Abstract
Neuronal loss in the ipsilateral thalamus after focal cortical infarction participates in post-stroke cognitive deficits, and enhanced angiogenesis in the thalamus is expected to reduce neuronal damage. We hypothesize that novel translocator protein (TSPO) ligand, 2-Cl-MGV-1, can promote angiogenesis, attenuate neuronal loss in the thalamus, and ameliorate post-stroke cognitive deficits. Cortical infarction was induced by distal middle cerebral artery occlusion (dMCAO) in stroke-prone renovascular hypertensive rats. 2-Cl-MGV-1 or dimethyl sulfoxide was administered 24 h after dMCAO and then for 6 or 13 days. Spatial learning and memory were assessed using the Morris water maze. Neuronal loss, TSPO expression, angiogenesis, and intrinsic pathway were determined by immunofluorescence and immunoblotting 7 and 14 days after dMCAO. Cortical infarction caused post-stroke cognitive deficits and secondary neuronal loss with gliosis in the ipsilateral thalamus within 14 days of dMCAO. Increased angiogenesis and elevated expression of vascular TSPO were detected in the ipsilateral thalamus, and treatment with 2-Cl-MGV-1 enhanced angiogenesis by stimulating the PI3K-AKT-mTOR pathway. The effects of 2-Cl-MGV-1 on angiogenesis coincided with reduced neuronal loss in the thalamus and contributed to improvements in post-stroke cognitive deficits. Our findings suggest that 2-Cl-MGV-1 stimulates angiogenesis, ameliorates neuronal loss in the thalamus, and improves post-stroke cognitive deficits.
Introduction
Post-stroke cognitive deficits occur in approximately one-third of patients with stroke; however, the underlying mechanisms and effective treatments have not been fully elucidated. 1 Focal cortical infarction leads not only to direct pathologic damage in the primary ischemic site but also causes secondary neurodegeneration in the non-ischemic areas that are synaptically connected to the primary location of damage (thalamus, hippocampus, and corpus callosum).2 –5 Secondary neuronal damage in the thalamus has been demonstrated to be involved in post-stroke cognitive deficits.3,5 Thalamic pathology secondary to cortical infarction involves retrograde or anterograde axonal degeneration, neuro-inflammation, apoptosis, β-amyloid (Aβ) deposition, and excessive autophagy.2,3,5,6 Moreover, focal cortical infarction triggers endogenous protective mechanisms, especially a newly formed vascular network and facilitates angiogenesis in the remote thalamus. Still, this response is insufficient to rescue neuronal loss and related functional neurological deficits. 7 Therefore, strategies dedicated to stimulating vascular regeneration in the thalamus are expected to ameliorate secondary damage and cognitive impairment after ischemic stroke.
The translocator protein (TSPO) is a transmembrane domain protein (18 kDa) located in the outer mitochondrial membrane that participates in neurosteroid synthesis, the mitochondrial apoptosis cascade, inflammation, and nerve regeneration.8 –12 Within the brain, TSPO is expressed in 30% of the vessels in the cortex and white matter. 13 Elevated TSPO expression was induced by chronic hypoxia, and cerebral angiogenesis was confirmed by positron emission tomography (PET) imaging of 11C-PK11195 (a TSPO ligand) after chronic hypoxia in a mouse model. 14 TSPO knockout or TSPO ligands triggered angiogenesis in in vivo or in vitro models of glioblastoma.15,16 These findings suggest a critical role of TSPO in cerebral angiogenesis. A novel synthetic ligand, 2-(2-chlorophenyl)quinazolin-4-yl dimethylcarbamate (2-Cl-MGV-1) regulates TSPO function and was designed to confer neuro-protection against brain damage.5,17 Our preliminary studies have shown that 2-Cl-MGV-1 reduced neuronal apoptosis in the thalamus after distal cortical infarction, 5 but very little is known about its effect on thalamic angiogenesis and post-stroke cognitive deficits, and the underlying mechanisms still need to be elucidated.
The present study was designed to investigate alterations in TSPO expression and the effects of 2-Cl-MGV-1 on angiogenesis and secondary neurodegeneration in the ipsilateral thalamus after focal cortical infarction. Furthermore, we sought to explore the possible mechanisms of 2-Cl-MGV-1-mediated angiogenesis in the thalamus after cortical infarction.
Materials and methods
This experiment was approved by the Institutional Animal Ethical Committee of Sun Yat-sen University. All experiments were conducted in accordance with the Guide for the Care and Use of Laboratory Animals, and reported according to the ARRIVE guidelines.
Animal models and experimental groups
The stroke-prone renovascular hypertensive rat (RHRSP) model (n = 114) was established in male Sprague-Dawley rats weighing 80–100 g (21∼28-day-old) using a two-kidney two-clip method, as described in our previous studies. 18 Twelve weeks later, RHRSP whose systolic blood pressure was steadily >180 mmHg and without stroke symptoms were randomly assigned to distal middle cerebral artery occlusion (dMCAO) or sham surgery groups. Briefly, under anesthesia, the right middle cerebral artery was exposed (above the olfactory tract) and then occluded distal to the origin of the striatal branches using a bipolar electrocoagulation device, which resulted in a permanent cerebral infarction in the right hemisphere. 19 The sham-operated rats underwent the same surgery, except for the occlusion of the middle cerebral artery. Body temperature was maintained at 37 ± 0.5 °C during the surgical and recovery periods using a heat lamp. Ninety-six rats with successful dMCAO were randomly assigned to the 2-Cl-MGV-1 or vehicle groups (16 rats/group). Eighteen rats were excluded because of dMCAO failure (n = 12) or death during surgery (n = 6).
2-Cl-MGV-1 administration
2-Cl-MGV-1 (provided by Professor Veenman, Professor Gavish, and Professor Marek; Israel Institute of Technology, Haifa, Israel) was dissolved in dimethyl sulfoxide (25 mg/ml) and kept at room temperature protected from light. The compound (7.5 mg/kg) was initially administered 24 h after dMCAO by intraperitoneal injection, followed by once daily for 6 or 13 consecutive days, as described previously. 5 Vehicle-treated rats were administered an equal dose of dimethyl sulfoxide as a control, whereas the sham-operated group did not receive an injection.
In vivo BrdU labeling
To label the newly generated endothelial cells, 5′-Bromo-2′-deoxyuridine (BrdU, Sigma-Aldrich, 50 mg/kg), a thymidine analog that is incorporated into the DNA of dividing cells during the S-phase, was dissolved in saline and injected intraperitoneally twice daily for six consecutive days, starting 24 h after dMCAO or sham operation.
Behavioral test
Cognitive function assessment
Spatial learning and memory were examined using the Morris water maze from days 8 to 14 after dMCAO or sham operation (8 rats/group). 20 The test consisted of adaptive training, spatial acquisition training, and a probe trial. On day eight after dMCAO, the animals were allowed to swim freely in the pool for 240 s for adaptive exercise. From days 9 to 13, four acquisition trials in 90 s with a 10-cm diameter platform under the water in the southwest quadrant, were performed each day. The time spent to seek the platform (referred to as escape latency) and swimming distance, were recorded to determine spatial learning ability. On day 14, a probe trial was performed with the platform removed. The time spent in the target quadrant and the frequency of platform crossings within 60 s were recorded to evaluate the spatial memory ability.
Bederson scores
Whole motor function was tested at 7 and 14 days after dMCAO. Control rats extend both forelimbs toward the floor, whereas flexion of the forelimb contralateral to the lesioned hemisphere would occur in injured rats. Behavior was scored as follows: 1, forelimb flexed and no other abnormality; 2, reduced resistance to lateral push toward the paralytic side; 3, circled toward the paralytic side when allowed to move freely; and 4, spin along the longitudinal axis of the body when suspended by the tail.
Beam-walking test
Motor function balance was evaluated using the beam-walking test at 7 and 14 days after dMCAO. The beam was 122 cm long and 2.5 cm wide. Scoring was defined as follows: 0, immediately fell off the beam; 1, unable to traverse but remained sitting across the beam; 2, fell off while walking; 3, walked along the beam, but the affected hindlimb did not work in forward movement; 4, crossed the beam with more than 50% footslips; 5, crossed the beam with occasional footslips; and 6, crossed the beam without footslips.
Tissue preparation
At 7 and 14 days after dMCAO, eight rats from each group were selected to be sacrificed under deep anesthesia and then transcardially perfused with 0.9% sodium chloride at 4 °C followed by 4% paraformaldehyde in 0.1 M PBS (pH 7.4). The brains were then removed, kept in the same fixative for 4 h at 4 °C, and sequentially immersed in 20% and 30% sucrose in 0.1 M PBS overnight at 4 °C. Coronal sections were cut on a cryostat (CM1900; Leica) between +4.7 and −5.2 mm from the bregma, for histological analysis.
Nissl staining
Every 12th coronal section from the bregma +4.7 to −5.2 mm in each animal was selected for Nissl staining to determine the volume of the primary infarction and secondary thalamic neuronal loss. Nissl staining was performed using 0.3% cresyl violet (Sigma-Aldrich) and images were captured using a computer-connected microscope (Olympus, BX51). The primary infarct volume was determined by calculating the percentage of infarct volume relative to contralateral hemisphere volume.
Immunofluorescent staining
A series of coronal sections between −2.8 and −4.4 mm relative to the bregma were selected from each group to perform immunofluorescence analysis to assess secondary thalamic damage, TSPO expression, and angiogenesis. Sections were rinsed in 0.01 M PBS (pH 7.4) and blocked with 5% normal goat-derived serum (Solarbio) at room temperature for 60 min. Then, the sections were incubated at 4 °C overnight with the following primary antibodies: rabbit anti-TSPO/PBR (1:100; Abcam), mouse anti-NeuN (1:300; Millipore), mouse anti-GFAP (1:500; CST), chicken anti-GFAP (1:1000; Abcam), mouse anti-Iba-l (1:400; Abcam), mouse anti-RECA-1 (1:400; Abcam), rabbit anti-Laminin (1:300; Sigma-Aldrich), mouse anti-BrdU (1:250; Sigma-Aldrich), rabbit anti-PI3K (1:250; Abcam), rabbit anti-AKT (1:200; CST), and rabbit anti-mTOR (1:100; CST). Negative control sections were incubated with 0.01 M PBS instead of the primary antibody. The sections were then rinsed in 0.01 M PBS and incubated with the following secondary antibodies at room temperature for 60 min: Alexa Fluor 488-conjugated goat anti-mouse (1:400; CST), Alexa Fluor 488-conjugated goat anti-rabbit (1:400; CST), Alexa Fluor 555-conjugated goat anti-mouse (1:400; CST), Alexa Fluor 555-conjugated goat anti-rabbit (1:400; CST), and Alexa Fluor 647-conjugated goat anti-chicken IgY (1:1000; Abcam). The images were captured under a fluorescence microscope (Olympus, BX51).
Western blotting
Seven and 14 days after dMCAO, the remaining eight rats from each group were sacrificed as described above and subjected to intracardiac perfusion with 0.9% sodium chloride. The brain was removed, and the ipsilateral thalamus was rapidly separated. Total protein was extracted using western blot lysis buffer with 1% protease inhibitor cocktail (Pierce) and the homogenate was centrifuged at 14,000 × g for 15 min at 4 °C. Overall protein concentration was determined using the BCA protein assay system (Pierce). Eighty micrograms of protein extract from each sample were separated by SDS-PAGE and transferred to a polyvinylidene fluoride membrane (Millipore). The membranes were blocked with 5% non-fat milk for 60 min at room temperature and then incubated with the following primary antibodies at 4 °C overnight: rabbit anti-TSPO/PBR (1:300; Abcam), rabbit anti-PI3K (1:1000; Abcam), rabbit anti-AKT (1:1000; CST), rabbit anti-p-AKT (1:1000; CST), rabbit anti-mTOR (1:1000; CST), rabbit anti-HIF-1α (1:300; Abcam), mouse anti-VEGF (1:200; Santa Cruz Biotechnology), mouse anti-VEGFR2 (1:200; Santa Cruz Biotechnology), rabbit anti-GAPDH (1:1000; CST), and rabbit anti-β-actin (1:1000; CST). The membranes were washed with tris-buffered saline plus Tween-20 (0.1%; TBST, pH 7.4) and incubated with the following secondary antibodies: goat anti-mouse IgG (1:2000; CST) or goat anti-rabbit IgG (1:2000; CST) for 60 min at room temperature. Thereafter, the membranes were washed with TBST and visualized using an enhanced chemiluminescence system (Millipore, WBKLS0100).
Statistical analysis
Every sixth section from the bregma −2.8 to −4.4 mm was used for quantification on Nissl-stained sections and immunopositive cells on immunostained sections. The number of intact neurons and NeuN+ cells in the ipsilateral thalamus was counted within three non-overlapping fields under ×400 magnification and was presented as the average cell number per field from each section. TSPO expression and the quantification of Laminin-positive vessels in the thalamus were determined as a percentage of positive immunoreactivity within the total thalamic area under ×400 magnification in each section. For western blotting, the ratios of band densities for p-PI3K/PI3K, p-AKT/AKT and for TSPO, mTOR, HIF-1α, VEGF, and VEGFR2, relative to GAPDH or β-actin, were calculated. All quantitative analyses were performed using NIH Image J software (National Institute of Mental Health, Bethesda, Maryland, USA).
Statistical analysis was performed using the GraphPad Prism 9.0 and SPSS software 25.0. The Kolmogorov-Smirnov normality test was performed on all the datasets firstly to examine data distribution. Data are presented as mean ± standard deviation (SD) for variables with normal distribution and as median ± interquartile for those with non-normal distribution. Differences between groups with normal distribution were compared using one-way ANOVA followed by Student’s t-test or LSD-t test. Differences between groups with non-normal distribution were analyzed by Wilcoxon rank sum test. Repeated measurement ANOVA followed by LSD-t test was performed to compare the differences in escape latencies in Morris Water Maze among the three groups during days 9–13 after dMCAO. Spearman rank correlations were used to determine the relationships between angiogenesis and the number of intact neurons in the ipsilateral thalamus or cognitive scores across different groups. Result was corrected for multiple comparisons using statistical hypothesis testing (Dunnett). Statistical significance was set at P < 0.05.
Results
2-Cl-MGV-1 improved post-stroke cognitive deficits without reducing infarct volume
The initial infarct was consistently restricted to the right somatosensory cortex, and the thalamus was not involved in the primary damage observed in dMCAO-exposed rats (Figure 1(a)). The relative infarct volumes were 14.8% ± 4.6% and 16.4% ± 4.2% on day 7, 16.0% ± 4.9% and 15.6% ± 3.7% on day 14 in the vehicle and 2-Cl-MGV-1 groups, respectively. No significant difference was observed between the groups at either time point (P > 0.05; Figure 1(e)).
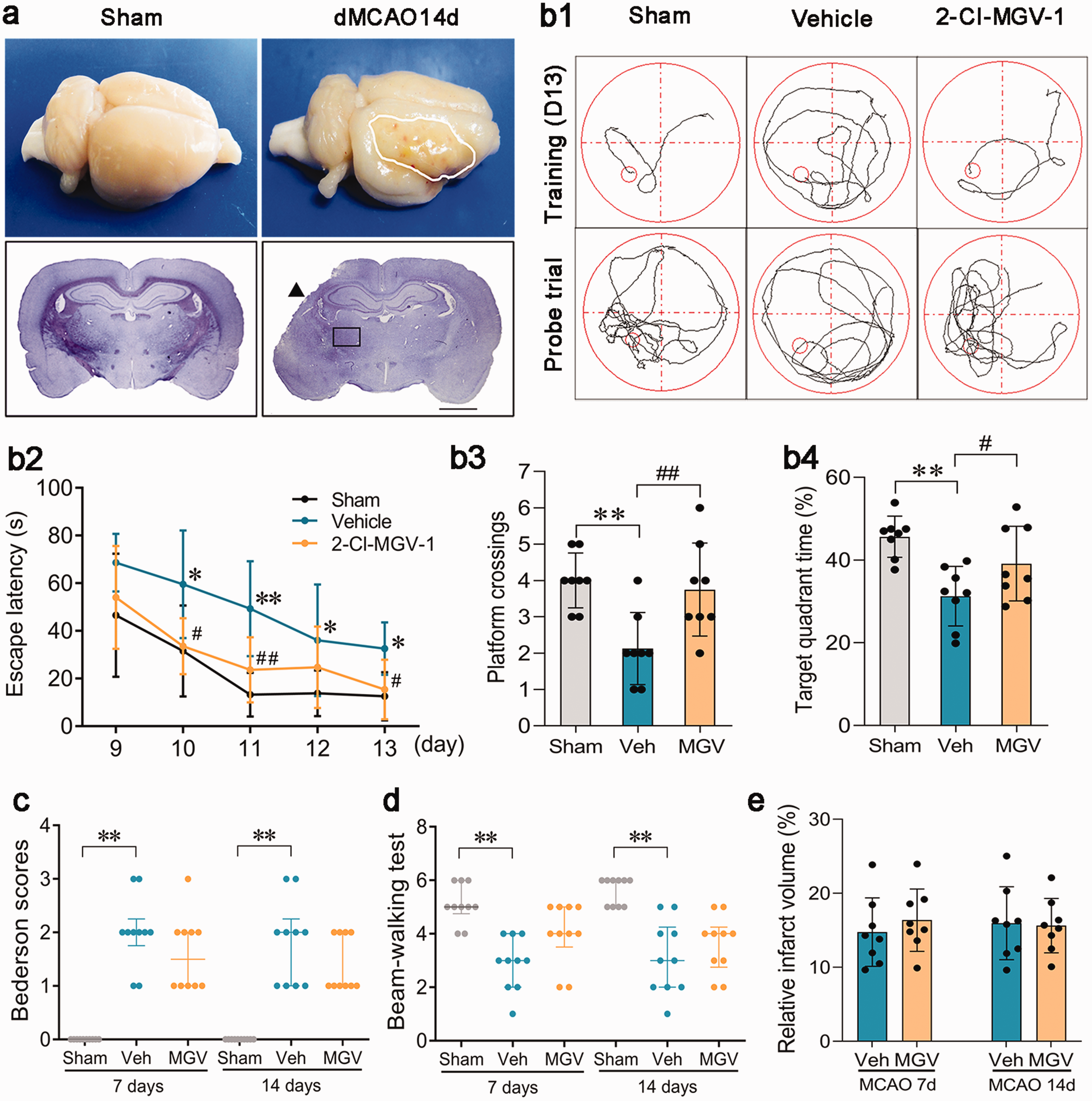
2-Cl-MGV-1 ameliorated cognitive deficits tested by Morris Water Maze after focal cortical infarction without reducing infarct volume. (a) The initial infarct (arrowhead) was restricted to the right somatosensory cortex, and the thalamus (rectangle) was not involved in the primary damage. Scale bar: 2 mm. (b1) Swimming paths in the training trials on day 13 and in the Probe trials on day 14. (b2) Rats in the vehicle group spent longer latencies to locate the hidden platform compared to the sham-operated rats in the training trials on day 10∼13, and rats treated with 2-Cl-MGV-1 needed shorter latency to find the platform compared to those treated with vehicle on day 11∼13. n = 8, *P < 0.05 and **P < 0.01 compared with the sham-operated control, #P < 0.05 and ##P < 0.01 compared with the vehicle-treated group, by repeated measurement ANOVA followed by LSD-t test (mean ± standard deviation). (b3) Vehicle-treated rats had less platform crossings than the sham-operated rats in the probe trial, and rats in the 2-Cl-MGV-1 group achieved more platform crossings than those in the vehicle group. n = 8, **P < 0.01 compared with the sham-operated controls and ##P < 0.01 compared with the vehicle-treated group, by one-way ANOVA followed by LSD-t test (mean ± standard deviation). (b4) Vehicle-treated rats spent less time in the target platform than the sham-operated rats in the probe trial, and rats in the 2-Cl-MGV-1 group spent more time in the target platform than those in the vehicle group. n = 8, **P < 0.01 compared with the sham-operated controls and #P < 0.05 compared with the vehicle-treated group, by one-way ANOVA followed by LSD-t test (mean ± standard deviation). (c-d) Quantitative analysis of the Bederson scores and Beam-walking test scores. n = 10, **P < 0.01 compared with the sham-operated controls by Wilcoxon rank sum test (median ± interquartile). (e) Quantitative analysis of the relative infarct volumes. n = 8, by Student’s t test (mean ± standard deviation).
The Morris water maze results revealed a longer latency to locate the hidden platform in the vehicle-treated rats than in the sham-operated rats on days 10–13 after dMCAO, during the acquisition trials (all P < 0.05; Figure 1(b1) and (b2)). Moreover, in the probe trial on day 14, the vehicle group spent less time in the target quadrant and had fewer platform crossings (both P < 0.01; Figure 1(b1), (b3) and (b4)). Treatment with 2-Cl-MGV-1 significantly shortened the latency on days 10, 11, and 13 (all P < 0.05; Figure 1(b1) and (b2)), increased the number of platform crossings (3.8 ± 1.3 vs. 2.1 ± 1.0, P = 0.005; Figure 1(b3)), and prolonged the percentage of time spent in the target quadrant (39.1% ± 9.0% vs. 31.3% ± 7.2%, P = 0.042; Figure 1(b4)) in the probe trial on day 14.
Regarding motor function, the vehicle-treated rats developed significant motor function deficits, as demonstrated by higher Bederson scores and lower Beam-walking test scores compared to the sham-operated rats 7 and 14 days after dMCAO (all P < 0.01; Figure 1(c) and (d)). Compared with the vehicle-treated rats, rats in the 2-Cl-MGV-1-treated group did not exhibit better Bederson and beam-walking test scores at either time point (all P > 0.05; Figure 1(c) and (d)).
Focal cortical infarction caused neuronal loss and induced angiogenesis in the ipsilateral thalamus
Progressive secondary neuronal degeneration in the ipsilateral thalamus, characterized by reduced NeuN+ neurons and increased Iba-1+ microglia and GFAP+ astrocytes by immunofluorescence staining, was observed in the vehicle group when compared with the sham-operated group on days 7 and 14 after dMCAO (all P < 0.001; Figure 2(a1) to (c2)).
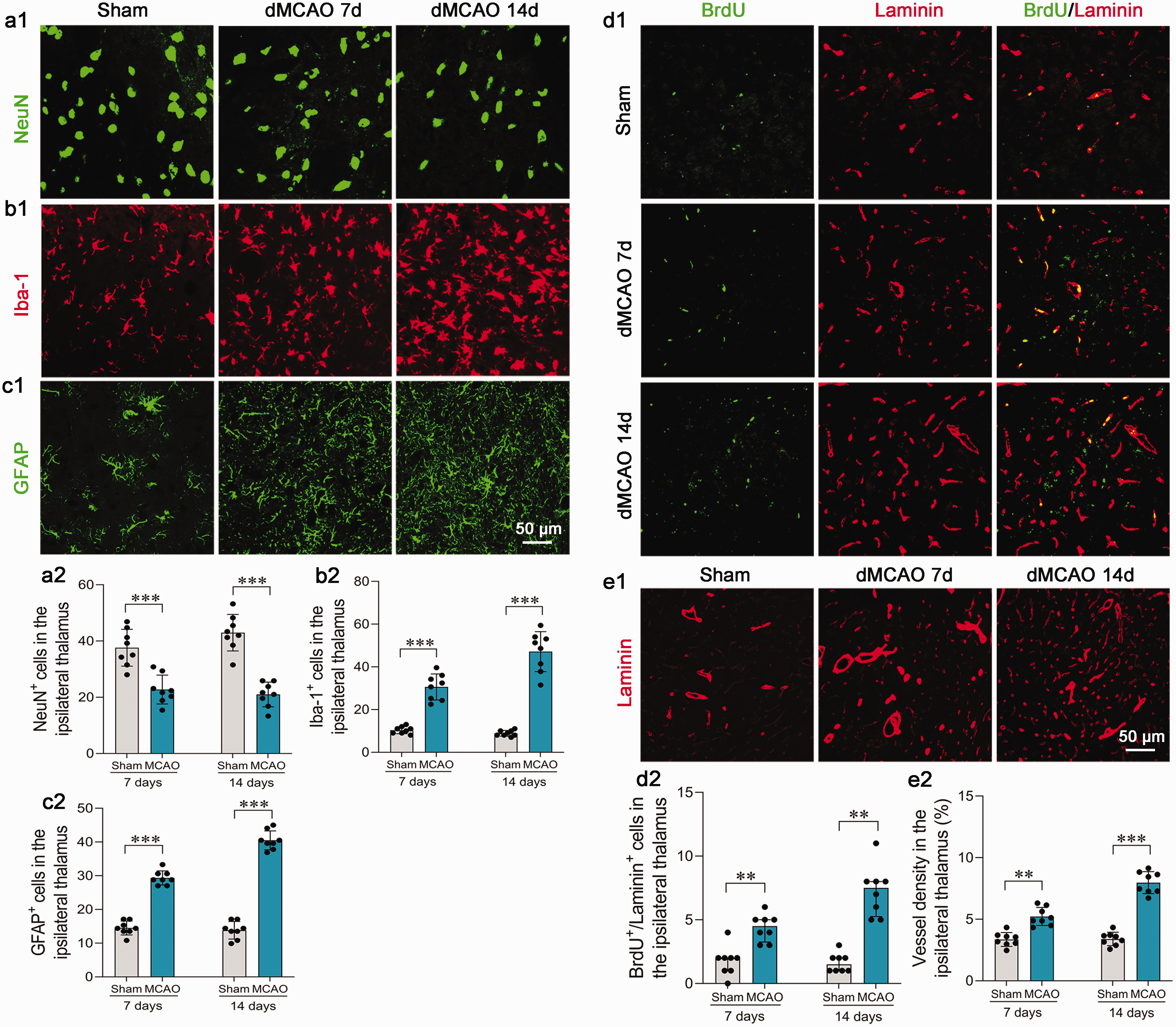
Focal cortical infarction caused secondary damage and induced angiogenesis in the ipsilateral thalamus. Focal cortical infarction led to neuronal loss (a1), microglia activation (b1), and astrocytes proliferation (c1), and induced increased newly formed vessels (d1) and high density of vessels (e1) tested by immunostaining in the ipsilateral thalamus. Scale bar: 50 μm. (a2-c2) Quantitative analysis of the number of NeuN+, Iba-1+, and GFAP+ cells. n = 8, ***P < 0.001, by Student’s t test (mean ± standard deviation). (d2-e2) Quantitative analysis of the number of BrdU+/Laminin+ and Laminin+ vessels. n = 8, **P < 0.01, ***P < 0.001, by Wilcoxon rank sum test (median ± interquartile) in d2 and by Student’s t test (mean ± standard deviation) in e2.
After dMCAO, very few BrdU-positive cell nuclei were visualized in Laminin-labeled endothelial cells within the ipsilateral thalamus of the sham-operated rats. There was a progressive increase in the number of BrdU+/Laminin+ cells in the thalamus of the rats exposed to dMCAO on days 7 and 14 (both P < 0.01; Figure 2(d1) and (d2)). Consistently, the density of Laminin-labeled blood vessels was remarkably higher within the ipsilateral thalamus in rats with dMCAO than sham-operated rats at the same time points (both P < 0.01; Figure 2(e1) and (e2)). These results implicate a self-repair mechanism by angiogenesis, in addition to neurodegeneration in the ipsilateral thalamus remote from the primary infarction.
TSPO expression and effects of 2-Cl-MGV-1 on angiogenesis in the thalamus after cortical infarction
TSPO expression was significantly elevated in the ipsilateral thalamus 7 and 14 days after dMCAO compared with that in the sham-operated controls (both P < 0.001; Figure 3(a)). Immunoblotting analysis confirmed a continuous increase in the level of TSPO in the thalamus within 14 days after cortical infarction when compared with the sham-operated groups (P < 0.05, dMCAO 7d compared to sham-operated rats, and P < 0.05, dMCAO 14d compared to dMCAO 7d; Figure 3(b)), and 2-Cl-MGV-1 treatment did not alter the thalamic expression of TSPO (Figure 3(b)). Moreover, TSPO-positive immunostaining was observed to be increasingly distributed in RECA-1-immunoreactive vascular endothelial cells 7 and 14 days after dMCAO when compared with the sham-operated controls (all P < 0.05; Figure 3(c)). TSPO-positive immunostaining was observed more frequently in the blood vessels with large lumens and intact structures. Meanwhile, TSPO was also co-stained with Iba-1+ microglia, GFAP+ astrocytes, and NeuN+ neurons (Supplemental Figure S1).
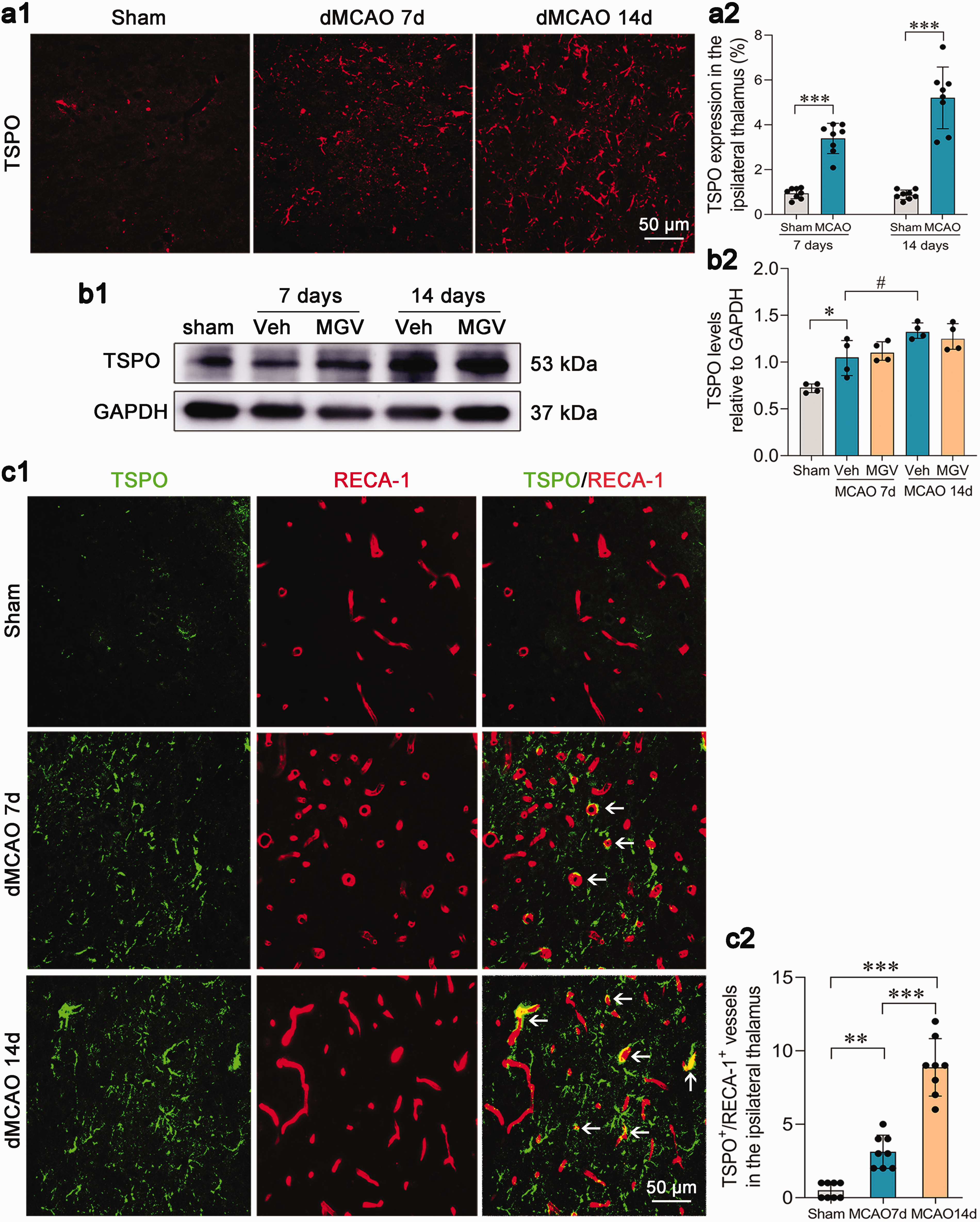
Increased vascular expression of TSPO in the ipsilateral thalamus after focal cortical infarction. (a1-a2) Increased expression of TSPO in the ipsilateral thalamus 7 and 14 days after dMCAO. n = 8, ***P < 0.001, by Student’s t test (mean ± standard deviation). Scale bar: 50 μm. (b1-b2) Immunoblot showing TSPO levels in the thalamus of the sham-operated controls, vehicle, and 2-Cl-MGV-1 groups. n = 4, *P < 0.05 compared with the sham-operated controls and #P < 0.05 compared with the vehicle-treated rats 7 days post-dMCAO, by Wilcoxon rank sum test (median ± interquartile). (c1-c2) Increased positive immunostaining of TSPO with RECA-1-labeled vessels in the ipsilateral thalamus 7 and 14 days after dMCAO. n = 8, **P < 0.01, ***P < 0.001, by one-way ANOVA followed by LSD-t test (mean ± standard deviation). Scale bar: 50 μm.
Treatment with 2-Cl-MGV-1 progressively increased the number of BrdU+/Laminin+ vessels within the ipsilateral thalamus when compared with the vehicle group 7 days (P < 0.01) and 14 days (P < 0.001) after dMCAO (Figure 4(a) and (c)). In parallel, the density of Laminin-labeled blood vessels in the thalamus was higher in the 2-Cl-MGV-1-treated group than in the vehicle-treated group 7 days after dMCAO (P < 0.01), and was progressively elevated 14 days post-dMCAO (P < 0.001; Figure 4(b) and (d)).
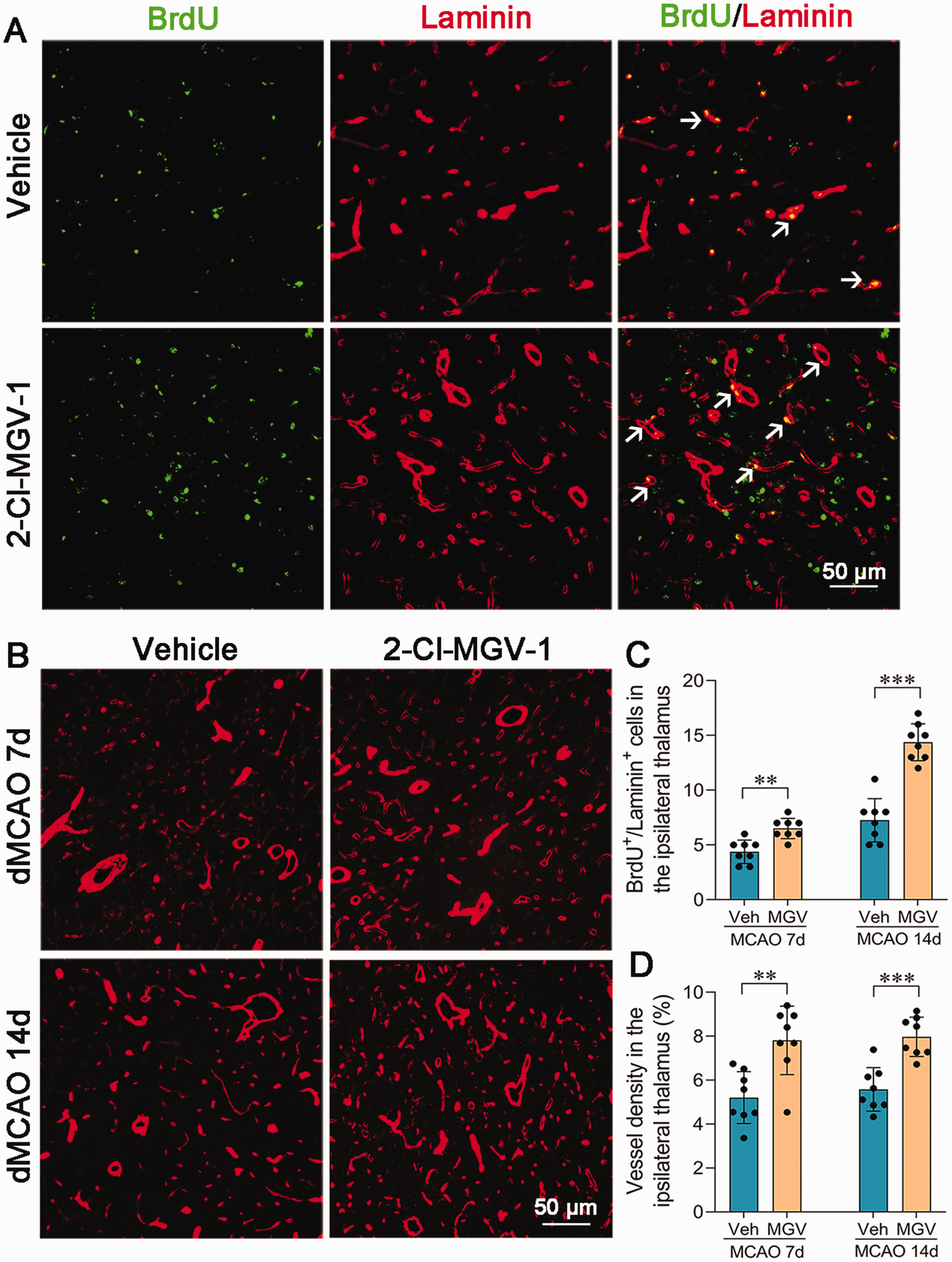
2-Cl-MGV-1 promoted angiogenesis in the ipsilateral thalamus after focal cortical infarction. (a) Immunostaining for BrdU and Laminin in the ipsilateral thalamus of the vehicle and 2-Cl-MGV-1 groups on 7 days after dMCAO. 2-Cl-MGV-1 treatment markedly increased BrdU+ cells (green) that co-stained for Laminin (red). Scale bar: 50 μm. (b) Laminin-labeled blood vessel significantly elevated in the ipsilateral thalamus of the 2-Cl-MGV-1 group on 14 days after dMCAO. Scale bar: 50 μm. (c-d) Quantitative analysis of the number of BrdU+/Laminin+ cells and density of blood vessel. n = 8, **P < 0.01, ***P < 0.001, by Student’s t test (mean ± standard deviation).
2-Cl-MGV-1 might enhance angiogenesis through PI3K-AKT-mTOR pathway
We investigated the signaling pathways involved in 2-Cl-MGV-1-induced angiogenesis in the remote thalamus secondary to cortical infarction. The expression of p-PI3K/PI3K were significantly elevated in the ipsilateral thalamus within 14 days post-dMCAO in the 2-Cl-MGV-1-treated groups compared to those in the vehicle-treated groups (both P < 0.05; Figure 5(a1) and (a2)). In parallel, higher ratios of p-AKT/AKT were observed in the ipsilateral thalamus after 2-Cl-MGV-1 treatment than after vehicle therapy 7 and 14 days after dMCAO (both P < 0.05; Figure 5(b1) and (b2)). Additionally, 2-Cl-MGV-1 treatment significantly increased the expression of mTOR (P < 0.05; Figure 5(c1) and (c2)) and its downstream factor HIF-1α (P < 0.05; Figure 5(d1) and (d2)) 7 days post-dMCAO, when compared with vehicle treatment. Higher levels of VEGF in the ipsilateral thalamus were detected in the 2-Cl-MGV-1 treatment groups compared to the vehicle-treated groups 14 days after dMCAO (P < 0.05; Figure 5(e1) and (e2)). In contrast, no remarkable differences were detected in the levels of the VEGFR2 – a VEGF receptor, in the thalamus between the 2-Cl-MGV-1 and vehicle treatment groups (both P > 0.05; Figure 5(f1) and (f2)).
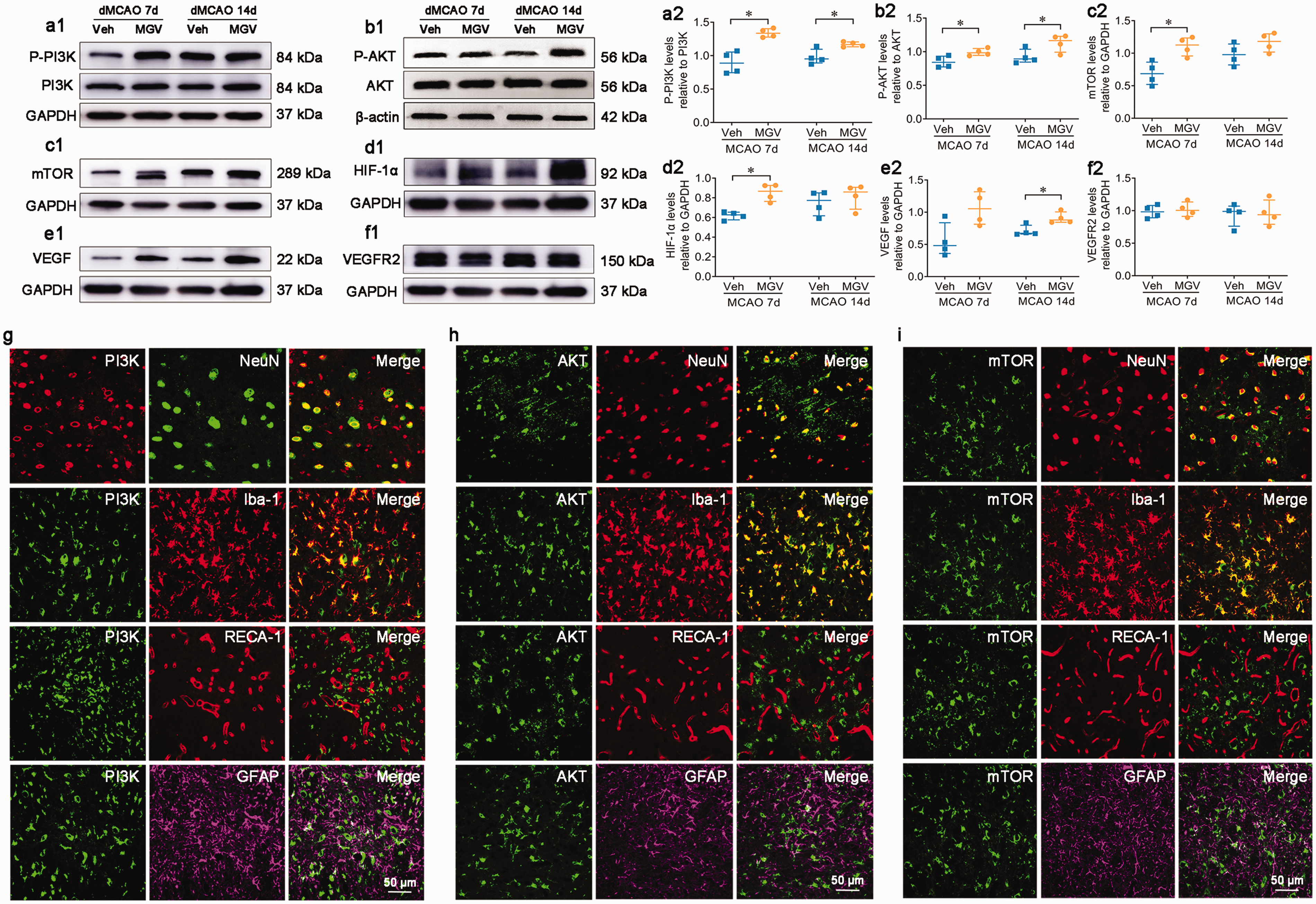
2-Cl-MGV-1 enhanced angiogenesis through PI3K-AKT-mTOR pathway. Immunoblot showed that 2-Cl-MGV-1 treatment upregulated the levels of P-PI3K/PI3K (a1), P-AKT/AKT (b1), mTOR (c1), HIF-1α (d1), and VEGF (e1) in the ipsilateral thalamus as compared with vehicle treatment. The levels of VEGFR2 were not significantly different between the two groups (f1). (a2-e2) Quantitative analysis of the levels of P-PI3K/PI3K, P-AKT/AKT, mTOR, HIF-1α, VEGF, and VEGFR2. n = 4, *P < 0.05 by Wilcoxon rank sum test (median ± interquartile). (g-i) Immunofluorescence staining showing the colocalization of PI3K/AKT/mTOR and NeuN+ neurons, Iba-1+ microglia, GFAP+ astrocytes, and RECA-1+ vascular endothelial cells, respectively. Scale bar: 50 μm.
PI3K-positive, AKT-positive, and mTOR-positive immunostaining were mainly distributed in NeuN+ neurons and Iba-1+ microglia (Figure 5(g) to (i)). There was no positive co-staining of PI3K/AKT/mTOR with GFAP+ astrocytes. Some PI3K-positive immunostaining was seen in RECA-1+ vascular endothelial cells, especially in those with large lumens and an intact structure (Figure 5(g)), but there was no positive co-staining of AKT and mTOR with RECA-1+ vascular endothelial cells (Figure 5(h) and (i)).
2-Cl-MGV-1-induced angiogenesis was associated with decreased neuronal loss and improved cognitive deficits
We further examined whether angiogenesis mediated by 2-Cl-MGV-1 reduced secondary neurodegeneration in the thalamus and improved post-stroke cognitive deficits. Nissl staining showed abnormal neurons, characterized by a shrunken cytoplasm and pyknotic nuclei, in the vehicle group. Treatment with 2-Cl-MGV-1 obviously increased the number of intact neurons in the thalamus as determined by Nissl staining, compared to the vehicle-treated rats on days 7 and 14 after dMCAO (both P < 0.01; Figure 6(a)). In addition, a 39.5% (day 7) and 51.1% (day 14) reduction of NeuN+ neurons in the ipsilateral thalamus of the vehicle group was observed, when compared with the sham-operated group, and 2-Cl-MGV-1 treatment resulted in a marked increase in the number of NeuN+ neurons on days 7 (P < 0.01; Figure 6(b)) and 14 (P < 0.001; Figure 6(b)) after dMCAO. Moreover, activated neuroinflammation, as demonstrated by increased Iba-1+ microglia and GFAP+ astrocytes in the thalamus, was obviously alleviated by 2-Cl-MGV-1 compared with vehicle treatment on 7- and 14-days post-stroke (both P < 0.001; Figure 6(c) and (d)).
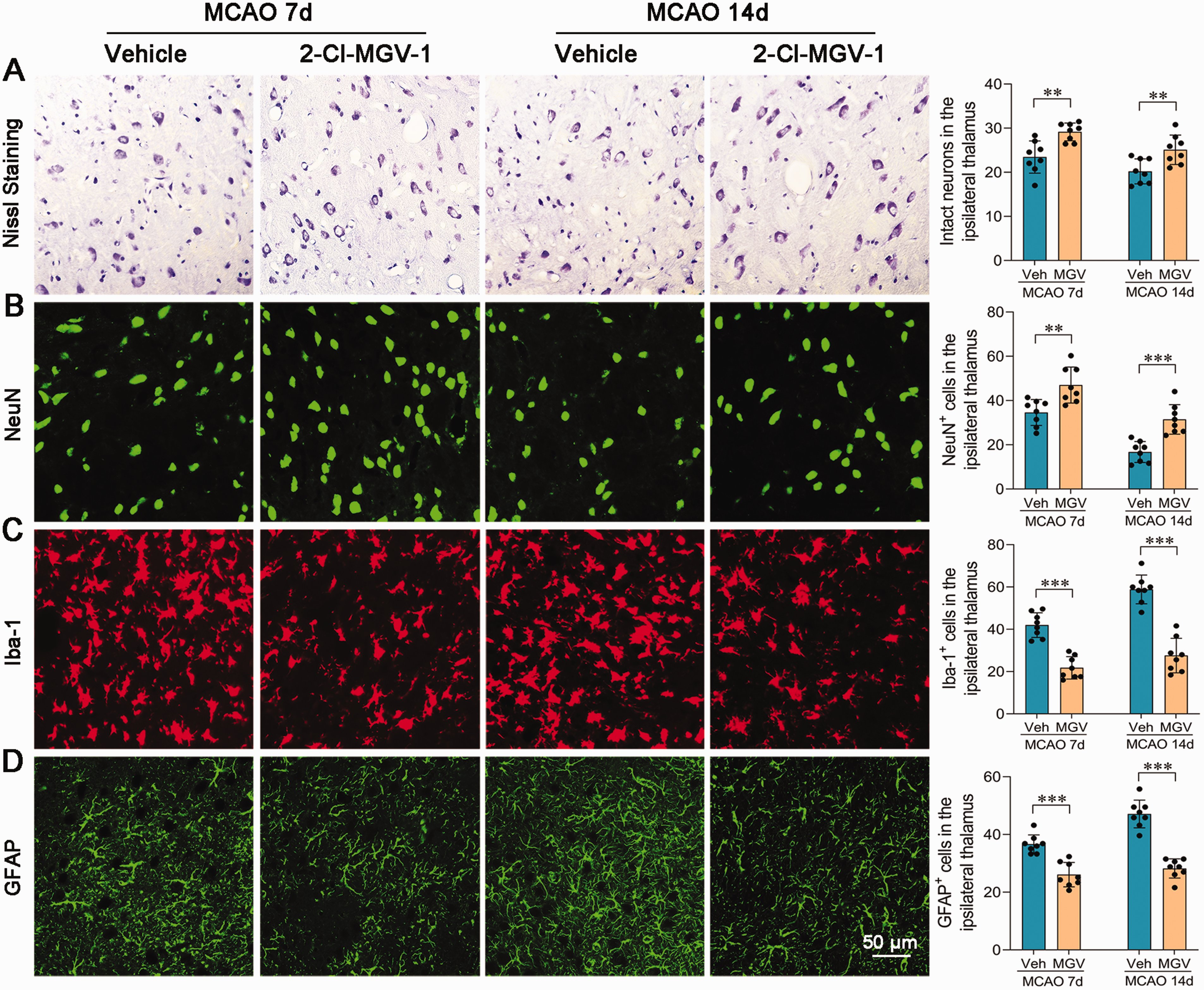
2-Cl-MGV-1 ameliorated secondary damage of the ipsilateral non-ischemic thalamus after focal cortical infarction. 2-Cl-MGV-1 increased the number of intact neurons tested by Nissl staining (a), increased the number of NeuN+ neurons (b), reduced the proliferation of Iba-1+ microglia (c) and GFAP+ astrocytes (d) tested by immunostaining in the ipsilateral thalamus. n = 8, **P < 0.01, ***P < 0.001 by Student’s t test (mean ± standard deviation). Scale bar: 50 μm.
A correlation analysis revealed a positive relationship between the number of BrdU+/Laminin+ cells and that of NeuN+ neurons (r = 0.800, P < 0.001) in the ipsilateral thalamus, across the two groups on 14 days after dMCAO. There was also a positive relationship between the number of BrdU+/Laminin+ cells in the ipsilateral thalamus and shorter escape latencies (r = 0.530, P = 0.042), longer target quadrant time (r = 0.669, P = 0.006), and more platform crossings (r = 0.846, P < 0.001). These findings suggest 2-Cl-MGV-1 enhances angiogenesis and ameliorates secondary neuronal loss in the ipsilateral thalamus, thereby, improving cognitive deficits following focal cortical infarction.
Discussion
Secondary neuronal damage in remote brain regions (such as the thalamus) is associated with post-stroke cognitive deficits but lacks effective treatment. The results of this study reveal elevated TSPO expression in the ipsilateral thalamus following focal cortical infarction. Treatment with the novel TSPO ligand, 2-Cl-MGV-1, enhanced angiogenesis and reduced neuronal loss in the thalamus, accompanied by an improvement in cognitive function after focal cortical infarction. Mechanistically, 2-Cl-MGV-1 stimulated the PI3K-AKT-mTOR signaling pathway and its downstream factor HIF-1α, resulting in increased VEGF levels and elevated vessel density. Taken together, these findings suggest that TSPO plays an important role in thalamic angiogenesis and promoting vascular regeneration by 2-Cl-MGV-1 in the thalamus helps ameliorate secondary neurodegeneration and improves cognitive deficits after stroke.
In agreement with previous studies,3,4,7,21,22 we showed that BrdU/Laminin double-positive cells and Laminin-labeled vascular density were markedly increased in the ipsilateral thalamus within 14 days after focal stroke, suggesting compensatory endogenous vascular repair in addition to progressive secondary neurodegeneration in the thalamus. However, it is not sufficient to solely promote recovery of neurological function; therefore, strategies to promote angiogenesis have been attempted.3,4 Increase of vascular density in the thalamus by activating ephrinB2 4 or enhancing vascular autophagy with blockade of RTN4-S1PR2 interaction 3 could rescue remote damage and alleviate cognitive deficits after cortical infarction. However, the role of TSPO and TSPO ligands on cerebral angiogenesis has not been fully investigated.
A recent TSPO PET study demonstrated that in humans, TSPO was expressed in 30% of the vessels in the cortex and white matter. 13 In the early stages of a mouse model of chronic hypoxia, there was enhanced TSPO expression and cerebral angiogenesis in the cortex, thalamus, hippocampus, and cerebellums. 14 TSPO deficiency or a TSPO ligand (PK11195) induced angiogenesis and promoted the growth of glioblastoma both in vitro and in vivo.15,16 Moreover, the TSPO-NOX1 axis controlled phagocyte-triggered pathological angiogenesis in age-related macular degeneration. 23 These findings demonstrate the importance of positive expression of TSPO in cerebral vessels and highlight the protein as a drug target for angiogenesis and neuro-protective therapies in brain disorders. In this study, administration of the novel synthetic TSPO ligand 2-Cl-MGV-1 increased vascular density and alleviated neurodegeneration in the thalamus at an early stage after dMCAO, suggesting a therapeutic effect of 2-Cl-MGV-1 on angiogenesis.
TSPO has drawn much attention over the years owing to its role in regulating neurosteroid synthesis, neuronal mitochondrial apoptosis, and microglial activation and as being a biomarker for reactive gliosis in cortical infarction and brain injury.5,8,24 –26 2-Cl-MGV-1 shows therapeutic potential in stroke, traumatic brain injury, and neurodegenerative diseases, however, the underlying mechanisms have not been fully elucidated.5,17 2-Cl-MGV-1 counteracted the release of chemokines and inflammatory factors (IL-6, IL-β, IFN-γ, and TNF-α) and suppressed the activation of LPS-stimulated microglial cells, and its inhibitory effect was larger than that of the classical TSPO ligand PK11195.27,28 2-Cl-MGV-1 also inhibited cigarette smoke-induced cytotoxicity to H1299 lung cancer cells through prevention of ATP synthase reversal, ROS generation, depolarization of the mitochondrial membrane, and elevation in LDH. 29 Furthermore, MGV-1 promoted differentiation of progenitor cells to mature neurons and weeded out non-differentiating progenitor cells. 17 Further, our previous study suggested that 2-Cl-MGV-1 reduced neuronal apoptosis by inhibiting the mitochondrial-dependent pathway. 5
However, mechanisms underlying cerebral angiogenesis induced by 2-Cl-MGV-1 were not clear. The classical TSPO ligand PK11195 was part of the mitochondria-to-nucleus signaling pathway and activated gene expression of genes related to proliferation and angiogenesis in U118MG glioblastoma cells within 1 h of exposure. 30 TSPO knockout or XBD173 (TSPO ligand) treatment prevented the reactivity of retinal microglia and reduced inflammation-associated pathological angiogenesis in a laser-induced mouse model of age-related macular degeneration. 23 Angiogenesis regulators, including VEGF-A, interleukin-8, and matrix metalloproteinase-2 (MMP-2) levels were significantly increased in TSPO-knock out gliomas compared with wild-type tumors, whereas no significant changes were observed in VEGF-C and MMP-9 expression. 15 In our study, VEGF levels were significantly increased in the ipsilateral thalamus with 2-Cl-MGV-1 treatment following cortical infarction. VEGFs and their receptors (VEGFRs) are pivotal regulators of angiogenesis. 21 The lack of a single VEGF allele results in abnormal blood vessel development and leads to embryonic lethality.31,32 VEGF-A (also known as VEGF) is a major member of the VEGF family that promotes angiogenesis by binding to VEGFR2.33,34 The PI3K/AKT/mTOR pathway, which is an upstream pathway of VEGF, is essential for angiogenesis, including endothelial cell survival, migration, and new vessel formation.35 –38 Activated PI3K/AKT/mTOR signaling activates its downstream factor HIF-1α, thereby regulating VEGF expression.34,37,39 2-Cl-MGV-1 significantly increased the expression of p-PI3K/PI3K, p-AKT/AKT, mTOR, and HIF-1α in the ipsilateral thalamus following cortical infarction, revealing its potential involvement in cerebral angiogenesis. Microglia or macrophages serve as important pro-angiogenic cells in blood vessels development.34,40 PI3K-positve, AKT-positive, and mTOR-positive staining were mainly distributed in microglia in our study, suggesting that 2-Cl-MGV-1 might stimulate the pro-angiogenic cells (mainly microglia) to promote angiogenesis via the PI3K/AKT/mTOR pathway. The distribution of PI3K/AKT/mTOR in neurons has been reported to exert crucial effects on apoptosis. 41 Moreover, the PI3K/AKT pathway in the endothelial cells can promote endothelial cell survival and vascular permeability. 34 We observed that PI3K-positive staining was also distributed in the relatively large blood vessels, suggesting that 2-Cl-MGV-1 might also exert effects on maintaining the integrity of the vascular structure.
Accumulating evidence shows that secondary damage in the thalamus after focal cerebral infarction is involved in post-stroke cognitive infarction.42 –44 There are extensive fiber connections between the thalamus and prefrontal cortex, hippocampus, dentate gyrus and striatum, which play an important role in learning, memory, execution, adaptive decision-making, and integration of associative information. 45 A Focal iron accumulation 42 and microstructural abnormalities 43 in the thalamus remote to infarct was associated with poor cognitive outcome after ischemic stroke. Our previous study found that secondary damage to the thalamus, especially the left subregions connected to specific cortices, was associated with early verbal memory impairment at 12 weeks after stroke onset in patients with acute subcortical infarct. 44 These findings suggest a significant role of the thalamus in cognitive activities.
Angiogenesis can provide a suitable microenvironment for triggering axonal outgrowth and may induce neurogenesis. Angiogenesis in the thalamus coincides with neurogenesis following the same time course after dMCAO, 7 and we hypothesized that angiogenesis in the remote area helped promote the proliferation of neurons. However, the number of BrdU/NeuN double-positive cells and the expression of DCX in the thalamus were not significantly increased after treatment with 2-Cl-MGV-1 compared to the vehicle-treated group (data not shown). Focal cortical infarction also induces evident Aβ deposition in the ipsilateral thalamus, leading to neuronal damage and secondary neurodegeneration. However, our previous study demonstrated that 2-Cl-MGV-1 did not promote the degradation of Aβ or reduce the Aβ(x-40) or Aβ42 burden in the ipsilateral thalamus compared with vehicle treatment. 5 Our data showed that 2-Cl-MGV-1 increased the number of intact neurons and rescued the neuronal loss within the thalamus. There was a significant relationship between vessel density and the number of NeuN+ cells in the ipsilateral thalamus in the 2-Cl-MGV-1 and vehicle groups. In addition, VEGF exerts neurotrophic or growth-promoting effects on neurons, which can provide protection from a range of insults. VEGF has been reported to promote both axonal outgrowth and cell survival in cultured superior cervical and dorsal route ganglion neurons and CA3 hippocampal neuronal development, and to improve neuronal survival in organotypic midbrain explant cultures.41,46 Collectively, these data suggest that the effect of 2-Cl-MGV-1 on angiogenesis in the thalamus results in a marked reduction in neuronal loss; thereby, ameliorates cognitive deficits after cortical infarction.
In this study, we showed increased TSPO expression in the remote thalamus after cortical infarction and revealed the effects of TSPO ligand 2-Cl-MGV-1 on thalamic angiogenesis after cortical infarction and its potential underlying mechanism. Our findings indicate that strategies dedicated to promoting angiogenesis in the non-ischemic thalamus are expected to ameliorate secondary damage and cognitive impairment after ischemic stroke. One limitation of the present study was that no specific knockdown or knockout of TSPO in the thalamus to evaluate the effects and mechanisms of 2-Cl-MGV-1 on angiogenesis after cortical infarction. However, we found that the level of TSPO did not change after 2-Cl-MGV-1 treatment compared to vehicle, and 2-Cl-MGV-1 regulated the function of TSPO rather than change its expression to exert neuroprotective effects.
Conclusions
Our findings demonstrate that the novel TSPO ligand, 2-Cl-MGV-1, promotes angiogenesis through the PI3K-AKT-mTOR signaling pathway, reduces neuronal loss in the thalamus, and ameliorates cognitive deficits following focal cortical infarction. This highlights TSPO as a potential therapeutic target in angiogenesis to rescue secondary thalamic neurodegeneration and improve post-stroke cognitive impairment.
Supplemental Material
sj-pdf-1-jcb-10.1177_0271678X231214671 - Supplemental material for Enhanced angiogenesis in the thalamus induced by a novel TSPO ligand ameliorates cognitive deficits after focal cortical infarction
Supplemental material, sj-pdf-1-jcb-10.1177_0271678X231214671 for Enhanced angiogenesis in the thalamus induced by a novel TSPO ligand ameliorates cognitive deficits after focal cortical infarction by Yicong Chen, Leo Veenman, Mengshi Liao, Weixian Huang, Jian Yu and Jinsheng Zeng in Journal of Cerebral Blood Flow & Metabolism
Supplemental Material
sj-pdf-2-jcb-10.1177_0271678X231214671 - Supplemental material for Enhanced angiogenesis in the thalamus induced by a novel TSPO ligand ameliorates cognitive deficits after focal cortical infarction
Supplemental material, sj-pdf-2-jcb-10.1177_0271678X231214671 for Enhanced angiogenesis in the thalamus induced by a novel TSPO ligand ameliorates cognitive deficits after focal cortical infarction by Yicong Chen, Leo Veenman, Mengshi Liao, Weixian Huang, Jian Yu and Jinsheng Zeng in Journal of Cerebral Blood Flow & Metabolism
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by grants from the Natural Science Foundation of China (Key Program: 82130035; General Program: 82371308, 81971103; Youth Program: 81901077), the Basic and Applied Basic Research Foundation Natural Science Foundation of Guangdong Province (2021A1515012216), Sun Yat-sen University Clinical Research 5010 Program (2018001), the Kelin Star Talent Support Program of the First Affiliated Hospital, Sun Yat-sen University (R08014), the Guangdong Provincial Key Laboratory of Diagnosis and Treatment of Major Neurological Diseases (2020B1212060017), Guangdong Provincial Clinical Research Center for Neurological Diseases (2020B1111170002), Southern China International Joint Research Center for Early Intervention and Functional Rehabilitation of Neurological Diseases (2015B050501003 and 2020A0505020004), Guangdong Provincial Engineering Center for Major Neurological Disease Treatment, Guangdong Provincial Translational Medicine Innovation Platform for Diagnosis and Treatment of Major Neurological Disease, Guangzhou Clinical Research and Translational Center for Major Neurological Diseases (201604020010).
Acknowledgements
We thank Professor Gavish and Professor Marek (Israel Institute of Technology, Haifa, Israel) very much for providing 2-Cl-MGV-1 for this study.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Authors’ contributions
CYC conceived the study and drafted the manuscript, CYC, LMS, and HWX conducted the experiment, LMS and HWX contributed to the analysis of the data, CYC, LV, JY, and JSZ contributed to the data interpretation and editing of the manuscript. All authors contributed to editing the manuscript.
Supplementary material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
