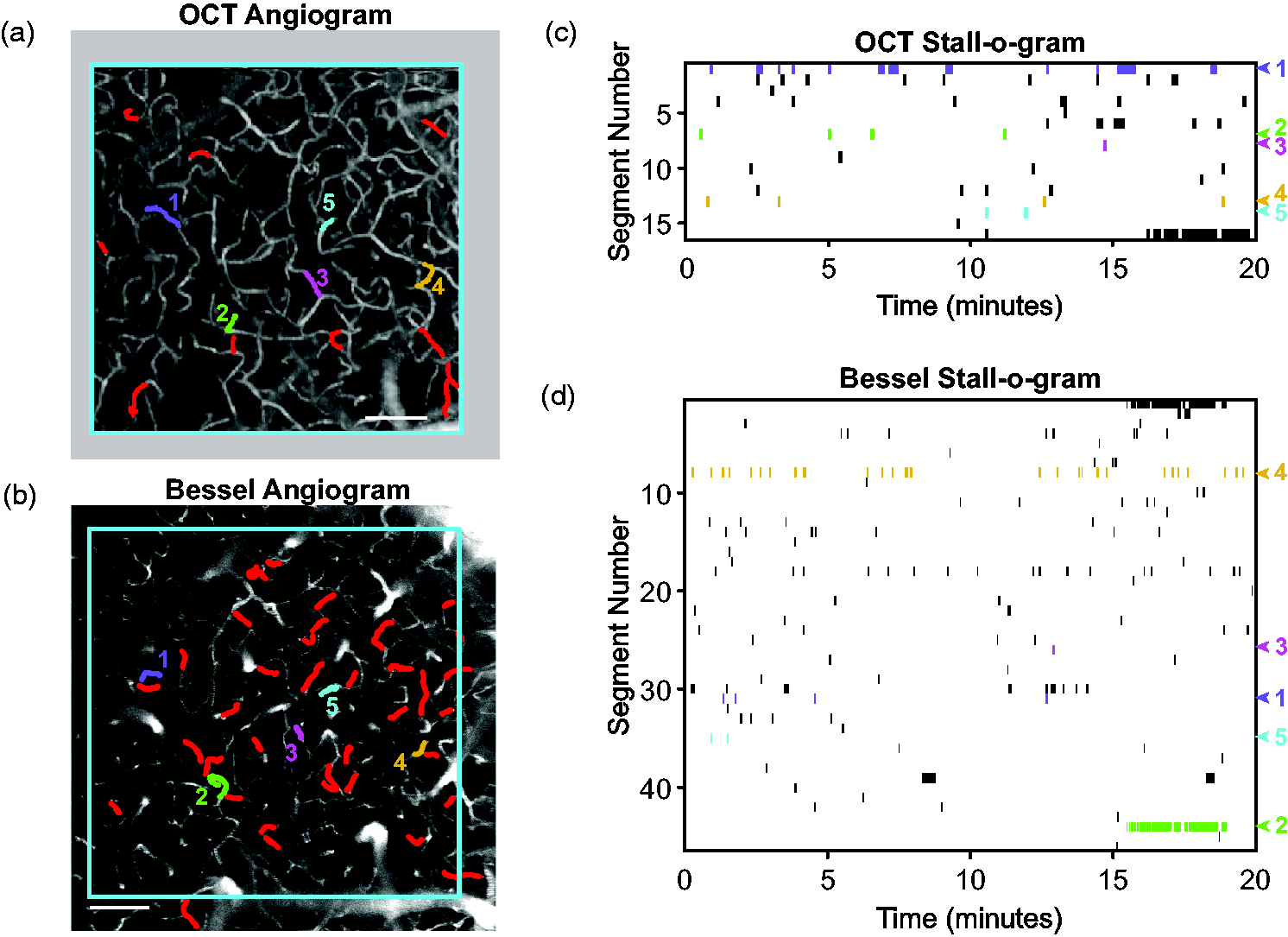
Representative (a) OCT and (b) Bessel-2PM angiograms, with highlighted capillaries indicating those that experienced at least one stalling event during the 20 min recording. Scale bar 100 µm. (c) OCT and (d) Bessel-2PM stall-o-grams showing the stalling dynamics for each highlighted capillary. Capillaries which stalled in both methods are highlighted with colored numbers in the angiograms and stall-o-grams. The same color/number in each subplot identifies the same capillary.
Cumulative incidence curves were calculated for each dataset by taking the cumulative sum of capillaries that stalled at least once and dividing the resulting timeseries by the total number of detected capillaries.
5
The group average cumulative incidence curve was fit to an exponential model using the MATLAB fit function. For the single exponential function (Supp. Eq. S1), lower bounds were set to 0 for all coefficients. Upper bounds were set to 100 for
Compensation for duty cycle differences (Figure 2(c)) was performed by sampling every fourth frame of the original Bessel stall-o-gram, resulting in four down-sampled stall-o-grams that were used to compute cumulative incidence curves. The cumulative incidence curves for stalls lasting >8 s (Figure 2(c)) were estimated by first removing stalls that last in less than 4 consecutive frames (<7.25 s) in Bessel-2PM or lasted in only 1 frame in OCT (<7.5 s) before calculating the cumulative incidence curves.
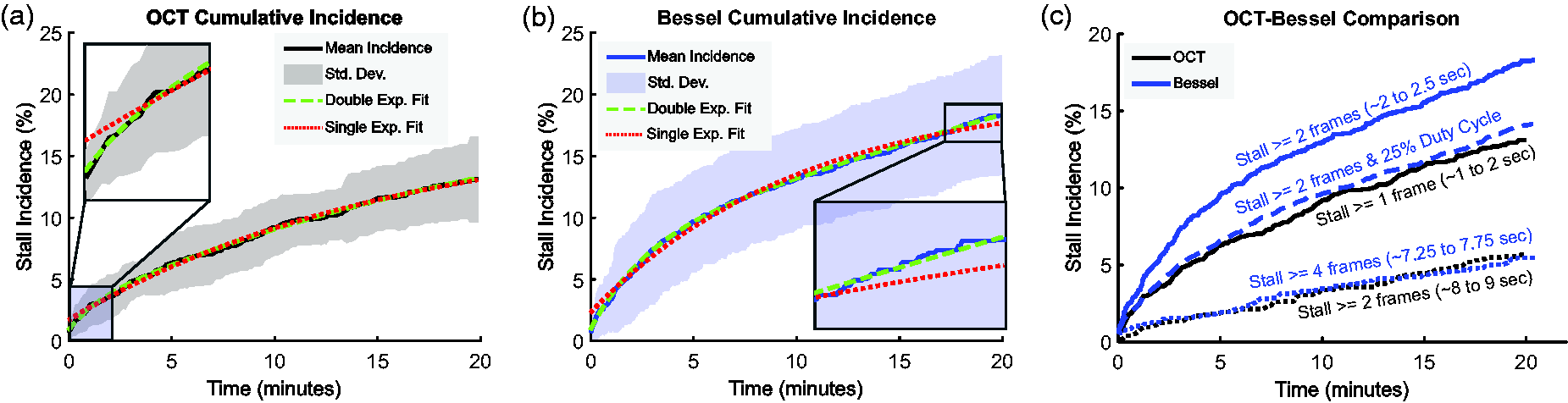
Average cumulative incidence curve for (a) Bessel-2PM (n = 12 datasets) and (b) OCT (n = 13 datasets) with the fit with the double exponential model (green, dashed), and the single exponential model (red, dotted). Insets show regions of the graph where the single exponential model does not adequately predict the data. Shaded regions are standard deviation. Fit coefficients are available in Supp. Tables S2 and S3 and (c) cumulative incidence curves for OCT and Bessel-2PM with selective filtering of stalls by either dropping the duty cycle of image frames for Bessel-2PM to mimic the frame repeat rate of OCT (dashed blue line), or by analyzing only stalls that last for more than ∼8 s for both Bessel-2PM and OCT (dotted lines).
The rate of stalls for each capillary was computed by dividing the number of stalls by the 20-minute recording time. The normalized cumulative sum was calculated for each dataset before performing a pairwise t-test between the Bessel-2PM and OCT datasets at each stall rate bin. The bins of the Bessel-2PM duration histogram were grouped to form new bins equivalent to OCT duration histogram bins based on whether the mean of each Bessel-2PM bin was within the OCT bin boundaries.
Repeatability between datasets was quantified using equation (4). The adjusted total number of capillaries is the number of capillaries visible in both recordings, excluding segments that were not captured in both volumes.
Results
We used Optical Coherence Tomography (OCT) to capture repeated microvascular angiograms over a region of interest (ROI) spanning 600 × 600 × 100 µm for a duration of 20 minutes in awake, head-fixed mice (Figure 1(a)). On the same day, we performed Bessel-2PM to capture a 713 × 713 × 100 µm volume containing the same ROI for 20 minutes (Figure 1(b)). Stalls were identified in each image series as described in Methods, producing the stall-o-grams shown (Figure 1(c) and (d)). Capillaries that stalled across both measurements were manually identified for repeatability analysis below.
Our initial analysis of temporal stall dynamics used conventional cumulative incidence curves, which describe the fraction of total capillaries which have stalled in a given time period. The average curve over all Bessel-2PM (Figure 2(a)) or OCT (Figure 2(b)) datasets was initially fit with a single exponential model, as previously reported in the literature using 10-minute recordings.
5
However, this model did not fit these 20-minute recordings as well as expected (Figure 2(a) and (b)), and a two-population double exponential model (equation (3)) was empirically found to be a more accurate fit (Figure 2(a) and (b)). The fit time constants describe the expected time interval between stalls. The short time constant
We investigated whether differences between Bessel-2PM and OCT cumulative incidence curves could be due to differences in the timing of the image scanning and in the image analysis. First, the longer frame period of OCT results in an ∼4-fold longer off time for detection of each capillary when that capillary is not being scanned. To compensate for this longer OCT off time, each Bessel-2PM stall-o-gram was partitioned into four subsets containing every fourth frame of the stall-o-gram to effectively match the OCT off time. The resulting cumulative incidence dynamics from the down-sampled data are similar to the OCT dynamics (Figure 2(c)), suggesting that differences in absolute detection off time can account for differences in the quantified stalls between the two methods. Second, to compare Bessel-2PM and OCT with matched stall durations, we considered only stalls lasting longer than approximately 8 seconds (i.e. >= 4 frames on Bessel-2PM and >= 2 frames on OCT) as these stalls are equally detected by the two methods given their respective scan parameters. The resulting cumulative incidence curves for these longer duration stalls are similar, suggesting that there are no differences in each method’s capability of capturing long-duration stalls (Figure 2(c)).
Due to the limitations of cumulative incidence analysis for informing us about the stalling rates for different capillaries, we expanded the temporal stall dynamics analysis to consider the distribution of individual capillaries. Calculating the stalling rate for each capillary revealed an exponential probability distribution of different rates as observed by both OCT and Bessel-2PM (Figure 3(a)). This further emphasizes that a minority population of capillaries has a high rate of stalls while the majority of capillaries have a low rate of stalls. The cumulative distributions of stalling rates indicate that OCT detects a lower proportion of low-rate stalls compared to Bessel-2PM (Figure 3(a) and (b)). We suspect that OCT with its longer off time between sampling a given capillary is missing many of the low-rate stalls that are of short duration. We suspected that capillaries with a higher rate of stalls may tend to have shorter average stall durations than capillaries with a lower rate of stalls, but no significant differences were observed (Supp. Fig. S3). To rule out confounding factors such as fluorophore injection for 2PM, sequential imaging, or animal stress as causes of differences between OCT and Bessel-2PM stalls, an identical imaging schedule was repeated in n = 3 mice but taking a second OCT recording in place of Bessel-2PM after the injection of the fluorophore. The average cumulative incidence curves and stall rate distributions of the two groups were identical (Supp. Fig. S4), providing support that our observed differences between OCT and Bessel-2PM stall dynamics are not due to systematic changes in physiology between the two imaging sessions.
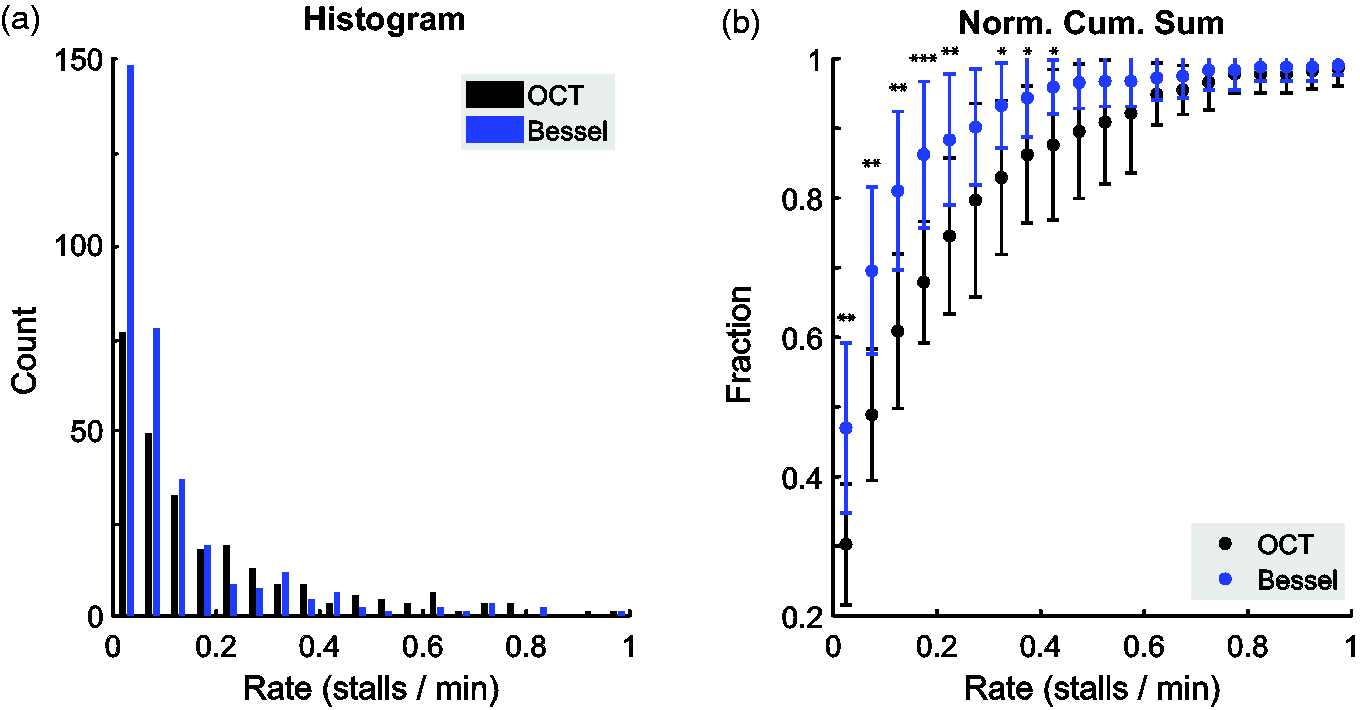
(a) Histograms of stalling rate for OCT (n = 247 capillaries) and Bessel-2PM (n = 320 capillaries). Normalized histogram available in Supp. Fig. S2 and (b) normalized cumulative sum of stalling rate (mean and standard deviation over n = 12 datasets each), with a t-test comparison between OCT and Bessel-2PM. *p < 0.05 **p < 0.01 ***p < 0.001.
The histogram of average stall durations for all capillaries showed that a large majority of stalls are of short duration, with 80% lasting less than 10 s (Figure 4(a) and (b)). The distributions of each method were comparable after the narrower Bessel-2PM histogram bins were stacked to group those that were within the same OCT histogram bin. While longer stall durations can be reliably caught with both, the methods have different sensitivity to the shortest stalls in the first histogram bin because OCT has a longer off-detection period between measuring each capillary and because Bessel-2PM must measure a stall across two image frames (Supp. Fig. S6). Therefore, careful consideration of these biases must be taken when analyzing stall data by duration.
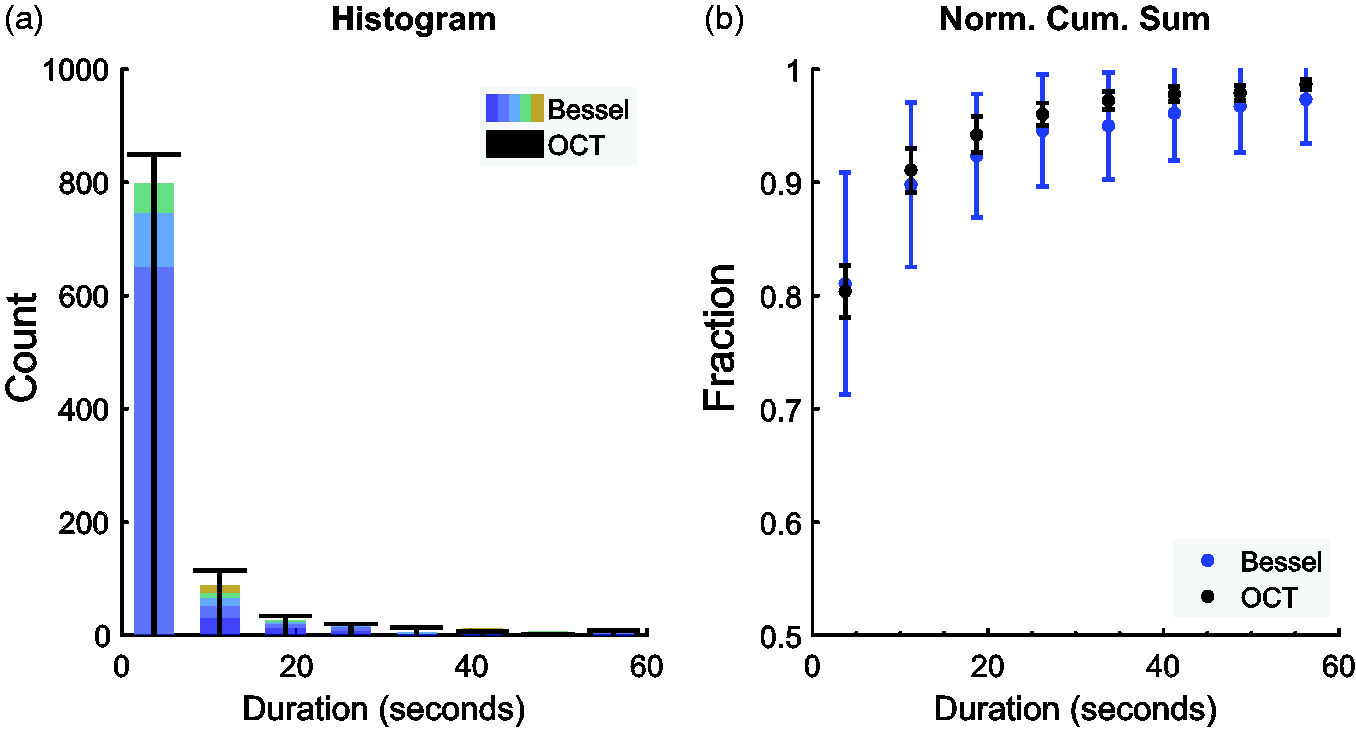
(a) Histogram for Bessel-2PM (n = 662 stalls) and OCT (n = 809 stalls) stall durations, with stacked Bessel bars which group bins that were within the same OCT histogram bin together (each original Bessel bin is a separate color in the stack). Normalized histogram available in Supp. Fig. S5 and (b) normalized cumulative sum of stall durations (mean and standard deviation over n = 12 datasets each), with a t-test comparison between OCT and Bessel-2PM.
The repeatability (fraction of total capillaries that stalled in both of the two sequential recordings) is a metric which can elucidate relationships between stalling tendencies of specific capillaries over longer separations in time. We used repeatability to further analyze stall dynamics by comparing the first and last 10 minutes of each measurement for a given imaging method, or comparing the first 10 minutes from 0-week with the first 10 minutes of the 1-week repeat measurement. Consistent with prior results, OCT showed a 67% repeatability in consecutive 10 min periods and a 50% repeatability one week later (Figure 5(a)). 5 However, Bessel-2PM had lower repeatability, at 47% and 39% respectively (Figure 5(a)). This suggested that repeatability may be different between the two methods due to their differing sensitivity to certain temporal dynamics of stalls discussed above. When the full 20-minute recordings were compared, both methods had a 1-week repeatability of ∼50%. However, the repeatability between methods with data taken sequentially was only 35%. After accounting for biological variability, this may still be lower than expected from data taken sequentially with a single method, owing again to differences in sensitivity to stall dynamics arising from the image scan parameters of each imaging method.
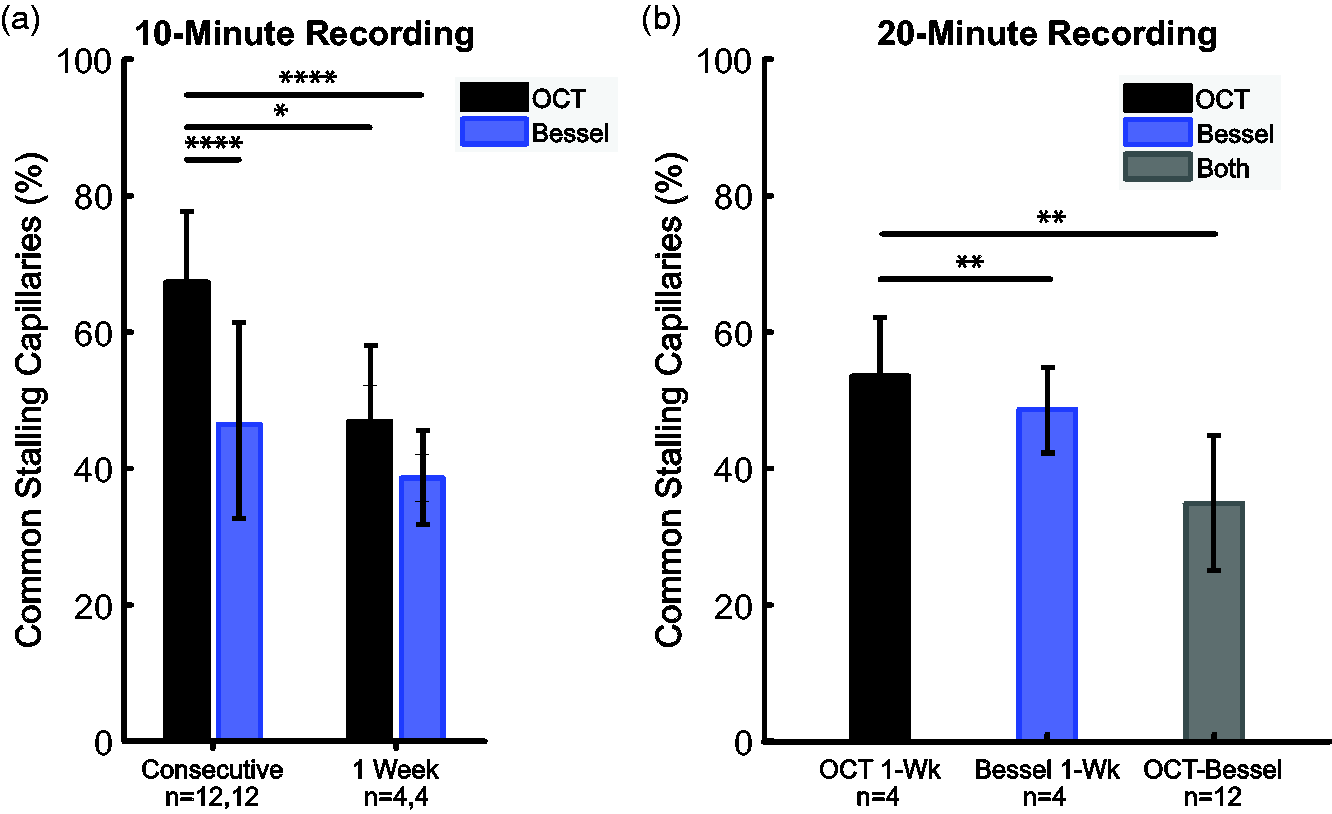
(a) Repeatability of consecutive (n = 12 datasets each) or 1-week separated (n = 4 datasets each) recordings of 10-minute duration and (b) repeatability of 1-week separated (n = 4 dataset pairs) recordings of 20-minute duration or between sequential OCT and Bessel-2PM recordings (n = 12 dataset pairs). Standard deviation is marked with error bars. *p < 0.05 **p < 0.01 ****p < 0.0001 (Welch’s t-test).
In order to delve deeper into the relationship between stall rates and repeatability, we calculated the heatmap of the number of stalls experienced by a given capillary in the first 10 minutes versus the second 10 minutes of each recording. Only capillaries that stalled at least once in either period were considered. For both OCT and Bessel-2PM, high stall rate capillaries tended to stall in both periods (Figure 6(a) and (b)). Furthermore, capillaries which had a higher stalling rate in the initial 10 minutes were more likely to stall in the second period (Figure 6(c)). The survival curves of capillaries based on stall count showed that OCT detected a higher proportion of these frequently stalling capillaries—or conversely a lower proportion of infrequently stalling capillaries—than Bessel-2PM (Figure 6(d)). These results support that higher repeatability of frequently stalling capillaries, which Bessel-2PM are less sensitive to, can explain the lower-than-expected repeatability between OCT and Bessel-2PM in Figure 5.
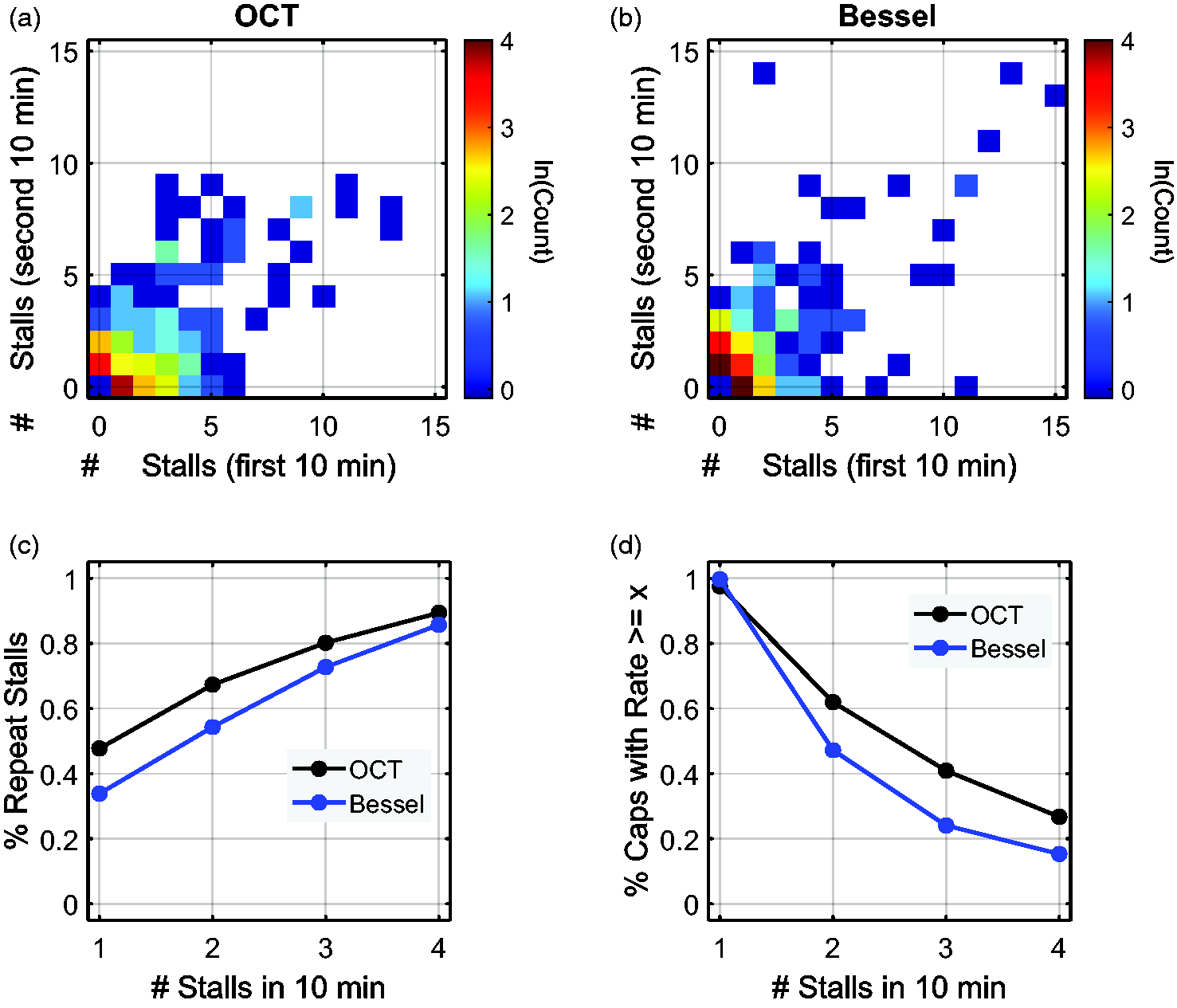
Heatmap of stall count in the first and second 10-minute periods of (a) OCT (n = 247 capillaries) and (b) Bessel-2PM (n = 320 capillaries) recordings for repeatedly stalling capillaries. (c) Repeatability of capillaries separated by stall count for capillaries that had stalls in either 10-minute period and (d) survival function of proportion of total capillaries based on stall count for capillaries that had stalls in either 10-minute period.
To demonstrate the utility of temporal stall dynamics, we applied our analysis to an ischemic stroke model. Photothrombosis was used to induce stroke in the somatosensory cortex of mice (n = 8) and 10-minute recordings of OCT stalling were collected at baseline prior to stroke induction and at 2 days, 1-, 2-, and 4-weeks post-stroke. The cumulative incidence curves were significantly different from baseline at 2 days and 1-week post-stroke by weighted log-rank test (p = 0.0001 and p = 0.005, respectively) (Figure 7(a)). The stall rate distributions for each post-stroke timepoint provided more information, indicating a higher proportion of frequently stalling capillaries at 2 days and 1 week (Figure 7(b))—the proportion of stall rates <0.5 stalls/min was significantly decreased compared to baseline (Figure 7(c)). In addition, the histograms of stall duration showed that while the total number of stalls increased at 2 days and 1 week post-stroke (Figure 7(d)), short stalls were disproportionately affected and accounted for a smaller fraction of total stalls even at week 2 (<30 s stalls) and week 4 (<10 s stalls) compared to baseline (Figure 7(e)). These results suggest that distributions of stall rate and duration can offer a more nuanced biomarker of stalls in disease beyond simply measuring the percent of stalling capillaries.
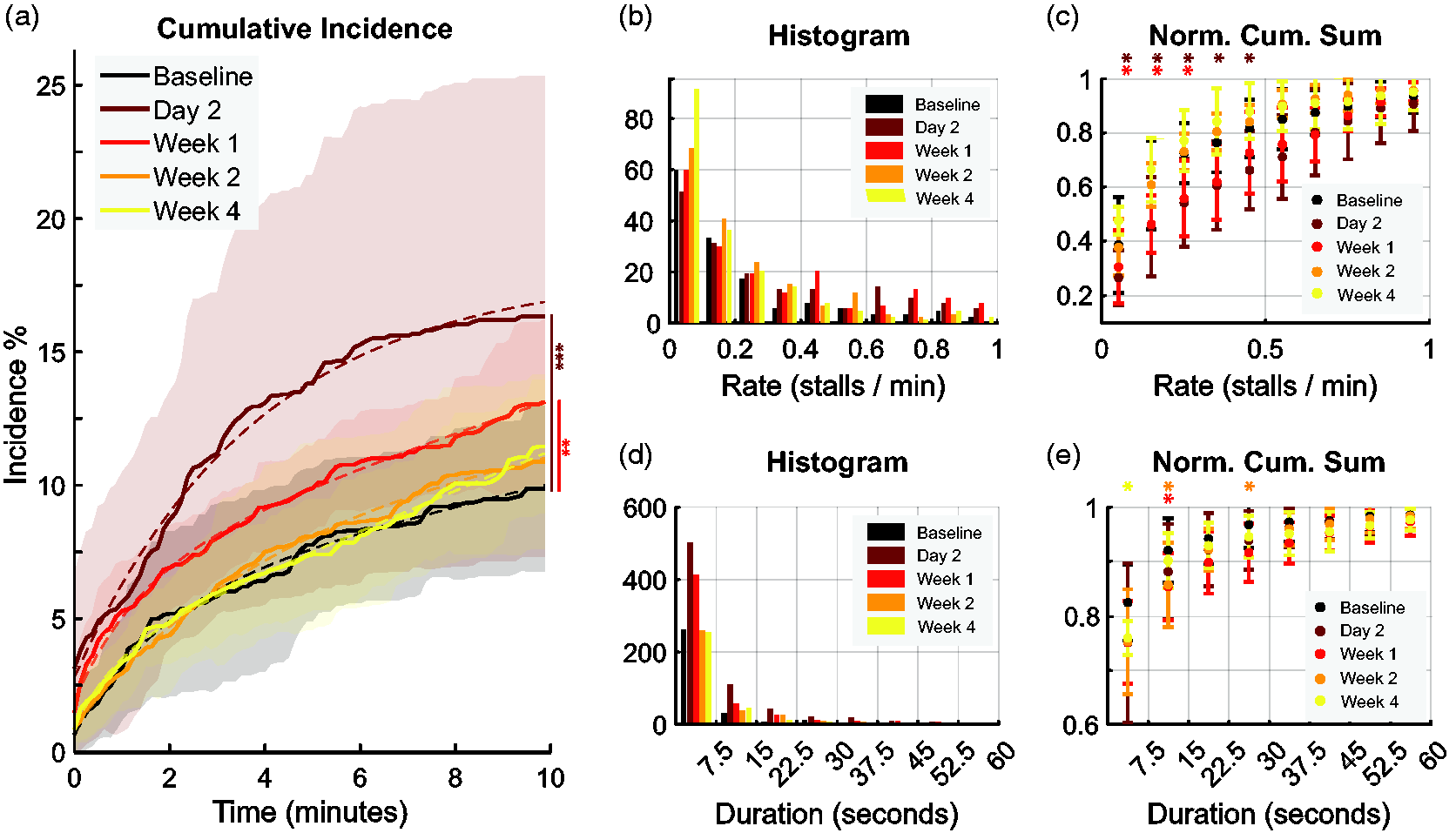
Stalling dynamics under photothrombosis stroke model. (a) Cumulative incidence curves measured at different post-stroke timepoints (mean of n = 8 mice) (weighted log-rank test). Shaded regions are standard deviations, and dotted lines are double exponential fits. Fit coefficients available in Supp. Table S4. (b) Histogram of stalling rate for each post-stroke timepoint (Baseline n = 140; Day 2 n = 181; Week 1 n = 179; Week 2 n = 170; Week 4 n = 180 capillaries). (c) Normalized cumulative sum of stalling rate distribution (mean and standard deviation) with t-test comparison to baseline. (d) Histogram of stall durations for each post-stroke timepoint and (e) normalized cumulative sum of duration distribution (mean and standard deviation) with t-test comparison to baseline. *p < 0.05 **p < 0.01 ***p < 0.001.
Discussion
Our study of the stall dynamics with both OCT and Bessel-2PM revealed that a minority of capillaries have frequent stalls while most capillaries experience infrequent stalls. Further, both methods reveal that most stalls are of a short duration. This novel stall analysis provides a new biomarker for quantifying the temporally dynamic physiology of stalls and can aid interpretations of the biology underlying different pathologies, as we demonstrate in a stroke model. Observing these different populations of stalling capillaries with both OCT and Bessel-2PM strengthens the observation, and at the same time highlights that methodological differences in the image scanning parameters can result in differences in the specific quantities of frequent and short duration stalls observed by each method. These differences from scan parameters must be considered when comparing results between different studies.
Limitations of exponential models of cumulative incidence of stalls
Erdener et al.
5
and Giblin et al.
15
showed that a single exponential model was able to fit cumulative incidence curves well for 18 minutes of OCT or 10 minutes of Bessel-2PM data, respectively. However, a double exponential model resulted in a better fit of our 20-minute data, with a larger improvement on the 20-minute Bessel-2PM data than on the OCT data. This is consistent with the finding that Bessel-2PM is more sensitive to infrequent stalls that form the
Comparison of stall rate and duration distributions between OCT and bessel-2PM
Deeper analysis of the distributions of stall rate and duration revealed nuances about the physiology of stalls as well as differences between the two imaging methods. The stalling rates were exponentially distributed, with a large majority of infrequently stalling capillaries (Figure 3). Bessel-2PM detected a higher proportion of infrequently stalling capillaries compared to OCT, suggesting that Bessel-2PM is able to detect infrequently stalling capillaries with a shorter duration of recording than OCT. The distributions of stall duration for both OCT and Bessel-2PM were similar after Bessel-2PM histogram bins were merged to match OCT bins. This was despite differences in the methods’ sensitivities to stalls in the first histogram bin (<8 s). OCT is sensitive to some stalls below the minimum duration for detection by Bessel-2PM (2 s). However, OCT also has a longer off time between repeat measures of the same capillary, which lowers the probability of detecting stalls less than an image frame period (∼8 s for OCT) in general compared to Bessel-2PM, which will capture all stalls lasting more than 2 frames (∼2–2.5 s).
While many causes for longer stall durations (above ∼10 s) have been found, such as leukocytes and platelets, 23 causes for stalls of shorter durations are less known. Long stalls have been linked to negative outcomes in disease models.20,23,30 However, even short stalls can be physiologically consequential, especially because they make up the majority of stall events. Modeling of tissue oxygenation during passage of RBCs through capillaries suggests that an interval of even ∼1 s without RBC passage can lower local oxygenation by more than 30%, or even greater in tissue up to 15 µm from the capillary. 31 Therefore, we suspect that the large proportion of short duration stalls can alter oxygen tension and prevent adequate perfusion of the brain.
The causes for capillaries to have frequent or infrequent stalls have also not been uncovered. Stalling capillaries tend to have lower RBC and plasma velocities compared to capillaries without stalls. 5 Additionally, flow reversals are known to occur in the capillary network, which can result from changes in the local pressure gradient. 4 We speculate that a large proportion of frequently stalling capillaries experience low pressure gradients due to morphology, diameter, and path length through the capillary network, which result in lower RBC velocities and thereby a greater tendency to stall. We performed an analysis of the capillary branch order from the closest arterioles or venules in Bessel-2PM (Supp. Fig. 8). The branch order distribution was heavily venous, similar to OCT reported in literature 5 (Supp. Fig. 8a,b). These capillaries on the venous side likely experience low pressure conducive to flow reversals and low-pressure stalls. Capillaries with high rate and long average duration of stalls tended to be on the venous side (Supp. Fig. 8c,d), but permutation test (p = 0.20) and Fisher’s exact test (p = 0.20) comparing arterial and venous capillaries did not show significant differences, likely due to low numbers of capillaries on the arterial side (n = 10). In summary, stalls due to low pressure gradients could be consistent with why the majority of stalls are short and infrequent, and why the majority of capillaries are susceptible to stalling. While the data collected in this study do not facilitate analysis of RBC flux and capillary diameter, future work can elucidate these relationships.
Repeatability comparison between OCT and bessel-2PM
The repeatability of 10-minute OCT recordings corroborated prior work 5 (Figure 5(a)). However, Bessel-2PM had a consistently lower repeatability than OCT and only 34% commonality of stalling capillaries between methods, despite more stalling capillaries typically being detected in Bessel-2PM (Figure 1). We attribute the low repeatability of Bessel-2PM to both its insensitivity to <1.8 s stalls (Figure 5(b)) and its higher detection probability for infrequent, less repeatable stalls (Figure 6). These sub-2 s stalls may also explain why stall duration distributions are similar despite differences in the methods’ rate distributions. OCT is able to detect some sub-2 s stalls, but its longer detection off-time causes it to miss many short duration stalls. OCT’s low temporal resolution also classifies these short stalls invariably as 1-frame stalls (Supp. Fig. S6). Within the bin of <8 s stalls, Bessel-2PM can more successfully capture >2 s stalls due to its temporal resolution. While the methods are sensitive to different durations of short stalls, they ultimately detect the same total number of short stalls.
When considering 20-minute measurements spaced 1 week apart, repeatability with both methods increased, but the increase was only significant for Bessel-2PM (p = 0.037, Welch’s t-test), and was lower than the 82% previously predicted based on a single exponential model. 5 We believe this is due to the longer recording duration allowing more infrequently stalling capillaries—that are less repeatable—to be detected, lowering the overall repeatability. In summary, repeatability is lowered within each method due to less repeatable low-rate stalls, and discrepancies between the methods are due to temporal resolution.
Increase in high stalling rate capillaries and long duration stalls after ischemic stroke
Temporal stall dynamics revealed differences in specific stall rates and durations that emerged after ischemic stroke. The distribution of stall rates showed a shift toward higher stall rates at 2 days and 1 week post-stroke (Figure 7(b)), and the proportion of stall rates <0.5 stalls/minute was significantly lower than baseline (Figure 7(c)). Interestingly, the distribution of stall durations indicated an increase in stalls of every duration (Figure 7(d)), but the proportions were not significantly different at 2 days and 1 week post-stroke compared with baseline (Figure 7(e)). Instead, only some durations of shorter stalls were significantly decreased at Week 1, 2, or 4, indicating a possible shift toward longer duration stalls.
Recent work by Williamson et al. has reported a 10-fold increase in incidence even at 28 days post-stroke compared to baseline. 32 However, the incidence measured in our data at Week 4 is comparable to baseline levels, and this discrepancy cannot be explained by distance from the infarct border, as Williamson et al. show no change in incidence up to 1.5 mm away. 32 It is possible that differences may arise from their use of Gaussian-2PM which scans each capillary for 5.4 s. 32 Due to stalls having to persist for the entire capillary’s scan time, a low volume rate, and a low duty cycle, this method may miss shorter, infrequent stalls similarly to OCT but additionally being only sensitive to >5.4 s stalls. Without additional information about the duration of recording, adequate comparisons of incidence measured by Williamson et al. and in the present study cannot be made. Overall, the analysis of stall dynamics applied to stroke suggests that the dominant high-rate stalls may be a physiologically relevant biomarker in disease models.
Considerations
This study is limited by acquiring OCT and Bessel-2PM data sequentially rather than simultaneously, which could have allowed us to register the same stalls in both methods and increase our confidence in identifying stalls that missed detection in one of them. While an integrated system is possible, we deemed sequential acquisition to be adequate due to the high repeatability of stalls over long time periods, which supports our assumption that the dynamics and distribution of stalls will not change drastically between sequential OCT and Bessel-2PM measurements. With sequential imaging, differences in the depth of field between OCT and Bessel-2PM may also arise. While the distribution of stalls is known to depend on cortical depth, 32 we capture consistent cortical depths by cropping the OCT angiogram in depth to match the depth range where Bessel-2PM is sensitive enough to measure stalls, and by matching the volume during acquisition of Bessel-2PM. In cross-modality comparisons, we only consider capillaries visible in both techniques. Therefore, we do not expect depth variations to influence the findings of this paper.
A future improvement to our study would be the addition of cellular labelling to differentiate the causes of frequent and infrequent stall dynamics, as stalls have been shown to be associated with RBCs, leukocytes and platelets. Leukocytes could be labeled with Rhodamine-6G or Hoechst dyes, or with fluorescent anti-Ly6G antibodies for neutrophils, for Bessel-2PM detection. This highlights an advantage of Bessel-2PM over OCT, which is label-free. In practice, however, Rhodamine-6G was found to leak out of vasculature and made Bessel-2PM images unsuited for stall analysis, and Hoechst was incompatible with our system’s spectral configuration. Anti-Ly6G antibodies are likely to alter stalling dynamics, as they have been shown to reduce neutrophil-associated stalls. 8 In the future, more labeling and filtering strategies can be optimized for Bessel-2PM to enable deeper investigation into stalls with relation to temporal dynamics.
Cell labeling would also allow investigation into stalls that may be classified on OCT but not Bessel-2PM, such as slow-moving leukocyte stalls or “plasma-only” stalls in which there are no RBCs visible in the capillary by Bessel-2PM, but the vessel will appear as a typical stall on OCT. “Plasma-only” stalls were not included in the Bessel-2PM analysis because the flowing plasma may still provide some tissue oxygenation. In either case, these events were infrequent, contributing an ∼3% increased stall incidence on average, or a 16% increase in the number of stalling capillaries, and their exclusion did not significantly affect our main findings.
Finally, inflammatory cells which are upregulated after ischemic stroke may be implicated in the increase specifically of high-rate stalls post-stroke. Future directions may focus on improving acquisition rate for both methods to improve sensitivity to frequently occurring short stalls, as well as applying the cell-specific labeling possible with Bessel-2PM in stroke and Alzheimer disease models.
Currently, OCT and Bessel-2PM are both well-suited for imaging of dynamic stalls. OCT requires less intensive manual labeling and has commercially available imaging systems. Although Bessel-2PM has a higher frame rate and opens the possibility for other fluorescent signals to be simultaneously acquired, the intensive manual data analysis that is required is a significant shortcoming compared to OCT. Being label-free, OCT imaging additionally enables measurement of capillary stalling sub-acutely post-stroke where dye leakage through the damaged blood-brain-barrier prevents the use of Bessel-2PM. Additionally, penetration depth and image quality of Bessel-2PM are further reduced post-stroke due to increased tissue scattering and sequestration of leaked dye by microglia, which degrade image quality so that stall analysis cannot be performed. However, with future advances in frame rate, 21 automated analysis of Bessel-2PM data, and fluorescent labeling strategies to overcome these challenges, Bessel-2PM may soon pose a better option to probe basic science questions involving capillary stalling dynamics.
Supplemental Material
sj-pdf-1-jcb-10.1177_0271678X251353141 - Supplemental material for Capillaries susceptible to frequent stall dynamics revealed by comparing OCT and bessel-2PM measurements
Supplemental material, sj-pdf-1-jcb-10.1177_0271678X251353141 for Capillaries susceptible to frequent stall dynamics revealed by comparing OCT and bessel-2PM measurements by Rockwell P Tang, Shannon Kelley, Gülce Küreli, Emily A Long, Piergiulio R Bressan, Shashwat Shah, Şefik E Erdener, John Jiang, John T Giblin, Sreekanth Kura, Matthew G Simkulet, Bradley C Rauscher, Chris B Schaffer, Nozomi Nishimura, Martin Thunemann, Anna Devor and David A Boas in Journal of Cerebral Blood Flow & Metabolism
Supplemental Material
sj-pdf-2-jcb-10.1177_0271678X251353141 - Supplemental material for Capillaries susceptible to frequent stall dynamics revealed by comparing OCT and bessel-2PM measurements
Supplemental material, sj-pdf-2-jcb-10.1177_0271678X251353141 for Capillaries susceptible to frequent stall dynamics revealed by comparing OCT and bessel-2PM measurements by Rockwell P Tang, Shannon Kelley, Gülce Küreli, Emily A Long, Piergiulio R Bressan, Shashwat Shah, Şefik E Erdener, John Jiang, John T Giblin, Sreekanth Kura, Matthew G Simkulet, Bradley C Rauscher, Chris B Schaffer, Nozomi Nishimura, Martin Thunemann, Anna Devor and David A Boas in Journal of Cerebral Blood Flow & Metabolism
Footnotes
Ethical considerations
All experiments and animal procedures were conducted according to protocols approved by the Boston University Institutional Animal Care and Use Committee. We used the ARRIVE checklist when writing our report. 29
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Research reported in this publication was supported by the National Institute of Neurological Disorders and Stroke of the National Institutes of Health under award numbers R01NS127156, T32NS136080, R01NS128947, and R01AG049952. RPT is grateful for support from the National Institute of General Medical Sciences under award number T32-GM145455. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
Acknowledgements
We acknowledge support from the Boston University Neurophotonics Center.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author contributions
DAB, AD, NN, and CBS conceived the study. ShK, RPT, GK, SS, EL, PB, and JJ performed animal preparation and data collection. ShK, RPT, GK, EL, and PB analyzed the data. JTG, SrK, BR developed the imaging system and code. RPT and ShK wrote the paper, and DAB, ŞEE, MT, and MGS critically revised the article. All authors approved the final version.
Supplementary material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.