Abstract
Current metabolomics technologies can measure hundreds of chemical entities in tissue extracts with good reliability. However, long-recognized requirements to halt enzyme activities during the initial moments of sample preparation are usually overlooked, allowing marked postmortem shifts in levels of labile metabolites representing diverse pathways. In brain many such changes occur in a matter of seconds. These comments overview the concern, contrast representative studies, and specify approaches to consider as standards in the field going forward. Comparison with established metabolite signatures of in vivo brain is an essential validation step when implementing any collection method.
Introduction
Study of brain metabolism is central to the scope of the Journal of Cerebral Blood Flow and Metabolism (JCBFM). A deeply rooted focus on energy utilization and its responsiveness to functional demand has broadened to encompass essentially all processes contributing to the integrated physiology and pathophysiology of this ever-remarkable organ. Key to progress is reliable measurement of the compounds comprising pathways of interest. Analytical methods have progressed from fluorometric enzymatic assays of specific metabolites 1 to unbiased metabolomic screens that can assess hundreds of compounds by liquid chromatography-mass spectrometry (LC-MS) 2 and map their distributions using quantitative imaging approaches. 3
Unfortunately, these advances in measurement technology have not been accompanied by general awareness of procedural details essential to proper brain harvest. Figure 1 illustrates the frequency with which different tissue collection approaches were applied in a selection of publications reporting brain metabolomics studies, with a timeline showing the fixation delay for each and changes in representative endpoints occurring during the respective intervals. All but a few studies (∼5% of 900 publications tallied in Supplemental File 1) are confounded by intrinsically flawed methodologies, introducing significant postmortem artifact. The text below will briefly summarize experimental evidence relating to the consequences of postmortem enzymatic activity and the established approaches to minimize such impact.
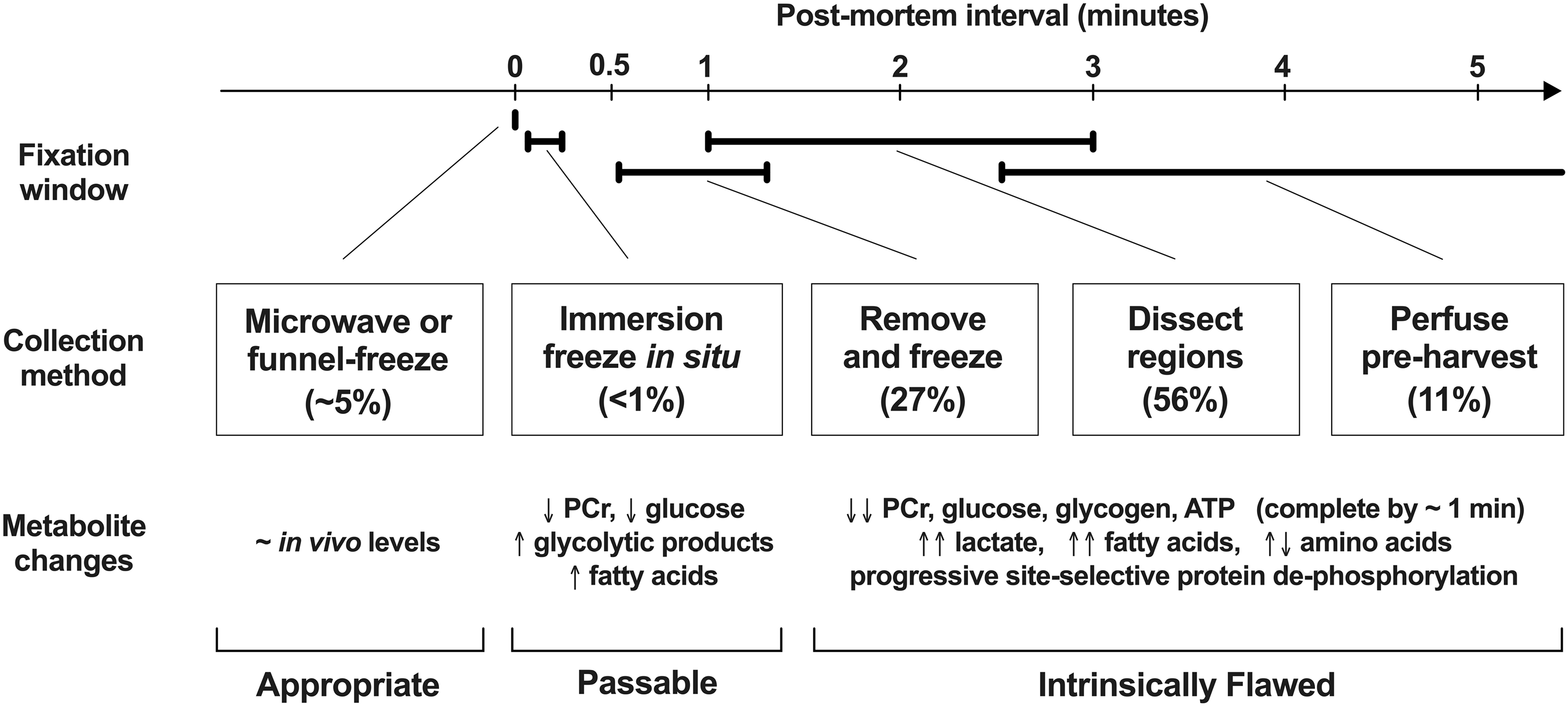
Harvest procedures, timelines, and accompanying endpoint changes. Literature reporting brain metabolomics data was screened to document use of available tissue collection methods (900 source publications are tabulated in Supplemental File 1). This figure summarizes the course of events during the initial minutes postmortem, identifying the minimum intervals at which metabolism can be stopped by each approach, and providing an overview of the range of endpoint changes known to occur within these intervals. The proportion of studies in which each category of harvest procedure was used is indicated in parentheses. Of ∼780 publications specifying an identifiable approach, just 6% employed microwave fixation or in situ freezing methods that alone are sufficiently rapid to approximate in vivo metabolite profiles (metabolism stopped within seconds). All others are confounded by progressive postmortem ischemia, with recognized impacts on components of diverse metabolic pathways. 12 These changes have been most extensively characterized for substrates and intermediates of energy metabolism, but lipids also are rapidly impacted, and protein phosphorylation at some sites is affected within minutes. It should be emphasized that euthanasia involving CO2 is particularly problematic due to its prolonged interval of hypercapnia and hypoxia/anoxia followed by decapitation ischemia, impacting numerous endpoints. 25 Perimortem variables such as anesthesia can alter some metabolites (see text), although effects would be considered modest compared to postmortem changes.
Postmortem changes during tissue collection
In the 1930s studies by Kerr and colleagues4,5 built on still-earlier work to confirm the need to rapidly halt brain enzymatic activity to prevent loss of labile compounds such as phosphocreatine (PCr), ATP, glucose and glycogen, and the accompanying increase in lactate. This established in situ freezing of the living brain with liquid air/nitrogen as a standard approach in the field, adapted to rodents as funnel-freezing. 6 (Details of this and other methodologies are considered below.)
The classic study of Lowry et al. 7 examined an expanded range of metabolites, employing a rapid postmortem freezing method in which mice were decapitated, after which heads were frozen at varied intervals in liquid Freon at −150°C. Within 30 seconds after decapitation, when brain glucose was nearly depleted, the downstream metabolite fructose diphosphate had already undergone marked upward and downward oscillation. Precise time courses varied with age, anesthesia, and temperature, but the seconds to minutes over which changes occurred demonstrated the futility of introducing any brain removal and processing steps whatsoever prior to freezing if in vivo levels were to be reliably inferred. An ultra-rapid freeze-blowing method that extrudes and freezes brain homogenate in less than one second subsequently set the standard against which other approaches can be compared.8,9
Importantly, concerns about lability apply not only to intermediates of energy metabolism but quite generally to diverse biomolecules. The spectrum of postmortem artifact and its implications have been extensively reviewed,10–12 and explicitly addressed by metabolomic approaches in brain and other tissues.13–15 As examples: substantive increases in fatty acids and many lipid mediators occur within seconds to minutes;16–18 most neurotransmitters decrease during the time required for postmortem dissection, whereas GABA increases; 19 levels of many neuropeptides are reduced; 20 protein phosphorylation changes markedly, some sites already maximally dephosphorylated within 2 minutes.21–23 The adverse impact of CO2 euthanasia has been well documented for a wide range of metabolites.19,24,25 Note that many studies utilized microwave irradiation, a contemporary method to inactivate brain enzymes that compares favorably with freeze-blowing,26,27 provided the device achieves comparable sub-second fixation. This approach has been rediscovered in various settings,15,19,24 and as will be argued below is probably the method of choice for most metabolomics studies.
There is sometimes an implicit assumption that changes occurring during brain collection will be comparable in direction and magnitude among experimental groups within a given study, which might allow valid comparison even under conditions of less-than-optimal fixation. This is a flawed assumption. One striking example emerges in a study of brain prostaglandins in wild type and α-synuclein knockout mice, collected after either microwave fixation or decapitation with a delay of 30 seconds for dissection. 28 No strain differences were evident with rapid fixation, but increases occurring during the brief dissection procedure were two-fold greater in the knockout strain. This result has implications regarding postischemic release of arachidonic acid as a precursor for prostaglandin synthesis, but if unrecognized would simply lead to wrong conclusions about baseline prostaglandin levels. Similarly, fixation delay impacts genotype-dependent differences in the mouse liver metabolome. 14 A more general situation involves group comparisons following experimental manipulations, e.g., a recent study examining time-dependent metabolome changes in peri-infarct territory after distal middle cerebral artery occlusion in mice. 29 Brain tissue was dissected after saline perfusion, and metabolite levels were expressed in relative units. Not surprisingly, glucose was essentially undetectable following delayed extract preparation from control brain. The variably greater relative abundance seen on subsequent days post-stroke must largely reflect attenuated postmortem loss, unrelated to any potential in vivo differences. For most metabolites the complex course of known postmortem variation precludes even such limited interpretation. Reported order-of-magnitude increases in itaconate may well reflect the suggested microglial localization. However, until postmortem stability is established for a metabolite there is simply no circumstance in which delayed fixation permits reliable inferences regarding in vivo levels. Further confounds arise when expressed relative to unpredictably changing baselines.
Practical considerations for brain collection and processing
The ultra-rapid freeze-blowing technique provides a gold standard against which other methods can be compared. We exclude it from detailed consideration here since it involves unique instrumentation and precludes anatomical resolution. This leaves in situ freezing which requires anesthesia, or microwave fixation which routinely does not, as approaches suitable for general use in metabolomics studies. Both are routinely approved by animal oversight committees with appropriate justification, which is straightforward given the unambiguous scientific requirement for rapid fixation if meaningful data are to be obtained. Indeed, if properly informed they would reject all other forms of euthanasia for such studies. At their best the methods yield comparable levels of most metabolites in head-to-head comparisons (shown for ATP and PCr in Figure 2), the sometimes-reported superiority of one over the other usually reflecting failures to meet distinct technical challenges of the other approach. Table 1 summarizes basic considerations in implementing collection procedures for metabolomics analyses, detailed below.
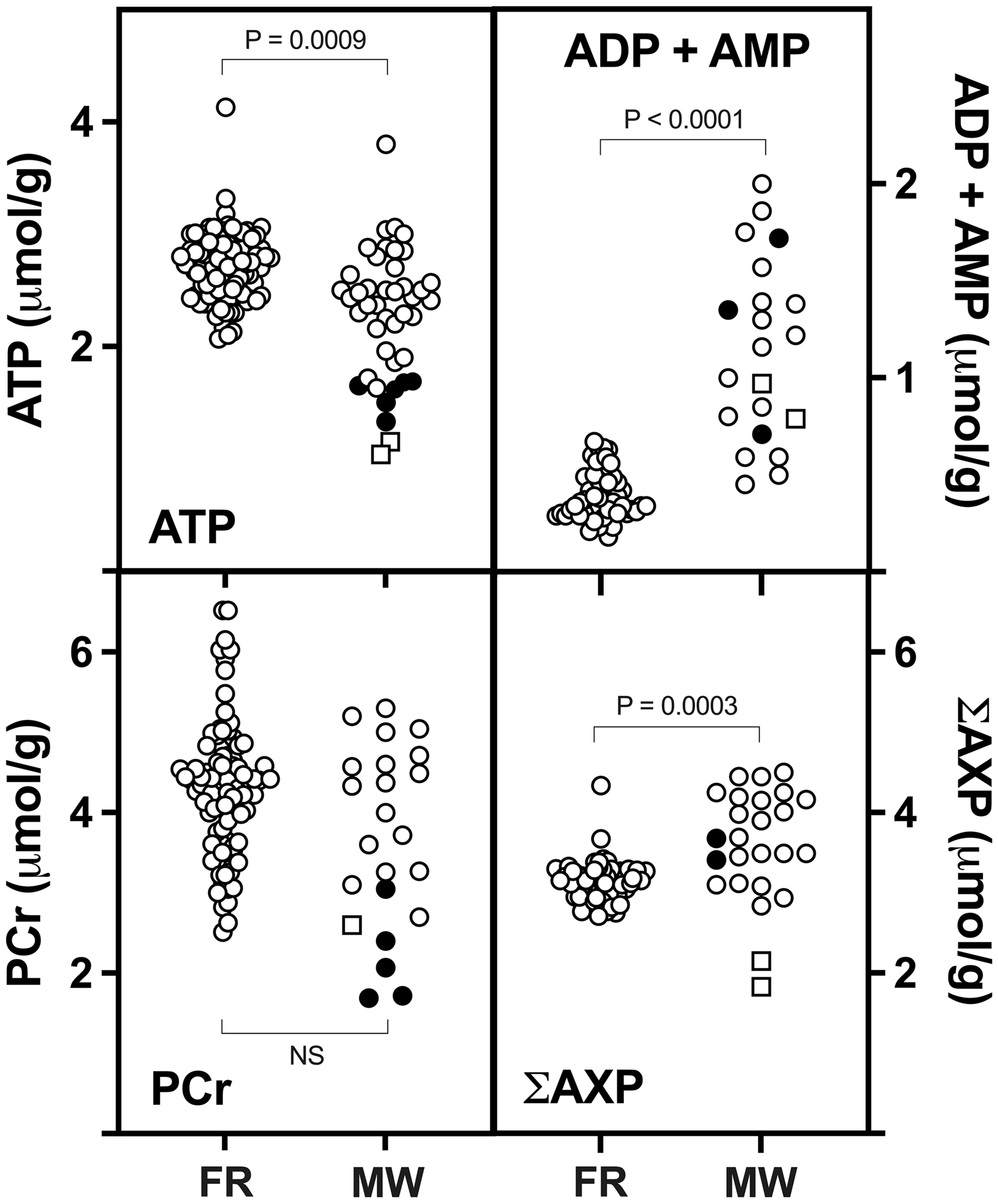
ATP and PCr as indicators of fixation quality, establishing accepted values for in vivo brain and showing the general equivalence of rapid freezing and microwave irradiation. Each point represents the mean value reported for the indicated endpoint in a published study using either rapid freezing (FR) or microwave fixation (MW) to preserve metabolite levels (source data from 120 papers are provided in Supplementary File 2; statistical comparisons utilized the nonparametric Mann-Whitney test). Levels obtained after rapid fixation are 2.7 ± 0.3 for ATP and 4.3 ± 0.8 for PCr (µmol/g, mean ± SD; pooled results for freeze-blowing, funnel and rapid postmortem freezing). ATP averages lower after microwave fixation, not entirely attributable to older (pre-1980) studies using less powerful devices (closed circles) that were excluded from statistical analysis, although some yielded results equivalent to those obtained with rapid freezing or later microwave instruments (open circles). Microwave fixation is associated with increased ADP + AMP, and total adenine nucleotide levels (∑AXP) are also significantly higher. The coinciding transition from enzymatic to chromatographic analyses may be a contributing factor. PCr levels are expectedly more variable than ATP and do not differ significantly with fixation method, but the lowest values were reported in a subset of older microwave studies, consistent with postmortem artifact due to slow fixation. Notably, the two lowest ATP levels involved modern microwave instrumentation (open squares). This was associated with only moderately lower PCr in the study in which it was reported, and somewhat elevated ADP + AMP, but greatly reduced ∑AXP, indicating that variables other than ischemic artifact are likely at play. Given the potential residual activity of adenylate kinase and other enzymes, details of postmortem dissection, storage and extraction procedures clearly require careful consideration even in the setting of microwave fixation. Robust method validation is possible based on established metabolite signatures, as provided by ATP and PCr. Absolute values for metabolite concentrations are needed to permit reliable comparison to the literature.
Key considerations for brain collection and processing in metabolomics studies.

In situ freezing
Whether as a method of euthanasia or immediately postmortem, this is accessible to almost any laboratory via funnel-freezing or direct immersion in liquid nitrogen. By intent, funnel-freezing of the living brain permits sustained local perfusion prior to arrival of the freezing front. Early rodent studies confirmed this to be the case for deeper structures, whereas cortex lost its blood supply via superficial arteries prior to tissue freezing. 6 Nevertheless, since effective ischemia duration in cortex is on the order of seconds and metabolism slows with cooling, in practice the approach provides reliable fixation of most brain structures. Maintaining stable physiology is critical, particularly for rats, since this determines the metabolic integrity of more slowly freezing regions. At minimum it requires avoiding hypoxia due to inhalation of nitrogen vapor and might optimally include physiological monitoring and control.
For immersion freezing, delay in cooling with tissue depth limits reliable application to mice and young rats, and even in these restricts stringent sampling to cerebral cortex. 30 When freezing is performed following decapitation, 7 cervical dislocation 31 or other forms of euthanasia, labile metabolite levels are less-well preserved, 9 particularly in deeper structures. 30 Whole animal freezing without prior euthanasia lessens subcortical ischemia by prolonging circulation and avoiding brain activation (e.g., post-decapitation), 30 making it the preferred approach if regional comparisons are to be attempted. Nevertheless, despite limitations, rapid postmortem in situ freezing can be a reasonable alternative for sampling cerebral cortex, effectively minimizing the changes that otherwise occur during tissue removal and dissection. All freezing approaches require that thawing be avoided until stable extracts are achieved, usually accomplished by dissection in a cryostat and disruption in a medium that remains liquid at subzero temperatures.
Microwave irradiation
Irreversible heat inactivation of enzymes can provide rapid fixation and eliminate many of the tedious and time-consuming procedures required when handling frozen tissues. In principle, its disadvantages are few: commercial instruments are available (at some cost), established procedures heat rapidly yet maintain tissue integrity, thermal instability is not a recognized concern for most metabolites, and diffusion does not present a significant confound for typical regional measurements. In practice, microwave fixation can yield levels of ATP, PCr and lactate approximating those obtained by freeze-blowing,26,27 although this has not always been the case.8,9,32 Deficiencies are often attributed to insufficient power of early devices.17,27 However, the problem is not trivial and, as discussed below, ATP loss appears to present a particular challenge.
Anesthesia
Perimortem variables come into play regardless of brain harvest method, but with care these can have relatively modest impacts on metabolite profiles. Terminal hypercapnia causes large alterations in levels of energy metabolites, neurotransmitters, lipid signaling molecules, and other compounds,19,24,25,33 so CO2 euthanasia is clearly inappropriate for metabolomic analyses. Microwave fixation is often done without anesthesia, permitting most direct comparison with historical freeze-blowing and immersion freezing data. In contrast, surgical preparation for funnel-freezing, and today’s standards for whole-animal freezing, require anesthesia. Such agents affect physiological variables and brain activity,34–38 and can slow postmortem PCr loss,7,39 but there are other potential effects. Most clearly documented are several-fold increases in lactate after exposure to isoflurane or other halogenated volatile anesthetics,40,41 and decreased glucose. 41 These and other changes have emerged in global comparisons of anesthesia effects on metabolite profiles.42,43 Isoflurane use also may have contributed to differences in a cluster of metabolites distinguishing brains frozen under anesthesia from those microwave-fixed. 13 Impacts will differ with agent, dose and duration, establishing the importance of reporting such procedural details to permit reliable cross study comparisons. These considerations strengthen the rationale for awake microwave fixation as the preferred euthanasia method.
Verifying fixation quality
Two key metabolites provide valuable assessments of method performance. Criterion levels for ATP and PCr are 2–3 and 3–5 µmol/g, respectively, based on freeze-blowing data,8,9,44,45 with ATP comprising ∼90% of total adenine nucleotides and [PCr] ≈ [creatine]. Comparable values are achieved with funnel-freezing6,46 or in cortex after immersion of intact animals,30,47 and these standards are frequently met by microwave fixation (Figure 2; data from 120 studies, Supplemental File 2). Reduced ATP levels are evident in a subset of early studies, accompanied by lower PCr, consistent with ischemic artifact. However, the two lowest ATP levels are from recent publications,25,48 indicating that current instrumentation alone has not consistently resolved all issues. PCr is the more sensitive index of postmortem metabolic activity per se, and its preservation indicates the general adequacy of focused microwave irradiation. ATP loss remains particularly prominent as a proportion of total adenylates, reflecting increased levels of ADP and AMP, which are higher in most microwave studies. Mechanisms underlying this sensitivity are unclear. Adenylate kinase is more heat stable than most enzymes. 49 ATP is also markedly more labile after tissue disruption 50 than in the intact brain, 7 which could translate to more rapid loss after suboptimal heat fixation. This illustrates the importance of absolute measurements that can be unambiguously compared with established values. Results expressed in relative units are of more limited utility, but ATP ≥80% of total adenylates and PCr ≥35% of total creatine would together indicate reasonable preparation quality. Lactate and glucose also have been monitored, e.g., to confirm stability following microwave fixation, 51 and could remain useful in some settings. 52 Their reliability is limited by above-noted anesthesia effects. More generally, brain levels of both are driven by blood concentration,53–56 and can be increased by stress, activity, and sensory stimulation prior to brain harvest. As mentioned in Table 1, a range of labile metabolites representing diverse pathways are available for potential validation as fixation standards.12–14
Metabolomics studies in JCBFM
Since its inception in 1981, at least 115 reports in JCBFM have described assessments of potentially labile endpoints in brain (Supplemental File 3; inserted plot documents use of acceptable/unacceptable methods over time). Prior to 2005 all involved targeted assays and appropriate rapid fixation methods when required. At least 13 studies since then can be identified as ‘metabolomic’ in approach. Two utilized in situ freezing, five microwave fixation, and the remaining six postmortem brain dissection. Takenouchi et al. 31 immersed one-week-old rats in liquid nitrogen within 1 second after cervical dislocation, removed the brain at −30°C, and either prepared extracts or collected sections for quantitative imaging mass spectrometry. Frozen tissue was disrupted in ice-cold methanol, or sections were rapidly thaw-mounted on slides. ATP and PCr in whole-brain extracts were reduced by 2- and 3-fold, respectively, relative to optimally frozen cortex, and imaging results confirmed lower ATP and elevated AMP in deeper structures. Although experiencing brief ischemia during postmortem freezing and possibly transient thawing during processing, this represents a good faith, if imperfect effort to capture the in vivo condition. Similarly, Koning et al. 57 froze rat pup heads in −80°C isopentane following decapitation. Cortex samples were dissected frozen, and extraction took place at −10°C so thawing was avoided. Relative measures were used, but control and hypoxic-ischemic groups showed expected reciprocal variation in PCr and lactate levels, indicating that gross postmortem changes had been minimized.
Of studies using microwave fixation,48,58–61 three measured ATP and/or PCr. McDonald et al. 48 reported ATP and total adenylates of 1.15 and 2.15 µmol/g, respectively, among the lowest found in microwave studies. Smeland et al. 58 obtained values of 2.29 and 4.15 µmol/g; PCr was 35% of total creatine, consistent with minimal postmortem ischemia. Both found glucose in an expected range and lactate perhaps slightly elevated, but in both ATP represented only 55% of total adenylates, illustrating the lingering concern regarding adenine nucleotide measurements in microwave studies. Notably, an ex vivo spectroscopy study used PCr as 40% of total creatine to confirm the efficacy of microwave fixation, 61 implementing a quality control measure recommended here.
Clearly, postmortem changes are not entirely avoided even with documented efforts to achieve rapid fixation, illustrating the technical challenges of all methods. One study of brain glycogen metabolism made specific use of its lability, comparing levels after rapid vs. delayed brain collection. 62 Brains in the remaining reports experienced profound ischemia with partially-predictable but quantitatively unknowable impacts on the measured endpoints.29,63–67 Looking broadly at post-2005 studies involving labile endpoints, 50% (20/40) appear to give full consideration to brain collection procedures (Supplemental File 3). In this respect JCBFM far outperforms the literature average but must do better.
Conclusions
Most published brain metabolomics studies are intrinsically flawed, despite a century of experience with freezing methods that effectively limit postmortem artifact, and some decades of microwave availability. Even when these proven methods are employed, essential technical details are too often overlooked or unreported. No fixation method is perfect. Freezing approaches are preferred based on physical principles but are demanding to implement. Microwave irradiation is simplest to perform, and available evidence supports this as the practical alternative, but instrumentation and procedural limitations remain to be fully resolved. Notably, issues concerning ATP loss merit further attention, and similar challenges might arise for other metabolites. Fortunately, the measurements themselves provide indices of fixation quality. Ideally, assessments would be based on absolute levels but with caution ratios could be used, and to an extent retrospectively applied to data from prior studies. Most importantly, these considerations must drive implementation of valid methodological pipelines for future work; appropriate harvest procedures must be required for funding and publication. It will take concerted efforts to inform generations of researchers and journal/grant reviewers. If properly motivated, commercial entities carrying out metabolomic analyses could contribute to practical enforcement. An apparent resurgence in use of focused microwave irradiation bodes well for the quality of such studies going forward provided the above-suggested standards are rigorously validated and applied.
Supplemental Material
sj-xlsx-1-jcb-10.1177_0271678X251314331 - Supplemental material for Setting standards for brain collection procedures in metabolomic studies
Supplemental material, sj-xlsx-1-jcb-10.1177_0271678X251314331 for Setting standards for brain collection procedures in metabolomic studies by Gerald A Dienel and Thaddeus S Nowak in Journal of Cerebral Blood Flow & Metabolism
Supplemental Material
sj-xlsx-2-jcb-10.1177_0271678X251314331 - Supplemental material for Setting standards for brain collection procedures in metabolomic studies
Supplemental material, sj-xlsx-2-jcb-10.1177_0271678X251314331 for Setting standards for brain collection procedures in metabolomic studies by Gerald A Dienel and Thaddeus S Nowak in Journal of Cerebral Blood Flow & Metabolism
Supplemental Material
sj-xlsx-3-jcb-10.1177_0271678X251314331 - Supplemental material for Setting standards for brain collection procedures in metabolomic studies
Supplemental material, sj-xlsx-3-jcb-10.1177_0271678X251314331 for Setting standards for brain collection procedures in metabolomic studies by Gerald A Dienel and Thaddeus S Nowak in Journal of Cerebral Blood Flow & Metabolism
Footnotes
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplementary material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
