Abstract
Animal models make an important contribution to our basic understanding of the pathobiology of human brain aneurysms, are indispensable in testing novel treatment approaches, and are essential for training interventional neuroradiologists and neurosurgeons. Researchers are confronted with a broad diversity of models and techniques in various species. This systematic review aims to summarize and categorize extracranial aneurysm models and their characteristics, discuss advantages and disadvantages, and suggest the best use of each model. We searched the electronical Medline/PubMed database between 1950 and 2020 to identify main models and their refinements and technical modifications for creation of extracranial aneurysms. Each study included was assessed for aneurysm-specific characteristics, technical details of aneurysm creation, and histological findings. Among more than 4000 titles and abstracts screened, 473 studies underwent full-text analysis. From those, 68 different techniques/models in five different species were identified, analyzed in detail, and then grouped into one of the five main groups of experimental models as sidewall, terminal, stump, bifurcation, or complex aneurysm models. This systematic review provides a compact guide for investigators in selecting the most appropriate model from a range of techniques to best suit their experimental goals, practical considerations, and laboratory environment.
Introduction
Brain aneurysms are a cerebrovascular disease in which a weakening of a cerebral artery causes an abnormal focal dilatation. Microsurgical and endovascular treatment aims to eliminate brain aneurysms from cerebral circulation and prevent rupture. Despite rapid advances in the development of endovascular treatment, complete and long-lasting aneurysm occlusion remains a challenge, and the biological mechanisms that predispose brain aneurysms to grow and recanalize are not yet fully understood.1,2 Although strong histological similarity exists between the cerebral arteries of humans and animals, the prevalence of naturally developed cerebral aneurysms in animals is extremely low.3,4 Models with artificial induction of aneurysm formation are therefore needed for preclinical studies of the pathobiology of human brain aneurysms as well as to evaluate and invent novel endovascular devices and medical therapies that prevent rupture and recurrence after endovascular therapy.
Current models can be divided in two main groups: first, intracranial aneurysm models serve to evaluate induction, growth, and rupture of brain aneurysms and, second, extracranial aneurysm models that are mainly designed to test novel endovascular treatment options. Animal models of intracranial aneurysm formation induced by flow manipulation, hypertension, and impaired collagen synthesis developed by Prof. Nobuo Hashimoto et al. 5 are the most physiological models in terms of reproducing human morphology, histology, hemodynamics, and brain aneurysm vessel surroundings. In all other models, including the induction of intracranial aneurysms by intrathecal elastase injection, 6 aneurysms are created by direct vessel manipulation of intra- and extracranial arteries. This review focuses exclusively on extracranial aneurysm models for the study of endovascular therapies in the most often used species, that is, mouse, rat, rabbit, dog, and swine. Extracranial animal models in sheep7 and monkey8,9 have also been described but have never undergone detailed methodological analysis and are seldom used today.
Although the number of studies using animal aneurysm models has steadily increased in recent years, no model has yet been established as the generally accepted standard for preclinical testing.10,11 To the contrary, confronted with a diversity of animal models and techniques, investigators now face increasingly complexity in choosing the appropriate model for any given research question. Specifically, endovascular technology is progressing rapidly, traditional surgically constructed models have been adapted, and novel models and techniques have been designed. This review aims to provide a contemporary detailed overview of available extracranial aneurysm models and discusses advantages and disadvantages of specific species and techniques.
Materials and methods
Search strategy
The literature was reviewed to identify main animal models of experimental saccular aneurysm, their refinements, and technical modifications. We searched the database Medline/PubMed on 10 January 2020 using the key words “mouse,” “rat,” “rabbit,” “dog,” and “swine” in combination with “aneurysm” with the Boolean operator (AND). The search was restricted to “animals.” Two investigators (SM and FS) independently screened titles and abstracts for eligibility based on our predefined criteria, reviewed the full text of eligible studies, and confirmed articles for inclusion. Any disagreement about a particular study’s eligibility was resolved with consensus of the other authors. Additionally, we identified studies cited in previous reviews and added select articles by cross-reference checking until no other publications were found. Figure 1 outlines the applied search algorithm applied and reasons for exclusion in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines. 12
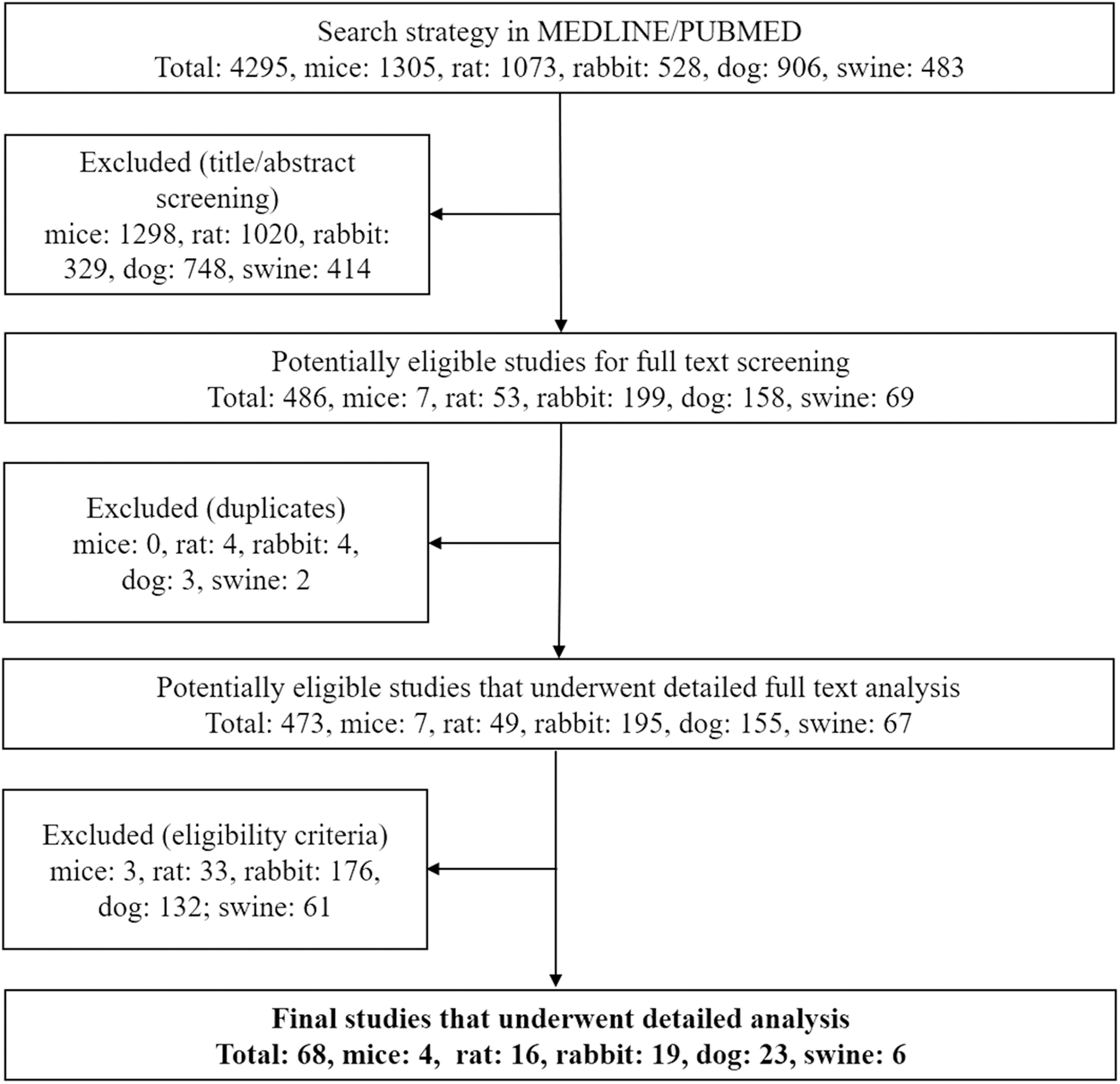
PRISMA flowchart of PubMed search strategy and selection process. Among more than 4000 titles and abstracts screened, 473 studies underwent detailed full-text analysis. From those, 68 models, including refinements and technical modifications, were identified in five different species.
Eligibility criteria and analyzed features
We considered in vivo extracranial aneurysm models in mice, rats, rabbits, dogs, and swine but excluded non-English publications and studies on intracranial aneurysms. In the rare case of a model in which an extracranial vessel was transposed or transplanted into the intracranial space, the model was, despite its anatomical location, assessed as an extracranial aneurysm model. For each included study, we recorded authors; year of publication; detailed technique of aneurysm creation; number of created aneurysms; time for creation; patency rate; histological findings; mortality and morbidity; and shape, size, and location of the aneurysms.
Animal models were defined with consideration to the large differences that occur in the natural course of aneurysms among species. Therefore, we considered the animal model to be unique when identical techniques of aneurysm induction were performed in either different species or a different anatomical location within the same species or with any variation in the aneurysm graft (arterial or venous pouch) or with modification by chemical or mechanical means. Besides differences in species, anatomical location, or graft characteristics, a model was defined as novel only if modifications of an existing technique were major, that is, resulting in significant changes in aneurysm size or shape, patency rate, time for creation, or morbidity and mortality. Any uncertainty about the novelty of a particular study was resolved by two authors based on the predefined criteria listed above.
Results
From 4295 publications related to the main animal models of experimental saccular aneurysm, including refinements and technical modifications, our further screening of titles and abstracts and removal of duplicates identified 473 studies for detailed full-text analysis. Among these, a total of 68 techniques were found that included mice (n = 4), rats (n = 16), rabbits (n = 19), dogs (n = 23), and swine (n = 6). For each technique, details of the animal model, localization, time for creation, size of the aneurysm, patency rate, and morbidity and mortality are outlined in Supplementary Tables. We defined five main groups of aneurysm models as (1) sidewall, (2) terminal, (3) bifurcation stump, (4) natural and artificial bifurcation, and (5) complex aneurysm models (Figure 2).
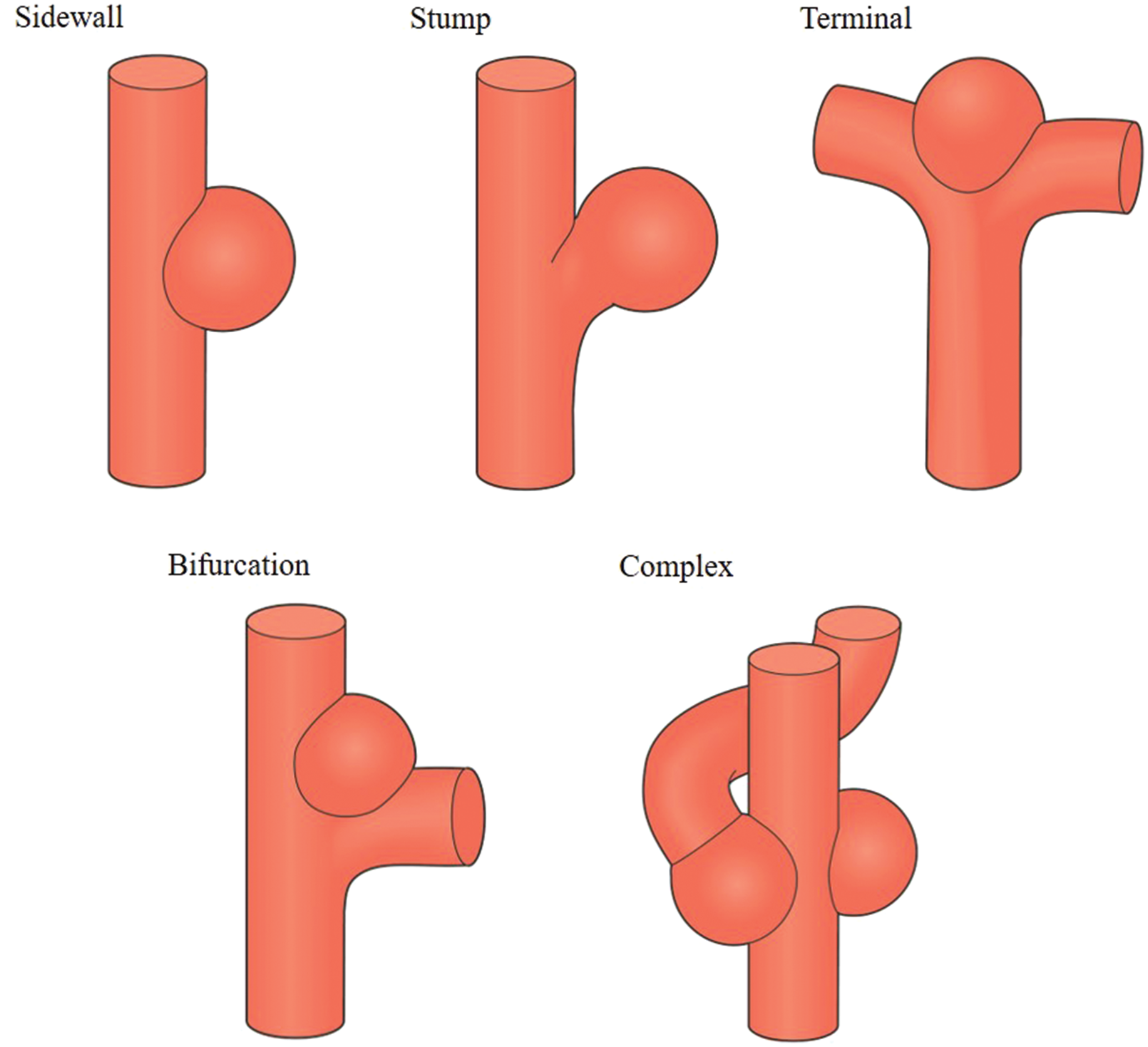
Main groups of extracranial aneurysm models. Preclinical extracranial aneurysm models in different species can be categorized into one of the five main groups. Top form left to right: (1) sidewall aneurysm models, (2) bifurcation stump aneurysm models, and (3) terminal aneurysm models. Down from left to right: (4) natural and artificial bifurcation aneurysm models and (5) complex aneurysm models.
In these aneurysm models, aneurysm walls consisted of either a venous or an arterial vessel segment (autogenic and allogenic grafts). Creation was performed surgically, by inducing vessel wall weakening (chemical or physical), or a combination of both. Methods to weaken the vessel walls consisted of treatment with elastase, papain, collagenase, CaCl2, or sodium dodecyl sulfate; mechanical destruction (e.g., arteriotomy) followed by laser sealing or glue application; or mechanical transluminal or external destruction of various parts of the vessel wall. Among all species, aneurysms were created in anatomical locations that included the femoral artery, common iliac artery, external and internal iliac artery, aortic bifurcation, abdominal aortic artery, renal artery, brachiocephalic artery, subclavian artery, vertebral artery, common carotid artery (CCA), CCA bifurcation, ascending cervical artery, external carotid artery (ECA), ascending pharyngeal artery, lingual artery, maxillary artery, superior thyroid artery, ascending cervical artery, basilar artery, and middle cerebral artery (Figure 3), respectively. Subtypes of these models are given in Figures 4 and 5, and their method depicted in step-by-step illustrations (Supplementary Figures).
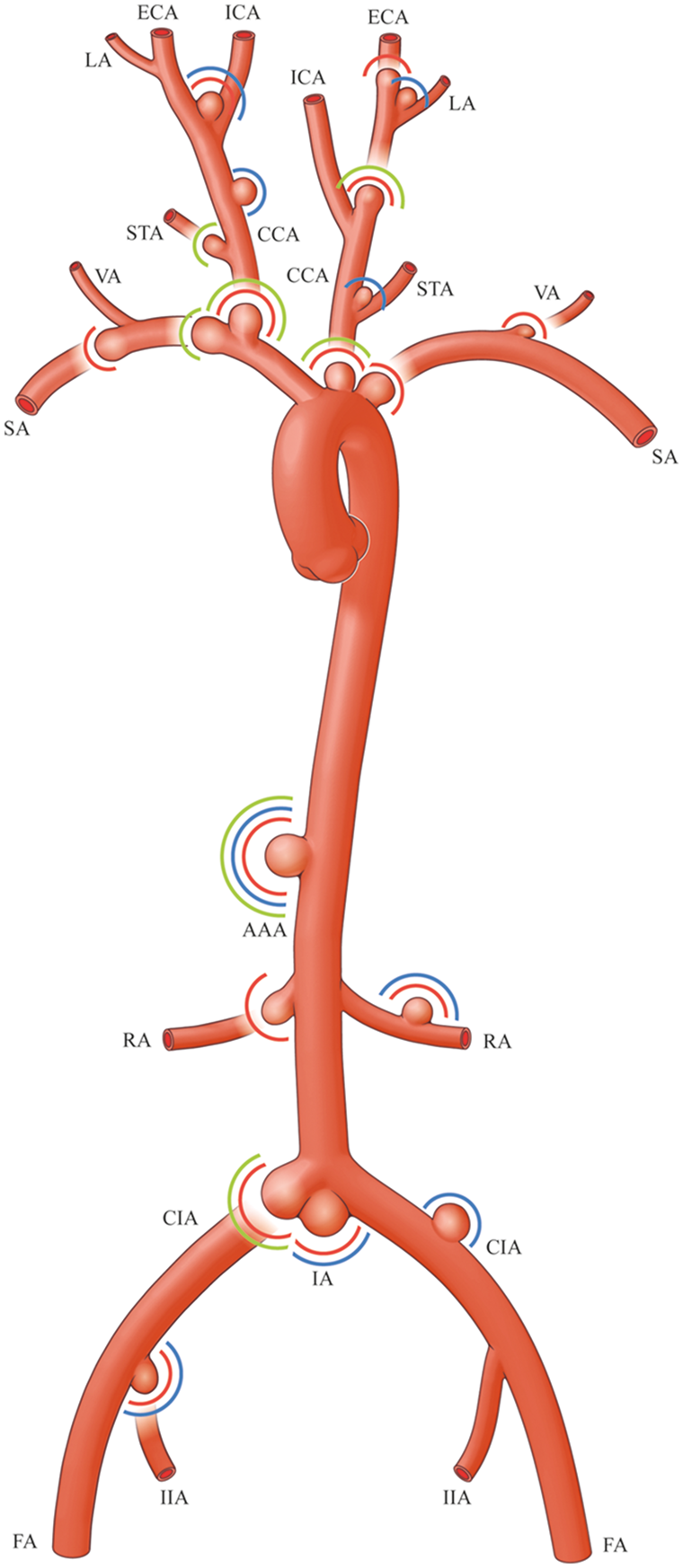
Anatomical locations of extracranial aneurysm models. Green circle: modified pouch; blue circle: venous pouch; red circle: arterial pouch. AAA: abdominal aortic artery; CCA: common carotid artery; CIA: common iliac artery; ECA: external carotid artery; FA: femoral artery; IA: iliac artery bifurcation; ICA: internal carotid artery; IIA: internal iliac artery; LA: lingual artery; RA: renal artery; SA: subclavian artery; STA: superior thyroid artery; VA: vertebral artery.
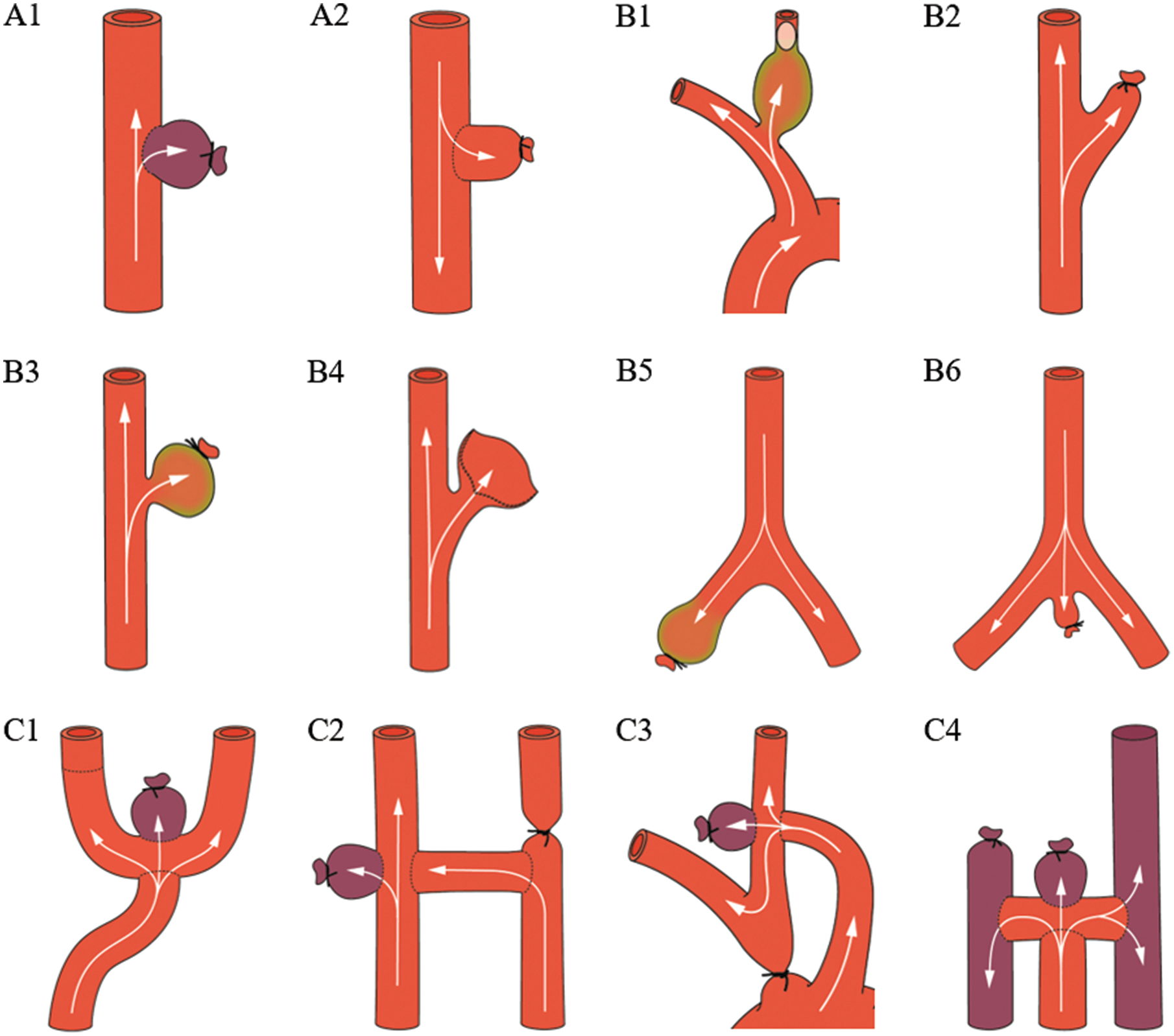
Sidewall, stump, and terminal aneurysm models. Sidewall aneurysms are created by suturing either a venous (A1) or arterial (A2) unmodified pouch onto a parent artery. Stump aneurysms are created by endovascular occlusion, ligature, or flap construction of a branching artery (B1–B5). Modification of the aneurysm wall (green) allows for growth of the stump into a more saccular shape. The tail artery stump represents a unique true arterial bifurcation model (B6). Terminal aneurysm models are designed to simulate flow conditions similar to human basilar and internal carotid artery bifurcation aneurysms (C1–C4).
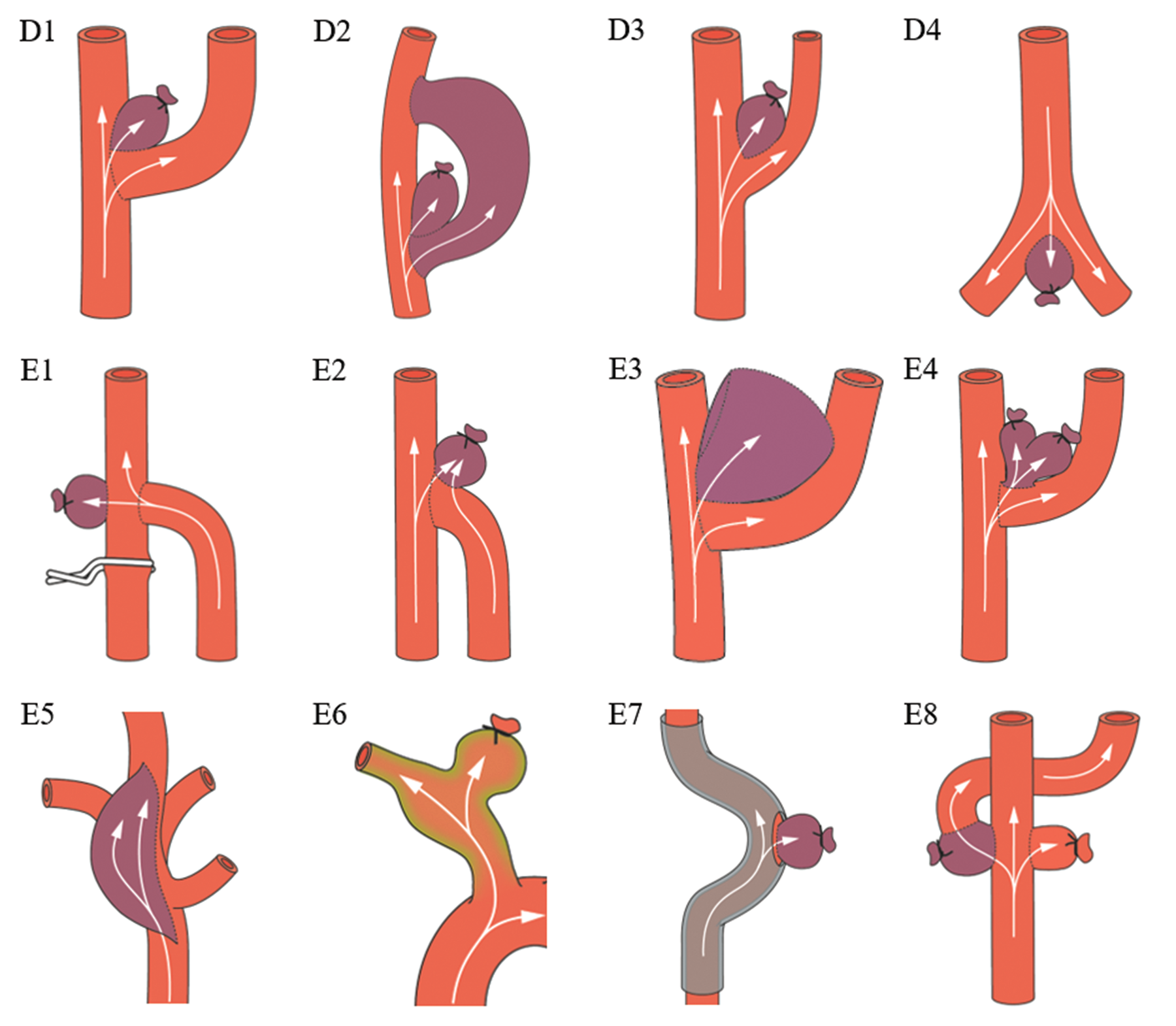
Bifurcation and complex aneurysm models. Bifurcation aneurysms are created by suturing a venous pouch into an artificially created bifurcation (D1 and D2). A natural bifurcation aneurysm model is created when a venous pouch is sutured into an already existing bifurcation (D3 and D4). Complex aneurysm models comprise a heterogenous group of microsurgically created aneurysms that mimic rare forms of intracranial aneurysms such as confluence (E1 and E2), giant sized (E3), multilobulated (E4), fusiform (E5 and E6), curved (E7), or aneurysms with a side branch (E8).
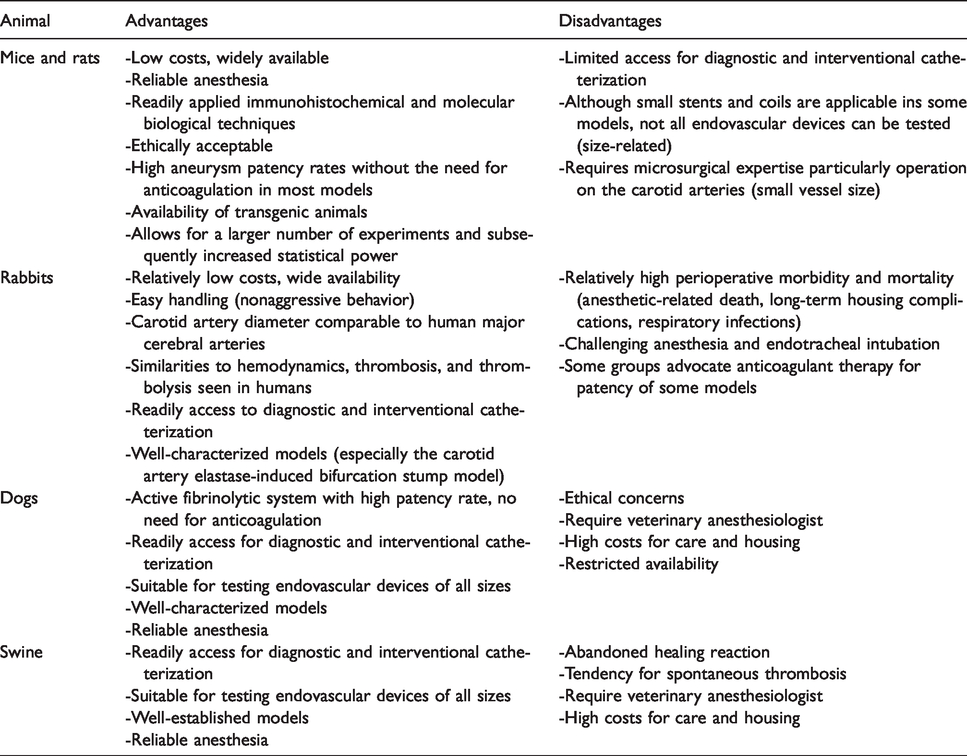
Compared to dogs and pigs, smaller animals such as mice, rats, and rabbits have the advantages of lower costs and easier handling. Although not all endovascular devices can be tested in mice and rats, transgenic animals are widely available and immunohistochemical and molecular biology techniques are easier to apply. Unlike all other species, pigs show an unfavorable strong tendency for spontaneous thrombosis and excessive healing reaction. Sidewall, untreated stump, and natural bifurcation models are the simplest and quickest techniques for creating extracranial aneurysms. In contrast, the terminal, artificial bifurcation, treated stump, and complex models are resource intensive, surgically demanding, and time-consuming. The sidewall model allows creation of most standardized aneurysm shapes with good reproducibility. However, the hemodynamic condition differs significantly from most human aneurysms. Artificial bifurcation and complex models exhibit comparably worse reproducibility but do allow for great variability in aneurysm shape and have flow characteristics like those seen in human aneurysms. Advantages and disadvantages of each animal and the five main extracranial aneurysm models are summarized in Tables 1 and 2.
Advantages and disadvantages of each animal.
Methods of aneurysm creation: Advantages and disadvantages of main models.

aAdvantages and disadvantages mainly relate to the right common carotid artery elastase bifurcation stump aneurysm model.
Sidewall aneurysm model
The sidewall aneurysm model, described by German and Black, is the oldest microsurgical technique to produce experimental aneurysms (Figure 4, A1). 13 The two variations of this technique have used (1) an isolated vein graft sutured end-to-side onto the parent artery (Supplementary Figure A1A) and (2) an arteriovenous fistula created by end-to-side or side-to-side suturing of a vein onto the parent artery followed by immediate or delayed ligation of the fistula (Supplementary Figure A1B).14,15 Modifications include weakening of the parent artery with the use of nitrogen mustard, elastase, or mechanical destruction and subsequent aneurysmal outpouching at the site of the damaged vessel segment. Although the size of arteriotomy can be standardized, the size of the venous pouch itself and ultimately the aneurysm volume varies greatly. Only use of an arterial graft can ensure the standardization of aneurysm size.
To date, the most standardized aneurysm model in terms of graft origin, aneurysm shape and dimensions, volume-to-orifice ratio, and parent vessel to aneurysm long axis angle is the rat sidewall aneurysm created by an arterial pouch (Figure 4, A2; Supplementary Figure A2). 16 The patency rate of sidewall aneurysms depends to a large extent on the animal used. Untreated sidewall swine aneurysms have a tendency for spontaneous thrombosis.17–19 Unlike the tendency for spontaneous thrombosis in swine,17–19 the sidewall venous pouch aneurysm models in the rat, rabbit, and dog achieved excellent rates of long-term patency, that is, 100% (84/84), 16 95% (38/40), 20 and 94% (81/86), 21 respectively, without need for an antithrombotic regimen.
Stump aneurysm model
Fingerhut and Alksne 22 were among the first to ligate a major muscular branch of the femoral artery to create an arterial stump. Unlike all other bifurcation stumps, the tail artery stump in dogs (abdominal aortic trifurcation), described by Roach, 23 represents a natural arterial bifurcation (Figure 4, B6; Supplementary Figure B6). Following these original descriptions, others have reported untreated arterial stump models at different anatomical locations and in various species. Untreated stumps are the simplest, fastest method to create an arterial pouch (even in relatively small species) and therefore are ideal models to screen potential embolization materials (Figure 4, B2; Supplementary Figure B2).24–26
Influenced by previous experimental work of elastase-induced abdominal aortic aneurysms in rats, 27 Cawley et al. 28 infused elastase (via microcatheter from the femoral artery) into unilateral and bilateral surgically created stumps of the ECA in rabbits (Figure 4, B3; Supplementary Figure B3). Cloft et al. 29 elaborated on a purely endovascular-based arterial aneurysm model. After blocking the origin of the left CCA 2 cm distal to the vessel origin using a detachable balloon, they infused the stump with elastase for 30 min (Figure 4, B1; Supplementary Figure B1). Altes et al. 30 modified a version of this model that reduced creation time (<1 h) and procedural morbidity and mortality. They surgically exposed the right CCA, blocked its origin retrograde with a pliable balloon, and infused the distal stump with elastase. The group proposed several modifications to their right CCA model to customize resultant aneurysm morphology.31–33 Because the right CCA model is more of a sidewall-type aneurysm (origins as bifurcation from the subclavian artery), Ding et al. 34 adapted their previous modifications to the left CCA (originates as a trifurcation between the brachiocephalic artery and aortic arch) to create a more bifurcation type elastase-induced aneurysm in 2013.
Others proposed improvements for the elastase-induced CCA bifurcation stump model. Krings et al. 35 advocated fluoroscopic guidance of elastase infusion to prevent leakage into aberrant tracheal arteries in effort to reduce mortality and morbidity (mainly attributed to hemorrhagic necrosis of the trachea). 36 One year later, Hoh et al., 37 also concerned by reports of failure of the original model due to aberrant branches from the right CCA, 38 proposed temporary clip placement at the origin of the right CCA and infusion of the lumen under visual control (aberrant branches are ligated or coagulated). The technique reduced procedure time by 35 min, eliminated the need for fluoroscopy, and minimized the use of endovascular supplies. More recently, Kainth et al. 39 proposed to ligate the left or right CCA 4–6 mm distal to the bifurcation, precisely brush the apex of the blind pouch using a 10/0 round-tip paint brush, and anchor the aneurysm to influence its orientation. This extravascular elastase application supposedly eliminates the need for angiography and endovascular supplies, preserves the femoral arteries for follow-up imaging or endovascular procedures, and allows for variation in the orientation of the aneurysm relative to the parent vessel.
The elastase-induced ECA bifurcation stump model also underwent modifications. Wang et al. 40 proposed to ligate the ECA temporally using an aneurysm clip and infuse the ECA stump with elastase for 20 min. This surgical procedure is simple, fast, and requires no interventional support. Elastase-induced bifurcation stump models have also been introduced in the right 41 and left42 CCA in mice and in the abdominal aortic bifurcation in rats. 43
The original elastase-induced right CCA bifurcation stump model in rabbits is one of the best characterized saccular aneurysm models. Serial angiography on days 1, 3, 5, 7, 14, and 21 demonstrated progressive enlargement, but thereafter the aneurysm size remained stable on one, two, three, and four months imaging. 44 An excellent long-term patency of 92% was confirmed in a five-year follow-up study. 45 Mortality rates associated with aneurysm creation and the embolization procedure were 8.4% (59/700) and 8.1% (43/529), respectively. 46 Hemodynamic analysis revealed that most features of the elastase-induced right CCA model are qualitatively and quantitatively similar to many of human cerebral aneurysms. 47 In a dose-escalation study, native bifurcation stumps failed to create saccular aneurysms. 48 However, increased elastase concentration or incubation time or both demonstrated a trend for widening parent arteries but did not result in larger aneurysms cavities at three weeks follow-up. Rather, the aneurysms were mainly caused by the initial infusion of elastase and not by ongoing wall remodeling and degradation by endogenous proteases. 49 Nevertheless, histological analysis has demonstrated that this model mimics wall types that have been identified in human intracranial aneurysms.50,51
Terminal aneurysm model
Terminal aneurysms are aimed at simulating human basilar tip and internal carotid artery (ICA) bifurcation aneurysms. Originally described by Strother et al. in 1992, they were constructed by forming an arch with end-to-end anastomosis of both CCAs (Figure 4, C1; Supplementary Figure C1). 52 One should be aware that the volume increases as the aneurysm matures during the first few weeks but thereafter remains stable (up to six months follow-up). 53 With its distinctive hemodynamic stress, terminal aneurysms demonstrate 100% patency in rat (9/9) 54 and rabbits (9/9) 55 and 85% to 100% (11/13) 56 to (16/16) 53 in dogs. Even in swine where long-term patency is a concern, terminal aneurysms have been reported to have direct intraluminal blood flow with a 100% (3/3) patency rate at three-month follow-up. 19
Bifurcation aneurysm model, natural, and artificial variations
Natural bifurcation aneurysm model
Stehbens first created a natural bifurcation aneurysm model in rabbits by suturing a venous pouch into the bifurcation of the abdominal aorta (Figure 5, D4; Supplementary Figure D4A). 57 Ujiie et al. 58 modified the arteriotomy and venous pouch ligature to achieve narrow or broad-based aneurysms shaped as dumbbell, oval, and lobular. In delivering elastase topically to the bifurcation of either the right CCA or the right superior thyroid artery bifurcation, Miskolczi et al. 59 found that aneurysm formation was mainly at the latter location. Nishikawa et al. 60 and Young et al. 61 induced natural bifurcation models in rats by suturing a vein pouch or inducing external damage to the vessel wall at the site of CCA/ECA bifurcation.
Since mechanical destruction at the apex of the CCA/ECA bifurcation from the outside failed to produce aneurysmal bulging, it was concluded that the internal elastic lamina represents a critical structure in aneurysm formation. 62 Accordingly, van Alphen et al. 63 achieved aneurysm formation in rats by transluminal (via arteriotomy proximal to the CCA/ECA bifurcation) removal of the tunica intima and media. Mucke et al. 64 harvested a CCA vessel segment and sutured this arterial graft into the aortic bifurcation in rats (Supplementary Figure D4B). In dogs, Sekhar et al. 65 , Macdonald et al. 56 , and Shin et al. 66 created an aneurysm by suturing a venous pouch in the natural bifurcation between the CCA and superior thyroid artery, lingual artery, and cranial thyroid artery, respectively (Figure 5, D3; Supplementary Figure D3). Boulos et al. 67 used the ECA-lingual artery bifurcations to create bilateral aneurysms. In swine, Massoud et al. 68 created natural bifurcation aneurysms at the CCA-ascending pharyngeal artery.
Artificial bifurcation aneurysm model
Forrest and O’Reilly were the first to create an artificial bifurcation aneurysm by suturing a venous pouch into the end-to-side anastomosis of the left to right CCA in rabbits (Figure 5, D1; Supplementary Figure D1). 69 Despite an excellent 97% patency rate (34/35) at 1–10 weeks follow-up, the 22% morbidity rate (10/45) was rather high. Subsequent modifications of the surgical technique (i.e., preservation of laryngeal nerves, tensionless anastomosis, reduction of number of sutures and procedural time) and improved anticoagulation and peri- and postoperative management successively reduced the mortality/morbidity rate from 24%, 70 <20% 71 to 0% 72 and maintained high patency rates (100% 71 and 87.5% 72 ) at one-month follow-up.
Creation of such aneurysms requires profound microsurgical skills. Accordingly, operative times decreased during the experimental series (from 210 to 90 min 71 and from 225 to 115 min 72 ). The same venous pouch technique for artificial bifurcation aneurysms performed at the same location in the rat, dog, and swine species achieved excellent patency rates (100% in 12/12). 73
Complex aneurysm model
This group represents microsurgically created aneurysms that mimic rare forms of intracranial aneurysms such as giant sized, broad-based, no-neck, fusiform, blister-like, bisaccular, bi- or multilobular, or confluence artery aneurysms. Nishikawa et al. 74 had already reported confluence aneurysms in dogs by inclusion of a vein graft into an intracranial lingual-basilar artery anastomosis. Jiang et al. 75 designed a dog confluence aneurysm model that used both the CCA and a vein graft to closely mimic human vertebral confluence aneurysms (Figure 5, E2; Supplementary Figure E2). Complex shaped bilobular, bisaccular, and broad-neck microsurgical aneurysms were created using various combinations of vein grafts in the rabbit artificial bifurcation model (Figure 5, E4; Supplementary Figure E4A–E4C). 76 Despite the complex angioarchitecture, the aneurysms proved excellent long-term patency. 77
Varsos et al. 14 first attempted to create a giant aneurysm by inducing a fistula between the CCA and external jugular vein and ligating this fistula one week later to form a giant sidewall aneurysm pouch (Supplementary Figure E3B). Yapor et al. 78 presented a modification with a one-stage approach (Supplementary Figure E3C). Almost two decades later, giant aneurysms (approximately 1 × 2.5 cm) were created in the canine terminal 79 and rabbit artificial bifurcation 80 models (Supplementary Figure E3A).
With the emergence of novel endovascular technologies that extended the indications for treatment to a broader range of intracranial aneurysm (IA) morphologies, new preclinical models were needed. More recently fusiform 81 and curved sidewall 82 aneurysm models were designed in rabbits and dogs. Darsaut et al. 83 introduced a giant fusiform model that used a rhomboid venous patch sutured to the anterior aspect of the lingual artery that allowed testing of flow diverters (Figure 5, E5; Supplementary Figure E5A).
Based on this groundwork, Greim-Kuczewski et al. 84 proposed to surgically include a venous patch as a sidewall aneurysm to mimic some morphological aspects of human intracranial dysplastic, fusiform, and blood blister-like aneurysms (Supplementary Figure E5B). To test flow diverter treatment of aneurysms with side branches, Darsaut et al. 85 created complex terminal aneurysms that featured a side branch originating from the aneurysm sac (Figure 5, E8; Supplementary Figure E8). Similarly, Fahed et al. 81 created a complex fusiform rabbit aneurysm model that permitted more comprehensive testing for flow diverters (Figure 5, E6; Supplementary Figure E6). Avery et al. 86 presented a refined fusiform aneurysm model by periarterial application of combined elastase and CaCl2 in a rabbit CCA. Yan et al. 87 and Nakayama et al. 88 created canine CCA curved-artery models (Figure 5, E7). First, they printed artificial hollow tortuous tubes (based on human ICA stereolitography data) and then transposed the animal’s CCA into this hollow rod (Supplementary Figure E7A-C).
Complex aneurysm models uniquely address a very narrow research questions about a specific aneurysm shape or flow condition. To date, these models are typically technically difficult, time-consuming to create (some even require two surgeons 79 ), and are not well standardized or characterized.
Discussion
Our systematic review confirms the great variety in vivo extracranial aneurysm models used for a range of applications from basic biological concepts to screening of endovascular materials to final testing of endovascular devices for human application. After screening more than 4000 potential publications, the 68 studies included in this review represented five major groups of extracranial animal models and techniques. Our results highlight each model’s evolution and, in recent years mainly, the modifications and refinements that improved morbidity and mortality rates, reduced operation time, improved patency rates, and kept pace with the implementation of novel endovascular techniques and devices (broadened clinical indications).
An ideal extracranial aneurysm model would require the following features: (1) size of aneurysm and parent artery similar to larger cerebral arteries (enables realistic microcatheter interventions), (2) long-term patency of the aneurysm complex without spontaneous thrombosis and no need for anticoagulation or anti-aggregation therapy, (3) standardized method with good reproducibility for comparison of treatment strategies, (4) hemodynamics, coagulation profiles (clotting and thrombolytic system), and tissue and immunologic reactions similar to those of human IA, (5) wide availability of the animal and easy handling, (6) low costs, and (7) surgical or endovascular creation technique that is simple, fast, and associated with a gentle learning curve.89–92 None of the identified preclinical extracranial aneurysm models that are currently available combine all these ideal characteristics. In contrast with intracranial aneurysm models where aneurysms develop in cerebral arteries without direct vessel manipulation, creation of extracranial models always requires a vascular injury. Researchers should be aware of the differences in vascular biology and hemodynamic characteristics between intra- and extracranial arteries. Furthermore, extracranial aneurysms are surrounded by cell-rich connective tissue and the aneurysm wall histology differs greatly from their intracranial counterparts.
Localization
The perianeurysmal space differs greatly between that of extracranial models and human brain aneurysms. With few exceptions, aneurysms models are not created in the subarachnoid space but in the soft tissues of the mediastinum,29,30 neck,28,93 leg,22,94 retroperitoneal space58,95 or within the abdominal cavity.96,97 Nishikawa et al. created an intracranial sidewall aneurysm by suturing an autologous vein graft aneurysm onto the main branch of the middle cerebral artery (via transzygomatic approach), 60 and sidewall and arterial confluence aneurysms were formed on the basilar and lingual arteries (via a transclival approach) in dogs. 74 O’Reilly et al. 98 relocated a rabbit vein pouch aneurysm via polyvinyl tubing from the right CCA into the subarachnoid space. However, technical difficulties, high mortality, and low patency rates prevented the further use of these techniques.
All other microsurgically created aneurysm models are surrounded by tissue that might affect intraluminal thrombosis and the healing response. The peritoneal cavity has the advantage of being less restrictive than the neck or other subcutaneous regions. Therefore, this region will potentially allow an aneurysm to grow if the wall is weakened. One specific advantage of aneurysm location on the renal artery is the possibility to control for potential ischemic embolic complications by postmortem assessment of renal infarction. 95 Besides the location of aneurysm creation, one must also consider the diameter of parent arteries. Comparable to the diameter of great cerebral arteries where most human brain aneurysms are located is the size of the CCA in rabbits and the abdominal artery in rats.
Creation time
The simplest and fastest surgical techniques are the sidewall, bifurcation stump, and natural bifurcation models; these take 30–60 min for creation based on whether the aneurysm pouch needs additional modification. In comparison, terminal and artificial aneurysm models need up to 3 h for creation. The most demanding and time-consuming are complex aneurysm models, which require advanced microsurgical skills, an operating microscope, and occasionally two operators to create. 79
Costs
Small experimental animals, such as mice and rats, are associated with lower costs than larger animals that require specialized experimental equipment, anesthesia, postoperative care, and housing. Additionally, surgery on small animals eliminates the need for a veterinary anesthesiologist and costs less for animal purchase, surgery, postoperative care, and housing. Surgery can usually be performed in the research laboratory, and general anesthesia is easily maintained by the operator with non-inhalational methods. Dogs and swine are the most expensive experimental animals. Operations are usually performed under costly general inhalational anesthesia by a veterinary anesthesiologist and additional technical assistants and must be performed in a designated animal operating theater.
Patency rate
Long-term patency without spontaneous thrombosis is a key characteristic and concern of any aneurysm model. Large series in rats, rabbits, and dogs demonstrated patency rates of 92.5%, 96 95%, 99 94%, 21 respectively, in 90° angle sidewall aneurysm models without any anticoagulation. However, when considering these excellent patency rates achieved by experienced researchers, it remains a matter of debate to what extent other factors are important measures in preventing thrombosis. These include extensive microsurgical training and associated technical factors (suture line, vein valves, badly placed sutures, or constricted aneurysm neck),100,101 shape and size of arteriotomy, 98 aneurysm volume-to-neck ratio, 102 angle to long axis of grafted venous pouches, 103 number of sutures, tensionless anastomosis, and perioperative and postoperative management (compensation for fluid loss, pain management, antibiotics, vitamin complexes).71,72 In rats and dogs, patency rates are better in artificial bifurcation models than in sidewall aneurysm models.21,60,74 In dogs and swine, terminal aneurysm models exhibited better patency than sidewall aneurysm models.19,56,66
Anticoagulation
Although most groups use local irrigation of parent arteries and storage of grafts in heparinized saline, systemic application of systemic anticoagulant58,80,104 or antiplatlet39,75,76 drugs is also common. In a rare direct comparison of effect of systematic antiplatelet medication on the patency rate of venous sidewall CCA aneurysms in dogs, Kerber and Buschman observed greater success with aspirin than without; patency rates were 74% versus 45%, respectively, but the difference was not statistically significant. 100 Excellent long-term patency rates in large series of sidewall, terminal, bifurcation stump, and arterial bifurcation models in rat, rabbit, and dog provide evidence against the use of systematic anticoagulation. One must be especially careful with the administration of aspirin because of its potential influence on aneurysm wall biology and recurrence after endovascular therapy.105,106
Histopathology and aneurysm healing
Debate continues about whether the morphological and histological characteristics of human cerebral aneurysms are more accurately modeled by elastase-digested arterial stump models or by surgically created vein or arterial pouch aneurysms.51,107 Nonetheless, there is consensus that models with a strong tendency for spontaneous thrombosis and excessive neointima formation are inappropriate for study of healing after embolization. Sidewall aneurysms (especially in swine) have demonstrated favorable healing reactions after coil embolization compared to artificial bifurcation models. 108 However, bifurcation models may also demonstrate healing rates that far exceed those seen in human aneurysms after coil embolization. 109 Recent research revealed that aneurysm healing and recurrence after endovascular treatment also depend on the aneurysm wall condition.1,110–112 Therefore, models that depict various types of wall conditions will gain interest and become appropriate models for testing novel endovascular therapies.50,110
Reproducibility
Standardized and reproducible aneurysm creation is vitally important to improve preclinical assessment of novel endovascular devices and enhance comparability of results between laboratories. To date, the most standardized aneurysm models in terms of graft origin, aneurysm shape and dimensions, volume-to-orifice ratio, and parent vessel to aneurysm long axis angle are created by an arterial pouch.55,96 To a certain degree, neck size and aneurysm volume can also be controlled and adjusted in elastase-induced aneurysms31,33,113,114 and modified techniques have achieved more consistent/standardized aneurysm diameters. 32 In venous pouch aneurysm techniques (especially complex models), the angioarchitecture (size, shape, and flow condition) is less standardized. However, the heterogeneity of aneurysm angioarchitecture increases the external validity of the model. Therefore, both are indispensable: standard models can test and refine the basic characteristics of a novel endovascular device, whereas models with high aneurysm variability can provide more exacting tests.
Ethical considerations
Depending on the location, most sidewall and some bifurcation stump and natural bifurcation models allow for the creation of two or more aneurysms in the same animal. Such models not only reduce the overall number of animals needed but also allow testing of two treatment modalities side-to-side within a single animal. Except for a three- and four-aneurysm dog model, most artificial bifurcation and complex aneurysm models allow creation of only one aneurysm per animal.66,67,76,79 Some aneurysm models depend on syngeneic grafts, that is, using two animals to produce one aneurysm.16,61,96 In contrast, aneurysm models with simple and fast aneurysm creation facilitate research with a larger number of experiments, thus ensuring adequate statistical power with increased significance and ultimately fewer animals. With the exception of some historical models and thanks to modified techniques over the past decades, currently used aneurysm models harbor low rates of morbidity and mortality.
Limitation of the study
Despite systematic approach by following the PRISMA guidelines and two investigators who independently screened the literature, this review might not be exhaustive based on our inclusion and exclusion criteria. Especially in the case of recognition of a new model related to a major modification of an existing technique, we cannot exclude a certain arbitrariness in the selection process despite strict application of predefined criteria.
Taking the potential confounding effects of the chosen species and techniques into consideration, basic biological concepts of novel intracranial aneurysm therapies can be tested in a great variety of models available today. Our categorization of models into five main groups and discussion of advantages and disadvantages allows a quick and comprehensive insight into the field. Detailed submodel analysis facilitates prediction of specific aneurysm model characteristics and therefore supports researchers in study planning, the execution of experiments, and interpretation of the results.
In summary, this systematic review of PubMed database, covering the period between 1950 and 2020, identified five main models of extracranial saccular aneurysms in mice, rats, rabbits, dogs, and swine, and their subsequent refinements and technical modifications. This review may serve as a compendium for investigators to identify the most appropriate model from a wide range of different techniques that suits best their experimental goals, practical considerations, and laboratory environment.
Supplemental Material
JCB908363 Supplemental Material1 - Supplemental material for Preclinical extracranial aneurysm models for the study and treatment of brain aneurysms: A systematic review
Supplemental material, JCB908363 Supplemental Material1 for Preclinical extracranial aneurysm models for the study and treatment of brain aneurysms: A systematic review by Serge Marbacher, Fabio Strange, Juhana Frösén and Javier Fandino in Journal of Cerebral Blood Flow & Metabolism
Supplemental Material
JCB908363 Supplemental Material2 - Supplemental material for Preclinical extracranial aneurysm models for the study and treatment of brain aneurysms: A systematic review
Supplemental material, JCB908363 Supplemental Material2 for Preclinical extracranial aneurysm models for the study and treatment of brain aneurysms: A systematic review by Serge Marbacher, Fabio Strange, Juhana Frösén and Javier Fandino in Journal of Cerebral Blood Flow & Metabolism
Supplemental Material
JCB908363 Supplemental Material3 - Supplemental material for Preclinical extracranial aneurysm models for the study and treatment of brain aneurysms: A systematic review
Supplemental material, JCB908363 Supplemental Material3 for Preclinical extracranial aneurysm models for the study and treatment of brain aneurysms: A systematic review by Serge Marbacher, Fabio Strange, Juhana Frösén and Javier Fandino in Journal of Cerebral Blood Flow & Metabolism
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding text goes here: This study was supported by a research grant from the Kantonsspital Aarau, Aarau, Switzerland (1400.000.054). Dr Marbacher was supported by a grant from the Swiss National Science Foundation (310030_182450/1).
Acknowledgements
We thank Mary Kemper (Glia Media) for medical editing and express our gratitude to Ruth Angliker for illustrations.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author contributions
Dr Marbacher contributed to the study concept, screened titles and abstracts, analyzed the data, prepared the figures, obtained personal funding, and wrote the article. Dr Strange screened titles and abstracts, extracted full-text data, and prepared the tables. Drs Fandino and Frösén directed the project and helped to write the article.
Supplementary material
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
