Abstract
Contrast-enhanced near-infrared spectroscopy (NIRS) with indocyanine green (ICG) can be a valid non-invasive, continuous, bedside neuromonitoring tool. However, its usage in moderate and severe traumatic brain injury (TBI) patients can be unprecise due to their clinical status. This review is targeted at researchers and clinicians involved in the development and application of contrast-enhanced NIRS for the care of TBI patients and can be used to design future studies. This review describes the methods developed to monitor the brain perfusion and the blood–brain barrier integrity using the changes of diffuse reflectance during the ICG passage and the results on studies in animals and humans. The limitations in accuracy of these methods when applied on TBI patients and the proposed solutions to overcome them are discussed. Finally, the analysis of relative parameters is proposed as a valid alternative over absolute values to address some current clinical needs in brain trauma care. In conclusion, care should be taken in the translation of the optical signal into absolute physiological parameters of TBI patients, as their clinical status must be taken into consideration. Discussion on where and how future studies should be directed to effectively incorporate contrast-enhanced NIRS into brain trauma care is given.
Introduction
Near-infrared spectroscopy (NIRS) represents a valuable complementary non-invasive monitoring tool for use in the context of traumatic brain injury (TBI).1,2 However, to date, measurements on these patients in this context with commercially available NIRS devices have not shown parity with the invasive techniques regarding their ability to detect episodes of ischemia.3–5 Changes in optical signal due to the passage of a contrast dye, such as indocyanine green (ICG), are potentially more readily detected than changes due to endogenous chromophores alone (e.g. hemoglobin). 6
The use of contrast-enhanced NIRS optodes applied to the scalp has definite advantages over other contrast-enhanced imaging techniques (e.g. computerized tomography (CT), magnetic resonance imaging (MRI), positive emission tomography) by potentially allowing continuous, repeatable bedside-monitoring as well economical, safety, logistical and radiation exposure benefits.7–10 Despite these advantages, studies on TBI patients using contrast-enhanced NIRS have been limited and the utility of this technique in brain trauma care is still subject to investigation. This review aims to analyze the methods adopted so far to assess cerebral blood flow (
The dye kinetic in dynamic-contrast enhanced images can be divided into two phases: the first phase can be used for the evaluation of brain perfusion, as expressed by the Stewart-Hamilton equation, because the dye stays predominantly in the vascular space; subsequently, in the following 2–10 min, the passage of the dye into the extravascular space can be used for the assessment of vascular permeability.11,12 It should be noted that these two phases are not rigidly separated, but they overlap at different time rates. 13
Cerebral blood flow
Since the first description of the technique was made by Colacino et al.,
14
multiple methods have been introduced to assess
Central volume principle
The convolution formula for the central volume principle allows measurement of 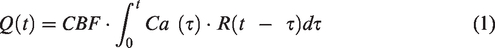
If a non-diffusible contrast dye is injected into the arteries so that all of it reaches the tissue instantaneously (i.e.
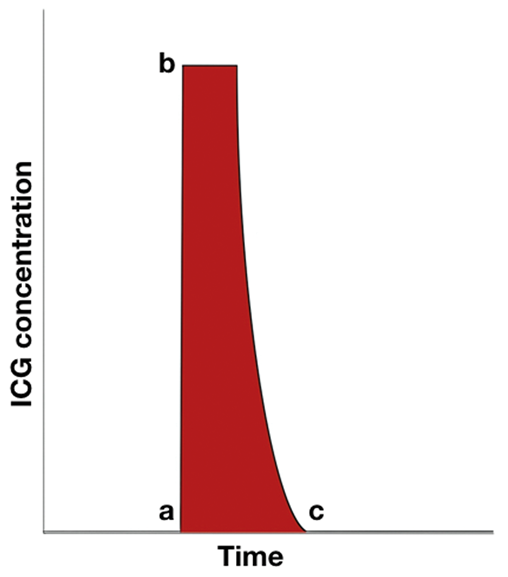
Graph of the ICG concentration in the tissue (
If instead the contrast agent is injected into the tissue from the peripheral venous system, the measurement of 
In different studies,
Diop et al.
22
enhanced this method and calculated the absolute values of
Modified Fick principle
The Fick principle states that the flow passing through a system in a steady state can be measured from the tracer concentration’s inlet and outlet and its uptake from the system
24

This principle can be used to estimate the 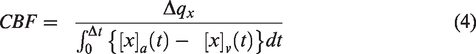
Edwards et al.
25
measured a sudden increase of oxyhemoglobin concentration in the brain to analyze the 
Similarly, other published studies measured the
Regional cerebral blood volume and regional mean transit time
Keller et al.
29
introduced a method to measure the 
This method was used on an optical signal collected with probes on the scalp and inside the skull.30–32
Regional cerebral blood volume
Wolf et al.
33
separated the signal from the arteries and veins based on the fluctuation of the optical density due to the cardiac and respiratory rates respectively. To measure the ICG concentration in the blood (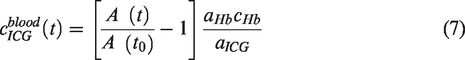
Based on the modified Beer–Lambert law, the ICG concentration in the tissue 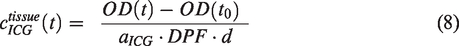
The optical properties of ICG upon injection are unstable due to the inhomogeneous distribution of the dye in the blood, which affects the 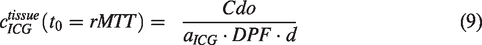
Finally, the 
Since the venous compartment does not show a pulsation related to the heart rate, it is not comprised within
Regional mean transit time
Based on the Fick principle, Keller et al.
29
calculated the difference between the inflow and outflow rates of ICG by measuring the ICG concentration inside the VOI
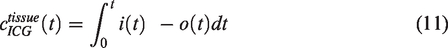
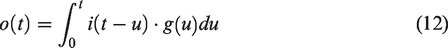
The transport function is the function of particles’ transit times from the beginning to the end of the VOI.
34
Blood flow index
Based on a model introduced by Perbeck et al., Kuebler et al. evaluated the blood flow index (BFI) in a study on piglets.35,36 The BFI is the ratio between the peak of ICG concentration in the examined tissue and the time interval between 10% and 90% of this peak (Figure 2). The value obtained is an arbitrary value linked to the CBF with an unknown coefficient. 37
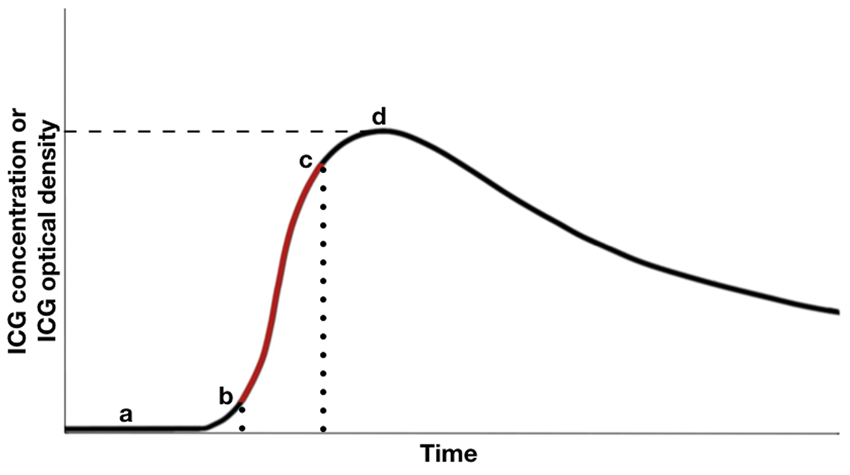
Graph of measurements of the BFI derived from the changes of ICG optical density or ICG brain tissue concentration during dye passage. After the venous injection (point “a”), there is an increase of ICG signal from the brain until a peak (point “d”). The ratio between this peak and the time for concentration/optical density to vary from 10% to 90% of its maximum value (points “b” and “c”, respectively), highlighted in red, forms the BFI.
This method has been tested in humans and translated into clinical practice on infants and adults.20,37–41 It should be noted that Wagner et al., 37 unlike others, considered the optical density changes without transforming them into ICG concentration.
Different results demonstrated method limitations. In a study on healthy volunteers, the BFI values derived did not correlate with those measured using the method described in section ‘Regional cerebral blood volume and regional mean transit time’. 30 In adults who had suffered an ischemic stroke, the BFI differed significantly between the affected and unaffected hemispheres, but not compared to the controls.38,39 In a study of TBI patients undergoing decompressive craniectomy, a surgical microscope saw no abnormal BFI via ICG fluorescence on the visible brain parenchyma. 42 In similar patients, Rothoerl et al. 43 found no correlation between the values obtained using the BFI and a radioisotopes technique. However, in the aforementioned study, there were two elements that differed from the methodology introduced by Kuebler et al. Firstly, the optical data were not converted into ICG concentration but into an oxygen saturation index instead. Secondly, the time interval considered was from the point of contrast dye injection to the peak of tissue concentration.
Distribution of time of flight of photons
TD-NIRS allows one to measure the distribution of time of flight of photons (DTOF).
Liebert et al.
44
separated the absorption changes in the intracranial tissue (ICT) and extracranial tissue (ECT) during the ICG passage using three statistical moments of DTOF: total number of detected photons, mean time of flight of photons and variance. This analysis is an estimation of
It should be mentioned that a comparison between ICG inflow into the ICT and into the ECT can also be performed on the distribution of times of arrival of fluorescence photons (DTA).50–52 The analysis on DTA serves to more accurately assess cerebral perfusion than that on DTOF.51,53
BBB damage
As is true for other contrast dyes (e.g. Evans blue), the ICG passage across the BBB could be ascribed to the multiple mechanisms that compound BBB damage. 54
The ICG passage into the extravascular space has been measured through the dye absorption or fluorescence properties. On animals, a comparison of the optical signal against CT perfusion scans and histological samples showed that the ICG retention in the interstitial brain tissue is linked to BBB damage.46,55–58 The high ICG tissue concentration is limited to the brain areas affected by the BBB damage rather than the whole brain.46,57 Ergin et al. 59 showed that certain drugs leak into the interstitial tissue as well as dye, which suggests potential clinical application of contrast-enhanced NIRS by means of optimizing end-organ dosage.
Similarly to these animal models, Liebert et al. 60 described a prolonged optical signal from the ICG in patients with identified BBB damage, due to a slower washing out of the dye from the brain tissue. These results are in agreement with Kamp et al., who measured the ICG retention in the parenchyma of severe TBI patients with acute subdural hematoma using a surgical microscopic during a decompressive craniectomy. 42 They reported a higher retention of the dye in the patients who went on to achieve less favorable outcomes.
Limitations
There are multiple limitations in the translation of the ICG optical signals into physiological parameters. These limits are further amplified in clinical scenarios where patients’ statuses can influence the values obtained with unpredictable variables.
Central volume principle's assumptions
The analysis of flow using the central volume principle can lead to an overestimation in the case of BBB damage. 17 This is because, in the case of a diffusible tracer, the concentration inside the blood volume must be considered as in equilibrium with the volume in which it diffuses. 15 In TBI patients, the presence of BBB damage can change the diffusive properties of the dye and so affect the validity of the central volume principle. These changes would depend on the extent of the areas affected by the BBB damage, and they would not be consistent between patients.
Fick principle's assumptions
The Fick principle’s assumptions are not completely fulfilled and accounted for in the modified Fick principle. The basic principle assumes a steady state, wherein a constant rate of indicator inflow and outflow exists.
24
However, the level of arterial ICG inflow changes over time (
Similarly to the ICG arterial inflow,
Indocyanine green as an indicator of plasma volume
The blood volume mainly comprises red blood cell volume and plasma volume. For a correct analysis of the blood volume, both these components have to be measured.61,62 Due to the Fåhraeus effect, the ratio between the two is different depending on the size of the vessels. 63 The brain volume illuminated would encompass vases of different sizes and consequently with different ratios between the cellular and plasma volumes. Since the ICG remains confined within the plasma, its optical signal is linked to the plasma volume only, and is proportional to the fraction of vessels of different sizes within the illuminated tissue.
Based on an experiment on healthy volunteers using single-photon emission computed tomography, Keller et al. assigned a cerebral-to-large-vessel hematocrit ratio to the plasma volume in the tissue illuminated.29,64 However, a meta-analysis showed that different disease states can result in large variations in the amount of hematocrit in the microcirculation, which makes the cerebral-to-large-vessel hematocrit ratio different in these patients. 62 Similarly, in trauma patients, changes in either the macro- or microcirculation can result in different cerebral-to-large-vessel hematocrit ratios than those obtained from analyses on non-trauma patients.65–67
Extracranial tissue interference
Analysis of the diffuse reflectance on the scalp can lead to an underestimation of the true
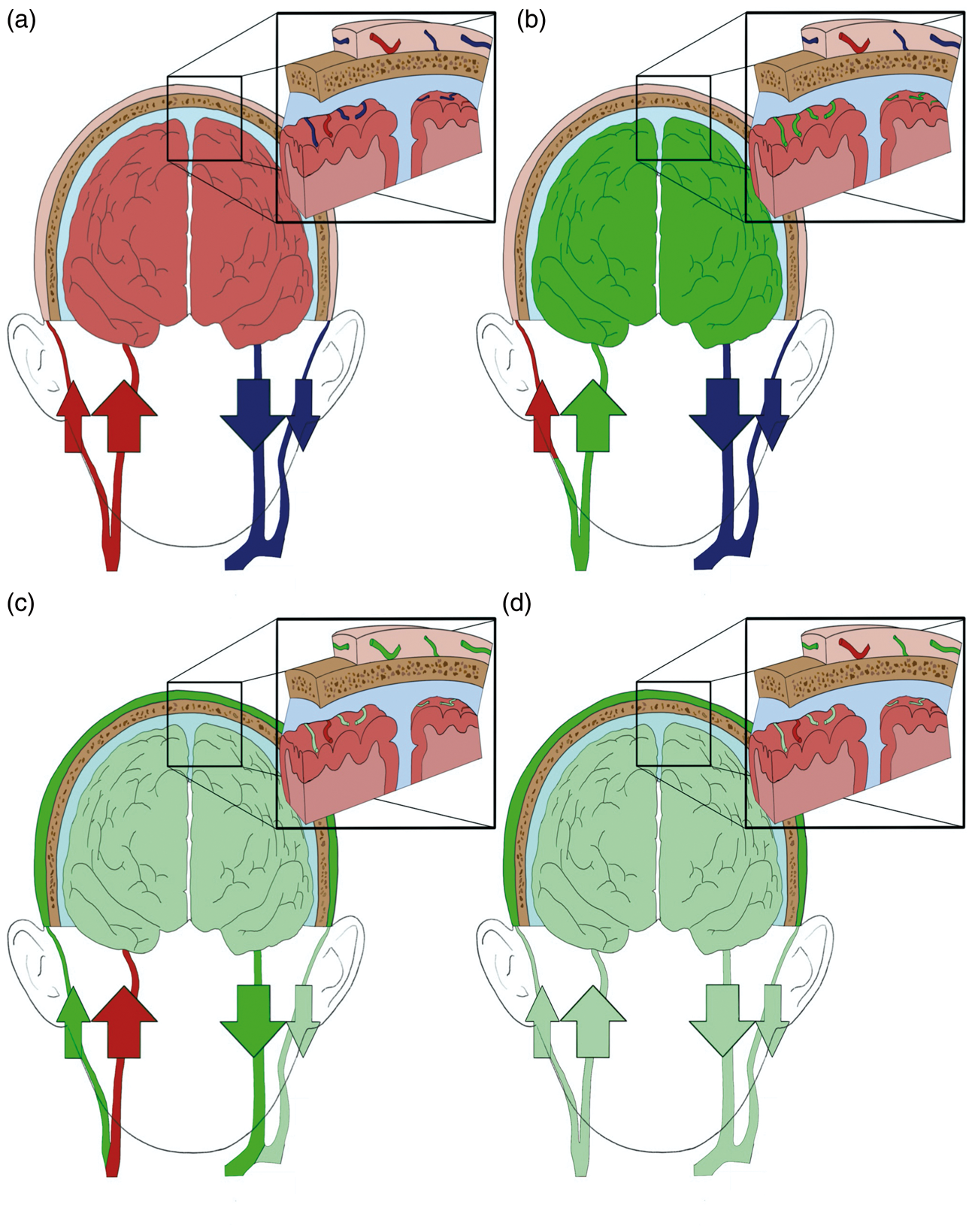
Representation of the ICG kinetic in the ICT and the ECT without a brain lesion. (a) Standard physiological state. (b) First ICG bolus passage. The ICG reaches the vessels in the ICT before the ECT due to the different perfusion levels between the two layers. (c) First ICG bolus passage. The ICG is being washed out from the ICT, while its concentration in the ECT is increasing due to the slower blood transit time there than in the ICT. (d) ICG kinetic a few minutes post-injection. The ICG blood concentration is reduced due to dilution and clearance; the ICG remains in the venous system of the microcirculation of the two layers with a higher concentration in the ECT than in the ICT.
Indocyanine green’s kinetics in the intracranial and extracranial tissue
The ICG kinetic within the two layers is related to their hemodynamic and vascular permeability. As described in section ‘Introduction’, the relevance of these components depends on the time from the injection.
First indocyanine green’s passage
Firstly, there is a single bolus ICG inflow into the ICT and then into the ECT, due to a different perfusion velocity in the two layers (Figure 3(b)).30,40
Secondly, once in the tissue, the ICG passage is quicker in the ICT than in the ECT due to the different mean transit times (Figure 3(c)).31,32
Due to the kinetics of the first passage, the optical density curve generated by the ICG is initially represented predominantly by a signal coming from the ICT alone, and subsequently, by a combination of the ICT and ECT, with an increasing component from the latter as the ICG leaves the ICT while still passing through the ECT. 46
The ICG kinetic between the two layers and the optical curve obtained are generally in agreement with the results observed from the gadolinium’s passage in an MRI reported in the literature. 41 A comparison between the passage of the bolus of gadolinium in the ICT and ECT showed a peak delayed by a few seconds, as well as a slower recovery to baseline in the former than in the latter. 41
As explained in section ‘Distribution of time of flight of photons’, abnormal cerebral perfusion can delay the passage of the bolus into the brain and eliminate the difference between the time of inflow into the ICT and ECT (Figure 4(b) and (c)). This different ICG kinetic would change the shape of the optical curve retrieved. 7
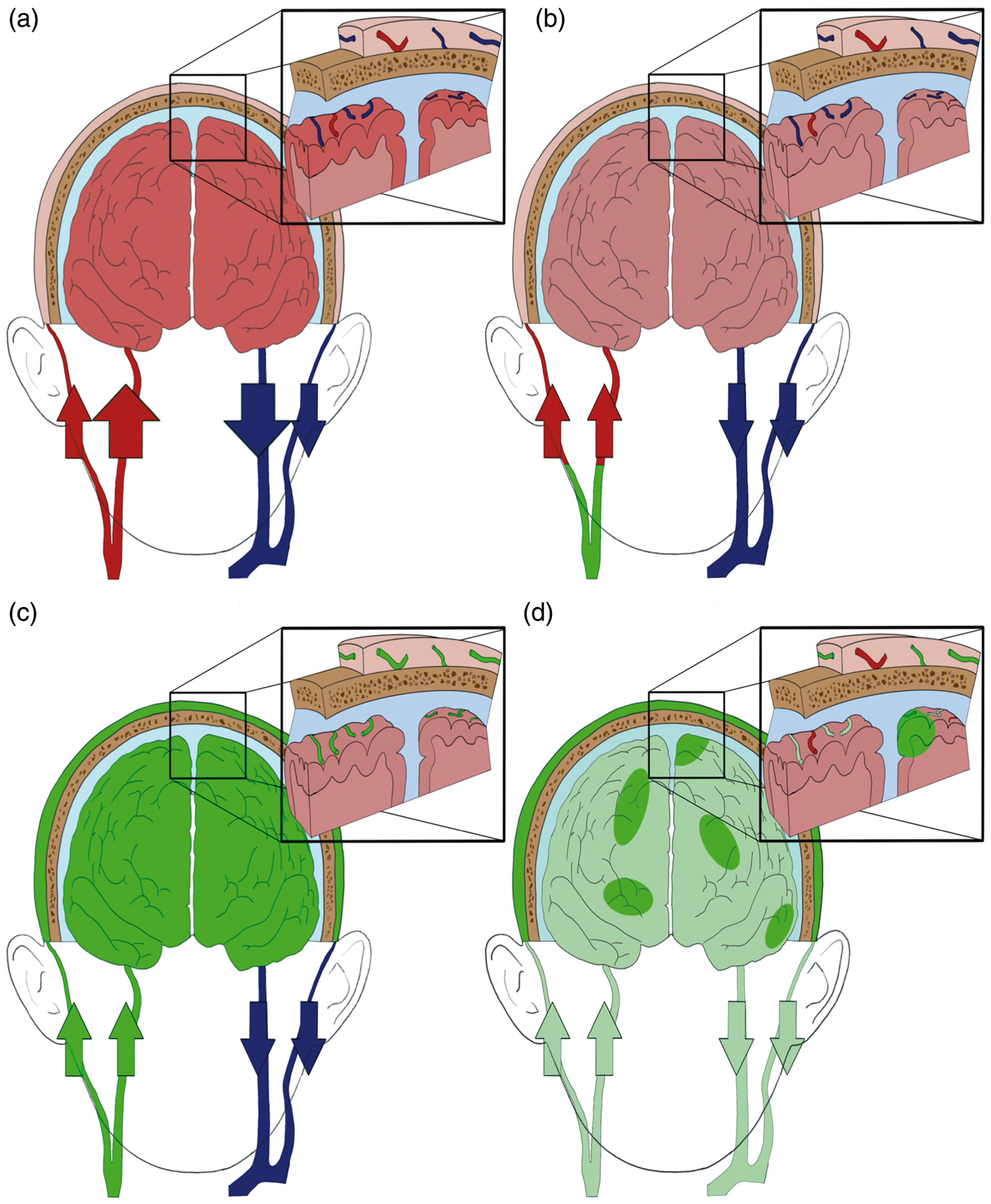
Representation of the ICG kinetic in the ICT and the ECT with a brain lesion. (a) Standard physiological state. (b) First ICG bolus passage. The ICG does not reach the ICT before the ECT due to a reduction of brain perfusion. (c) First ICG bolus passage. The ICG reaches the ICT and the ECT at approximately the same time. (d) ICG kinetic a few minutes post-injection. The ICG blood concentration is reduced due to the dye
Indocyanine green’s washing out and retention
After the peak of bolus passage, the optical signal from the ICG does not return immediately to the baseline. This suggests that the ICG is partially retained in the vascular space and is being slowly washed out (Figure 3(d)). This washing-out process can be delayed if the ICG leaks into the interstitial tissue potentially due to loss of BBB integrity (Figure 4(d)).
Washing out from the intravascular space
The ECT slower perfusion compared to that of the ICT results in a slower ICG washing out from the ECT than from the ICT (Figure 3(d)). The ICG is more likely to remain in the venous system (e.g. venules) where the blood pressure and transit velocity are lower (Figure 3(d)). 69 Due to the different washing-out rates, the percentage of optical signal coming from the ECT, compared to that from the ICT, increases over time. 46
Retention in the interstitial space
In case of BBB damage, the dye firstly inflows into the interstitial tissue and subsequently leaves once the concentration inside the blood is lower than that in the interstitial tissue. 13 Consequently, the ICG optical signals from injured brain areas are higher than the surrounding areas without BBB damage until the ICG has washed out from the interstitial tissue (Figure 4(d)). It should be mentioned that in the case of inflamed ECT, the ICG also outflows into its interstitial space.70,71
Quantification of the extracranial tissue interference
Monte Carlo simulations have been used on healthy volunteers’ MRIs at different physiological perfusion levels to simulate the diffuse reflectance obtained from the changes in optical density within ICT and ECT due to the ICG passage.72,73 The percentage of optical signal from the brain during the first ICG bolus passage was estimated to be approximately 70%, mainly due to the higher perfusion and blood volume in the ICT than in the ECT. The optical signal coming from the brain during the ICG passage was significantly higher than that estimated before injection, which led to the conclusion that the passage of dye enhances the brain signal. 7
However, the ratio assigned in the Monte Carlo simulations mentioned above is not representative of the entire ICG passage, because the fraction of ICG in the ECT progressively increases over the ICT (Figure 3(b) and (c)). Furthermore, this increase has a more profound effect on the percentage of diffuse reflectance from the brain than the simple proportional effect of different ICG ratios between the layers. This is because the optical properties in the ECT change the depth of the photon pathway and thus the volume of illuminated brain tissue. 74
Finally, brain-injury patients may have different perfusion levels and blood volumes than the physiological parameters considered by the Monte Carlo simulations. Consequently, the ICG ratio between the two layers would be different from the one assigned (Figure 4(b) and (c)).
Techniques to remove the extracranial tissue interference
The ECT contribution to the optical signal differs according to the SD distance of the probe and the type of NIRS method.
Single-channel continuous wave
A single-channel CW-NIRS has been used to measure the
Single-channel frequency-domain
Steinbrink et al.
40
used one source and one detector of an FD-NIRS device (e.g. ISS OxiplexTS) on four patients during cardiopulmonary bypass surgery to measure the ICG concentration change in the ICT and ECT. The analysis of the mean time of flight of photons allowed for the separation of the ICG concentration in each layer.40,77 While the FD-NIRS device monitored optical changes at different levels of
Multiple source-detector distances
Different studies have shown that multiple optodes at increasing SD distances can be a valuable tool to separate the optical changes in the ICT and ECT during the ICG passage.21,41,78 This is because the longer channels are more affected by the ICG passage through the brain than the shorter channels, which are mainly confined in the ECT. 79 The difference in intensity between channels allows for regression of the ECT signal from the brain signal. 80 Alternatively, plotting the decrease in intensity against the SD distances makes it possible to measure the optical properties of each layer. 81 This can be done using a CW- or a FD-NIRS. 82
The use of the FD-NIRS facilitates measurement of the phase-shift according to the SD distance as well as the intensity levels.83–87 Therefore, the ICG passage through the layers measured with a FD-NIRS can also be divided using the model described in section ‘Single-channel frequency-domain’ adapted to a multidistance approach system. 41
A comparison between multidistance depth-resolved NIRS and an intracranial probe showed no correlation between the results obtained, which led to the conclusion that this method has certain limitations in the brain signal measurement, possibly due to effects from the ECT. 32
Time-domain
The combined thickness of the ECT and the skull is not consistent between individuals and this can be further pronounced after surgical interventions (e.g. decompressive craniectomy).7,48 Consequently, the depth of the photon pathway differs between individuals.
One of the advantages of using the method described in section ‘Distribution of time of flight of photons’ with TD-NIRS instruments is that the depth of the photon pathway does not have to be assumed as it must be with CW- and FD-NIRS.21,44 This is because the latest photons have traveled via the deepest layers, which shape the characteristics of the DTOF.45,46,50
High-resolution diffuse optical tomography
Habermehl et al. 88 plotted the optical signal obtained from a CW-NIRS high-resolution diffuse optical tomography on an MRI mash to record the ICG passage on four healthy volunteers. The resultant brain 3D-reconstruction facilitated the separation of the ICG passage in the two layers.
It should be noted that by incorporating optical data into a structural image, the variables related to the ECT thickness can be measured, and the depth of the photon pathway estimated.
Indocyanine green’s clearance
Abnormalities in the liver perfusion or liver function can affect the rate of the ICG clearance from the blood. 89 Since liver injuries are relatively common in trauma patients, the rate and the amount of ICG leakage into the interstitial tissue in cases of BBB damage can be confounded by different rates of ICG clearance across TBI patients, or in the same patient at different time-points, independent of the status of the BBB.
Aims for future studies
The assessment of the
Appropriate clinical decisions are not necessarily based on analysis of absolute values. For example, a comparison between the outcomes derived from clinical decisions in brain trauma care based on absolute parameters of intracranial pressure (ICP) monitored through a bolt, and on an ICP assessment using serial CT scans and neurological testing, showed no significant differences.
99
Similarly to the ICP values, the transformation of the optical values obtained from the ICG passage into absolute parameters of
Conclusion
In conclusion, contrast-enhanced NIRS with ICG could be used to analyze the brain perfusion and the BBB in TBI patients. However, the translation of the optical signal acquired into quantitative values must account for the clinical variables present in TBI patients. Different solutions have been used to address these problems, either partially or totally. Further research is needed to implement these methods in clinical practice on trauma patients. Clinical studies with contrast-enhanced NIRS should be designed to meet specific current clinical needs and should consider the possibility of addressing them by measuring relative optical and physiological parameters rather than absolute values, as well as combining the results obtained with other neuromonitoring techniques.
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This article presents independent research funded by the project “Brain Injury and Trauma Monitoring Using Advanced Photonics”, financed by the European Union Horizon 2020 Research and Innovation Program, under grant agreement 675332, through the National Institute for Health Research Surgical Reconstruction and Microbiology Research Centre (NIHR SRMRC), partnership between University Hospitals Birmingham NHS Foundation Trust, the University of Birmingham, and the Royal Centre for Defence Medicine. The views expressed are those of the authors and not necessarily those of the NHS, the NIHR or the Department of Health.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
