Abstract
Remote ischaemic conditioning (RIC) is achieved by repeated transient ischaemia of a distant organ/limb and is neuroprotective in experimental ischaemic stroke. However, the optimal time and methods of administration are unclear. Systematic review identified relevant preclinical studies; two authors independently extracted data on infarct volume, neurological deficit, RIC method (administration time, site, cycle number, length of limb occlusion (dose)), species and quality. Data were analysed using random effects models; results expressed as standardised mean difference (SMD). In 57 publications incorporating 99 experiments (1406 rats, 101 mice, 14 monkeys), RIC reduced lesion volume in transient (SMD −2.0; 95% CI −2.38, −1.61; p < 0.00001) and permanent (SMD −1.54; 95% CI −2.38, −1.61; p < 0.00001) focal models of ischaemia and improved neurological deficit (SMD −1.63; 95% CI −1.97, −1.29, p < 0.00001). In meta-regression, cycle length and number, dose and limb number did not interact with infarct volume, although country and physiological monitoring during anaesthesia did. In all studies, RIC was ineffective if the dose was <10 or ≥50 min. Median study quality was 7 (range 4–9/10); Egger’s test suggested publication bias (p < 0.001). RIC is most effective in experimental stroke using a dose between 10 and 45 min. Further studies using repeated dosing in animals with co-morbidities are warranted.
Introduction
The paradigm of ischaemic conditioning conferring organ protection from a subsequent or ongoing ischaemic insult has been under investigation since the 1980s, 1 but its apparent pre-clinical benefit has yet to be translated consistently in randomised controlled trials. The potential to induce ischaemic tolerance in distant tissue beds by remote, transient, non-lethal limb ischaemia (remote ischaemic conditioning, RIC) is an attractive therapeutic strategy in terms of cost and ease of intervention delivery, performed simply by inflating a blood pressure cuff on an arm or leg.
Applying RIC before, during or after an ischaemic event (pre-conditioning [RIPreC], per-conditioning [RIPerC] or post-conditioning [RIPostC]) shows promise in multiple vascular diseases.2–4 However, although early trials of RIC prior to coronary artery bypass grafting demonstrated a reduction in peri-operative myocardial injury, larger phase III trials were neutral in improving long term outcomes,5,6 which is potentially explained by interactions with cardioprotective anaesthetic agents. 7 In the setting of protecting the brain from injury with RIC, multiple neuro-humoral mechanisms are implicated (see Hess et al. 8 ), but human clinical evidence is limited. In a large meta-analysis of randomised trials of ischaemic conditioning in all conditions, the risk of recurrent stroke was significantly reduced, though the evidence is of low quality. 9 Further, early proof-of-concept human trials assessing RIC in acute stroke (RIPerC and RIPostC),3,10,11 intracranial stenosis (RIPostC)12,13 and carotid stenting (RIPreC) have commenced. 14
Despite the move into human trials, there are a number of unanswered questions regarding the application of RIC, namely optimal method (e.g. one vs. two limbs), dose (number and length of cycles of limb ischaemia and reperfusion), and timing of intervention. We therefore systematically reviewed and meta-analysed the accumulating pre-clinical evidence in acute stroke models of RIPreC, RIPerC and RIPostC to help provide further insight and inform future work.
Methods
The systematic review was performed in accordance with Preferred Reporting Items for Systematic reviews and Meta-Analyses (PRISMA) guidelines. 15 The protocol was registered with PROSPERO, reference CRD42018095739. Preclinical (non-human) studies evaluating the effects of RIC in animal models with induced focal ischaemic stroke were searched up to December 2019 in Embase, Medline, Pubmed and Web of Science. Two authors independently performed the search and acquired the data. Search key words included: (stroke or cerebrovascular disease or brain infarction or brain ischemia or carotid artery disease or cerebral artery disease or cerebrovascular accident or (isch?emi$adj6 (stroke$or apoplex$or cerebral vasc$or cerebrovasc$or CVA))) AND (remote isch?emic conditioning or (remote adj3 (preconditioning or perconditioning or postconditioning)) or RIC or RIPerC or RIPostC or RIP or RIPC or RPC or IPerC or rIPC).
The identified abstracts and titles of the studies were checked and removed if they were not relevant to the study. If only the abstract of a study was available, it was excluded. The studies were included if the following criteria were met: (i) there was a control group; (ii) the study was completed in nonhuman subjects; (iii) a focal ischaemic stroke, not global; (iv) treatment was given in acute models (within seven days), not chronic; (v) RIC was the only treatment administered, not in conjunction with other treatments; (vi) RIC must be administered before, during or after the onset of an ischaemic stroke; (vii) there were measures on infarct size or neurological score and (viii) data were from original articles not review articles.
Risk of selection, performance, detection, attrition and reporting bias was assessed using SYRCLE’s risk of bias tool. 16
Data acquisition
The number of animals, mean outcome, standard deviation or standard error of the mean was collected for control and treatment groups. Studies providing summary data on the infarct size as a volume or area (mm 3 or as a percentage [%] of the whole brain size) and neurological score were gathered from all included papers along with species, gender, stroke model and quality. If data were not written, published graphs were enlarged and the position of the data points determined using Grab software (version 1.10) on Apple Mac. If studies conducted more than one experiment against a single control, the number of animals in the control group was divided by the number of comparison groups (to prevent double counting control animals). Data were independently extracted by three authors (PW, RM and TE).
The time of first dose was recorded relative to the time of ischaemia onset and not the time of reperfusion. For example, if RIC commenced 10 minutes after reperfusion in a transient model of 120 min middle cerebral artery occlusion (MCAo), a time of 130 min was recorded. 17 Time of treatment was categorised as either RIPreC (treatment started before ischaemia), RIPerC (after ischaemia onset but before reperfusion) or RIPostC (started after reperfusion). It was not possible to consistently separate RIPerC and RIPostC groups and these were combined to form one group.
Study quality
The quality of the article from included studies was assessed using the scoring system recommended by CAMARADES (range 1–10).18,19 A point was awarded to the study if it met the following criteria: (i) peer-reviewed publication (ii) statement of control of temperature, (iii) random allocation to treatment or control, (iv) blinded induction of ischaemia, (v) blinded assessment of outcome, (vi) use of anaesthetic without significant intrinsic neuroprotective activity, (vii) appropriate animal model (transient, permanent, embolic or photothrombotic models), (viii) sample size calculation, (ix) compliance with animal welfare regulation, (x) statement of potential conflicts of interest. Further, assessment of data quality was determined by the presence or absence of physiological monitoring during anaesthesia, including blood glucose, blood gas, cerebral blood flow (CBF) and blood pressure.
Data analysis
The data were analysed using Cochrane Review Manager (version 5.3, Copenhagen, The Nordic Cochrane Centre, The Cochrane Collaboration, 2014) according to pre-specified subgroups: species, model of ischaemia (permanent vs. transient), time of administration (RIPreC vs. RIPerC and RIPostC), dose (number, length and total length of cycles) and study quality. Continuous data are presented as a standardised mean difference (SMD) with 95% confidence intervals. Statistical significance was set at p < 0.05. Egger’s statistic and meta-regression of subgroups were performed using Stata/SE (version 15.1 for Mac). Data reliability was assessed through sensitivity analyses by re-analysing the dose–response relationship in all studies that provided a statement of physiological monitoring during anaesthesia.
Results
Study characteristics
The primary search for studies on Medline, Pubmed, Embase and Web of Science identified 804 studies (Supplementary Figure I). After the exclusion criteria were applied, 57 studies remained and were used in the meta-analysis. Studies were conducted across seven countries (Canada (n = 1), China (43), Italy (1), Japan (1), Russia (2), Slovak Republic (2) and USA (7)) across 41 laboratories. In 99 experimental paradigms including 1521 animals, RIPreC, RIPerC and RIPostC significantly reduced infarct volume, SMD −1.87 (95% CI, −2.18, −1.56), which was equivalent to a 34% reduction (weighted by number of animals per study) compared to control.
Eighty of the 99 experiments used Sprague–Dawley rats (n = 1311 animals), five tested Wistar rats (n = 50), two examined outbred rats (n = 43), eight studied C57BL/6 mice (n = 89), one studied CD1 mice (n = 12) and only three used primates (n = 14) (Supplementary Table I). The majority of publications (n = 81 experiments) induced transient focal cerebral ischaemia ranging from 10 to 120 min of MCAo. In three studies,20–22 ischaemia was induced by embolic MCAo and three studies used permanent models of middle cerebral artery occlusion.23–25 RIC was mostly administered by occlusion of the femoral arteries or hind limbs (Supplementary Table I), however, in one study RIC was achieved through infrarenal aortic occlusion (categorised as bilateral limb occlusion) 26 and another study occluded the unilateral renal artery. 27 The administration and frequency of RIC varied between studies and therefore allowed a comparison between different times of administration and the number and length of cycles. Timing of treatment was not clear in two of the experiments, which were consequently excluded from time-to-treatment analyses.28,29
All studies
RIC was significantly effective in both RIPreC and RIPerC/RIPostC models (Table 1, Figures 1 and 2), the greatest magnitude in the latter, though there was no interaction with infarct volume when the two groups were analysed in meta-regression (SMD [95% CI]: RIPreC −1.54 [−2.07, −1.01] vs. −2.0 [−2.38, −1.61], p = 0.368). Notably, there was significant statistical heterogeneity: I 2 = 71% in RIPreC studies and 80% in per/post conditioning experiments. Efficacy was evident in both transient and permanent stroke models though much fewer animals were assessed with permanent ischaemia (n = 140). RIC was not effective if the length of each cycle was less than 5 min, or if the total length of limb ischaemia was less than 10 minutes (Table 1). RIC also improved neurological function significantly (SMD −1.63 [−1.97, −1.29], p < 0.00001) in studies using the Garcia 18-point scale (by 2.5 points, p = 0.002), Longa 5-point scale (0.9 points), focal neurological score (9.7 points), the 12-point scale (1.4 points) and the Spetzler motor score in monkeys (1.5 points); but not in studies using the Neurological Severity Score or the 3-point scale (Table 1).
The effect of administration time, species, stroke model, and RIC administration method on (A) infarct volume in all preclinical studies of remote ischaemic pre- per- and post-conditioning, compared to control and (B) the effect of remote ischaemic pre- per- and post-conditioning, compared to control, on neurological score.
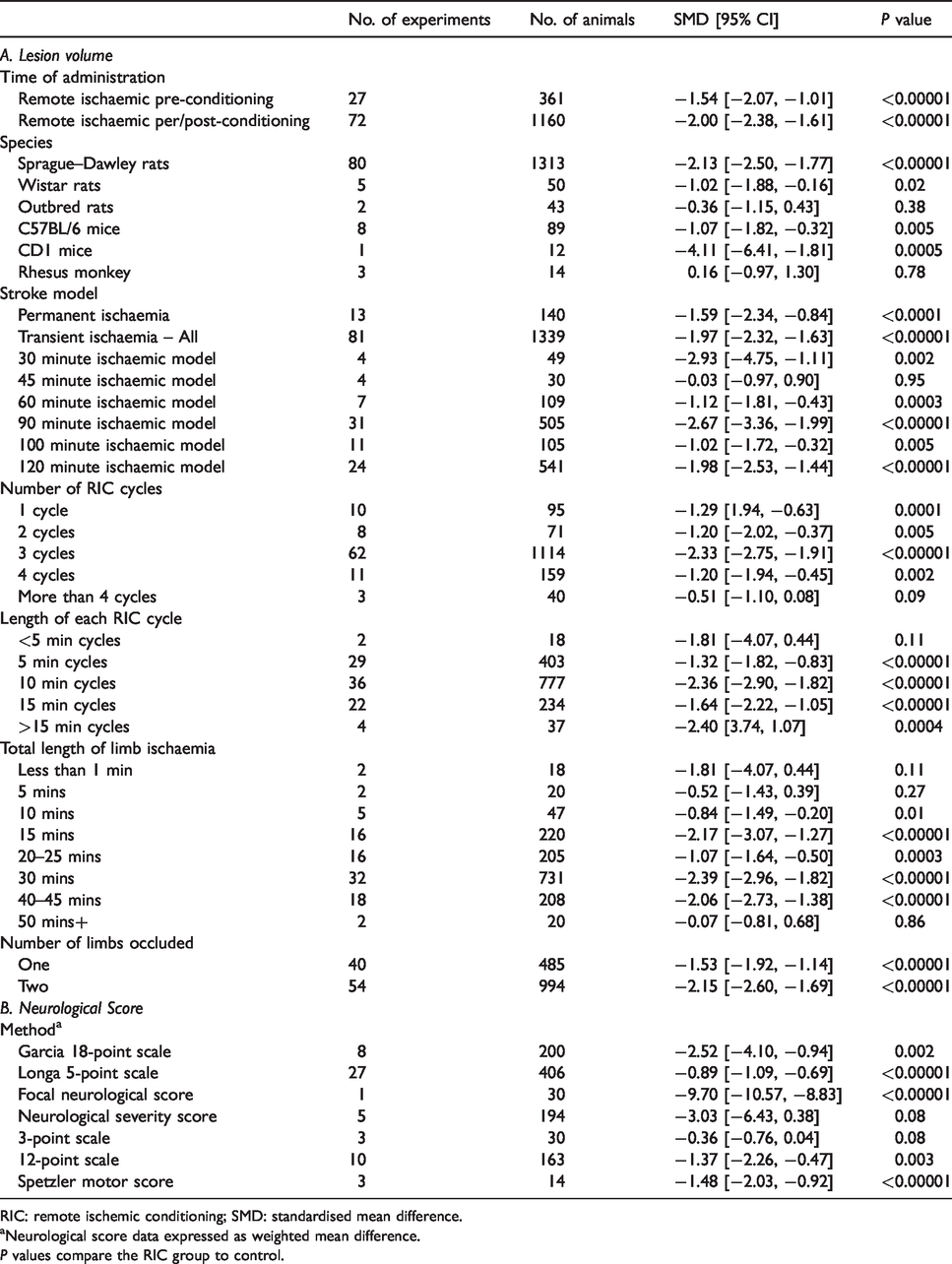
RIC: remote ischemic conditioning; SMD: standardised mean difference.
aNeurological score data expressed as weighted mean difference.
P values compare the RIC group to control.
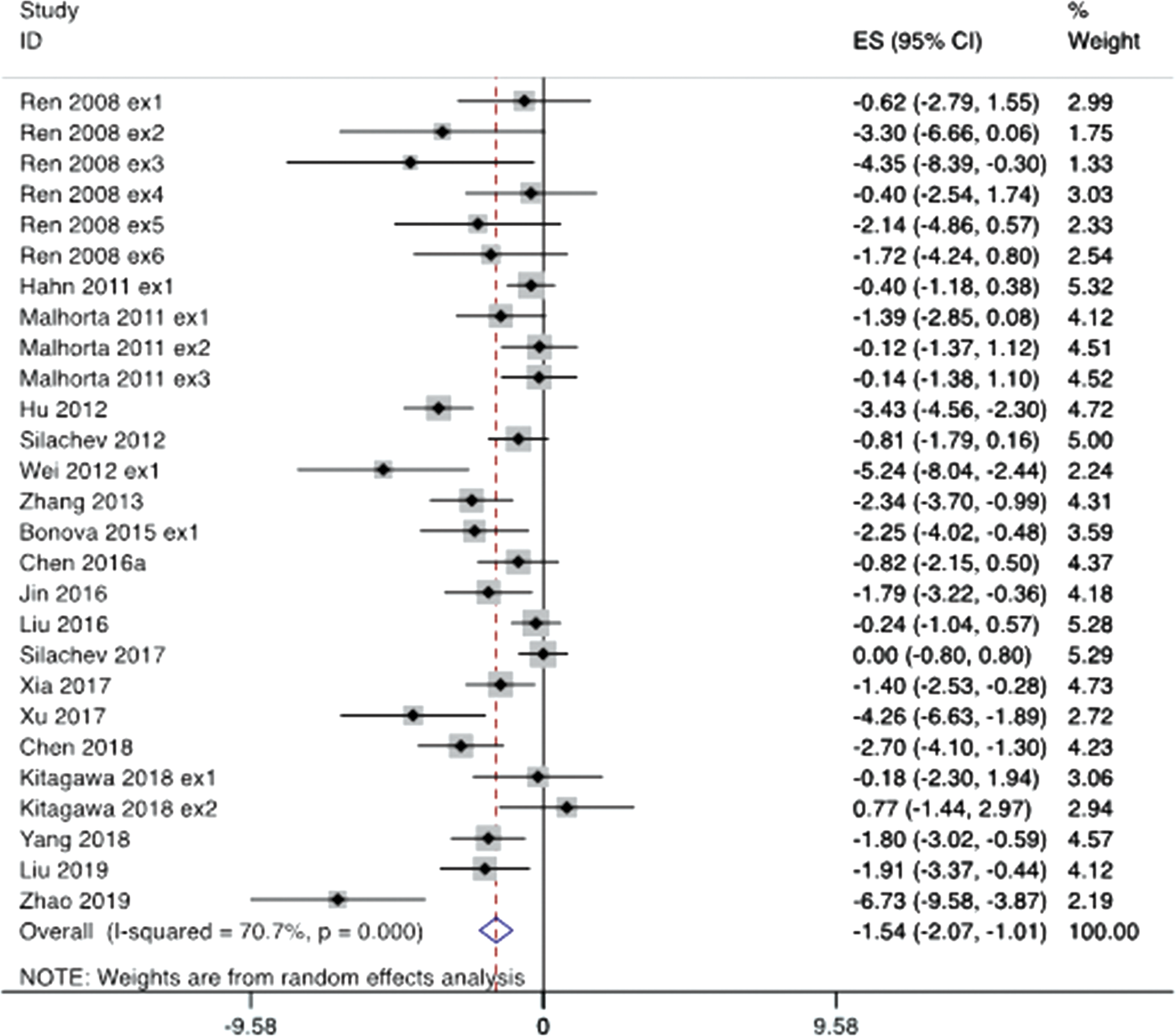
Effect of remote ischemic pre-conditioning (RIPreC) compared to control on infarct volume, expressed as a standardised mean difference, by individual publication experiment.
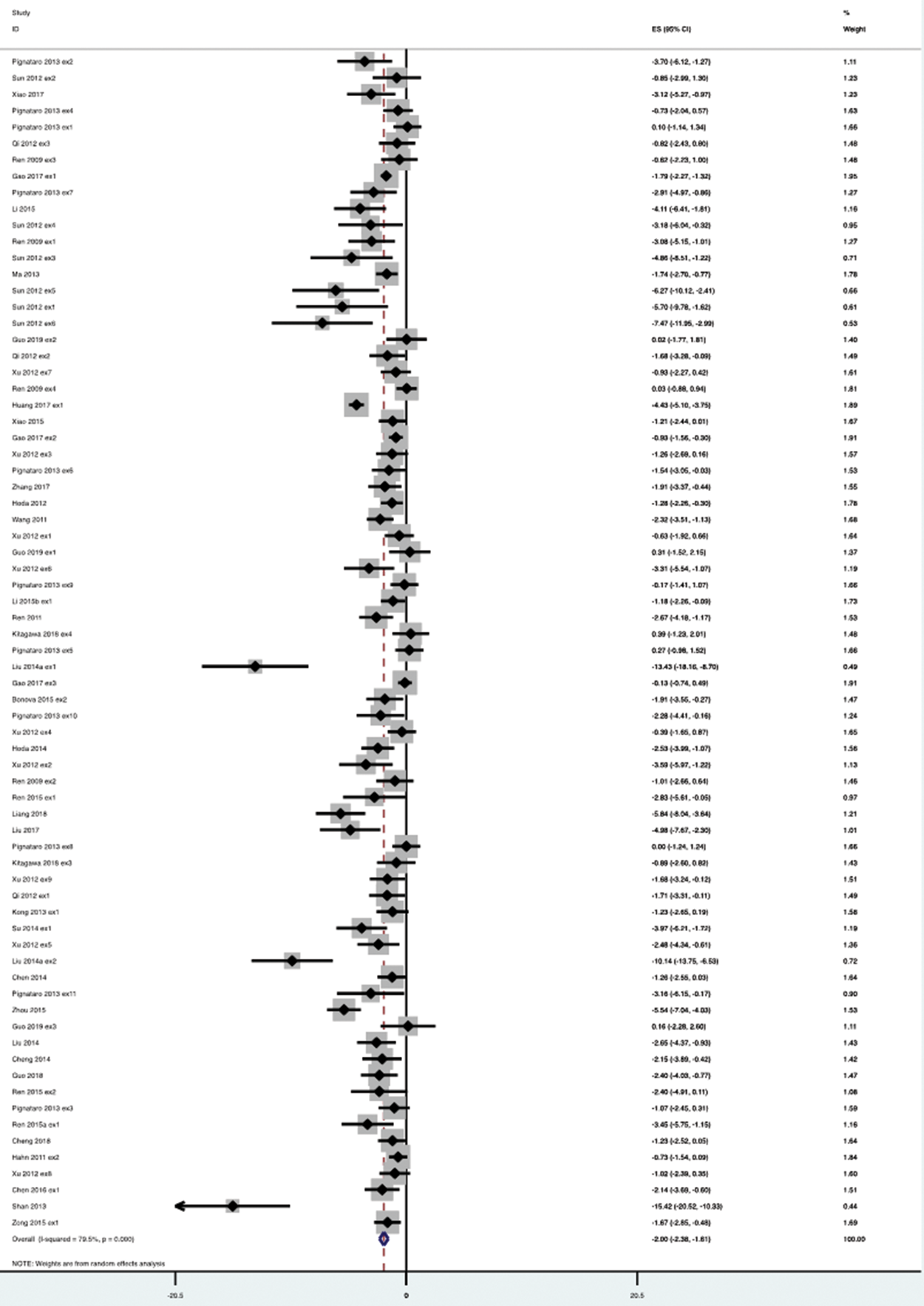
Effect of remote ischemic per- and post-conditioning (RIPerC and RIPostC) compared to control on infarct volume, expressed as a standardised mean difference, by individual publication experiment.
Pre-conditioning (RIPreC)
We assessed protocol variables against infarct volume change in the RIPreC studies using meta-regression. There was a significant interaction caused by species, with RIC effective in rats but not mice (p = 0.01, Figure 3(a)). RIC cycle length (up to 15 min) and total length of limb occlusion (≥30 min and up to 45 min) significantly reduced infarct volume, with longer periods of cycle length and total limb ischaemia leading to greater falls in infarct volume (although there was no significant interaction with meta-regression p = 0.115 and 0.102, respectively, Figure 3(a)). Using either one or two limbs to administer RIPreC reduced infarct volume, but using one limb was not significantly better than two limbs (SMD −2.00 [−2.76, −1.24] vs. −0.72 [−1.29, −0.16], p = 0.134).
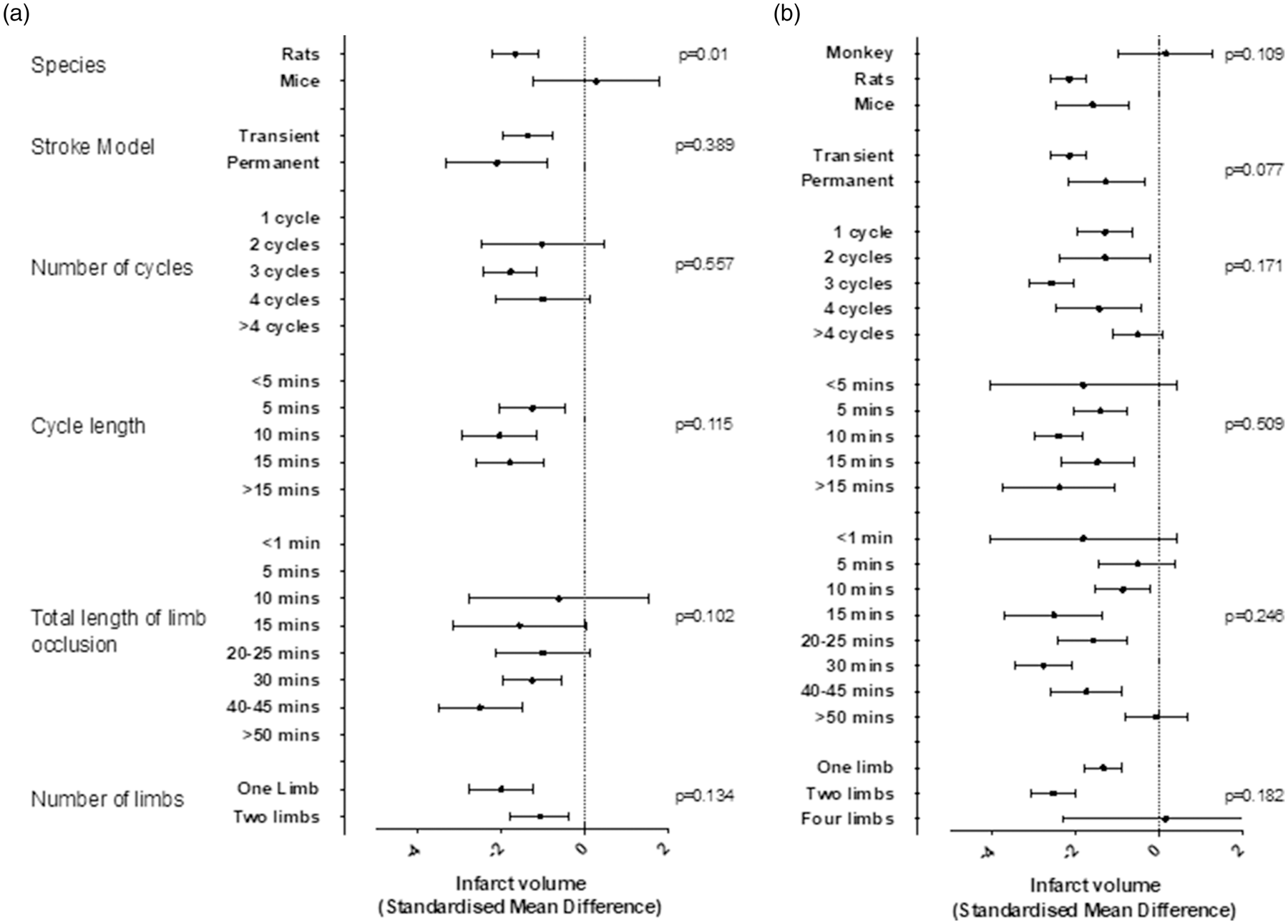
Subgroup analyses of RIC in experimental stroke. Each point estimate represents the change in infarct volume in treated animals compared to control, divided by subgroups according to animal model and RIC administration (pre-conditioning, RIPreC; per- and post-conditioning, RIPerC and RIPostC). The p values, obtained through meta-regression analyses, indicate whether the respective parameter has a significant interaction with infarct volume.
Per- and post-conditioning (RIPerC and RIPostC)
We assessed protocol variables against infarct volume change in the RIPerC and RIPostC studies using meta-regression (Figure 3(b)). Infarct volume was significantly decreased in all species except Rhesus monkeys, reduced in transient and permanent models, with one, two, three or four cycles of RIC, total length of limb occlusion was effective ≥10 min but not at 50 min and using one or two limbs. There was no interaction with species, model type, cycle number or length and total length of limb occlusion. Using two limbs might be more effective than one (SMD −2.53 [−3.07, −1.99] vs. −1.33 [−1.78, −0.89]) but the use of four limbs was worse than both meaning there was no significant interaction with limb number (p = 0.182).
Study quality and risk of bias
Quality of study median score was 7 (range 4–9, Supplementary Tables I and II). The study quality score did not impact on infarct volume estimate (meta-regression p = 0.495, Figure 4(a)). Median risk of bias score was 7 and ranged from 5 to 10 (Supplementary Tables I and III), the score also did not influence infarct volume (p = 0.672). The cohort of studies was of reasonable quality with 68% of publications giving statements on randomisation and blinded assessment of outcome, but only 6% using a sample size calculation and 57% provided a statement on conflicts of interest. Further, statements of physiological monitoring during anaesthesia were limited: blood pressure 25%, blood gas 19%, blood glucose 5% and regional CBF 40%. Monitoring of blood glucose and CBF interacted significantly with the infarct volume outcomes (p = 0.047 and p = 0.032, respectively), in that those studies which monitored glucose and CBF demonstrated greater reductions in infarct volume (Figure 4(b)).
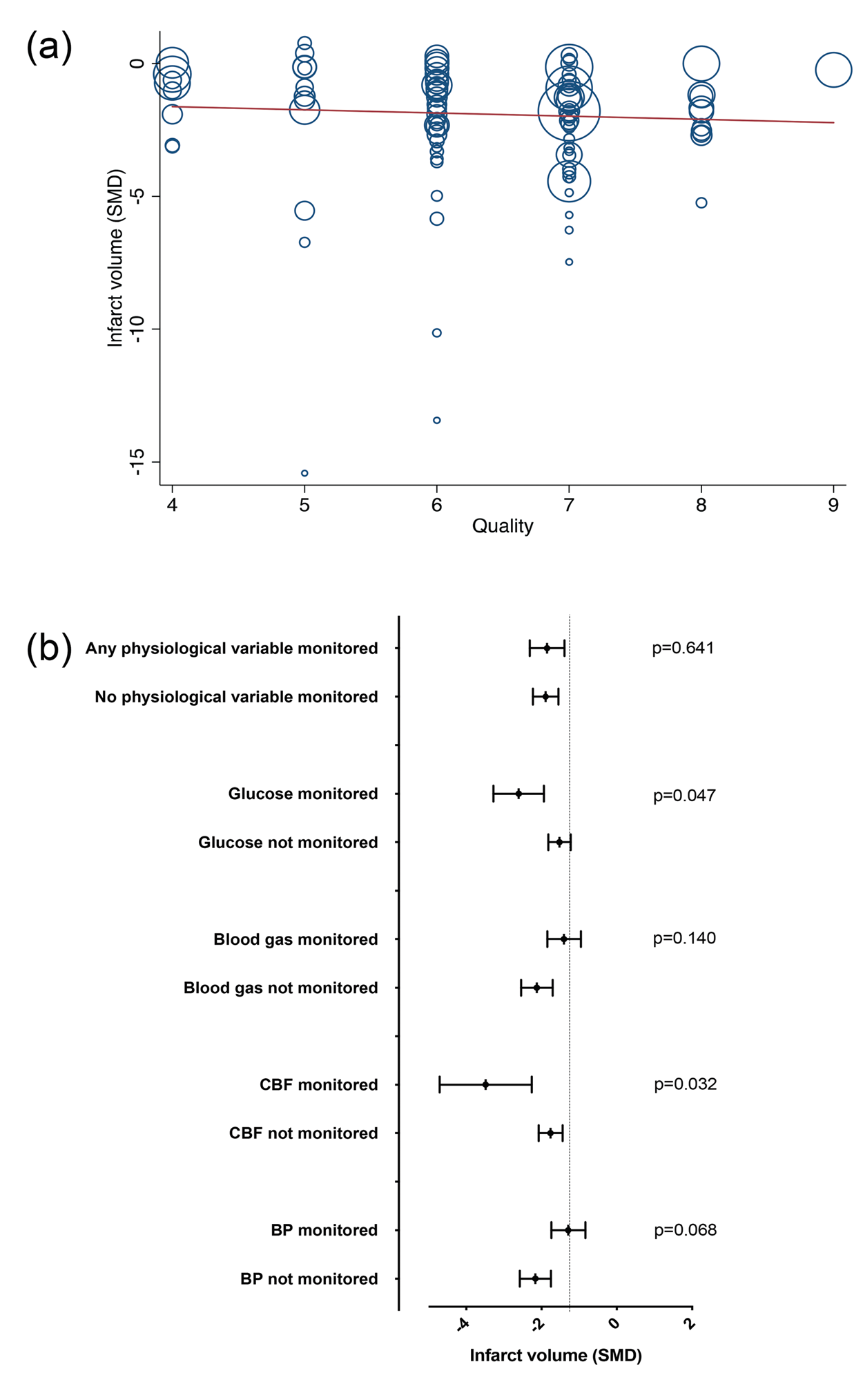
Impact of study quality on infarct volume by (a) CAMARADES criteria: each point represents one study, the size of the circle is proportional to the study size. The y-axis is infarct volume change expressed as the standardised mean difference (SMD) between RIC-treated animals and control. The was no statistical interaction with study quality and infarct volume (meta-regression p = 0.495) and (b) measurement of physiological monitoring (blood glucose, blood gas, cerebral blood flow (CBF) and blood pressure (BP)). The p values indicate whether the respective parameter has a significant interaction with infarct volume.
In sensitivity analyses (dose–response by studies with statements of physiological monitoring, n = 57 of 99 experiments), an effective dose range remained between 15 and 45 min of total limb occlusion time using two, three or four cycles of RIC (Supplementary Figure II). Analysing for further sources of statistical heterogeneity determined that there was no interaction by laboratory, but there was by country, with greatest efficacy seen in studies from China (Supplementary Figure III). Begg’s funnel plot (Figure 5) indicates an asymmetry in published studies, i.e. the possibility of missing data due to a publication bias (Egger’s statistic p < 0.001).
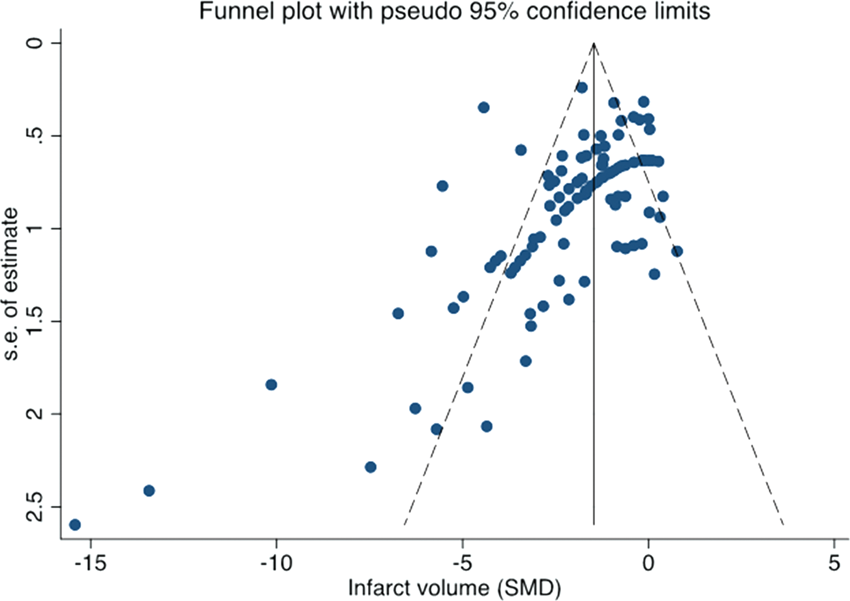
Begg’s funnel plot. An asymmetric funnel indicates a relationship between treatment effect estimate and study precision. Egger’s test suggested significant publication bias (p < 0.001).
Discussion
This comprehensive systematic review and meta-analysis of 1521 animals has confirmed the potent effect of RIC in improving infarct volume and neurological outcome in pre-clinical stroke models when applied before the insult (RIPreC) or during and after the stroke (RIPerC and RIPostC). In all studies, the average reduction in infarct volume in RIC groups compared to control was 34% and appeared to be efficacious in RIPerC/RIPostC studies more than RIPreC, in both rats and mice, in transient and permanent ischaemia, using one or two limbs and using a total length of limb ischaemia ≥10 min. Total length of limb ischaemia for greater than 50 min was ineffective with an optimal period between 15 and 45 min.
Both RIPreC and RIPerC/RIPostC groups demonstrated significant statistical heterogeneity and our pre-specified subgroup analyses in both experimental paradigms helped to explore the reasons for this. In RIPreC experiments, there was a significant interaction with species, suggesting that RIPreC was ineffective in mice; RIPerC/RIPostC was equally effective in rats and mice, however. This raises concern of treatment failure when moving into human clinical trials due to inter-species differences. There was only one study assessing larger gyrencephalic species, a recent study assessing the effects of RIPerC on stroke-related cardiac dysfunction in rhesus monkeys (five control, nine RIC) 22 ; multi-limb RIPC improved motor neurological scores (in addition to reducing cardiac enzymes, von-Willebrand factor and C-reactive protein) without affecting cerebral infarct volume, suggesting improvements might be mediated through improving endothelial injury and anti-inflammatory mechanisms. This study is confounded by a small sample size and the use of Propofol during anaesthesia, which interferes with RIC efficacy and a factor that may have contributed to the neutral findings in prior cardiac bypass surgery RIC trials. 7 Clinical trials assessing RIC in mechanical thrombectomy in hyperacute stroke (often using general anaesthesia) need to factor this into their design. Other trials of RIC in cerebrovascular disease are underway and small trials have been completed. 30 Interpreting results will, however, be challenging since they are fraught with heterogeneity in terms of RIC protocols and stroke subtype assessed.
RIPreC studies showed no significant interaction with the total length of limb occlusion (a product of cycle number and length of each cycle, reflecting the ‘dose’ of RIC), though only doses greater than 25 min reduced infarct volume significantly. Doses above 45 min were not tested in the RIPreC group, but were ineffective in the RIPerC/RIPostC group suggesting the presence of a therapeutic window. A higher dose may also be reflected by the number of limbs used to administer RIC, but here we obtained no statistical interaction with the number of limbs used in either of the subgroups. We did not find any differences in the number of cycles used or in the length per cycle, though analysis of all studies revealed the therapeutic window of total dose to be between 15 and 45 min. Whether repeated dosing provides additional benefit remains largely untested except in two studies, where RIPostC for up to 14 days was more effective at improving outcome than a single per-conditioning dose.31,32 Interestingly, delayed daily RIPostC, started at day 5, using three cycles of 10 min of limb ischaemia, improved neurological function and brain injury (without impacting on early lesion volume) through pleiotropic effects such as angioneurogenesis and modulation of the inflammatory response. 32
The mechanisms of RIC are still under exploration, appear to be multi-modal and not fully dependent on achieving reperfusion, which is important since only 50% of strokes achieve early recanalization after iv thrombolysis. 33 Beneficial mechanisms, in addition to attenuation of reperfusion injury, include anti-inflammatory, 34 anti-oedema,35,36 angioneurogenic,31,32,37 anti-platelet 38 and vasodilatory (enhancement of collateral microvascular circulation) effects, 39 mediated through numerous neurohumoral chemical messengers, 8 including release from endothelial derived exosomes. 40 This meta-analysis confirms that RIC reduces infarct volume in both transient ischaemic models (standardized mean difference, SMD 1.93, p < 0.0001) and permanent models (SMD 1.59, p < 0.001), suggesting that reperfusion is not necessary, though desirable, for RIC to achieve beneficial effects.
The majority of papers is this review utilised young male rodents with a notable absence of animals with co-morbidities such as age, hypertension and diabetes, factors which may inhibit the effects of RIC. 41 Of some concern is the absence of effect seen in an aged model of right MCAo occlusion treated with ‘direct’ ischaemic conditioning (not remote). 42 Studies are present testing RIC efficacy in female rodents (n = 46)40,43 which is important to examine considering the neuroprotective effects and potential interaction of female hormones. 44 In post hoc analyses, RIC studies in female rodents reduce infarct volume to a similar extent to that seen in all studies (SMD −1.76 95% CI −3.07 to −0.45, p = 0.009, excluding Xiao 2017 which contains both male and female rats). Other experimental paradigms important for translation into human trials have also been tested including co-administration of thrombolysis (a synergistic effect), use in large animals, 22 experiments specifically designed to address dose response 45 and the time window of administration. 32
The risk of bias in our findings exists considering the presence of significant statistical heterogeneity. This does not appear to be explained by differences in study quality (CAMARADES criteria) or risk of bias (SYRCLE criteria). Indeed, reporting of randomisation and blinding of outcome assessments were moderately high (68%) but, disappointingly, the use of sample size calculations (6%) is lacking despite calls to include these in animal study design. 46 Sources of heterogeneity were significant for the presence/absence of physiological monitoring of CBF and glucose, and also the country in which the experiment was performed (but not the laboratory). Somewhat reassuringly, in sensitivity analyses, an effective RIC dose range between 15 and 45 min remained. The presence of significant publication bias also raises concern, theoretically leading to an under- or over-estimation of effects due to unpublished neutral or negative data. It is also feasible that we missed publications in our literature search but this was comprehensively performed independently by two authors. Overall, however, this is a robust and comprehensive review of the current literature strengthened by pre-registration and pre-specified analyses.
In summary, RIC significantly reduces lesion volume and neurological impairment in experimental models of focal ischaemic stroke. Statistical heterogeneity may be explained by RIPreC cycle length, dose and number of limbs; monitoring CBF and glucose during anaesthesia and country in which the experiment was conducted. Dose analyses suggest a therapeutic window of between 10 and 45 min in RIPerC and RIPostC models. The presence of publication bias raises the possibility that neutral/negative studies have been performed but not published. Pre-clinical studies in animals with co-morbidities using protocols with repeated dosing that would be deemed feasible in humans are warranted.
Supplemental Material
sj-pdf-1-jcb-10.1177_0271678X20924077 - Supplemental material for A meta-analysis of remote ischaemic conditioning in experimental stroke
Supplemental material, sj-pdf-1-jcb-10.1177_0271678X20924077 for A meta-analysis of remote ischaemic conditioning in experimental stroke by Philippa Weir, Ryan Maguire, Saoirse E O’Sullivan and Timothy J England in Journal of Cerebral Blood Flow & Metabolism
Footnotes
Data sharing
Data will be shared on reasonable request through direct contact with the corresponding author.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Dr England is the Chief Investigator of the Remote Ischaemic Conditioning After Stroke Trial 3 (RECAST-3) funded by the National Institute of Health Research Efficacy and Mechanism Evaluation programme (Grant number NIHR128240).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
