Abstract
Ischemic stroke (IS) induces neurological damage due to cerebrovascular occlusion. Restoring blood perfusion to the ischemic brain area in a timely fashion is the most effective treatment strategy. Hypoxia is an effective way of restoring blood perfusion by improving cerebrovascular microcirculation, while the effect varies greatly depending on hypoxic mode. This study aimed to screen for the optimal hypoxic mode to improve cerebrovascular microcirculation and prevent IS. Here, we found that compared with continuous hypoxia (CH), intermittent hypoxia (IH) significantly improved cerebral blood flow and oxygen saturation in mice without causing neurological impairment. By analyzing cerebrovascular microcirculation from mice, we found that the IH mode (13%, 5*10) with 13% O2, 5 min interval, and 10 cycles per day significantly improved the cerebrovascular microcirculation by promoting angiogenesis without affecting the integrity of the blood-brain barrier. In addition, IH (13%, 5*10) treatment of distal middle cerebral artery occlusion (dMCAO) mice significantly alleviated neurological dysfunction and reduced cerebral infarct volume by improving cerebrovascular microcirculation. CH had none of these positive effects. In summary, our study screened for an appropriate intermittent hypoxic mode that could improve cerebrovascular microcirculation, laying a theoretical foundation for the prevention and treatment of IS in clinical practice.
Keywords
Introduction
Ischemic stroke (IS) is one of the leading causes of death and disability worldwide.1,2 Early course of IS, blood flow is interrupted. The irreversible damage in the microvasculature of the brain is a key cause of secondary brain injury in IS. 3 This can lead to impaired neurological function and decreased motor or cognitive function, which can severely impact the quality of life. 4 Although IS brain recovery is closely related to vascular protection, no successful therapies are available.
In the past few years, a number of studies have shown that treatments that promote angiogenesis can improve brain recovery after stroke. 5 Promoting angiogenesis in ischemic penumbra is considered a promising treatment for IS. 6 Angiogenesis is the formation of new microvessels from pre-existing microvessels. This includes vascular endothelial cell sprouting, extension, proliferation, and vessel wall cell (pericytes, astrocytes) coverage.7,8 Physiological vascular neovascularization not only has intact vascular structure but also can establish connections with neurons that together form neurovascular units and play a protective role. On the contrary, pathological vascularization is harmful to the organism.9,10 In addition, it has been recently found that blood vessels also have the function of influencing the structure of central nervous system cells and neuronal plasticity. 11 Therefore, studying therapies that promote angiogenesis and protect neurovascular function is essential for preventing and treating IS.
Hypoxia is the most critical factor in promoting angiogenesis. When exposed to hypoxia, hypoxia-inducible factors (HIFs) are stabilized, which directly or indirectly activate the transcription of multiple angiogenesis-related molecules to drive vascular angiogenesis.12 –14 It has been demonstrated that continuous hypoxia (CH) leads to intense angiogenesis in the brain, which can significantly reduce the infarct volume in IS.5,15 Despite this, the clinical translation of hypoxic therapy remains limited. This may be due to the risk of neurovascular injury in CH that may result from too low an oxygen concentration and too long a hypoxia duration. 16 In this regard, intermittent hypoxia (IH) offers significant advantages by virtue of its safety and ease of administration. 17 Because IH exposes the organism to hypoxia in small doses multiple times, the stress response is mild but long lasting. Therefore, modestly adjusting different factors affecting the efficacy of IH, including oxygen concentration, frequency, and duration, may affect IS treatment and avoid additional injuries. 18 However, current IH studies are limited, and the effects and mechanisms of resistance to IS are unclear.
Here, we characterized and compared different hypoxic modes and found that IH was able to increase cerebral blood flow (CBF) and oxygen saturation (SO2) no less than CH. We demonstrated that IH with 13% O2, 5-min interval, 10 cycles per day, for 14 consecutive days significantly improved cerebral microcirculation by inducing functional angiogenesis. Furthermore, the effect of IH can resist IS damage, including reducing infarct volume and mitigating neurological damage. Our results reveal that IH is a potential intervention for IS, which lays a theoretical foundation for further clinical translation and application.
Materials and methods
Animals
C57BL/6J wild male mice used in the study were purchased from SPF (Beijing) Biotechnology Co., Ltd. Mice were maintained on a 12 light/12 dark cycle. All animal procedures were approved by the Animal Care and Use Committee of Capital Medical University (permit no. AEEI-2021-058) and performed according to the National Institutes of Health Guide for the Care and Use of Laboratory Animals. All efforts were made to minimize animal suffering and the number of animals used. All animal experiments complied with the ARRIVE guidelines. 19
Models of hypoxia treatments
All hypoxia treatments were performed in mice at 8 weeks of age. The hypoxia models were carried out in a normo-depressional oxygen chamber at a stable and sufficient rate controlled by the gas control delivery system. The normo-depressional anoxic environment was produced with different ratios of 99.99% oxygen and 99.99% nitrogen gases. The hypoxia model included CH and IH carried out for 14 consecutive days. The oxygen concentration in the chamber was automatically controlled with the former constant at the set oxygen concentration and the latter alternating between the target hypoxic and normoxic concentrations. IH was divided into two types. The one type was a 5-min reoxygenation (21% O2) followed by a 5-min hypoxia with 10 consecutive cycles alternately. The other type was a 10-min reoxygenation followed by a 10-min hypoxia with 5 consecutive cycles. Control mice were placed in the same hypoxic chamber with normal air circulating continuously over the same time frame. Chronic intermittent hypoxia began each day at 7:00 p.m. During non-hypoxic hours, the mice were fed in a normoxic animal-rearing environment with free access to food and water.
Surgical approach for distal Middle cerebral artery occlusion
Mice were deeply anesthetized with 5% isoflurane and maintained anesthetized with 2% isoflurane in O2 (Anesthesia Apparatus, RWD Life Science Co., Ltd., China). The dMCAO model is described in the literature. 20 In short, the right common carotid artery was exposed via a midline pre-tracheal incision and was ligated permanently. After exposing the right distal branch of the middle cerebral artery (MCA) by a small craniotomy, the branch was permanently blocked by cautery with an electrical coagulator, taking care to avoid damage to the surrounding tissue. Body temperature was maintained between 36.5–37.5 °C during surgery. To confirm occlusion of the distal MCA, CBF was monitored in the mice before, during, and after surgery using laser speckle (LSCI) (RWD Life Sciences Ltd., China). CBF changes were measured by LSCI at pre-operation, post-operation, 1d and 3 d after dMCAO.In the sham group, the procedure was the same as above, except that the left common carotid artery was not ligated, and the distal MCA branch was not cauterized. Exclusion criteria: CBF was decreased by less than 30% compared with the baseline level after modeling; intraoperative subarachnoid hemorrhage; coagulation damaged the surrounding tissue and caused massive bleeding.
Detection of cerebral blood flow and oxygen saturation
The moor vascular monitoring system and laser doppler perfusion and temperature monitor (Moor Instruments Ltd., UK) was used to monitor the cortical local perfusion level and the local cerebral oxygen level in real time. After 2 weeks of hypoxia, the mice were anesthetized with 5% isoflurane and fixed in the prone position. The midline of the parietal lobe was disinfected, and a longitudinal incision was made to fully expose the skull. The probe was affixed 2 mm posterior to the anterior fontanelle of the mice, 4 mm laterally, and monitored at each spot for 15 s. The instrument measured CBF and cerebral SO2 in the vessels surrounding the probe by laser doppler technology.
Images of cerebral vessels in vivo
We first intravenously injected ICG (0.3 mg/ml, 0.3 ml/30 g; R018175, RhawnSeal) and allowed it to circulate for 30 minutes. In this duration, we prepared for other operations. The skull was exposed after hair removal and skin incision, and the mucosa was removed. Subsequently, a plastic holder with a 5-mm hole was glued onto the skull and an optical clearing skull window was established with skull optical clearing agents. 21 Finally, cerebral vessels were observed in vivo under a Near-Infrared-II Fluorescence microscope 22 (NIR2-MS, Sunny Optical Technology (Group) Company Limited, China).
Detection of cerebral vascular permeability
The permeability of sodium fluorescein (NaFlu) across brain blood vessels was determined. The brains of hypoxic mice were dissected completely, and the cortex, striatum, hippocampus, thalamus and hypothalamus were isolated and homogenized separately. The fluorescence was detected with a CLARIOstar fluorescence microplate reader (BMG LABTECH, Offenburg, Germany) at excitation/emission wavelengths =485/520 nm. Relative cell permeability was calculated by normalizing to the control group.
Behavioral tests
Behavioral experiments were conducted in a quiet room between 8:00 a.m. and 2:00 p.m. The behavior chamber was cleaned with 75% ethanol after each test to remove the smell of mice.
Rotarod test
The rotarod test (Panlab, LE8205) was conducted to assess motor coordination, balance, and learning. 23 The first test session came after a training session of three consecutive days. The mice were subjected to a total of three rotarod test sessions with accelerating speeds (4 to 40 rpm) on day 0, day 14 after initiation of hypoxia, and day 14 after reoxygenation. Each test session was composed of three trials on the rotarod, each with a maximum duration of 300 s, and an inter-trial interval of 1 h. The latency on the rotarod for each mouse was recorded, and the results from each trial were used for further analysis.
Novel object recognition test
As noted earlier, 24 the novel object recognition test is used to evaluate object recognition memory. For three consecutive days, each mouse was placed in an open testing chamber (40 cm × 40 cm) and allowed to explore for 30 min to adapt to the environment. After day 3 of acclimation training, the mice were removed, and two identical objects were placed about 10 cm from the wall and 15 cm apart. New object recognition was defined as spending more time or number of times sniffing new objects (N) than familiar objects (F). When a mouse's nose was within 2 cm of an object, it was rated as close. The identification index was calculated as (N − F) ÷ (N + F), with a positive discrimination index representing a preference for new objects. Recognition memory was scored using each mouse's identification index.
Modified Garcia neurological function score
Neurological deficits in the mice were assessed on day 3 after dMCAO. The neurological scoring system used was a modification of a well-established sensorimotor assessment system.25,26 Briefly, the modified Garcia neurological function score was judged mainly by five manifestations: Body Proprioception, Posture Symmetry, Forelimb Extension, Lateral Turning, Holding and Climbing ability. Scores of 0 to 3 for each test and maximum score is 15. Higher scores indicating less severe neurological impairment and 15 indicating no neurological deficits.
Adhesive removal test
The adhesive removal test is a classic method for measuring sensorimotor deficits. 27 Mice were kept in clean, transparent cages for 1 min to acclimatize them to their environment. A sticky tape (4 × 3 mm) was then applied to the volar side of the mouse's right forepaw with the same force. The mice were then gently returned to their cages. The time taken for them to touch and remove the sticky tape was recorded for up to 60 s.
Grid-walking test
The grid-walking test was as described previously. 28 Mice were placed on a grid (40 × 40 cm; small grid diameter: 2 × 2 cm; 50 cm above the ground). When the mice were allowed to walk freely on the grid, the error was defined as when the right forelimb stepped empty. The error rate was measured as a percentage of the total right forelimb steps.
Cortical tissue clearing and imaging
As previously described, the cortical tissue clearing process was performed to clear the whole brain tissue. 29 Briefly, brain tissue was post-fixed in 4% PFA for four days and immersed in 8% sodium dodecyl sulfate (SDS, Sigma-Aldrich, USA), diluted with 0.1 M PBS (pH 7.5), and shaken at 100 rpm and 37 °C. The clearing solution was replaced every few days until the tissue was clear. After clearing, the brain tissue was washed with 0.01% PBS and Tween-20 (PBST) for 24 h at 37 °C and 100 rpm. Then, the brain tissue was treated by Focus Clear (CelExplorer Labs, Taiwan, China) for 2 h before being photographed. We used light-sheet microscopy (Light-sheet Z.1, Zeiss, Oberkochen, Germany) to take pictures. In the software, the Z-stack was set at 2.5 μm. The images were scanned at 1800 × 1800 pixels in the X–Y direction. Images were stitched into a series of whole-brain transverse images and exported in raw format in Imaris software.
Immunofluorescence staining
Lectin (Vector Lab, DL-1178-1, 1:100; 100 μL/25 g) was used to label vascular endothelial cells. Mice were perfused with saline and 4% paraformaldehyde and then dehydrated in a gradient of 20% and 30% sucrose solution (prepared with 4% PFA). Coronal sections were cut to 100 μm thickness in a freezing microtome (Leica, Germany). Hyperthermia antigen repair was performed in sodium citrate solution. The cells were then blocked with 3% BSA for 60 min. For primary antibody staining (AQP4, Rabbit, CST, 59678S, 1:8000; PDGFRβ, Rabbit, Huabio, ET1605-20, 1:200), the primary antibody was added to the well-plate where the slices were placed and incubated overnight at 4 °C. The primary antibody was carefully aspirated, and the sections were washed before the secondary antibody (594, Goat anti-rabbit, Invitrogen, A-11012,1:500; 488, Goat anti-rabbit, Invitrogen, A-11008) was added and incubated for 1 h at room temperature. DAPI staining was performed for 15 min. After washing, the slices were sealed, and the sections were photographed in 3 D using a fluorescent confocal microscope (Leica TCS SP8, Germany).
Proliferating cell detection by bromodeoxyuridine
To label cells in the proliferative phase, animals received bromodeoxyuridine (BrdU) (CST, 5292S, 50 mg/kg, dissolved in 0.9% sodium chloride) intraperitoneally once daily for 7 consecutive days from 1 week after hypoxia. The control group was injected with BrdU at the same time. Then, 40 μm-thick brain sections were stained with fluorescence, stained with BrdU antibody, sealed, and photographed with a fluorescent confocal microscope (Leica TCS SP8, Germany).
Three-dimensional (3 D) rendered and quantification by imaris
Image processing and quantification have been described previously. 29 Briefly, confocal images were imported into Imaris software (9.7.1, Bitplane, Zurich, Switzerland). Stained images were remodeled to 3D images by the filament tool and surface tool. The source channel was selected, and the proper threshold was set manually to differentiate the target signal from the background. Filter was used to remove non-specific signals. Some parameters needed to be set during processing. The diameter was set at 4 μm and the branch length ratio was set at 3 of the filament tool. Voxels inside surface was set to 100 in the mask channel of the surface tool. Then, the 3 D-rendered images were constructed. All analyses were conducted in the statistics function of the filament tool. The reconstructed vasculature was observed by the surface tool.
Statistical analysis
Shapiro-Wilk test was used to determine the normality of the data. Normally distributed data are reported as mean ± SD, while non-normal variables are presented as median (interquartile range [IQR]). Statistical analyses were performed using Prism 9 (GraphPad Software, San Diego, CA). The two-tailed t-test (normal distribution) or Mann-Whitney U test (non-normal distribution) was used between two groups. One or two-way ANOVA was applied to multiple groups followed by Bonferroni post hoc test for normal distribution datasets. Non-normal distribution datasets were compared using Kruskal-Wallis ANOVA followed by Dunn’s post hoc test. The Bartlett test analyzed the constancy of variances, and a Welch correction was used if not satisfied. Quantitative data are expressed as the mean ± SD, and n indicates the biological replicates per experimental group. A p value less than 0.05 was considered statistically significant. All statistics are referenced in the figure legends.
Results
Intermittent hypoxia treatment with 13% O2 for 2 weeks effectively improved brain perfusion in mice without causing neurological impairment
Angiogenesis is a chronic change in the structure of blood vessels, and effective angiogenesis can be manifested by supplying more blood and oxygen to brain tissue. The pro-angiogenic effect of hypoxia has been widely used in studies of animal models of ischemic stroke. To determine the optimal hypoxia pattern, this study treated mice with different oxygen concentrations and different patterns to investigate whether there were differences in the effects on CBF and SO2 in mice. We exposed adult male mice to normoxic chambers at 7%, 10%, and 13% O2 for 2 weeks with daily alternating hypoxia-normoxia intervals of 5 min for 10 cycles (Figure 1(a)). Zhang et al. found that continuous hypoxia with 7% O2 for 14 days significantly increased collateral circulation in the mouse brain. 15 Therefore, we similarly compared the effects of this hypoxic mode.
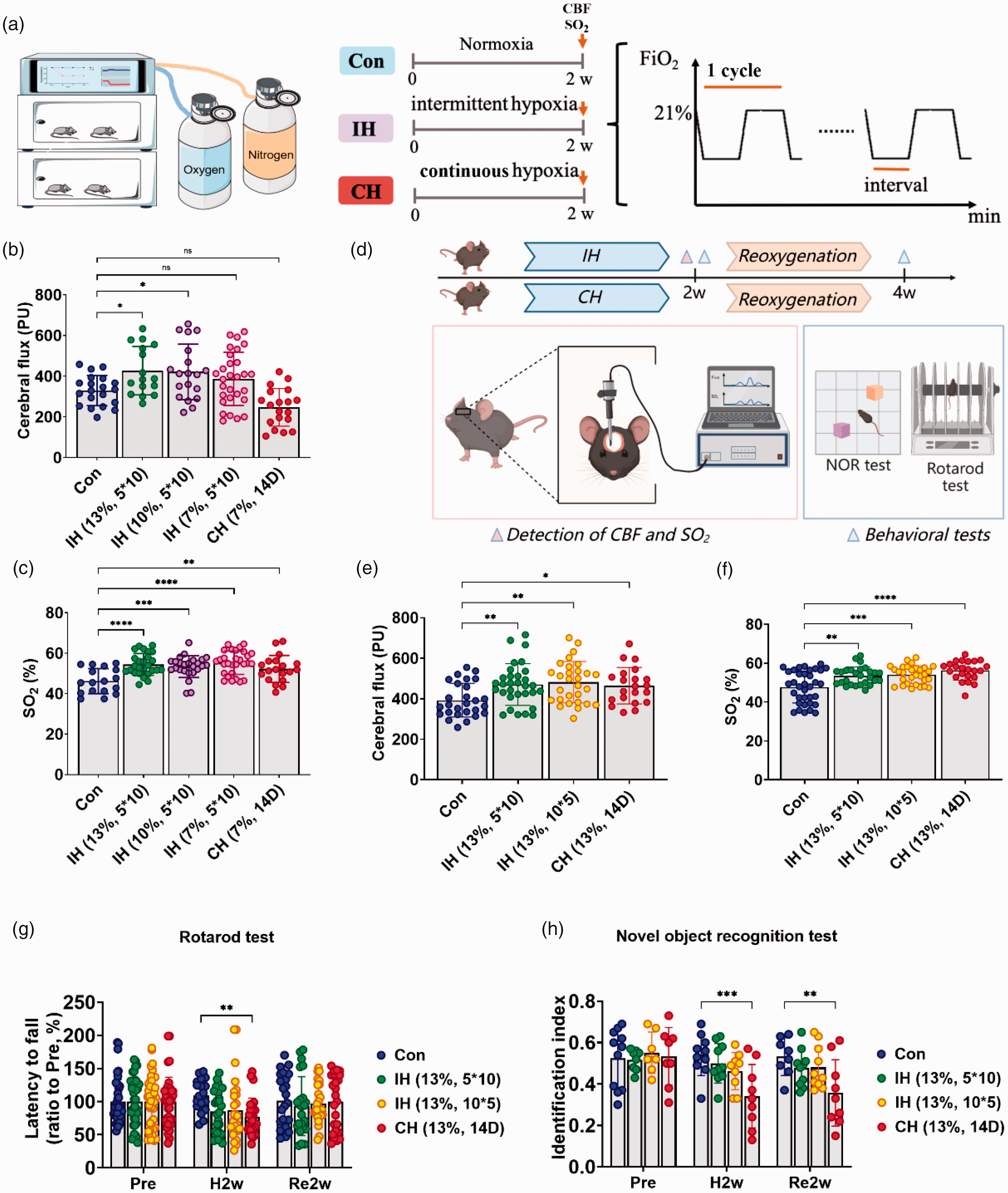
The screening of different hypoxic concentrations and modes. (a) A schematic diagram of the hypoxic chamber and the hypoxic experimental design. Continuous hypoxia (CH) is the maintenance of a constant oxygen concentration over a period of time, while intermittent hypoxia (IH) alternates between the target oxygen concentration and normoxia (21% O2) and ends automatically after a set number of cycles are completed. CBF: cerebral blood flow; SO2: oxygen saturation. CBF and SO2 were measured at the end of 2 weeks of hypoxia (arrows). (b and c) CBF (b) and SO2 (c) in mice treated with the same IH mode at different oxygen concentrations. (d) A schematic diagram of the screening hypoxic mode at 13% O2. Mice were reoxygenated for 2 weeks after 2 weeks of IH/CH. The corresponding tests were performed at the end of hypoxia and at the end of reoxygenation, respectively. NOR test: new object recognition. (e and f) CBF (e) and SO2 (f) in mice treated by different hypoxic modes at 13% O2. (g) Mouse motor function was tested by the rotarod test and (h) Mouse cognitive function was tested by the novel object recognition test. (b–f) Use of one-way ANOVA to compare with the control group. (g–h) Use of two-way ANOVA to compare with the treated group. Data are expressed as the mean ± SD; ns is considered as no significant differences; *p < 0.05, **p < 0.01, ***p < 0.001, ****p < 0.0001.
After 2 weeks of hypoxia, we examined blood flow and SO2 in the mouse cerebral cortex. We found 10% and 13% O2 increased CBF in mice compared with the control group (Figure 1(b)). There was little difference in the effect between the two groups. Compared with the control group, 7%, 10%, and 13% O2 significantly increased SO2 in mice. (Figure 1(c)). However, more severe hypoxia undoubtedly causes damage. 30 Therefore, considering the safety and efficacy, 13% O2 is more preferred to improve CBF and SO2 in mice than the other oxygen concentrations.
To clarify the effects of different hypoxia patterns on cerebral blood flow and SO2 in mice, we explored different modes of hypoxia with 13% O2. We exposed three groups of adult mice to different modes of hypoxia in a normobaric hypoxic chamber with 13% O2 for 2 w. IH (13%, 5*10) was the hypoxic mode in which there were alternating hypoxic-normoxic intervals of 5 min for 10 cycles per day. IH (13%, 10*5) was the hypoxic mode in which there were alternating hypoxic-normoxic intervals of 10 min for 5 cycles per day. CH (13%, 14 D) was continuous hypoxia for 2 weeks (Figure 1(d)). Compared with the control group, IH (13%, 5*10), IH (13%, 10*5), and CH (13%, 14 D) could significantly increase the level of cerebral blood flow and SO2 in mice with little difference in effect (Figure 1(e) and (f)).
To investigate the effects of different hypoxia patterns on neurological function in mice, we evaluated motor and cognitive function after 2 weeks of hypoxia and 2 weeks of reoxygenation. At 2 weeks of hypoxia, the continuous hypoxia group (CH (13%, 14 D)) showed a decrease in latency of the rotarod test (Figure 1(g)), a decrease in the identification index of new object recognition test, and did not recover after 2 weeks of reoxygenation (Figure 1(h)). In contrast, IH did not incur similar injuries. Overall, these data suggest that 13% O2 with IH for 2 weeks increased cerebral blood flow and SO2 in mice and without neurological impairment.
IH (13%, 5*10) improved the cerebrovascular microcirculation network in mice
Brain perfusion was significantly higher in mice after hypoxia, indicating changes in cerebral vascular structure. To investigate the effect of different hypoxic modes on cerebral vascular structure, we injected lectin into the tail vein of mice to label functional capillaries. Vascular reconstruction and quantitative analysis of confocal images were performed with Imaris software (Figure 2(a)). Because capillary diameters ranged from 4 to 10 μm,31,32 we focused our statistical analyses on vessel diameters in this range. We calculated the diameter, length, and volume of the vessels of seven brain regions, including the cortex (CTX), hippocampal CA1 (HPC CA1), hippocampal CA3 (HPC CA3), hippocampal DG (HPC DG), striatum (STR), thalamus (TH), and hypothalamus (HHA) (Figure 2(b) and (c)). No significant differences in vessel diameter changes were detected in each hypoxic mode (Supplementary Figure 1). While changes in length and volume in individual brain regions showed heterogeneity, IH (13%, 5*10) induced an increase in capillary length (Figure 2(d), (h) to (j)) and volume (Supplementary Figure 2) in CTX, HPC DG, TH, and HHA compared with the controls. In some brain regions, while there was homogeneity in length, there was a large distribution in volume, possibly due to changes in vessel diameter caused by vascular remodeling. This may also reflect brain region heterogeneity. CH (13%, 14 D) induced an increase in CTX and HHA capillary length compared to the control (Figure 2(d) and (j)). In STR, IH had no effect on vessel length compared with the control, but IH (13%, 10*5) and CH (13%, 14 D) significantly reduced vessel length (Figure 2(e)). In both HPC CA1 and HPC CA3, there was no significant difference in vessel length between the groups (Figure 2(f) and (g)). Overall, IH (13%, 5*10) and CH (13%, 14 D) increased whole-brain capillary length and volume, while IH (13%, 10*5) did not (Figure 2(k), Supplementary Figure 2H).
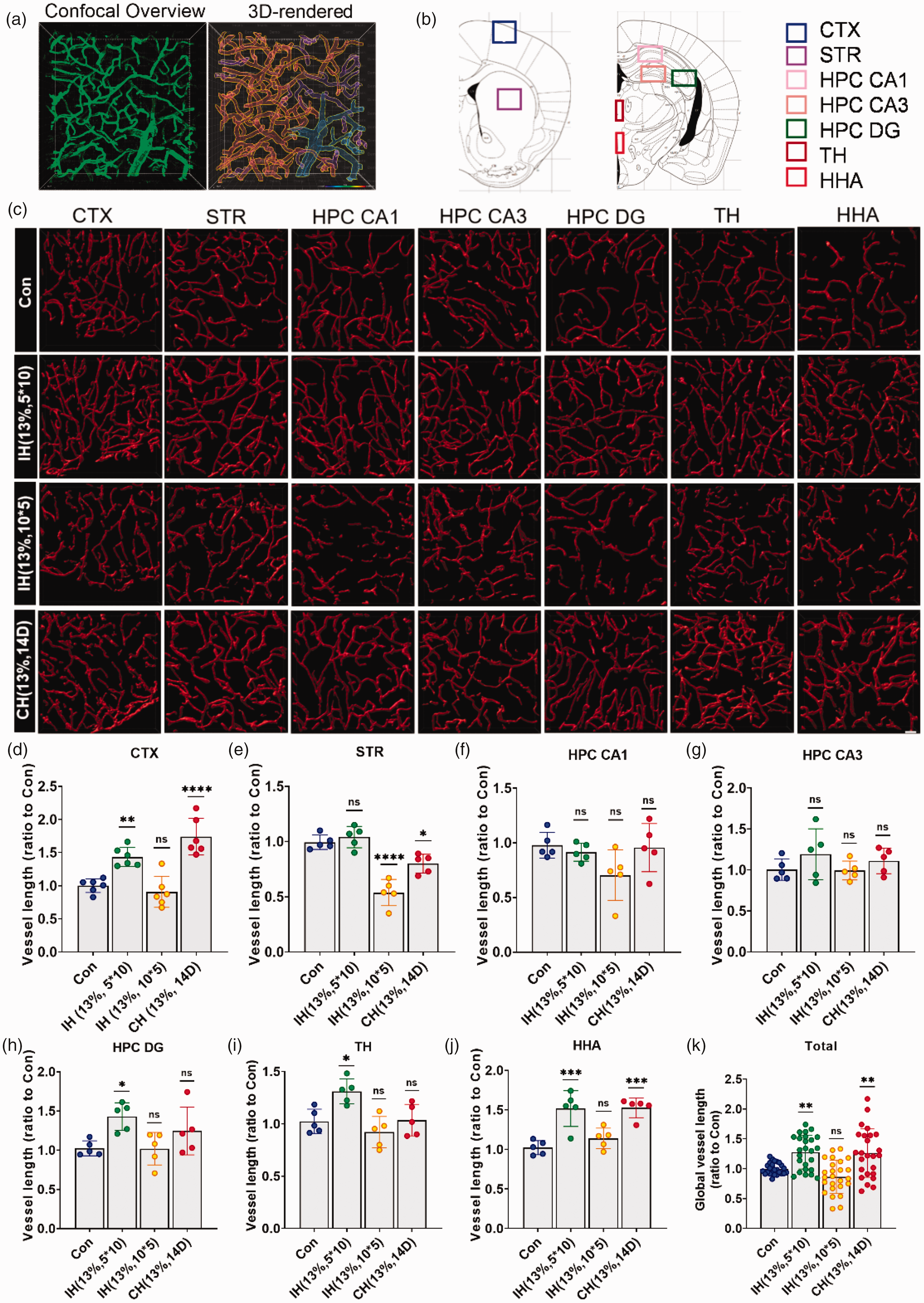
The vascular response to hypoxia in different brain regions is heterogeneous. (a) Confocal images were reconstructed in 3 D in the Imaris software. (b) Different brain regions were photographed by confocal microscopy. CTX: cortex; STR: striatum; HPC CA1: hippocampus CA1; HPC CA3: hippocampus CA3; HPC DG: hippocampus DG; TH: thalamus; HHA: hypothalamus. (c) 3 D reconstruction of representative vessels labeled by lectin in different brain regions. Scale bar = 40 μm. (d–k) Vessel lengths of CTX (d), STR I, HPC CA1 (f), HPC CA3 (g), HPC DG (h), TH (i), HHA (j), and global brain (k) in mice treated with different hypoxic modes. N = 5–6. Statistical methods used one-way ANOVA; data are expressed as the mean ± SD; ns is considered as no significant differences; *p < 0.05, **p < 0.01, ****p < 0.0001, compared with the control group.
To further compare the effects of IH (13%, 5*10) and CH (13%, 14 D) on the structure of perfused cerebral microvessels, hereafter referred to as IH and CH, respectively, we took a part of the cortical tissue (500 μm × 500 μm × 300 μm), which was clarified by the FACT method, and imaged it by a light-sheet microscope. The images showed the 3 D-reconstructed vessels by filament mode (Figure 3(a) and (b)), and we calculated the length, volume, straightness, and branch points of the vessels. Consistent with the previous results, IH and CH significantly increased the length and volume of the cortical vessels (Figure 3(c) and (d)). Meanwhile, although there was no significant difference in vessel straightness, we found that the CH mice had significantly tortuous vessels, whereas IH mice did not (Figure 3(b) and (e)). In addition, IH significantly increased the branch points of the vessels, suggesting a richer collateral circulation (Figure 3(f)). In conclusion, IH more effectively induced functional angiogenesis in mice to improve cerebral microvascular circulation.
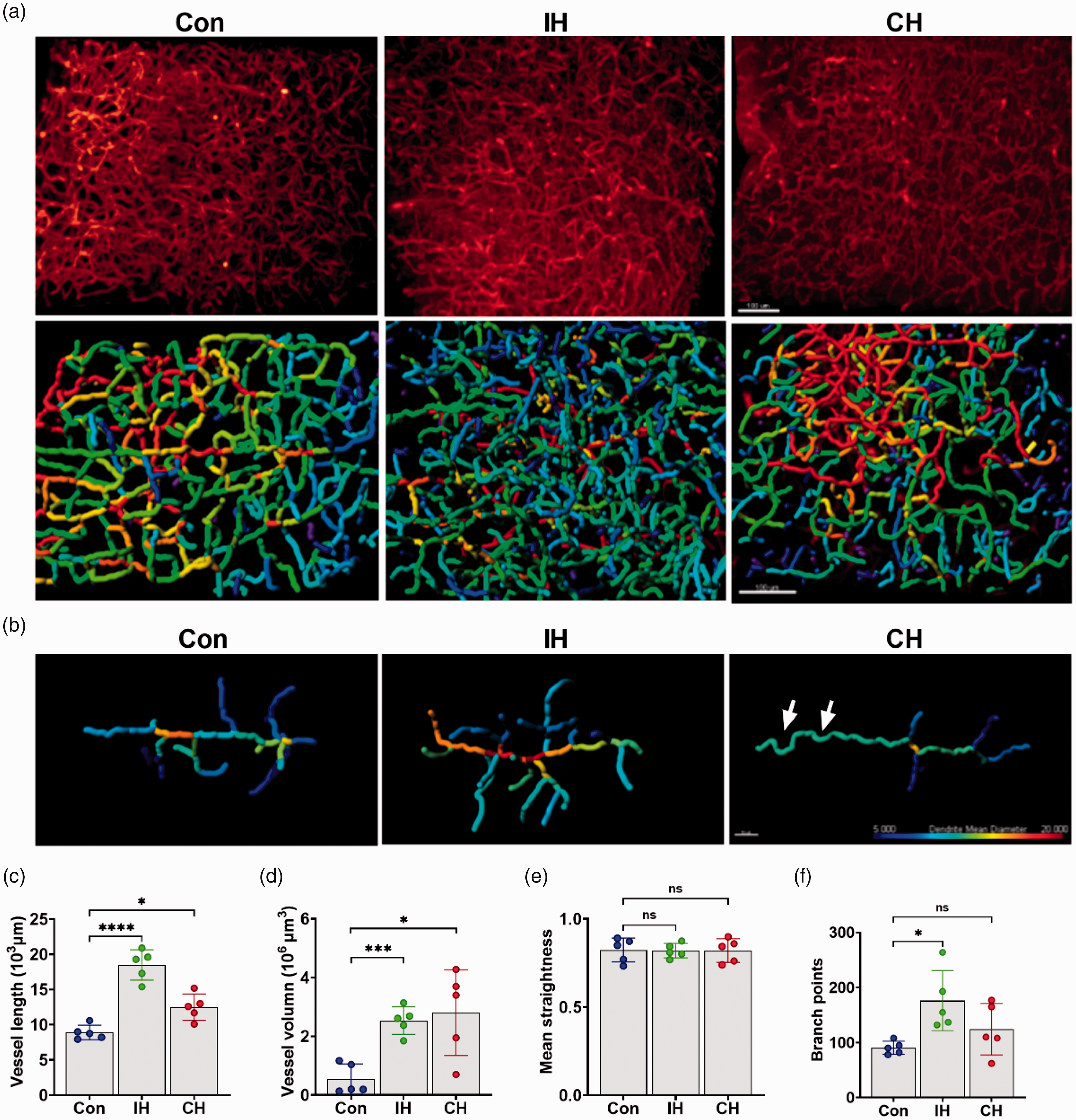
IH improves the cerebrovascular microcirculation network in mice. (a) Raw vascular images and 3 D-rendered images of the mice cortex of Con, IH and CH; Scale bar = 100 μm. (b) 3 D-reconstructed view of typical vessels from the Con, IH, and CH groups. Different colors represent vessel diameters. The CH group resulted in vascular tortuosity (arrows). Scale bar = 50 μm. (c–f) Quantification of vessel length (c), vessel volume (d), mean straightness (e), and branch points (f) in the Con, IH, and CH groups. N = 5. Statistical methods used one-way ANOVA or t test; data are expressed as the mean ± SD; ns is considered as no significant differences; *p < 0.05, **p < 0.01, ***p < 0.001, compared with the control group.
IH induced cerebral angiogenesis and did not affect the glial vascular structure in mice, whereas CH caused destruction
We injected BrdU intraperitoneally to label proliferating cells to investigate whether the hypoxia-induced increase in vessel length was associated with angiogenesis. Immunofluorescence staining of BrdU in blood vessels was performed to label angiogenesis. Compared to the control, BrdU+/Lectin+ was observed in the cortex of both IH and CH groups (Figure 4(a) and (b)). Additionally, in order to intuitively illustrate the new branches of blood vessels, we used in vivo imaging technology to observe the changes of cortical cerebral vessels in the same mouse before and after hypoxia (Figure 4(c)).
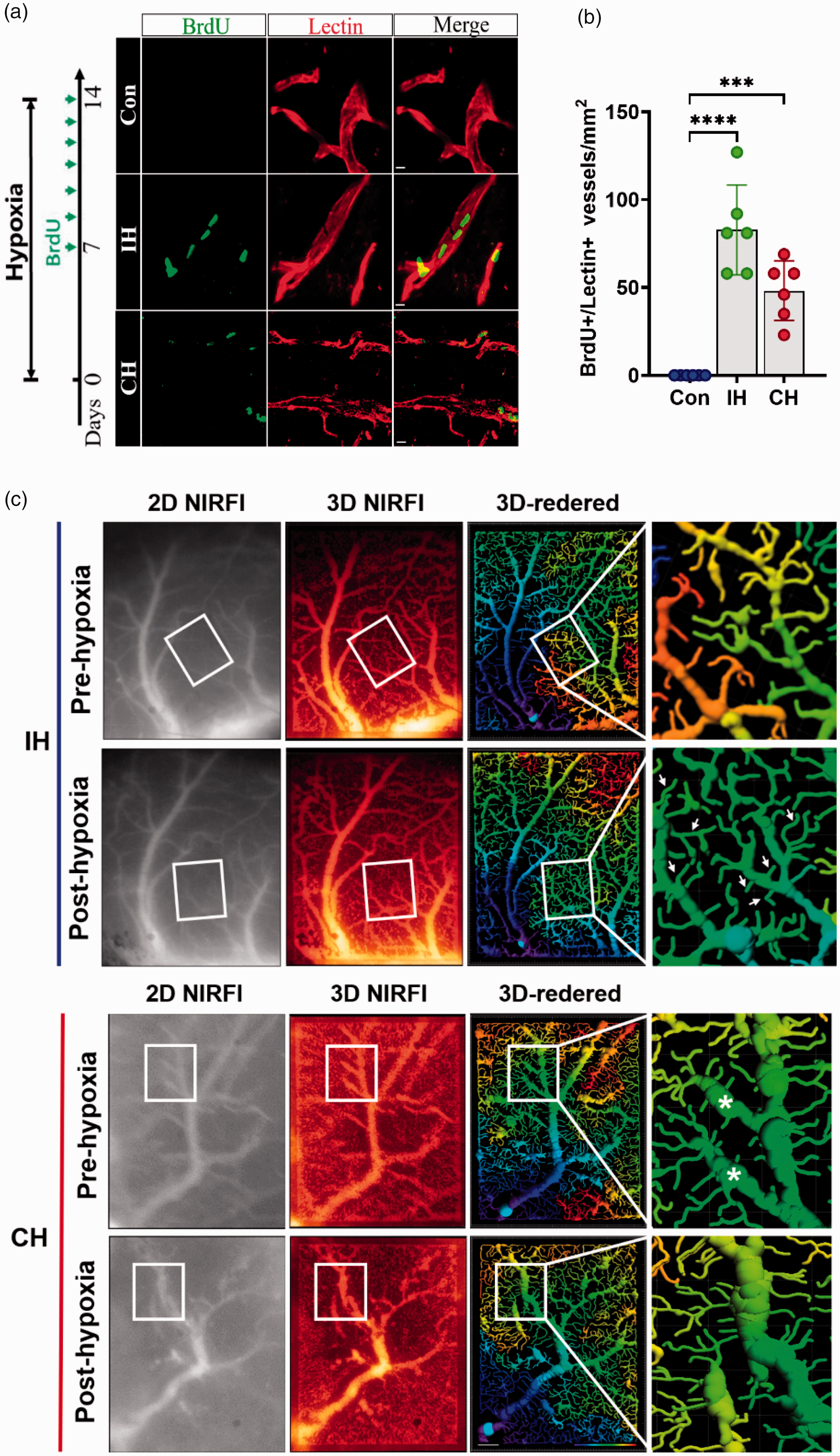
(a) BrdU was injected continuously for 7 days. Immunostaining of BrdU (green) and lectin-labeled vessels (red) within the cortex in the Con, IH, and CH groups at 14 days after hypoxia. Scale bar = 5 μm. (b) Quantitative analysis of BrdU+/Lectin+ cells. N = 6. (c) Near-infrared microscopy was used to observe the changes of cortical blood vessels in mice before and after hypoxia in vivo. Different colors represent branch level. The IH increased the number of vascular branches (arrows), but CH resulted in vascular ablation (asterisks). NIRFI: near-infrared fluorescence imaging; Scale bar = 100 μm. Statistical methods used one-way ANOVA and data are expressed as the mean ± SD; ns is considered as no significant differences; ***p < 0.001, ****p < 0.0001, compared with the control group.
Vascular units composed of endothelial cells, pericytes, and astrocytes are responsible for controlling cerebral blood flow and BBB properties, and the structurally intact vessels are required for proper function. 29 To investigate the coverage of blood vessels by pericytes and astrocytes, we performed immunofluorescence staining of blood vessels, pericytes, and astrocytes in IH, CH, and control mice (Figure 5(a) and (b)), which was quantified using the 3D co-localization function of Imaris. There was no significant difference in the coverage of blood vessels by pericytes after IH and CH (Figure 5(c)). The rate of wrapping of vessels by astrocytes did not differ significantly with IH treatment. However, the rate of wrapping vessels by astrocytes was significantly decreased after CH treatment compared to the controls (Figure 5(d)). To further illustrate the effect of IH and CH on vascular structure, vascular permeability was measured with sodium fluorescein. After 2 weeks of hypoxia, sodium fluorescein permeability was measured in different brain regions. We observed no significant change in vascular permeability in IH compared to Con, whereas CH was significantly increased. Thus, it indicated that CH had a risk of vascular damage (Figure 5(e)).
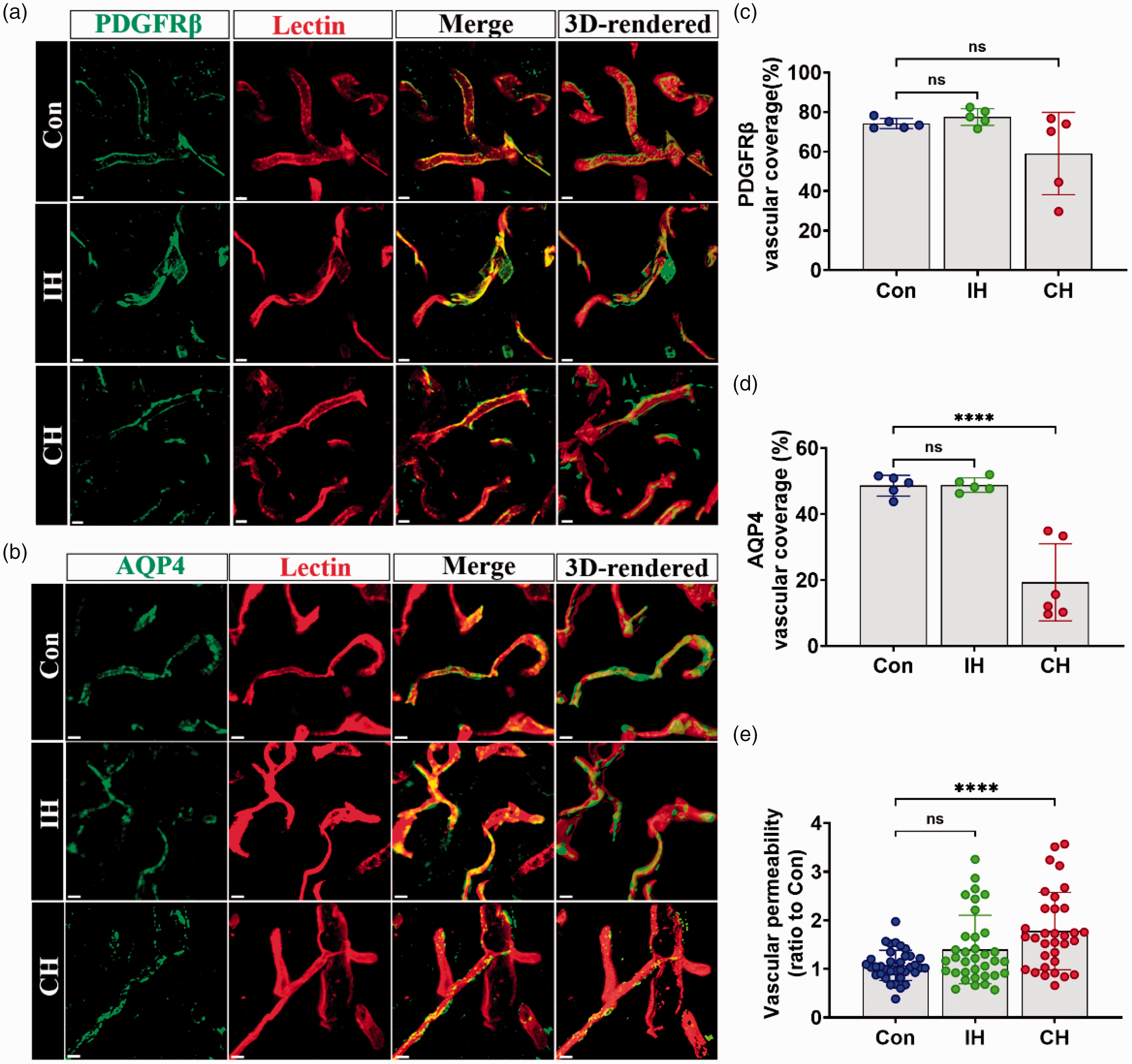
IH does not damage brain blood vessels. (a and b) Immunostaining and 3 D-rendered view of PDGFRβ or AQP4 (green) and lectin-labeled ves sels (red) within the cortex in the Con, IH, and CH groups at 14 days after hypoxia. Scale bar = 5 μm. (c and d) Coverage rate of PDGFRβ and AQP4 were quantitatively analyzed and (e) Vascular permeability was quantified. N = 5–6. Statistical methods used one-way ANOVA and data are expressed as the mean ± SD; ns is considered as no significant differences; *p < 0.05, **p < 0.01, ***p < 0.001, ****p < 0.0001, compared with the control group.
IH reduced neurological impairment in the dMCAO mouse model
The prevention of ischemic stroke by improving the cerebrovascular network has been reported. 33 To determine the preventive effect of IH on ischemic stroke in this study, we firstly investigated the effect of hypoxia on neurological function by the adhesive removal test, grid-walking test, and neurological function scores. We found that IH increased motor function while CH decreased sensory or motor function. This was demonstrated by the fact that CH prolonged the time that the mice took to remove the sticky tape (Supplementary Figure 3B), and IH increased the number of mouse-walking steps within a given time (Supplementary Figure 3C).
We then used a mouse model of ischemic stroke with distal middle cerebral artery occlusion (dMCAO) to investigate the effect of hypoxia treatments on neurological function in ischemic stroke. We administered IH and CH pretreatment for 2 weeks before the dMCAO model and performed dMCAO within 24 h of the end of hypoxia. We assessed neurological function and brain infarct volume 3 d after dMCAO (Figure 6(a)). IH attenuated the neurological impairment associated with ischemia and hypoxia. For the adhesive removal test, the fine sensory function of the mice was examined. Compared with the dMCAO group, IH significantly reduced the time mice felt the sticky tape at 3 d after surgery (Figure 6(b)), while there was no difference in the time of the sticky tape removal (Figure 6(c)). CH had no effect on the time the mice felt the sticky tape (Figure 6(b)), but prolonged the time of sticky tape removal (Figure 6(c)). There was no significant difference in the effect of both IH and CH on neurological function scores compared to the dMCAO group (Figure 6(d)). For the grid-walking test, the overall motor function of the mice was examined. IH increased the number of steps taken but significantly reduced the foot-fault rate (Figure 6(e) and (f)). CH decreased the total number of steps taken and increased the foot-fault rate (Figure 6(e) and (f)).
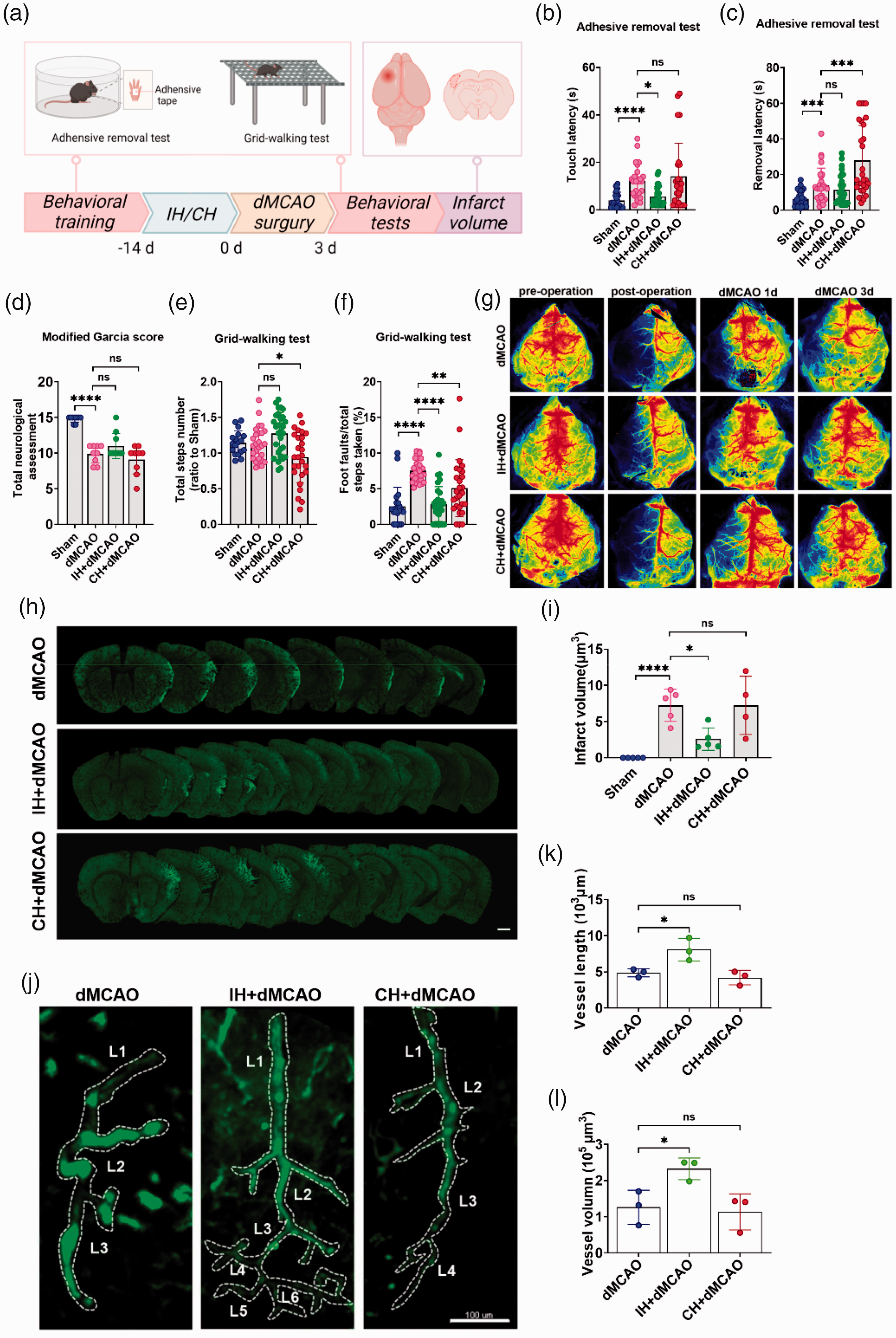
IH reduced neurological impairment in the dMCAO mice and reduced the infarct volume by improving the cerebral microcirculation network. (a) Experimental design of IH/CH effects on dMCAO mice. (b and c) Adhesive removal test was done at the 3rd day of dMCAO. The time of touching to sticky tape (b) and the time of removal (c) were recorded. (d) Neurological function scores were done at the 3rd day of dMCAO. (e and f) Grid-walking test was performed at the 3rd day of dMCAO. Total step number (e) and the foot fault rate were recorded (f). (g) CBF of mice at pre-operation, post-operation, 0d, 1d and 3d after dMCAO. (h and i) Fluorescent staining and quantification of the infarct areas in each group. Scale bar = 1 mm. N = 4–5. (j) Typical image of a single vessel around the infarct area in each group. L: branch level. Scale bar = 100 μm. (k and l) Quantification of vessel length and vessel volume in the infarct area. N = 3. Statistical methods used one-way ANOVA and data are expressed as the mean ± SD; ns is considered as no significant differences; *p < 0.05, **p < 0.01, ***p < 0.001, ****p < 0.0001. B-F.N = 7–10.
IH reduced the infarct volume by improving the cerebral microcirculation network in the dMCAO mouse model
We further explored the effects of IH/CH on stroke mice. For one thing, we observed the recovery of CBF at 1d and 3d after dMCAO. It was found that IH+dMCAO group significantly restored CBF in dMCAO 3d compared with dMCAO, while CH was less effective than IH (Figure 6(g)). For another, we used the immunofluorescent dye MAP2 to label neurons to calculate the infarct volume in the mice (Figure 6(h)). Compared with the dMCAO group, IH + dMCAO group reduced the infarct volume in dMCAO at 3 d, while CH exhibited increased infarct volume (Figure 6(i)). These data suggest that IH significantly reduced the infarct volume and neurological impairment in the dMCAO model mice. It is worth mentioning that we explored whether the protection from IH in stroke mice was related to cerebrovascular microcirculation. we evaluated the vascular density around the infarct area (Figure 6(j)). The IH intervention group exhibited longer vessel length, higher vessel volume, and more vascular branch levels than the dMCAO group (Figure 6(k) and (l)). In summary, IH might reduce infarct volume and neurological damage by improving cerebrovascular microcirculation.
Discussion
In conclusion, our findings confirmed that IH with 13% O2, 5-min intervals, and 10 cycles per day for 14 consecutive days induced functional cerebral microvascular angiogenesis with resistance to ischemic-hypoxic brain damage.
The sudden cessation of blood supply to parts of the brain is an important cause of IS, which can lead to neurovascular uncoupling and secondary neurological impairment. 3 Functional cerebrovascular microcirculation provides oxygen and nutrients to the central nervous system and improves the ischemic-hypoxic tolerance of brain tissue. This can restore perfusion to the infarcted area in time and prevent the occurrence of IS or improve the prognosis of IS in a more durable manner.5,34 Therefore, pro-angiogenic therapy to enhance brain tissue microcirculation in people at risk for stroke would enhance their resistance to ischemic-hypoxic injury. Hypoxia has been previously reported as an effective stressor for inducing angiogenesis. Previous studies found that 7%–13% O2 with hypoxia for 1–3 weeks reduced infarct volumes in rodent ischemic-hypoxic models, and all were associated with vascular neovascularization.15,35,36 However, some studies have also found negative effects of CH. Prolonged exposure to 8% O2 can lead to massive hippocampal neuronal death and vascular leakage.16,37 In contrast, there was no neurovascular damage with IH for 2–4 h per day compared to CH. 17 This is continuous with our findings, where even mild hypoxia with 13% O2 for 14 days led to neurovascular damage. However, IH did not lead to injury. More promisingly, we found that 13% O2 with a 5-min interval and 10 cycles per day for 14 days induced stronger vascular angiogenesis than continuous hypoxia with intact vascular structure. To assess the function of cerebrovascular microcirculation, we used the dMCAO model. We found that IH-intervened mice had significantly more vessel length, vessel branch levels, and branch points around the infarct area, and reduced brain tissue infarct volume and neurological impairment than dMCAO and CH-intervened mice. Instead, CH exacerbated the ischemic and hypoxic injury. This suggests that IH may induce richer cerebral perfusion.
In terms of improving cerebral microcirculation, IH showed multiple advantages over CH. First, for the effect on neurological function, we found that IH (13%, 5*10) did not affect motor and cognitive function in mice, while CH significantly impaired short-term cognitive function and had long-term effects. Second, for the effects on vascular angiogenesis (Figure 7), although both IH and CH increased CBF and SO2, IH induced angiogenesis more effectively, significantly increasing the vascular length in the cortex, hippocampus, thalamus, and hypothalamus. In addition, we found that CH caused cerebral vascular tortuosity similar to AD vascular pathology, which may suggest hemodynamic abnormalities. 38 Third, for the effect on the BBB, we found that IH maintained the structural integrity of the vasculature compared to CH. It is generally accepted that the major cellular units of the BBB are endothelial cells, pericytes, and astrocytes.9,39 The BBB interacts with neurons and together they form a functional neurovascular unit.9,40 Recent findings have suggested that microcirculatory collapse may underlie neurovascular uncoupling in ischemic peripheral brain regions after stroke.41,42 Therefore, we investigated the effects of IH and CH on the vascular coverage of pericytes and astrocytes. Compared with controls, IH did not alter the coverage of blood vessels by pericytes and astrocytes, whereas CH decreased the coverage by astrocytes.
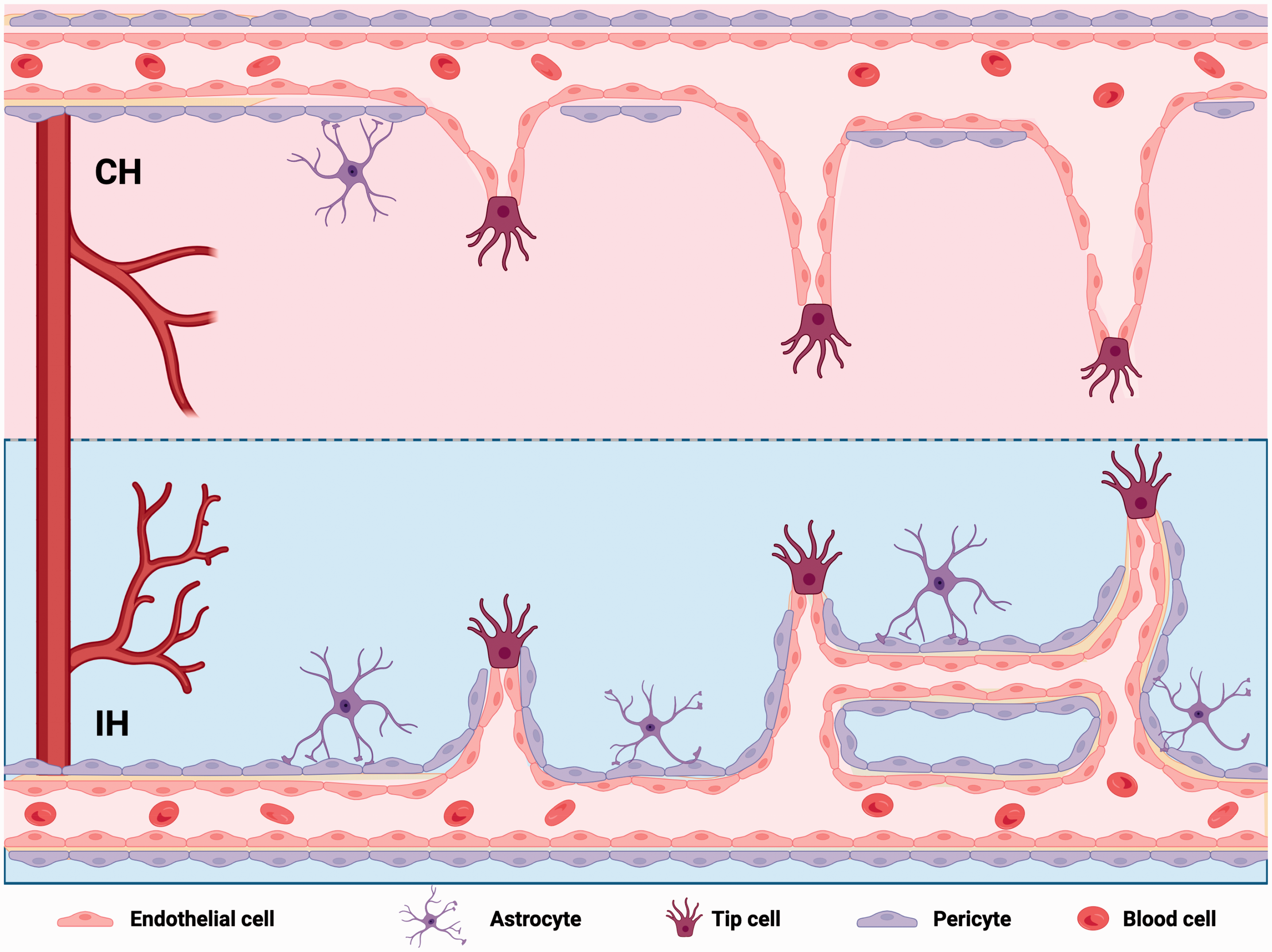
Different roles of IH and CH in promoting brain angiogenesis. IH promoted angiogenesis with regular cell arrangement and maintained vascular structural integrity. CH caused that cerebral vascular structural disrupted, the branch decreased, the diameter increased and less active endothelial cell sprouting.
The effect of IH to improve cerebrovascular microcirculation is influenced by various factors, such as oxygen concentration, frequency, cycles, and duration. For example, 10% O2 for 4 weeks for 15 h per day did not exert a protective effect in IS. 43 High-frequency IH (e.g., sleep apnea) increases the risk of IS. One review suggests that 9–16% O2, 3–15 times per day, will stimulate beneficial effects. 18 We compared different oxygen concentrations of 7%, 10%, and 13% IH with an interval of 5 min and 10 cycles. The 10% and 13% O2 significantly increased CBF and SO2. At 13% O2, we further screened different hypoxic modes and compared the hypoxic effects of IH (13%, 5*10), IH (13%, 10*5) and CH (13%, 14 D). The results showed that IH (13%, 5*10) produced beneficial effects by both increasing cerebral perfusion and promoting angiogenesis.
In summary, we demonstrated that IH (13%, 10*5) is a safe and effective intervention, providing a theoretical basis for clinical translation of IS. Current clinical interventions in IS are limited, and the related drug studies are largely translational failures. Compared to other interventions, IH is simple, inexpensive, and non-invasive, all of which facilitate its clinical translation. Although we have studied the effects of interventions for IS only, the current problems of vascular microcirculation are present in many diseases, including smog and neurodegenerative diseases.44 –47 Therefore, there may be broader applications for IH.
Although in this study, IH was found to protect against IS by improving cerebrovascular microcirculation, there are still several limitations. The protective effect might be affected by many factors, such as age, gender and comorbidities. Firstly, elderly population are more vulnerable to stroke, but preclinical experiments mostly use adult animals for IS intervention studies, 48 which may limit the clinical translation of intervention strategies. It is worth exploring the effect of IH on elderly IS mice models. Secondly, gender is a powerful risk factor for IS incidence and mortality.49,50 Many studies suggested that during most of the life span, men have a higher incidence of IS, 51 and over the age of 85 years more women suffer stroke. 52 Studies indicated that elderly women have worse recovery post-stroke, including sex-specific comorbidities, aggressiveness of acute treatment, which may increase mortality in women with IS.53,54 It is necessary to explore the effect of IH on the female IS model in future studies. Furthermore, in clinical practice, patients with IS are often accompanied by a range of comorbidities, such as hypertension, hyperglycemia, hyperlipidemia, overweight, etc. 53 It is worth exploring whether these comorbidities affect the protective effect of IH on IS models. Therefore, these factors should be considered to evaluate the impact of IH on complicated IS, offering a theoretical foundation for clinical translation.
In conclusion, our study revealed that IH can reduce infarct volume and protect neurological function by improving the cerebral microvascular network in a cerebral infarction model. This provides a basis for the clinical translation of IH against IS.
Supplemental Material
sj-pdf-1-jcb-10.1177_0271678X231185507 - Supplemental material for Intermittent hypoxia protects against hypoxic-ischemic brain damage by inducing functional angiogenesis
Supplemental material, sj-pdf-1-jcb-10.1177_0271678X231185507 for Intermittent hypoxia protects against hypoxic-ischemic brain damage by inducing functional angiogenesis by Yuying Guan, Yakun Gu, Haitao Shao, Wei Ma, Gaifen Li, Mengyuan Guo, Qianqian Shao, Yuning Li, Yingxia Liu, Chaoyu Wang, Zhengming Tian, Jia Liu and Xunming Ji in Journal of Cerebral Blood Flow & Metabolism
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This manuscript was supported by the Beijing Nova Program (Grant number: Z211100002121038), the National Natural Science Foundation of China (Grant number: 32100925, 82027802), Beijing-Tianjin-Hebei Basic Research Cooperation Project (Grant number: 22JCZXJC00190).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Authors’ contributions
J.L. and X.J. supervised the study. Y.Guan., Y.Gu., J.L., and X.J. conceived and designed the study. Y.Guan., Y.Gu., H.S., W.M., G.L., M.G., Q.S., Y.Li., Y.Liu., C.W., and Z.T. performed the experiments. Y.Guan., Y.Gu., J.L., and X.J. wrote the manuscript.
Supplementary material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
