Abstract
Aging is accompanied by a decrease in cerebral blood flow (CBF), especially in the presence of preclinical cognitive decline. The role of cerebrovascular physiology including regulatory mechanisms of CBF in processes underlying aging and subclinical cognitive decline is, however, not fully understood. We explored changes in cerebrovascular CO2 reactivity and dynamic cerebral autoregulation (dCA) through the eighth decade of life, and their relation with early cognitive decline. After 10.9 years, twenty-eight (age, 80.0 ± 3.5 years; 46% female) out of forty-eight healthy older adults who had participated in a previous study (age at baseline, 70 ± 4 years; 42% female), underwent repeated transcranial Doppler assessments. Linear mixed-model analyses revealed small reductions in cerebrovascular CO2 reactivity with aging (−0.37%/mmHg, P = 0.041), whereas dCA was modestly enhanced (gain: −0.009 cm/s/mmHg, P = 0.038; phase: +8.9 degrees, P = 0.004). These changes were more pronounced in participants who had developed subjective memory complaints at follow-up. Our observations confirm that dCA is not impaired in aging, despite lower cerebral perfusion and cerebrovascular reactivity. Altogether, this unique longitudinal study highlights the involvement of cerebrovascular health in preclinical cognitive decline, which is of clinical relevance in the development of dementia management strategies.
Introduction
Several studies, mostly cross-sectional, indicate that older age is linked to lower cerebral blood flow (CBF), 1 especially in the presence of cognitive disorders.2,3 In patients with established dementia, CBF is markedly lower compared to healthy controls, whereas patients with early stages of cognitive decline (i.e. mild cognitive impairment) show more subtle differences in cerebrovascular physiology.2 –4 A prospective study in a general population of 4,759 middle-aged and older adults showed that a lower CBF, measured using MRI, was related to future cognitive decline and dementia. 5 These observations suggest that cerebrovascular alterations occur already in the preclinical, often asymptomatic, stages of neurodegenerative diseases that underlie cognitive decline. Since these early cognitive symptoms are associated with higher risk for future cognitive decline,6,7 better understanding of early pathophysiological processes of cognitive decline is of utmost importance in the light of developing novel prevention and treatment strategies for cognitive decline.
We recently found that older adults who developed subjective memory complaints across a decade of follow-up demonstrated a reduction in CBF, and an increase in cerebrovascular resistance, compared to those who remained cognitively asymptomatic. 8 Although this finding is in line with the hypothesis that alterations in CBF are related to the early development of cognitive decline, it raises questions about potential pathways through which these reductions in CBF are mediated. It is therefore important to establish which mechanisms, relevant in the regulation of CBF, are affected in the process of cognitive decline. The regulation of CBF is comprised of a highly complex interplay of multiple factors, including the cerebrovascular response to changes in blood pressure (i.e. static and dynamic cerebral autoregulation) and responses to vasoactive stimuli such as partial pressure changes in arterial carbon dioxide (i.e. cerebrovascular CO2 reactivity).9,10 Previous studies reported conflicting results pertaining to the effect of older age on cerebral autoregulation and cerebrovascular CO2 reactivity, 9 possibly related to the cross-sectional design of these studies. This highlights the need for long-term follow-up studies to better understand the impact of older age on the regulation of CBF. As part of a unique longitudinal follow-up study, 8 we performed repeated comprehensive assessments of cerebrovascular CO2 reactivity under hypo- and hypercapnic conditions, and dynamic CA (dCA) in response to repeated sit-to-stand challenges. The aim of this work was to examine changes in cerebrovascular CO2 reactivity and dCA across a decade of aging, and to explore whether individuals who developed subjective memory complaints across follow-up demonstrate distinct changes in cerebrovascular CO2 reactivity and/or dCA.
Material and methods
Study design and participants
Forty-eight healthy older adults (age, 70 ± 4 years), who had participated in a study initiated in 2008 (ClinicalTrials.gov: NCT01417663), 11 were recruited for this follow-up study. At the time of inclusion between 2008–2010, participants had no history of cardio-/cerebrovascular disease, were not under medical treatment, and did not use any cardiovascular or psychotropic medication. Moreover, cognitive impairment was ruled out by a geriatrician during screening, based on clinical evaluation comprising of medical history, neurological and psychiatric evaluation, physical exam and cognitive testing (MMSE). Detailed information regarding inclusion criteria and health status of the original study population have been described previously. 11 As part of that study, participants underwent comprehensive cerebrovascular hemodynamic assessments. Approximately a decade later, we performed repeated assessments in these same individuals. Irrespective of health status, all surviving individuals from the original study population who possessed adequate visual and auditory acuity, and the capacity to provide informed consent, were eligible for inclusion in this follow-up study. Recruitment procedures for enrolment in this follow-up study are described elsewhere. 8 The study was conducted in accordance with the principles of the Declaration of Helsinki for medical research involving humans, and approved by the local Medical Research Ethics Committee (CMO Arnhem-Nijmegen). All participants provided written informed consent.
Participant characteristics
Participant characteristics were collected and assessed as described in detail previously, 8 including individual data on age, sex, height, weight and upper arm blood pressure at baseline and follow-up. To assess cognitive function, the Mini-Mental State Examination (MMSE) was administered at both timepoints, whereas the Montreal Cognitive Assessment (MoCA) was only administered at follow-up. Participants completed The Older Persons and Informal Caregivers Survey – Short Form (TOPICS-SF) at follow-up to evaluate various domains of health and well-being on a physical and psychological level. 12 Part of this questionnaire asked whether a participant experienced subjective memory complaints (yes/no). This was used to determine the presence of subjective memory complaints. Using the TOPICS-SF, a frailty index was calculated ranging from 0–1. As an additional measure for frailty, maximal handgrip strength was assessed at follow-up.
Instrumentation
Measurement protocols in 2008–2010 and at follow-up in 2019–2020 took place in the same laboratory with comparable equipment. Blood velocities in the right and left middle cerebral artery were continuously measured using two transcranial Doppler (TCD) probes (2 MHz; DWL Multi-Dop at baseline, DWL DopplerBox at follow-up). These were bilaterally placed over both temporal bones and fixed with a customized headband (Spencer Technologies, Seattle, WA). Control parameters, i.e. heart rate, beat-to-beat mean arterial blood pressure (MAP) and end-tidal CO2 (EtCO2), which was derived from the continuous respiratory CO2 waveform, were simultaneously measured using three-lead electrocardiography, finger photoplethysmography (Finapres Medical Systems, Amsterdam), and capnography (BIOPAC Systems, Goleta, CA), respectively. A data acquisition system (MP150, BIOPAC Systems, Goleta, CA) was used to capture all data signals at 200 Hz.
Procedures
Before reporting to the research lab, participants were instructed to adhere to standard dietary and physical restrictions (i.e. avoiding moderate-to-vigorous exercise for ≥24 hours, and refraining from smoking and the intake of alcohol, caffeine or vitamin C for ≥18 hours, and from any caloric intake ≥6 hours prior to the measurements) to standardize cerebrovascular measurements.13,14 Before recordings started, participants had rested at least 5 minutes with legs uncrossed. 14 Accordingly, all cerebrovascular measurement protocols described below were separated by recovery periods of at least 5 minutes in-between.
Hemodynamics and dynamic cerebral autoregulation
Resting normocapnic cerebral hemodynamics were assessed by recording data for at least 5 minutes with the participant in a comfortable seated position. These data were used to derive resting mean MAP and MCAv data (see ‘Data processing and analysis’). Subsequently, hemodynamic oscillations were enhanced at a frequency of approximately 0.05 Hz by repeated orthostatic challenges (10 seconds sitting, 10 seconds standing) for 5 minutes. This enabled assessment of dCA for induced oscillations in the very low frequency domain (0.02–0.07 Hz) using transfer function analysis (see ‘Data processing and analysis’).
Cerebrovascular CO2 reactivity
Cerebrovascular CO2 reactivity was assessed by testing responses in cerebral hemodynamics to changes in EtCO2. Specifically, hypo- and hypercapnic conditions were physiologically triggered by hyperventilation and CO2 inhalation, respectively. Regarding hyperventilation, participants were instructed to take deep breaths at a frequency of 0.5 Hz (1 second breathing in, 1 second breathing out). In 2008–2010, participants were instructed to hyperventilate for 1 minute, whereas at follow-up, participants were asked to hyperventilate for 30 seconds to induce hypocapnia. Hypercapnic conditions were reached by inhalation of a CO2 gas mixture. At follow-up, a 5% CO2 gas mixture was continuously inhaled for 5 minutes, whereas in 2008–2010, CO2 gas mixture concentrations were increased stepwise from 0 up to 7% in the fourth minute. 15
Peripheral endothelial function
Upon inclusion in 2008–2010, peripheral endothelial function was assessed by measuring bilateral blood flow in the upper legs using venous occlusion plethysmography during local intra-arterial infusion of vasoactive substances.11,16 –18 In fixed order, incremental doses of the following substances were administered unilaterally for 5 minutes: i) acetylcholine, an endothelium dependent vasodilator (at 1, 4, 16, 32 and 64 µg/mL/100 mL leg volume); ii) sodium nitroprusside, an endothelium independent vasodilator (at 0.06, 0.20 and 0.40 mg/mL/100 mL leg volume), and iii) NG-monomethyl-L-arginine (L-NMMA), a nitric oxide synthase inhibitor. A detailed protocol of these measurements is described previously. 11
Data processing and analysis
Raw data signals from all recordings were checked and pre-processed in MATLAB (MathWorks Inc, Natick, MA) using custom-written scripts, as described previously. 19 In accordance with international guidelines from the Cerebrovascular Research Network (previously Cerebral Autoregulation Network; CARNet),13,14 estimates of the mean bilateral (or unilateral in case TCD signal quality was sufficient for one side only) middle cerebral artery blood velocity (MCAv), MAP and mean EtCO2 during resting conditions were derived. MAP was calculated from the measured beat-to-beat systolic and diastolic pressures [MAP = 1/3*systolic blood pressure + 2/3*diastolic blood pressure]. To account for the confounding effects of EtCO2 on MAP, 20 the cerebrovascular conductance index (CVCi) was calculated (CVCi = MCAv/MAP). Pre-processed MCAv and MAP data from recordings during the 0.05 Hz repeated sit-to-stand manoeuvres were used as input for the CARNet MATLAB script for performing transfer function analyses. Output parameters from these analyses, i.e. gain, phase and coherence, represent measures of dCA, which were averaged over the very low frequency band (0.02–0.07 Hz).
Data from recordings during hyperventilation and CO2 inhalation were fitted using a smoothing spline (smoothing parameter p = 0.999) in MATLAB, and subsequently evaluated to derive the relevant outcome parameters. First, the minimum values (trough) for EtCO2 and MCAv during hyperventilation were identified, followed by the maximum values (peak) reached during CO2 inhalation. Subsequently, the corresponding MAP and CVCi values pertaining to the timepoint where the minimal/maximal value of MCAv was reached, were determined. For each parameter, i.e. MCAv, MAP and CVCi, the CO2 reactivity was expressed across the hypocapnic (i.e. normo- minus hypocapnic), hypercapnic (i.e. hyper- minus normocapnic), and full (i.e. hyper- minus hypocapnic) range. To account for the differences in CO2 protocol, and also to account for differences in normocapnic values between and within individuals, we corrected for timepoint related normocapnic values and changes in EtCO2. Therefore, the percentage change in MCAv, MAP and CVCi per absolute pressure change in EtCO2 (e.g. %ΔMCAv/ΔEtCO2 in %/mmHg) was calculated.
To assess peripheral endothelial function, blood flow ratios between the infusion and control leg were calculated for incremental doses of each administered vasoactive substance, which effectively corrects for potential systemic effects of the substances.
Statistical analysis
All statistical analyses were all performed using IBM SPSS (version 25.0), and statistical significance was set at P < 0.05. Normality of continuous data was visually checked by graphical interpretation. Linear mixed-model analyses were used that allowed for extrapolation of missing data, and correction for potential confounding factors. To address the primary objective regarding the impact of aging on cerebrovascular parameters as independent outcome variables, the effect of time as a fixed factor (baseline/follow-up) was evaluated with a random intercept, while correcting for baseline age, sex and time-by-sex interaction. After the total group was divided based on the presence of subjective memory complaints at follow-up (yes/no), general participant characteristics were presented as mean with standard deviations or median with interquartile range (IQR), and compared between groups using independent t-tests or Mann Whitney U tests as appropriate. To address the secondary study objectives, i.e. to examine the impact of aging on cerebrovascular parameters with versus without subjective memory complaints, linear mixed-model analyses as explained above were performed, with additional fixed group (subjective memory complaints, yes/no) effect and time-by-group interaction effect. Baseline group differences in blood flow ratio responses to incremental doses of vasoactive substances were similarly analysed using linear mixed-model analyses with a random intercept, fixed dose (used doses for each substance) and group (subjective memory complaints, yes/no) effects, and the dose-by-group interaction effect, without correction for other variables.
Results
Impact of aging
Twenty-eight older adults (46% female) who had participated in the 2008–2010 study responded positively (58%) and underwent repeated follow-up measurements after a median follow-up duration of 131 (IQR 122–138) months. In 2008–2010, participants were 69.1 ± 3.8 years old; at follow-up, they were 80.0 ± 3.5 years old. Body mass index did not change significantly from baseline to follow-up (26.3 ± 3.3 to 26.5 ± 4.4 kg/m2; P = 0.832). A CONSORT-style flowchart is presented in Figure 1. Detailed information on participant characteristics, health status and disease development across 10.9 years of follow-up have been reported previously. 8
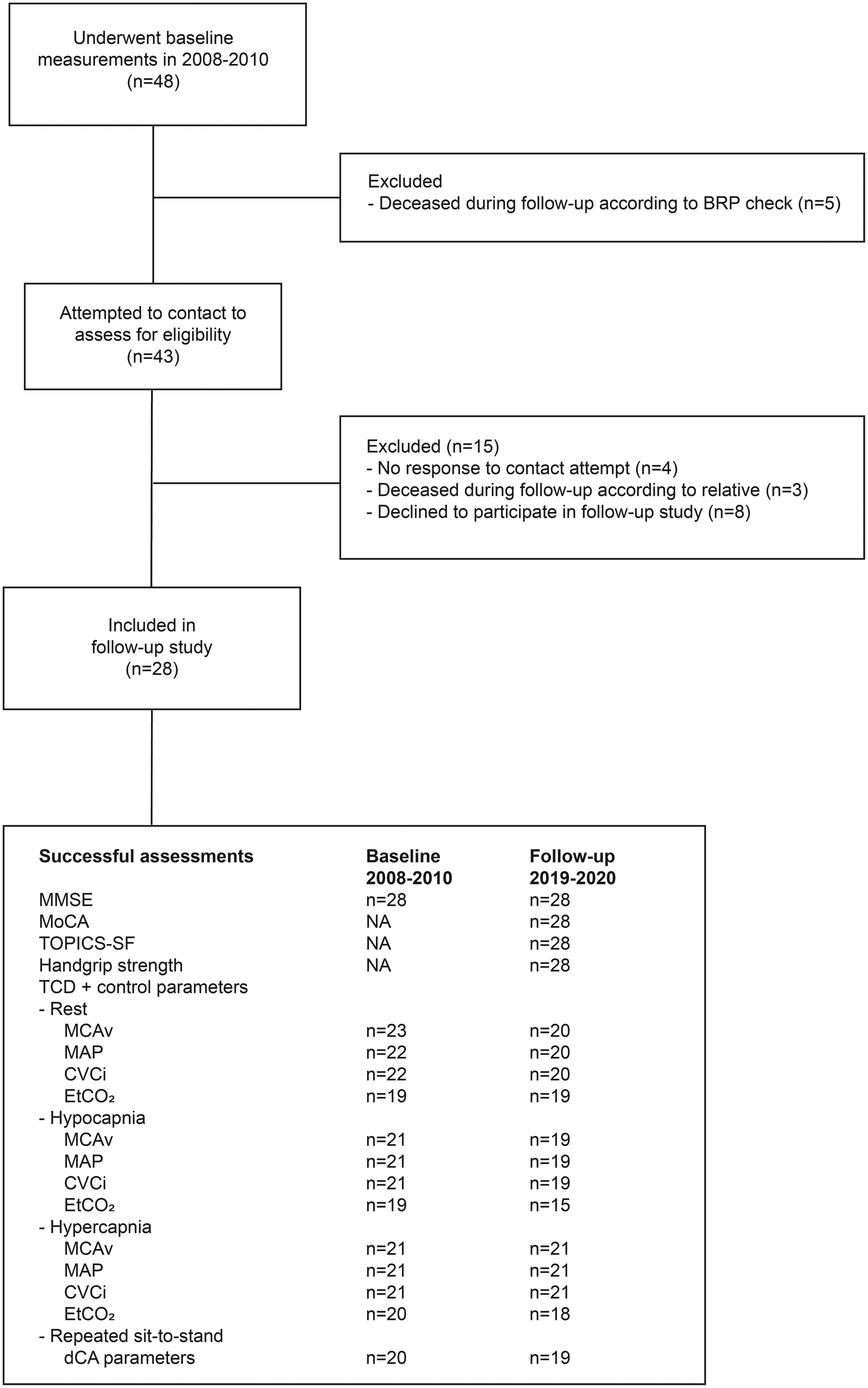
Flow of study participants. CONSORT-style flowchart diagram showing the flow of participants from the original study population at baseline to inclusion in the present follow-up study, together with numbers of successful assessments for each type of measurement at both baseline and follow-up. BRP: Dutch Personal Records Database; CVCi: cerebrovascular conductance index; dCA: dynamic cerebral autoregulation; EtCO2: end-tidal carbon dioxide; MAP: mean arterial pressure; MCAv: mean bilateral middle cerebral artery blood velocity; MMSE: Mini-Mental State Examination; MoCA: Montreal Cognitive Assessment; TCD: transcranial Doppler; TOPICS-SF: The Older Persons and Informal Caregivers Survey – Short Form. Figure created with Adobe Illustrator (2021).
Resting hemodynamics
After follow-up, there were no significant changes in resting, normocapnic cerebral hemodynamics (MCAv, MAP and CVCi) for the whole group (Table 1).
Effects of aging on cerebral hemodynamics, CO2 reactivity and dynamic autoregulation.
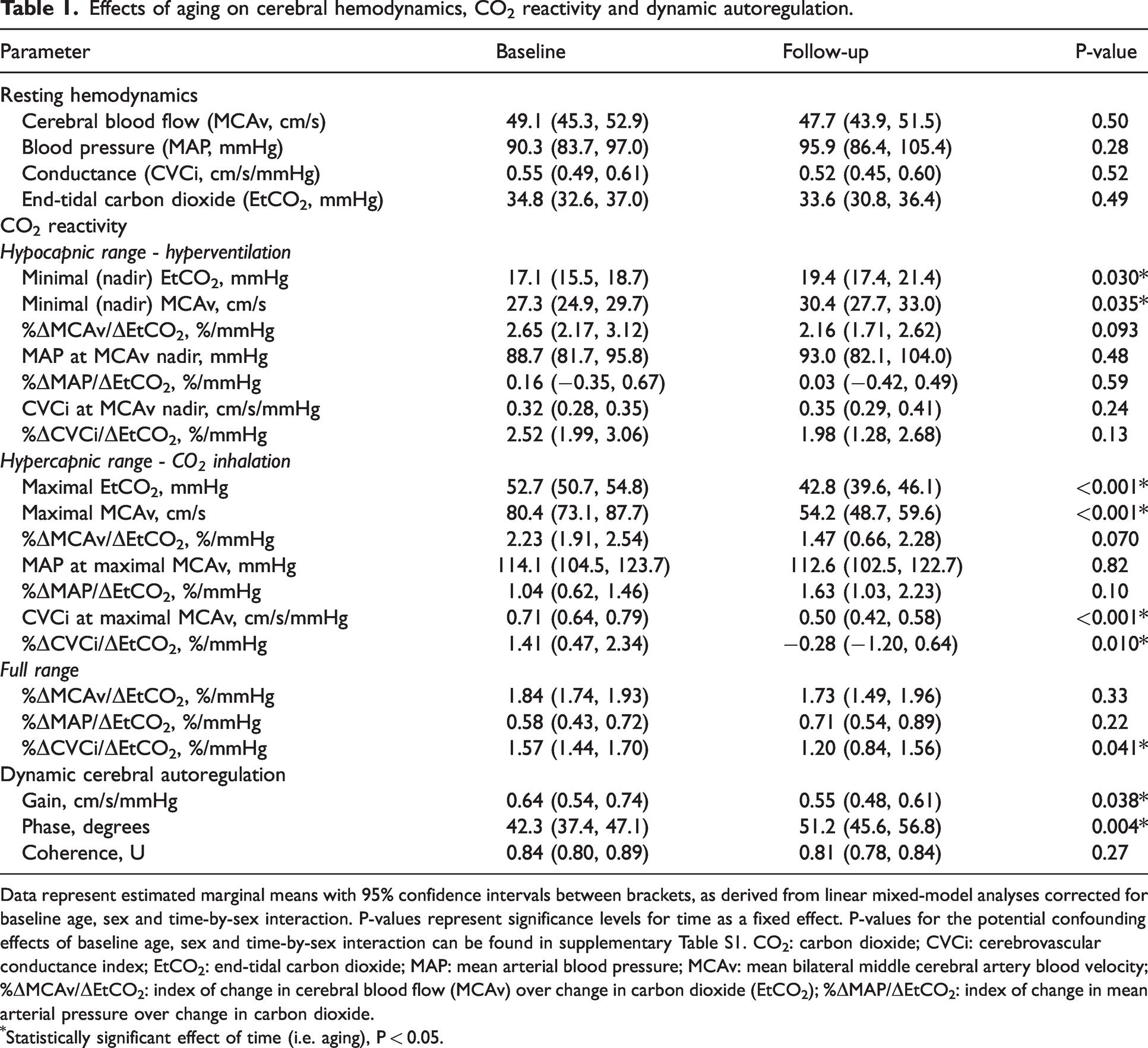
Data represent estimated marginal means with 95% confidence intervals between brackets, as derived from linear mixed-model analyses corrected for baseline age, sex and time-by-sex interaction. P-values represent significance levels for time as a fixed effect. P-values for the potential confounding effects of baseline age, sex and time-by-sex interaction can be found in supplementary Table S1. CO2: carbon dioxide; CVCi: cerebrovascular conductance index; EtCO2: end-tidal carbon dioxide; MAP: mean arterial blood pressure; MCAv: mean bilateral middle cerebral artery blood velocity; %ΔMCAv/ΔEtCO2: index of change in cerebral blood flow (MCAv) over change in carbon dioxide (EtCO2); %ΔMAP/ΔEtCO2: index of change in mean arterial pressure over change in carbon dioxide.
Statistically significant effect of time (i.e. aging), P < 0.05.
Cerebrovascular CO2 reactivity. During hyperventilation (hypocapnia), the minimum values reached for EtCO2 and MCAv at baseline were lower than after follow-up, while for hypercapnia the maximum values reached for EtCO2 and MCAv at baseline were higher than after follow-up (Table 1). The cerebrovascular CO2 reactivity index for MCAv (%ΔMCAv/ΔEtCO2, %/mmHg) in the hypo- and hypercapnic ranges were lower after follow-up, but these differences did not reach statistical significance (P = 0.093 and P = 0.070, respectively). However, the reactivity index based on CVCi was significantly reduced in the hypercapnic and full range at follow-up. Changes in MAP to hypo- or hypercapnia were not significantly different across follow-up, whilst CVCi was only altered after follow-up in response to hypercapnia (Table 1).
Dynamic cerebral autoregulation
For blood pressure oscillations using repeated sit-to-stand manoeuvres at 0.05 Hz, we found significant decreases in gain (−14%) and increases in phase (+21%) from transfer function analyses over the very low frequency band across time (Table 1). No significant changes after follow-up were observed for coherence (Table 1).
Relation with subjective memory complaints
Fifteen (54%) participants reported subjective memory complaints at follow-up. These participants did not significantly differ in age from participants without memory complaints, and had a comparable follow-up duration (Table 2). Participants with subjective memory complaints comprised more females, had a lower BMI at baseline, and at follow-up, had a higher frailty index and lower handgrip strength compared to those without subjective memory complaints (Table 2). No baseline group differences were found for blood flow responses to incremental doses of acetylcholine or sodium nitroprusside infusion (Figure 2(a) and (b)), whereas participants who developed subjective memory complaints demonstrated larger reductions in blood flow ratio responses to incremental doses of L-NMMA infusion (Figure 2(c)).
General characteristics for participants with and without subjective memory complaints.
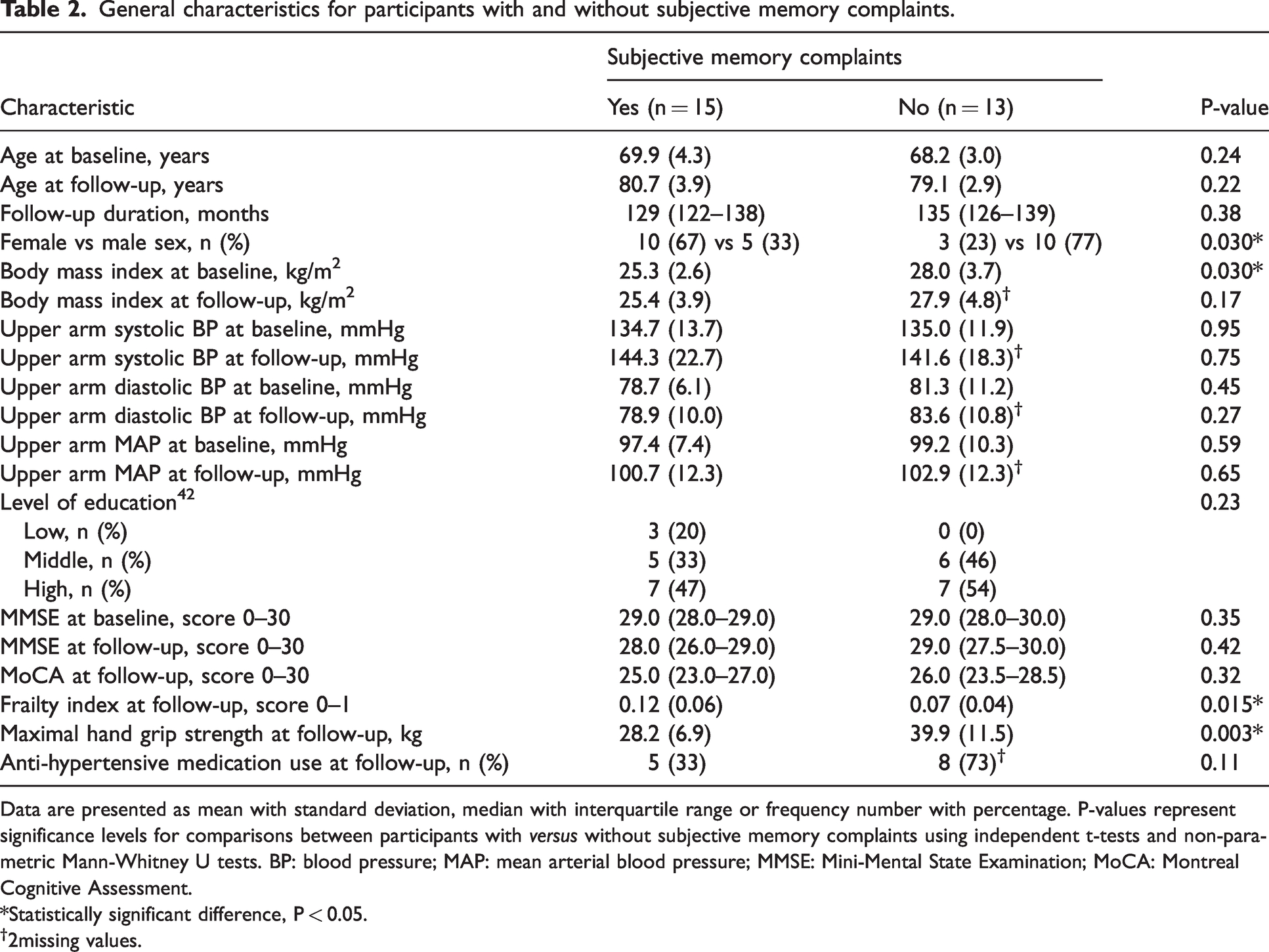
Data are presented as mean with standard deviation, median with interquartile range or frequency number with percentage. P-values represent significance levels for comparisons between participants with versus without subjective memory complaints using independent t-tests and non-parametric Mann-Whitney U tests. BP: blood pressure; MAP: mean arterial blood pressure; MMSE: Mini-Mental State Examination; MoCA: Montreal Cognitive Assessment.
*Statistically significant difference, P < 0.05.
2missing values.
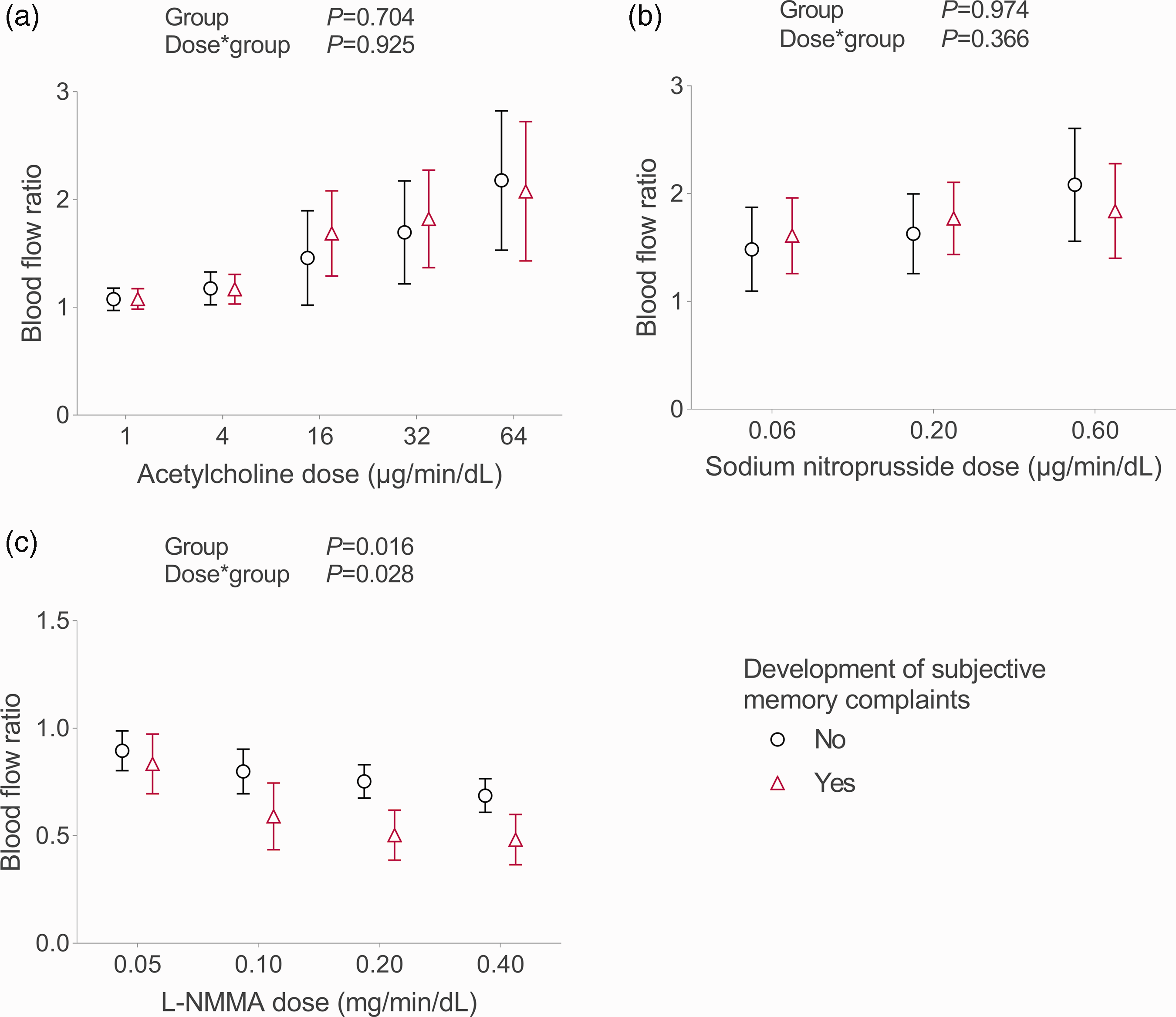
Baseline blood flow ratio responses to vasoactive substances. Figures present estimated marginal means with 95% confidence intervals from linear mixed-model analyses for baseline blood flow ratio responses to infusion of incremental doses of a) endothelium dependent vasodilator acetylcholine, b) endothelium independent vasodilator sodium-nitroprusside, and c) nitric oxide synthase inhibitor L-NMMA for participants with (red triangles) and without (black circles) development of subjective memory complaints. Figure created with GraphPad Prism 9.
Resting hemodynamics
Participants with subjective memory complaints demonstrated reductions in resting MCAv (⊟10%) and CVCi (⊟20%), while participants without subjective memory complaints had small increases in MCAv (+9%) and CVCi (+19%) after 10 years of follow-up (Table 3). No significantly distinct changes in MAP and EtCO2 were found across groups during follow-up.
Effects of aging with and without subjective memory complaints on cerebral hemodynamics, CO2 reactivity and dynamic autoregulation.
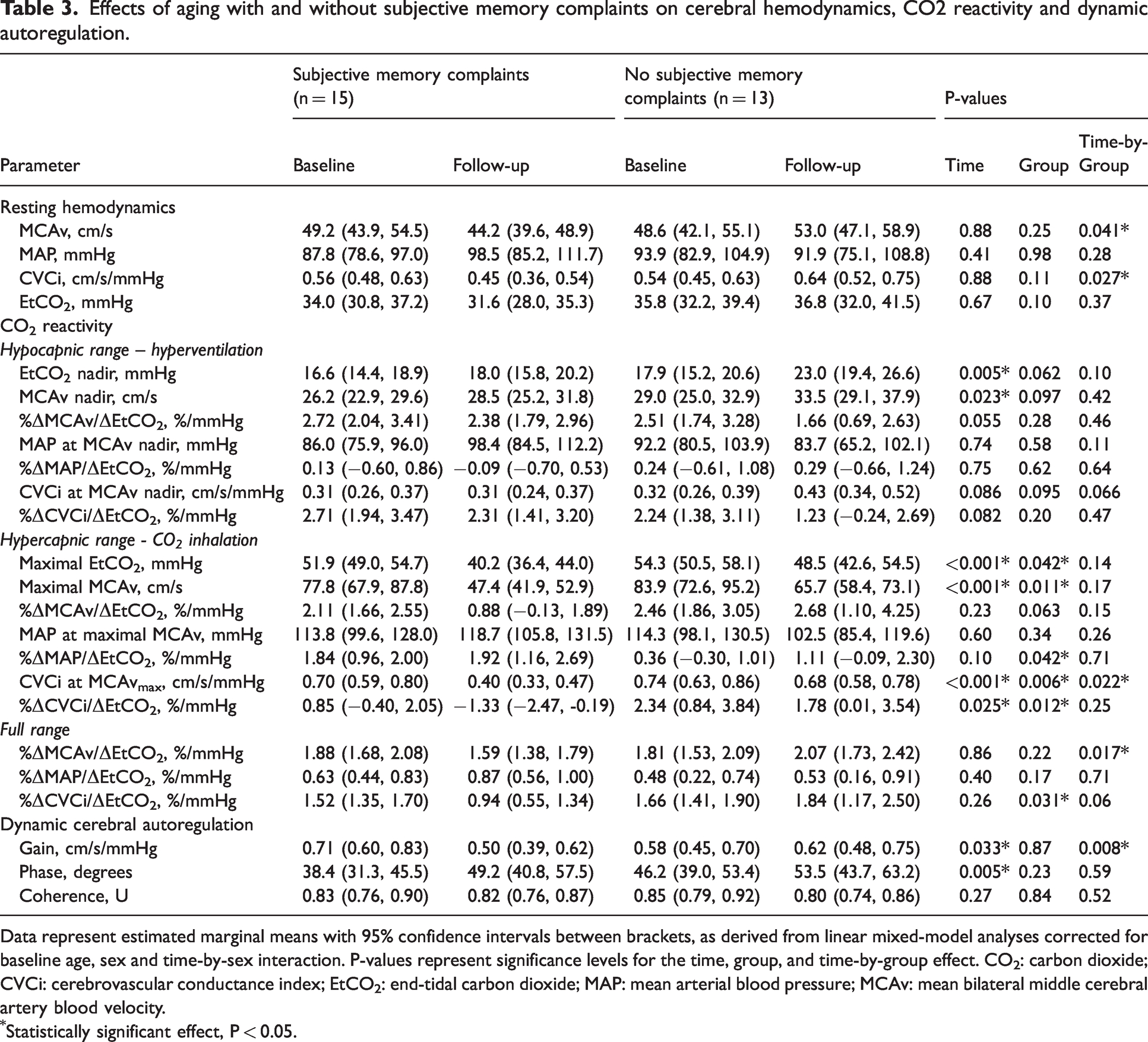
Data represent estimated marginal means with 95% confidence intervals between brackets, as derived from linear mixed-model analyses corrected for baseline age, sex and time-by-sex interaction. P-values represent significance levels for the time, group, and time-by-group effect. CO2: carbon dioxide; CVCi: cerebrovascular conductance index; EtCO2: end-tidal carbon dioxide; MAP: mean arterial blood pressure; MCAv: mean bilateral middle cerebral artery blood velocity.
Statistically significant effect, P < 0.05.
Cerebrovascular CO2 reactivity
For the full hypo-to-hypercapnic range, we found a significant time-by-group interaction effect in the cerebrovascular CO2 reactivity index for MCAv (P = 0.017), and a trend for CVCi (full range, P = 0.063, Table 3; significant effect for the hypercapnic range, P = 0.022, Table 3). Overall, data summarised in Table 3 suggest that the trends for age-related reductions in cerebrovascular CO2 reactivity observed for the pooled data are primarily driven by changes in the participants with subjective memory complaints, with marginal reductions in cerebrovascular CO2 reactivity in those without subjective memory complaints.
Dynamic cerebral autoregulation
Analyses revealed a larger reduction in gain in participants with subjective memory complaints compared to those without, whereas no significant time-by-group interactions were found for phase and coherence (Table 3).
Discussion
Supported by previous studies,1,21 –23 we recently suggested marginal (i.e. ≤1% annually) reductions in CBF across a decade of aging in twenty-eight healthy older adults who were ∼70 years old at baseline. 8 More importantly, we observed that these reductions appeared driven by a subset of healthy older adults who had developed subjective memory complaints. In these same individuals, the present study explored potential underlying mechanisms for these findings. Specifically, we investigated two important regulators of CBF, i.e. cerebrovascular CO2 reactivity and dynamic cerebral autoregulation, and their relation with the development of subjective memory complaints.
Impact of aging
The current literature provides discordant results regarding the effects of aging on cerebrovascular CO2 reactivity.9,15,24,25 The same is true for the literature regarding dCA in aging. While it is often suggested that aging is accompanied by impaired dCA, for example because age-related diseases such as stroke or dementia have been associated with impaired autoregulation, emerging evidence suggests that dCA remains intact in advanced age stages.9,15,26 Importantly however, the majority of previous work was limited to cohort studies and cross-sectional evidence, hence subject to various sources of bias. Therefore, our long-term follow-up study contributes to a better understanding of the true effects of healthy aging within individuals. Overall, our findings point towards subtle reductions in cerebrovascular CO2 reactivity, while confirming preserved, if not enhanced, dCA with aging in healthy older adults.
The concept of vascular aging describes age-related structural changes in the cardiovascular system (e.g. arterial wall thickening) that are accompanied by vascular dysfunction throughout the whole vascular tree, consequently leading to increased risk of cardio- and cerebrovascular disease. 27 In line with this concept, we observed reductions in cerebrovascular CO2 reactivity across follow-up, possibly mediated through endothelial dysfunction. 28 The lower reactivity may ultimately underlie, at least in part, the reduction in CBF observed with older age. Interestingly, we found that age-related reductions in cerebrovascular CO2 reactivity were mainly present during CO2 inhalation rather than in response to hyperventilation. This suggests that the vasodilatory ability of the cerebrovasculature is affected during aging rather than its ability to constrict, and highlights the importance of studying hypercapnic and hypocapnic conditions separately when assessing cerebrovascular CO2 reactivity.
Regarding dCA for induced oscillations through postural changes, transfer function analyses revealed decreases in gain and increases in phase. This combination of transfer function parameters suggests enhancement of dCA, wherein autoregulation has become more efficient across time in damping effects of blood pressure on CBF (gain) and is efficiently counteracting changes in blood pressure, increasing the phase shift between oscillations in blood pressure and CBF (phase). This finding at least debunks the suggestion that dCA declines with aging and would support findings from most recent cross-sectional studies that indicate preserved dCA with older age. Our findings are also in line with those of a recent trial with tadalafil, a drug aimed to increase CBF in older adults with vascular cognitive impairment. While this drug lowered both systolic and diastolic blood pressure, CBF showed small increases in grey and white matter, including in brain areas most affected by small vessel disease, suggesting preserved autoregulation. 29 This highlights the robustness of the autoregulatory mechanism. Importantly, dCA is dependent of smooth muscle cell responses to changes in intravascular pressure. 9 Therefore, our findings suggest that this smooth muscle response to arterial pressure is not affected by aging. They also indicate that dCA is well maintained regardless of the cerebrovascular changes in those with subjective memory complaints, who showed reductions in CBF and reductions in CO2 reactivity. Our observations raise the question how to interpret enhanced dCA with aging, specifically: does enhanced dCA represent a true improvement, or should it be considered a compensatory mechanism that aims to maintain sufficient CBF in aging, possibly with underlying neurological disease? Therefore, we have compared groups based on the presence of subjective memory complaints at follow-up, a possible early marker for neurodegenerative disease.6,7
Relation with subjective memory complaints
Examining participants who developed subjective memory complaints, we found a lower cerebrovascular CO2 reactivity index for CVCi, regardless of time, and larger reductions in cerebrovascular CO2 reactivity index for MCAv across follow-up, compared to peers who remained free from subjective memory complaints. Moreover, enhanced dCA was more pronouncedly found in those who developed subjective memory complaints, as expressed by larger decreases in transfer function gain.
It is commonly known that patients with Alzheimer’s disease and other vascular or mixed-type dementias demonstrate altered cerebrovascular physiology compared to cognitively healthy peers.2,3 Such observations were also made in individuals with a clinical diagnosis of mild cognitive impairment,2 –4 which represents a transition-stage between healthy age-related cognitive decline and progressive cognitive decline or dementia. 30 Accordingly, cerebrovascular pathology has been recognized as an early marker and risk factor for future cognitive decline.5,31 Possibly, cerebrovascular observations in healthy individuals may be subject to bias due to subclinical neurodegenerative disease. Indeed, we found differences in cerebrovascular parameters between healthy participants with versus without development of subjective memory complaints. Specifically, those with subjective memory complaints showed (more progressive) deterioration in cerebrovascular health, as indicated by impairments in cerebrovascular CO2 reactivity.
Our finding of enhanced dCA in participants who developed subjective memory complaints across follow-up reinforce conclusions from a recent meta-analysis, stating that dCA is at least preserved, and possibly even better, in patients with mild cognitive impairment or Alzheimer’s disease compared to controls. 32 Nonetheless, enhancement of dCA in (preclinical) cognitive decline requires explanation, and we propose some possible explanations. First, higher cerebrovascular resistance can enhance dCA,33 –35 expressed by lower gain and higher phase. The cerebrovasculature in individuals with preclinical cognitive decline may be in a more vasoconstrictive state. In support of this, a previous study demonstrated reduced transfer function gain in hypertensive participants who had cerebral vasoconstriction (i.e. lower CBF, higher cerebrovascular resistance), which increased to normal levels after anti-hypertensive treatment. 36 Secondly, our findings may relate to ‘hysteresis’ or directional sensitivity of dCA. Recent work has suggested that dCA is less efficient for reductions in blood pressure than for increases in blood pressure. 37 This may be best explained at the level of the vascular smooth muscle, where vasoconstriction occurs more swiftly than vasodilation. 37 Possibly, enhanced dCA may reflect a chronic shift towards the more sensitive direction of dCA. Third, ‘improved’ dCA may be recognized as a compensatory effect for reductions in CBF and cerebrovascular CO2 reactivity. Once compensatory effects of dCA ultimately fail, neurodegeneration and cognitive decline may accelerate due to critical insufficient levels of CBF. Taken together, although mechanisms are largely unknown and future research is warranted, our data support the presence of enhanced dCA with older age, especially in those with subjective memory complaints.
Examining lower limb peripheral vascular function, no differences were found between groups in endothelial dependent or independent function. In contrast to expectations, those who developed subjective memory complaints demonstrated a larger reduction in blood flow ratio responses to incremental doses of an nitric oxide synthase inhibitor, indicating a greater contribution of nitric oxide to peripheral vascular function. At least, our findings suggest that peripheral vascular function does not relate to changes in cerebrovascular perfusion or development of subjective cognitive decline across 10.9 years follow-up. This provides further support for peripheral and cerebrovascular arteries being regulated through distinct pathways.
Interestingly, participants who developed subjective memory complaints were more frequently female, and seemed to be more frail at follow-up, as indicated by the frailty index, and expressed by maximal handgrip strength. It is known that female sex is associated with a higher risk of the most common form of dementia, i.e. Alzheimer’s disease, accounting for 60–80% of dementia cases.38,39 Reasons for this remain uncertain. Current arguments include that men are less likely to get diagnosed because women care for them, and/or because women have a higher life expectancy than men. Alternatively, gender differences in personality or perception may explain the difference in sex proportions, as a result of which women may be more likely to admit and/or report the presence of subjective memory complaints compared to men. However, in this study, all these arguments do not explain the differences that were found, pointing towards more biological reasons. At least, observed differences regarding changes in CBF, cerebrovascular CO2 reactivity and dCA across follow-up between groups are unlikely driven by differences in sex, as our linear mixed-model analyses for the whole group revealed no significant effects of sex on changes in these parameters (supplementary Table S1). The fact that the group with subjective memory complaints comprised relatively more females may explain lower maximal handgrip strength in this group, as reference values for women are lower than for men. Unfortunately, our analyses on handgrip strength did not correct for sex (or age). The lower frailty index is however an interesting finding, that aligns with an underlying neurodegenerative process that explains both subjective memory complaints and, at least partly, frailty.
Based on the MAP values pertaining to the hemodynamic protocols, one may suggest that participants with subjective memory complaints had increased cardiovascular risk after follow-up, because this group demonstrated ∼10 mmHg increase in MAP. It is however important to realise that MAP values presented in Tables 1 and 3 were measured with finger photoplethysmography, that was used to relate changes in blood pressure to changes in CBF, and to assess measures of cerebrovascular CO2 reactivity and dCA. Finger photoplethysmography has not been validated to provide absolute values of blood pressure. Upper arm blood pressure values, which were assessed by clinically validated measurements to diagnose hypertension, are presented in Table 2. These values were comparable between groups at both timepoints. Specifically, both groups demonstrated ∼3 mmHg increase in upper arm MAP across time.
Limitations
An important contributing factor from this work is the long-term longitudinal design. However, few limitations should also be acknowledged. First, our study population was relatively small, which may have led to underpowered results. However, the proportion of original participants that reported to our study lab for repeated measurements (68%) exceeded expectations, whilst the strong, within-subject follow-up design detected strong effect sizes that reinforces previous work. For example, our finding of enhanced dCA across a decade of aging, especially in those who developed subjective memory complaints, strongly confirms that dCA is at least preserved in healthy aging. A second limitation is that presence of subjective memory complaints was reported in response to a single yes/no-question on the TOPICS-SF. Unfortunately, objective measures of cognitive function, i.e. the MoCA and MMSE, were not sensitive enough to explore the subtle differences and changes in cognitive function in a relatively healthy group. Future research in this area should consider an extensive questionnaire about memory complaints such as the Memory Functioning Questionnaire 40 or Prospective and Retrospective Memory Questionnaire. 41 Third, significant differences between baseline and follow-up in reached EtCO2 concentrations during hypo- and hypercapnia were found. This could be explained by discrepancies in measurement protocols between timepoint assessments, but also because of inability of participants for sufficient hyperventilation to reach lower EtCO2 concentrations. Nonetheless, we have corrected for differences in EtCO2 by calculating cerebrovascular CO2 reactivity indices expressed as a unit change in MCAv/CVCi per pressure change in EtCO2. On the other hand, as derived from transfer function analyses, the overall stronger relationships between MCAv and MAP during repeated sit-stand manoeuvres in this study compared to under previously examined resting conditions (coherence ∼0.8–0.9 versus ∼0.5–0.6), 8 emphasize the additional value of exploring dCA for induced blood pressure oscillations in addition to dCA for spontaneous oscillations. A final important limitation of this study was that baroreflex sensitivity was not taken into account in this study. Nonetheless, baroreflex sensitivity has been assessed as part of a previous study that used the baseline assessments in our study population. 19 In that study, our population were the part of the healthy control group. None of these healthy older adults demonstrated impaired baroreflex sensitivity at baseline. Also, baroreflex sensitivity did not differ between the control group and patients with mild cognitive impairment or dementia in that study. 19 Therefore, it may be reasonable to assume that baroreflex sensitivity does not play a significant role regarding the development of subjective memory complaints in our relatively small study population.
Conclusions
Our study provides important longitudinal insights in mechanisms potentially underlying reductions in cerebral blood flow with aging, and their relation with early cognitive decline. In line with the concept of vascular aging, we conclude that the subtle age-related reductions in CBF and cerebrovascular conductance are accompanied by reductions in cerebrovascular CO2 reactivity, which are related to the development of preclinical cognitive decline (Figure 3). Moreover, we confirm that dCA represents a robust mechanism that is not impaired in aging nor in early cognitive decline (Figure 3). Importantly, our findings even point towards enhancement of dCA in aging, especially in the presence of early cognitive decline (Figure 3). Future studies should confirm if dCA is indeed enhanced by age and/or underlying dementia pathology, and further investigate whether enhanced dCA serves as a compensation for decreased cerebrovascular health. Regarding the treatment and prevention of cognitive decline and dementia, prospective studies should focus on exploring effective strategies to maintain cardio- and cerebrovascular health, for example by physical (in)activity interventions and improved blood pressure management. Eventually, we recommend future studies that address individuals with possible early cognitive decline to use more objective and sensitive cognitive measurements for detecting preclinical cognitive manifestations such as subjective memory complaints.
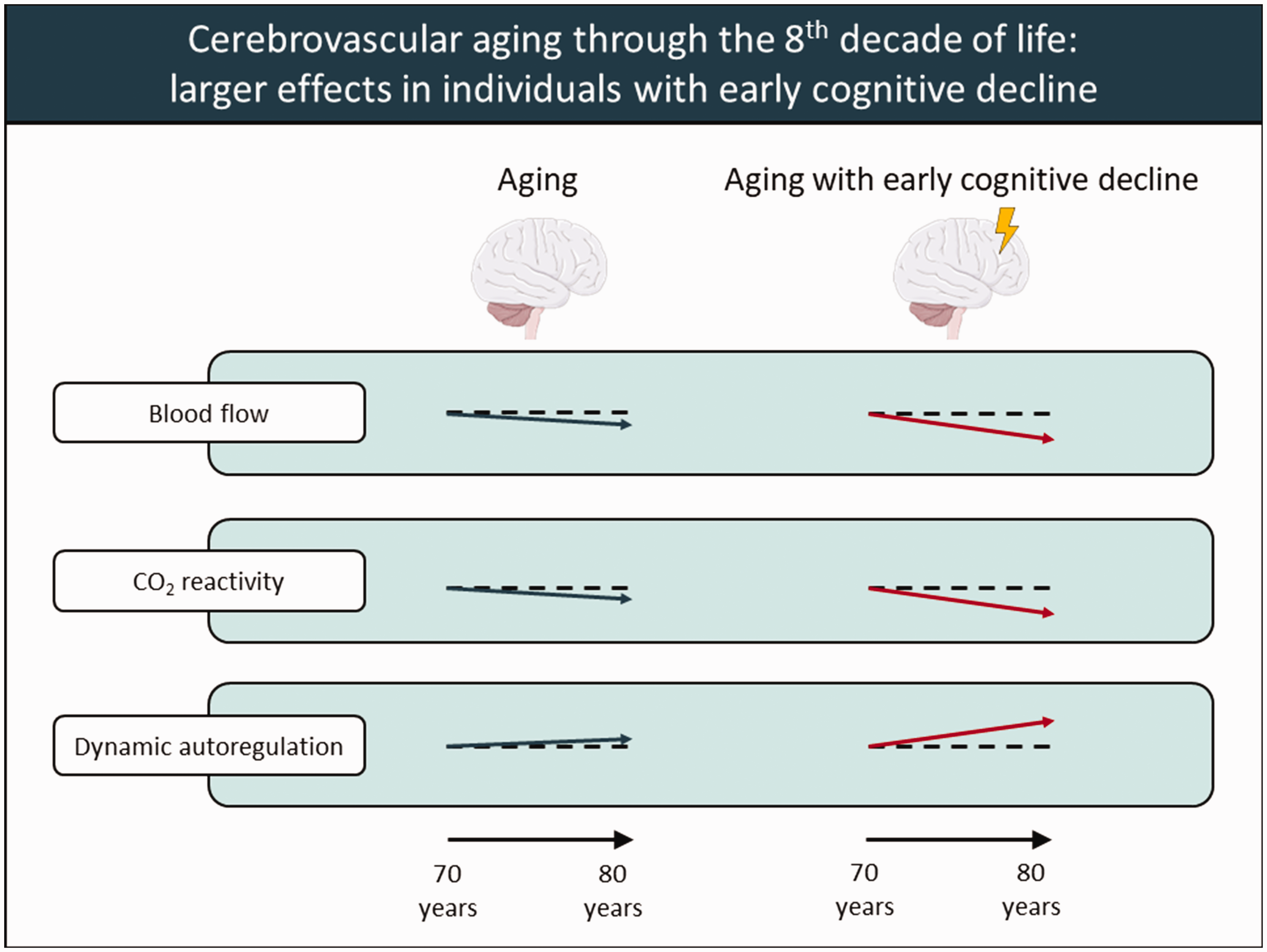
Illustrative summary of conclusions. Illustrative overview of conclusions regarding changes in cerebral blood flow, cerebrovascular CO2 reactivity and dynamic cerebral autoregulation with aging through the eighth decade of life without (left, blue arrows) and with (right, red arrows) early cognitive decline. Horizontal dashed reference lines indicate no change. Figure created with Powerpoint.
Supplemental Material
sj-pdf-1-jcb-10.1177_0271678X231219568 - Supplemental material for Cerebrovascular CO2 reactivity and dynamic cerebral autoregulation through the eighth decade of life and their implications for cognitive decline
Supplemental material, sj-pdf-1-jcb-10.1177_0271678X231219568 for Cerebrovascular CO2 reactivity and dynamic cerebral autoregulation through the eighth decade of life and their implications for cognitive decline by Ralf WJ Weijs, Madelijn H Oudegeest-Sander, Maria TE Hopman, Dick HJ Thijssen and Jurgen AHR Claassen in Journal of Cerebral Blood Flow & Metabolism
Footnotes
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Acknowledgements
The authors would like to thank everyone who has contributed to this follow-up study, including contributors who collected and analysed data as part of the baseline study in 2008–2010. The authors also thank the participants of this study, whose participation made this work possible. Finally, the authors would like to thank dr. R. Donders (biostatistician from the Radboud University Medical Center) for his helpful professional advice regarding the statistical analyses.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Authors’ contributions
Weijs, Hopman, Claassen and Thijssen contributed to the conception and design of the work. Weijs, Oudegeest-Sander and Claassen contributed to the acquisition and analysis of data for the work. All authors contributed to the interpretation of data for the work. Weijs drafted the work. All authors revised the work critically for important intellectual content. All authors approved the final version of the manuscript. All authors agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. All persons designated as authors qualify for authorship, and all those who qualify for authorship are listed.
Supplementary material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
