Abstract
Stroke remains a leading cause of death and disability with limited therapeutic options. Endothelial cell β1 integrin receptors play a direct role in blood-brain barrier (BBB) dysfunction through regulation of tight junction proteins and infiltrating leukocytes, potentially mediated by β1 integrins. Following tandem transient common carotid artery/middle cerebral artery occlusion on wild-type mice, we administered the integrin a5b1 inhibitor, ATN-161, intraperitoneal (IP) injection at 1 mg/kg acutely after reperfusion, on post-stroke day (PSD)1 and PSD2. Systemic changes (heart rate, pulse distension, and body temperature) were determined. Additionally, infarct volume and edema were determined by 2,3-triphenyltetrazolium chloride and magnetic resonance imaging, while neurological changes were evaluated using an 11-point Neuroscore. Brain immunohistochemistry was performed for claudin-5, α5β1, IgG, and CD45 + cells, and quantitative polymerase chain reaction (qPCR) was performed for matrix metalloproteinase-9 (MMP-9), interleukin (IL)-1β, collagen IV, and CXCL12. ATN-161 significantly reduced integrin α5β1 expression in the surrounding peri-infarct region with no systemic changes. Infarct volume, edema, and functional deficit were significantly reduced in ATN-161-treated mice. Furthermore, ATN-161 treatment reduced IgG extravasation into the parenchyma through conserved claudin-5, collagen IV, CXCL12 while reducing MMP-9 transcription. Additionally, IL-1β and CD45 + cells were reduced in the ipsilateral cortex following ATN-161 administration. Collectively, ATN-161 may be a promising novel stroke therapy by reducing post-stroke inflammation and BBB permeability.
Introduction
Acute treatment for ischemic stroke is currently limited to recanalization/reperfusion strategies only provided to a minority of patients: intravenous tissue plasminogen activator and/or endovascular thrombectomy.1–4 While these treatments remove the blood vessel-obstructing thrombus, they fail to impact secondary reperfusion injury. Reperfusion injury results from an influx of factors, including calcium, cytokines, reactive oxygen species, growth factors, and matrix metalloproteinases (MMPs), that induce extracellular matrix (ECM) degradation, leukocyte infiltration, and apoptotic cascades.5,6 These processes destabilize the blood-brain barrier (BBB), a vascular defense mechanism composed of non-fenestrated endothelial cells, tightly bound by tight junction (TJ) proteins and junctional adhesion molecules that are surrounded by astrocytic endfeet.5–7 Collectively, the BBB breakdown contributes to the expansion of the infarct volume from the site of initial injury (core) into the surrounding at risk tissue (peri-infarct). Therefore, targeting reperfusion injury could alter ischemic stroke outcomes.
One potential target is the integrin receptor family, a group of cell surface transmembrane glycoprotein receptors for the ECM. They are composed of non-covalently bound α and β subunits. Vital to stroke pathophysiology, integrins have been implicated in cell migration, cellular adhesion, and cellular survivability.8,9 One particular integrin, integrin α5β1, is of vital interest due to its increased expression in hypoxia and stroke models, 10 while exhibiting variable roles in tumor growth and metastisis, 11 atherosclerosis development, 12 and promotion of wound healing. 13 Previously, our laboratory has shown that elimination of the specific pro-angiogenic integrin α5β1 in endothelial specific knockout (α5KO) mice results in significant resistance to ischemic stroke injury. Furthermore, these α5KO mice maintained near-baseline pre-stroke levels of the TJ protein claudin-5 and exhibited little to no disruption of the BBB barrier. 14 These results, combined with previous studies demonstrating a sustained increase in the post-stroke expression of integrin α5β1,10,15 suggest that inhibition of α5β1 integrin could be therapeutic by stabilizing the BBB. Early intervention of the BBB following ischemic stroke is ideal, as its role in injury is not immediately apparent. 16 To that end, in this study we use the small peptide non-competitive integrin α5β1 inhibitor, ATN-161, in a mouse model of ischemic stroke. Importantly, ATN-161 was safe and well tolerated in a number of Phase I and Phase II clinical oncology studies, 17 supporting its potential as a “shovel-ready” clinical stroke therapeutic.
Materials and methods
The data that support the findings of this study are available from the corresponding author (Gregory J Bix) upon reasonable request.
Animals
All animal studies were approved by the Institutional Animal Care and Use Committee of the University of Kentucky prior to their performance. All experiments were performed according to the ARRIVE guidelines and the Guide for the Care and Use of Laboratory Animals of the National Institutes of Health. Three-month-old male C57/Bl6 (The Jackson Labs, Bar Harbor, Maine, USA) were housed in 14-h day/light cycles under free feeding conditions 10 days before being used in experiments.
Stroke surgery
Mice underwent transient tandem ipsilateral common carotid artery/middle cerebral artery (CCA/MCA) occlusion for 60 min as previously published, 14 followed by reperfusion for 1–3 days. Briefly, a small burr hole is created directly over the M2 branch of the MCA, and a 0.0005 inch metal filament is placed under the artery on both sides of the burr hole. The CCA was then isolated and occluded using an aneurysm clip. Blood flow was determined with the Laser Doppler Perfusion Monitor (Perimed, Ardmore, PA, USA). Animals that received less than 80% occlusion rate from baseline (i.e. pre-occlusion) were excluded from analysis. Sham animals underwent the listed procedure without vascular occlusion. All mice were allowed to recover to post-stroke day (PSD)3, during which daily body weights were measured.
Physiological measurements
Immediately following reperfusion, heart rate, pulse distension, and body temperature were recorded using MouseOx (Starr Life Science, Holliston, MA, USA). After 5 min, sterile phosphate buffered solution (PBS) (Vehicle) or ATN-161 was injected via IP (as described below); mice were continually observed for 15 min post reperfusion.
Animal treatments
Animals received an intraperitoneal (IP) injection of either Vehicle (sterile PBS) or ATN-161 (1 mg/kg, Medkoo Biosciences, Morrisville, NC, USA) immediately after reperfusion, on PSD1 and PSD2. Ischemic stroke was confirmed on 2 mm sections stained with 2,3-triphenyltetrazolium chloride (TTC; BD, Sparks, MD, USA) or magnetic resonance imaging (MRI) on PSD3. Accounting for edema volume, infarct size based on TTC images was analyzed with Image J (NH) software as
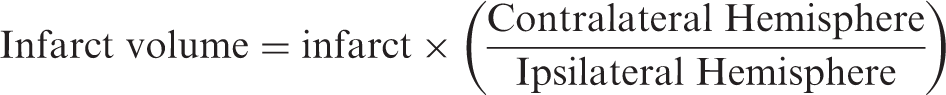
Animals were excluded from the study if the CCA and/or MCA was punctured during wire and clamp placement or removal.
Magnetic resonance imaging
PSD3 animals were anesthetized with isoflurane and placed on a heated bed where body temperature and breathing rate were carefully monitored throughout the scan. All images were performed by a Bunker/Siesman 7 T MR in the University of Kentucky Magnetic Resonance Imaging and Spectroscopy Center. T1 and T2 weighted images of the brain were obtained in the transverse plane without intravenous contrast. Additionally, apparent diffusion coefficient (ADC) images were produced in the transverse plane using diffusion tensor imaging (DTI). All images were analyzed with ITK-SNAP 18 (www.itksnap.org) by a blinded, independent neuroradiologist. Edema and infarct volume were determined independently by software-based manual segmentation. T2 weighted images and ADC images were examined by an experienced neuroradiologist. Areas of abnormally high-signal intensity on the T2 weighted images, representing edema, and areas of abnormally low-signal intensity on the ADC images, representing acute infarction, were visually identified and manually outlined or segmented using computer software.
Tissue histology and immunohistochemistry
On PSD3, animals were systemically perfused with PBS (1X) for 5 min. Brains were removed, flash frozen in liquid nitrogen, and stored at −20℃ until use. The brains were coronally sectioned by cryostat at 20 µm and mounted (four per slide, 2 mm separating each section) for staining. Negative controls (no primary antibody) were run in tandem with each target to determine thresholding values.
IgG Immunohistochemistry
Sections were fixed in 10% phosphate-buffered formalin. After washing, IgG 568 biotinylated antibody (1:500, Thermo-Fisher Scientific) in 1% bovine serum albumin was applied to the tissue for 2 h. Slides were then cover slipped (Vector Labs) for imaging on a Nikon Eclipse Ti and software (Nikon, Melville, NY, USA). Image J was used to isolate and count IgG positive pixels. Three separate 20 × images per core region were taken per section and averaged.
Immunohistochemistry
Sections were fixed in methanol:acetone (1:1), followed by blocking buffer (5% bovine serum albumin (Fisher Scientific) in PBS with 0.1% Triton-X). Sections were incubated in primary antibody overnight at 4℃ (anti-rabbit PECAM at 1:100, Fisher Scientific; anti-rat CD49e at 1:250, BD Pharminagen; anti-rat CD45 at 1:200, ThermoFisher Scientific, anti-rabbit Claudin-5 at 1:200, Abcam, anti-rabbit MMP-9 at 1:100, Abcam, and anti-rabbit fibronectin at 1:100, Abcam). Sections were washed and placed in fluorophore-conjugated secondary antibody, followed by NucBlue cell nucleus counterstain (Fisher Scientific) at room temperature (Vector Labs, 1:250), then cover slipped (Vector Labs) and imaged on a Nikon Eclipse Ti and software (Nikon, Melville, NY, USA). Image J was used to quantify all Immunofluorescence positive pixels.
Behavioral testing
Stroked mice underwent an 11-point behavioral neurological score behavioral assessment on PSD0 (Baseline prior to drug administration), PSD1, 2, and 3 to assess the following behavioral metrics: level of consciousness (LOC), gaze (G), visual field (VF), sensorimotor response (SR), grip strength, and endurance/paralysis paw hang (PPH). LOC was determined as natural movement prior to any cage disturbance. A severity score of 0–2 was assigned, 0 being alert and active, 1 alert upon stimulation, and 2 for being hunched, unstimulated, and loss of grooming. G was assessed by waving an object in front of each eye without disturbing the whiskers. Based on a severity score of 0–1, 0 was given if they recognized the sign by turning their head, and a 1 was given if no response was noted. VF was assessed holding the mouse vertically next to a platform by the tail (both right and left sides). A score of 0 was given if the mouse reached for the platform, and a 1 for failure to do so. SR was scored by pressing each paw in turn to elicit vocalization, paw retraction, or jumping as a reaction to pressure. A score of 0 was given for exhibiting a reaction, and a 1 for no reaction. Finally, PPH was scored by a paw hang test. Only front paws were used for gripping of a rod while being supported by a hold on the tail for 60 s. A score of 0 was given for completion of the full 60 s, a 1 for any dropping of a paw without full loss of contact, and a 2 for a fall. The total scores are tallied at the conclusion of the testing to assess overall function.
Gene expression
On PSD3, brains were removed and sectioned at 2 mm where the ipsilateral cortical tissue and corresponding contralateral cortical tissue were isolated. All sections per animal were combined and homogenized in Trizol (Life Technologies). RNA extraction was performed according to the RNA extraction kit provided by the manufacturer (Life Technologies). RNA was converted to cDNA using a high-capacity cDNA reverse transcriptase kit (Applied Bioscience, Grand Island, NY, USA). 18 S (control; Mm03928990_g1), collagen IV (Mm00483669_m1), interleukin (IL)-1β (Mm00434228_m1), CXCL12 (Mm00445553_m1), and MMP9 (Mm00442991_m1) Taqman genes were analyzed using Real-time quantitative polymerase chain reaction (qPCR) (ViiA7, Life Technologies). Data were analyzed as fold change comparing vehicle and ATN-161-treated mice as a % control (Sham).
Blinding and randomization
All experiments and data analysis were conducted in a blinded and randomized fashion using an online randomization generator.
Statistical analysis
All measureable values are presented as mean ± standard deviation (SD). Sample size was determined for 80% effect by power analysis. Comparisons between Vehicle and ATN-161 treatment were performed using a
Results
ATN-161-treated mice show reduced infarct size following experimental ischemic stroke
First, we examined whether acute, post-reperfusion IP ATN-161 administration was safe immediately following experimental ischemic stroke. ATN-161-treated animals did not show any differences in heart rate (Figure 1(a)), pulse distension (Figure 1(b)), body temperature (Figure 1(c)), and body weight (Figure 1(d)) following ischemic stroke compared to Vehicle-treated stroked controls.
ATN-161 is safe and effective following ischemic stroke. Following reperfusion, percent change of (a) heart rate (bpm), (b) pulse distension (μM), (c) body temperature (℃), and (d) weight (g). (e) Representative images of (f) TTC analysis of infarct volume at PSD3 (p = 0.0004; Vehicle 35.41 ± 14.04, n = 12; ATN-161 16.37 ± 14.25, n = 12). Images have been altered to align sections. (g) Injection schematic.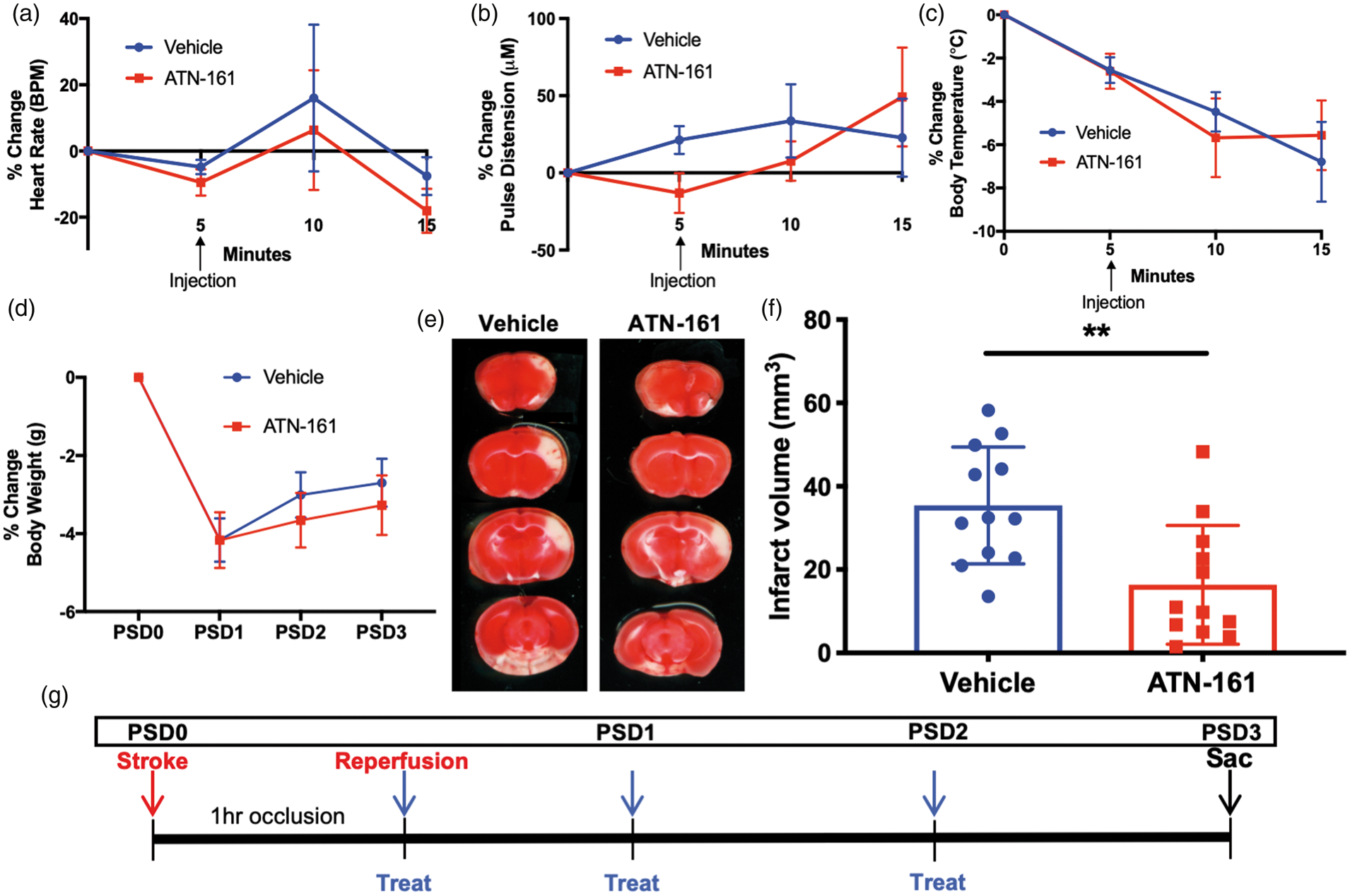
Once acute safety was determined, stroked mice were IP administered Vehicle or ATN-161 immediately after reperfusion, on PSD1, and PSD2 (Figure 1(g)). By TTC assessment, mice treated with ATN-161 had smaller infarcts when including edema, on PSD3 (Figure 1(E) and (F); p = 0.0004; ATN-161 16.37 ± 14.25, n = 12; Vehicle 35.41 ± 14.04, n = 12).
To further validate ATN-161 effects on infarct volume in a translationally relevant manner, stroked mice underwent DTI with ADC imaging on PSD3. Infarct volume, absent edema volume, was again shown to be reduced in ATN-161-treated mice (Figure 2(a) and (b); p = 0.0220; Vehicle 17.9 ± 4.677, n = 7; ATN-161 12.24 ± 3.758, n = 8). Collectively, the TTC and ADC analysis shows a decrease in infarct volume following ATN-161 treatment.
ATN-161 reduces MRI infarct volume and edema. (a) Representative ADC images. Arrows indicate infarcted region. (b) ADC analysis for infarct volume (p = 0.0220; Vehicle 17.9 ± 4.677, n = 7; ATN-161 12.24 ± 3.758, n = 8). (c) Representative T2-weighted MRI images. Arrows indicate edematous region. (d) T2-weighted MRI analysis for edema volume (p = 0.0097; Vehicle 15.12 ± 3.206, n = 7; ATN-161 8.777 ± 4.658, n = 8). Linear correlation of infarct volume and edema volume for (e) all treatments (p = 0.0001 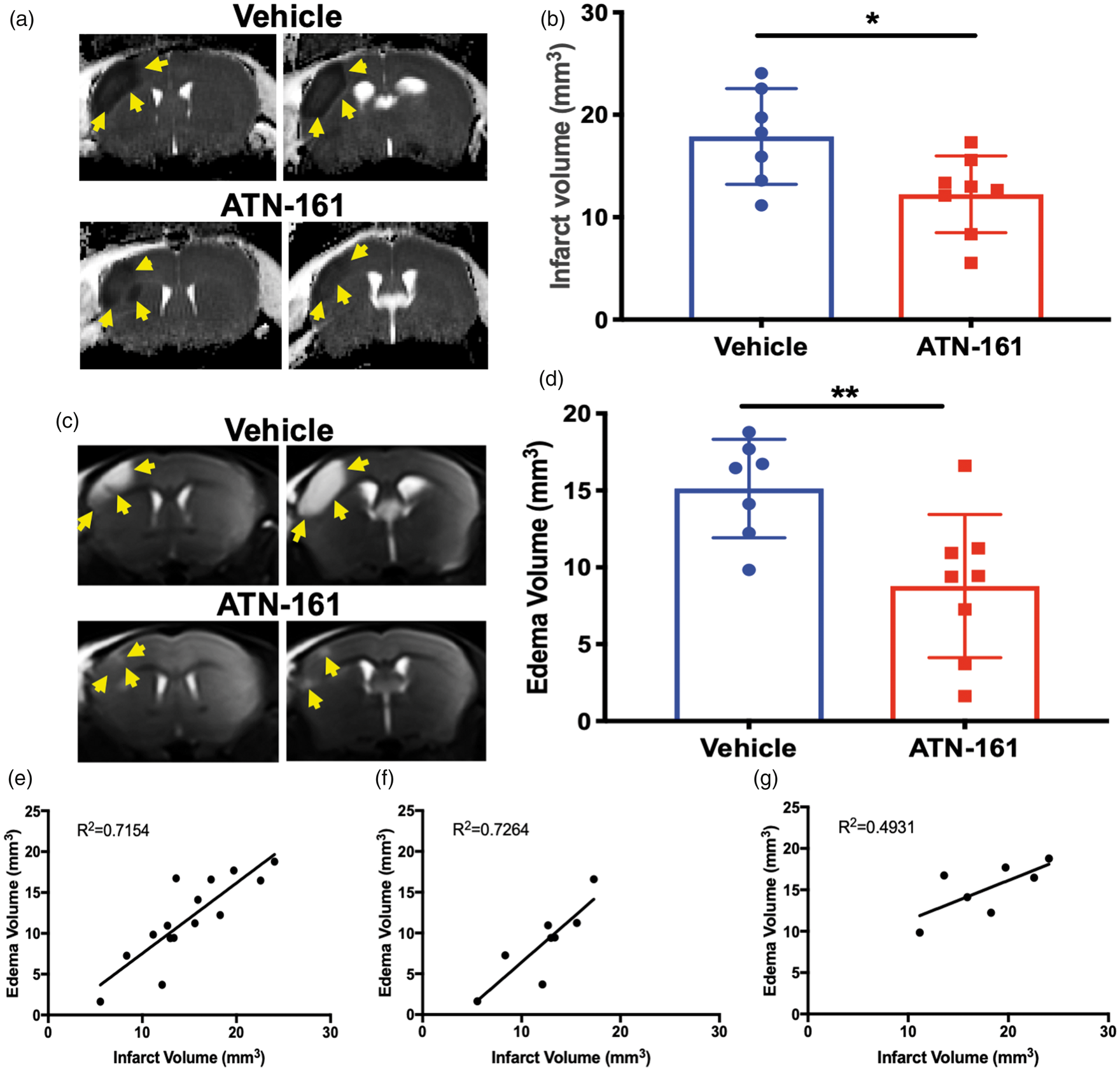
ATN-161 administration reduces edema following experimental ischemic stroke
In addition to infarct volume, we also determined the extent of post-stroke edema with T2-weighted MRI separate from infarct volume determination. Imaging showed a reduction in edema in ATN-161-treated mice (Figure 2(c) and (d); p = 0.0097; Vehicle 15.12 ± 3.206, n = 7; ATN-161 8.777 ± 4.658, n = 8). Comparison of all treatments collectively revealed a positive correlation between infarct and edema volume (all treatments p = 0.0001
ATN-161-treated mice have better functional outcomes
ATN-161-treated mice showed less functional deficit on PSD3 by Neuroscore (Figure 3(a); p = 0.0002; Vehicle 2.286 ± 0.8284, n = 14; ATN-161 3.286 ± 0.7263, n = 14; 95% confidence interval (Cl) −0.07218 to 0.4636), while there is no difference on PSD1.
ATN-161 reduces functional deficit and integrin α5β1 expression. (a) Analysis of 11-point Neuroscore (p = 0.0002; Vehicle 2.286 ± 0.8284, n = 14; ATN-161 3.286 ± 0.7263, n = 14; 95% Cl−0.07218 to 0.4636.). (b) Integrin α5β1 analysis at PSD3 in peri-infarct (p = 0.0835, Vehicle 308484 ± 53485, n = 7; ATN-161 162166 ± 23348, n = 7) and (c) core (p = 0.3138, Vehicle 332114 ± 209410, n = 7; ATN-161 233111 ± 134950, n = 7). (d) Representative images for (b) and (c) 10 × magnification, scale bar = 100 µm.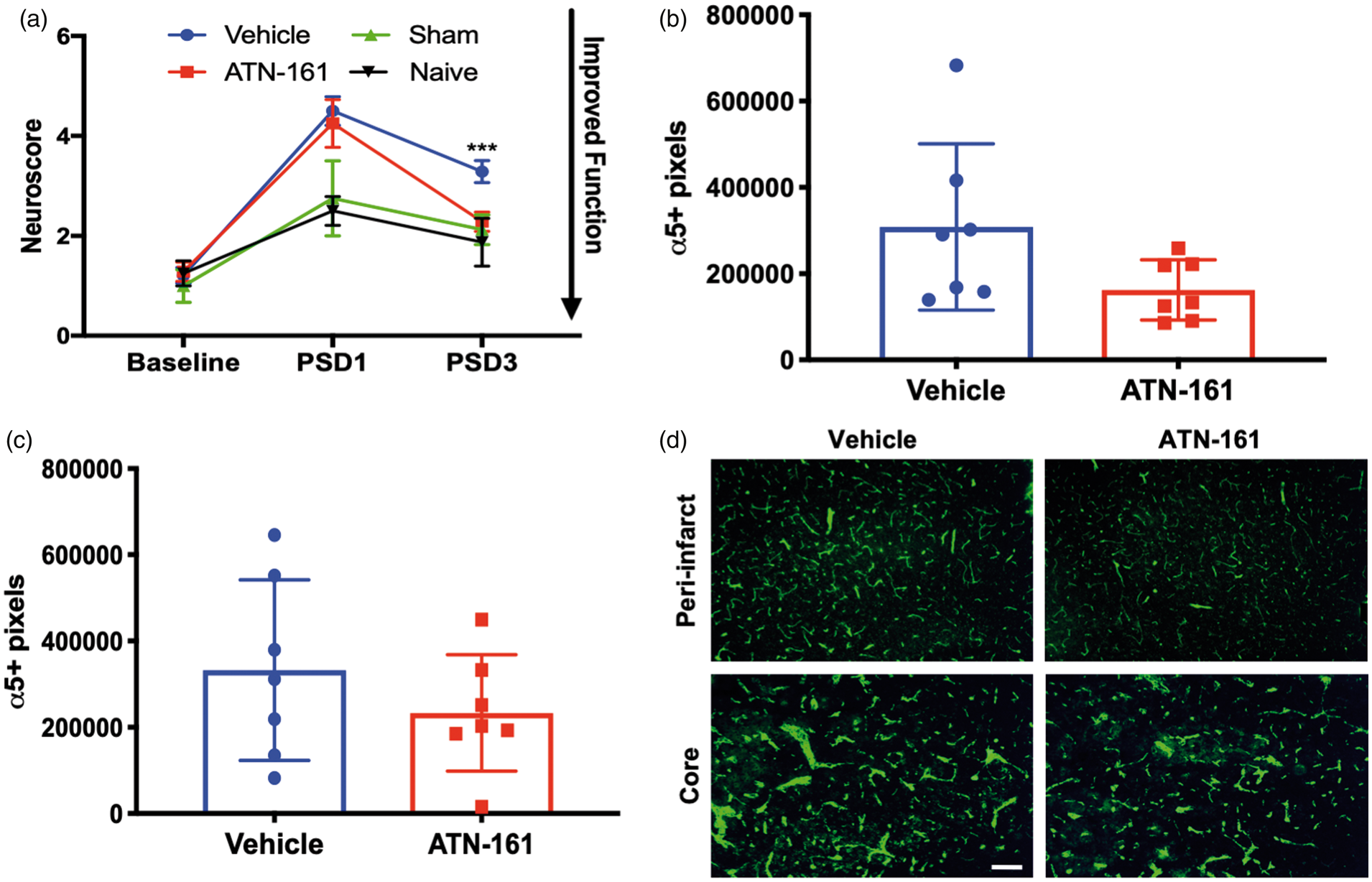
ATN-161 reduces α5β1 expression following ischemic stroke
α5β1 integrin expression decreased at PSD3 with ATN-161 administration in both the peri-infarct (Figure 3(b) and (d); p = 0.0835, Vehicle 308484 ± 53485, n = 7; ATN-161 162166 ± 23348, n = 7) and core (Figure 3(c) and (d); p = 0.3138, Vehicle 332114 ± 209410, n = 7; ATN-161 233111 ± 134950, n = 7) regions of the stroke, although these differences were not significant.
ATN-161 reduces BBB permeability following experimental ischemic stroke
Since α5KO mice showed a stabilized BBB after experimental stroke,
14
we next determined BBB stability via brain IgG extravasation following ATN-161 administration. ATN-161-treated animals had significantly less parenchymal IgG extravasation in the ischemic core (Figure 4(a) and (b); p = 0.0234; Vehicle 70125 ± 53485, n = 7; ATN-161 16753 ± 23348, n = 8). Interestingly, ATN-161-treated mice did experience a correlation between IgG extravasation (i.e. degree of BBB opening) and edema (Figure 4(d); p = 0.0590, ATN-161 reduces BBB permeability following ischemic stroke. (a) Representative images of (b) 20 × magnification, scale bar = 50 µm. (b) IgG positive pixel analysis in the core (p = 0.0234; Vehicle 70125 ± 53485, n = 7; ATN-161 16753 ± 23348, n = 8). Linear correlation between IgG positive pixels and edema volume after (c) Vehicle (p = 0.3705, 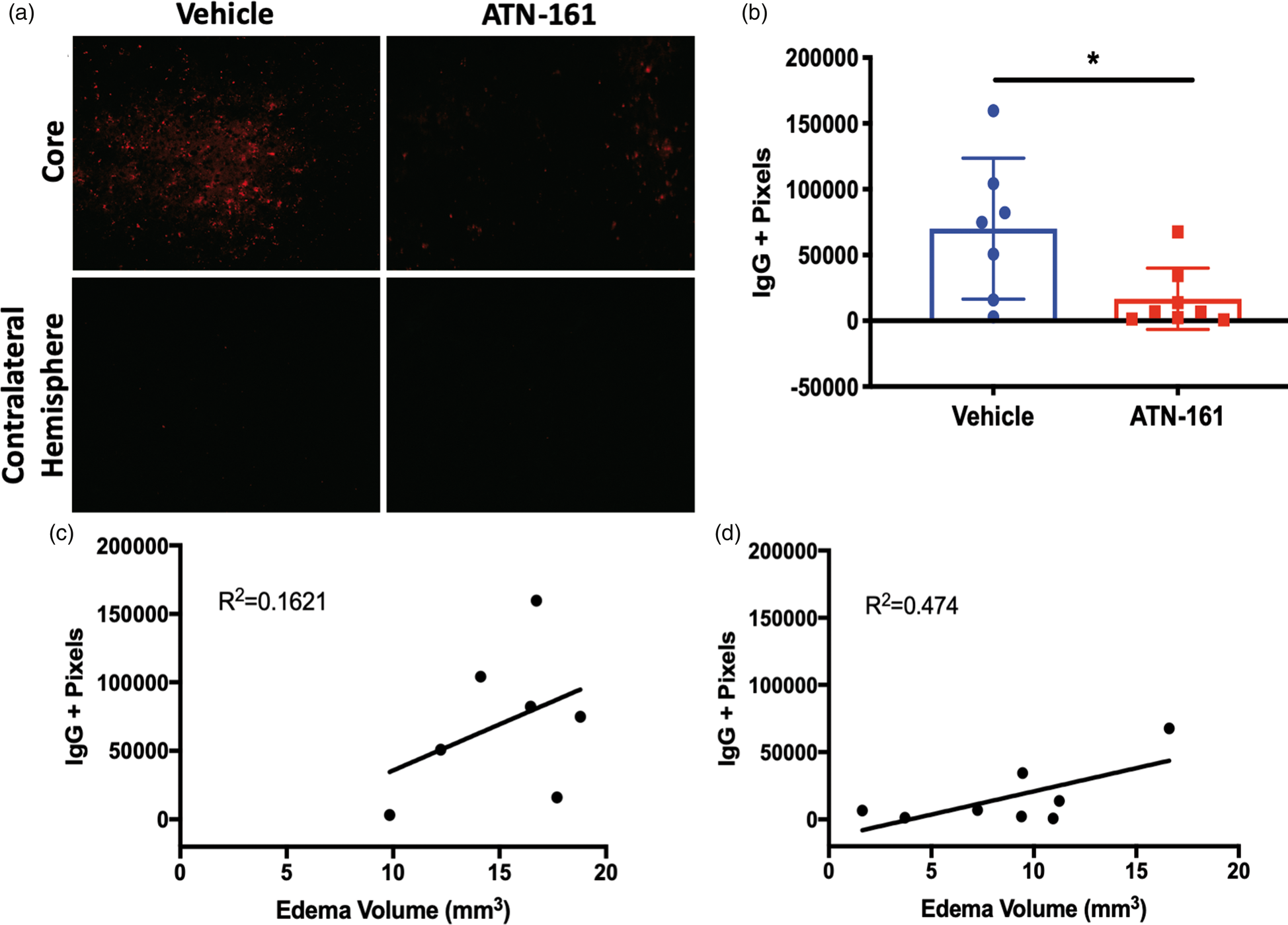
Next, to determine integrin α5β1’s role in cerebrovascular pathology after stroke, we analyzed changes in vascular ECM proteins, proteinases that degrade the ECM, and TJs. On PSD3, Vehicle-treated mice exhibit decreased collagen IV (an abundant ECM protein that is essential to BBB health) transcription compared to Sham and ATN-161-treated animals (Figure 5(a); p = 0.0115; Sham 1.018 ± 0.1573, n = 4; Vehicle 0.3543 ± 0.1013, n = 7; ATN-161 0.8563 ± 0.6898, n = 8; 95% Cl −0.6093 to −0.1379), while ATN-161-treated animals were recovered to Sham levels (p = 0.7272). Next, we determined that ATN-161 treatment resulted in an increase of claudin-5 expression (immunofluorescence) in the core (Figure 5(b) and (c); p = 0.0270; Vehicle 19830 ± 5538, n = 5; 44220 ± 19878, n = 6). However, there was no change in the ECM protein fibronectin in either the core (Figure 5(d) and (e); p = 0.5819; Vehicle 106153 ± 183136, n = 6; ATN-161 158938 ± 164906, n = 8), or peri-infarct (Figure 5(f) and (g); p = 0.4665; Vehicle 28782 ± 40979, n = 6; ATN-161 48028 ± 16730, n = 8) region between Vehicle- and ATN-161-treated animals.
ATN-161 stabilizes cerebrovasculature following ischemic stroke. (a) qPCR analysis of ipsilateral cortex at PSD3 for collagen IV (p = 0.0115; Sham 1.018 ± 0.1573, n = 4; Vehicle 0.3543 ± 0.1013, n = 7; ATN-161 0.8563 ± 0.6898, n = 8; 95% Cl − 0.6093 to −0.1379). (b) Representative images of (c) 10 × magnification, scale bar = 100 µm. (c) Analysis for claudin-5 positive pixels in the core at PSD3 (p = 0.0270; Vehicle 19830 ± 5538, n = 5; 44220 ± 19878, n = 6). (d) Representative images of (e) 10 × magnification, scale bar = 100 µm. (e) Analysis for fibronectin positive pixels at PSD3 in the core (p = 0.5819; Vehicle 106153 ± 183136, n = 6; ATN-161 158938 ± 164906, n = 8). (f) Representative images of (g) 10 × magnification, scale bar = 100 µm. (g) Analysis for fibronectin positive pixels at PSD3 in the peri-infarct (p = 0.4665; Vehicle 28782 ± 40979, n = 6; ATN-161 48028 ± 16730, n = 8).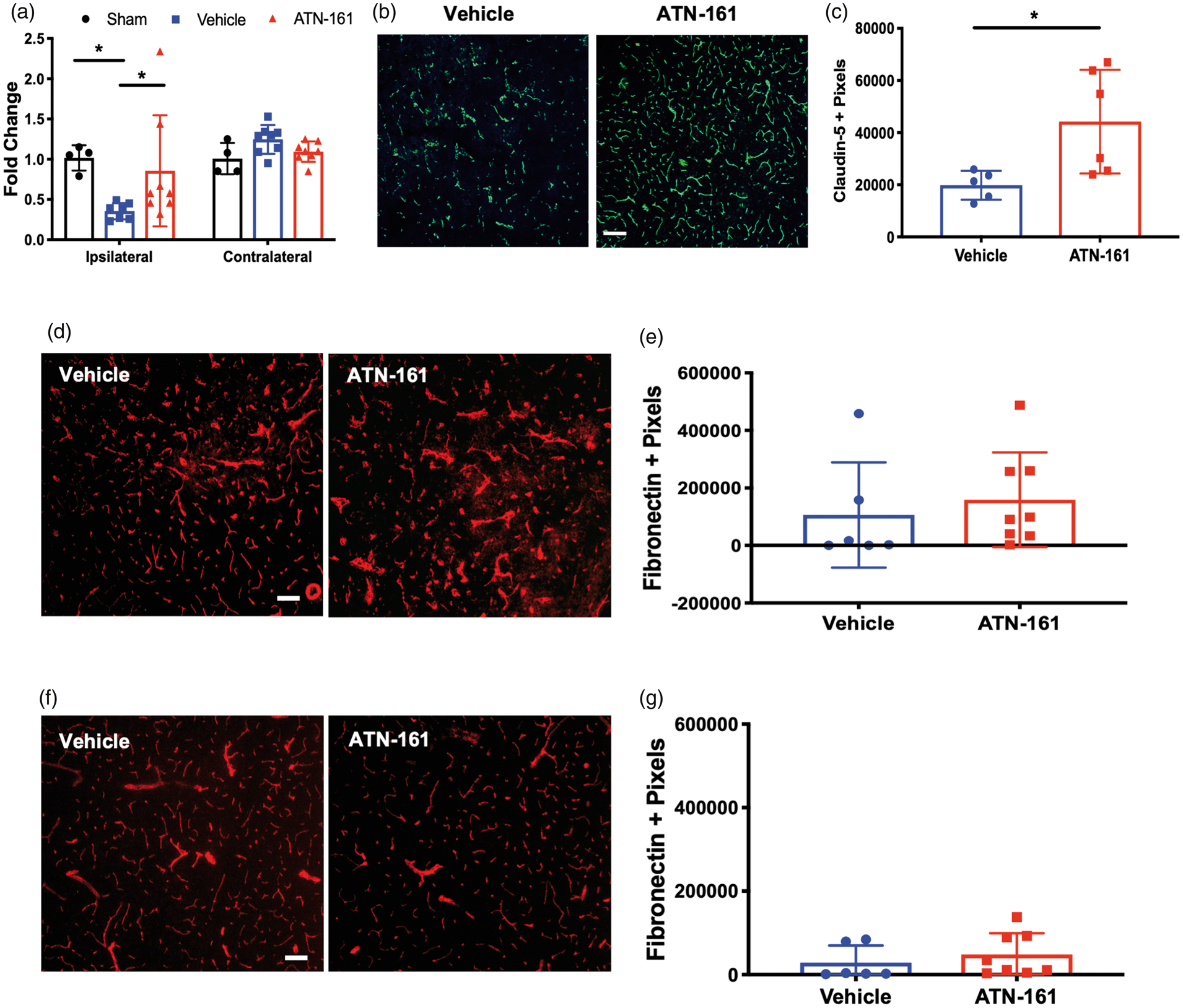
Finally, we determined that the transcription of MMP-9 (an ECM proteinase that typically increases following stroke) is increased in Vehicle-treated animals compared to Sham and ATN-161-treated mice (Figure 6(a); p = 0.0028; Sham 1.018 ± 0.1759, n = 4; Vehicle 6.761 ± 5.321, n = 7; ATN-161 2.757 ± 2.401, n = 7; 95% Cl 0.7299 to 4.232), though no significance difference was noted between Vehicle- and ATN-161-treated mice (p = 0.5238). We next determined the protein expression of MMP-9 through immunohistochemical staining. While the decrease of MMP-9 in ATN-161-treated mice remained consistent with qPCR results in the peri-infarct (Figure 6(d) and (e); p = 0.0450; Vehicle 189516 ± 170441, n = 7; ATN-161 42227 ± 180171, n = 7) with a significant decrease in MMP-9 in ATN-161-treated mice when compared with Vehicle-treated mice, no significant difference was found between Vehicle- and ATN-161-treated mice in the core of the infarct (Figure 6(b) and (c) p = 0.0734; Vehicle 443691 ± 220951, n = 6; ATN-161 232262 ± 68098, n = 7). Collectively, inhibition of α5β1 by ATN-161 stabilizes the vasculature at the site of injury (increased collagen IV, decreased MMP-9, and increased claudin-5 expression) and results in a more intact BBB (less IgG extravasation) following ischemic stroke.
ATN-161 results in reduced MMP-9 expression that is regionally dependent. (a) qPCR analysis of ipsilateral cortex at PSD3 for MMP-9 (p = 0.0028; Sham 1.018 ± 0.1759, n = 4; Vehicle 6.761 ± 5.321, n = 7; ATN-161 2.757 ± 2.401, n = 7; 95%Cl 0.7299 to 4.232). (b) Representative images of (c) 10 × magnification, scale bar = 100 µm. (c) Analysis for MMP-9 (green) positive pixels at PSD3 in the core (p = 0.0734; Vehicle 443691 ± 220951, n = 6; ATN-161 232262 ± 68098, n = 7). (d) Representative images of (e) 10 × magnification, scale bar = 100 µm. (e) Analysis for MMP-9 (green) positive pixels at PSD3 in the peri-infarct (p = 0.0450; Vehicle 189516 ± 170441, n = 7; ATN-161 42227 ± 180171, n = 7).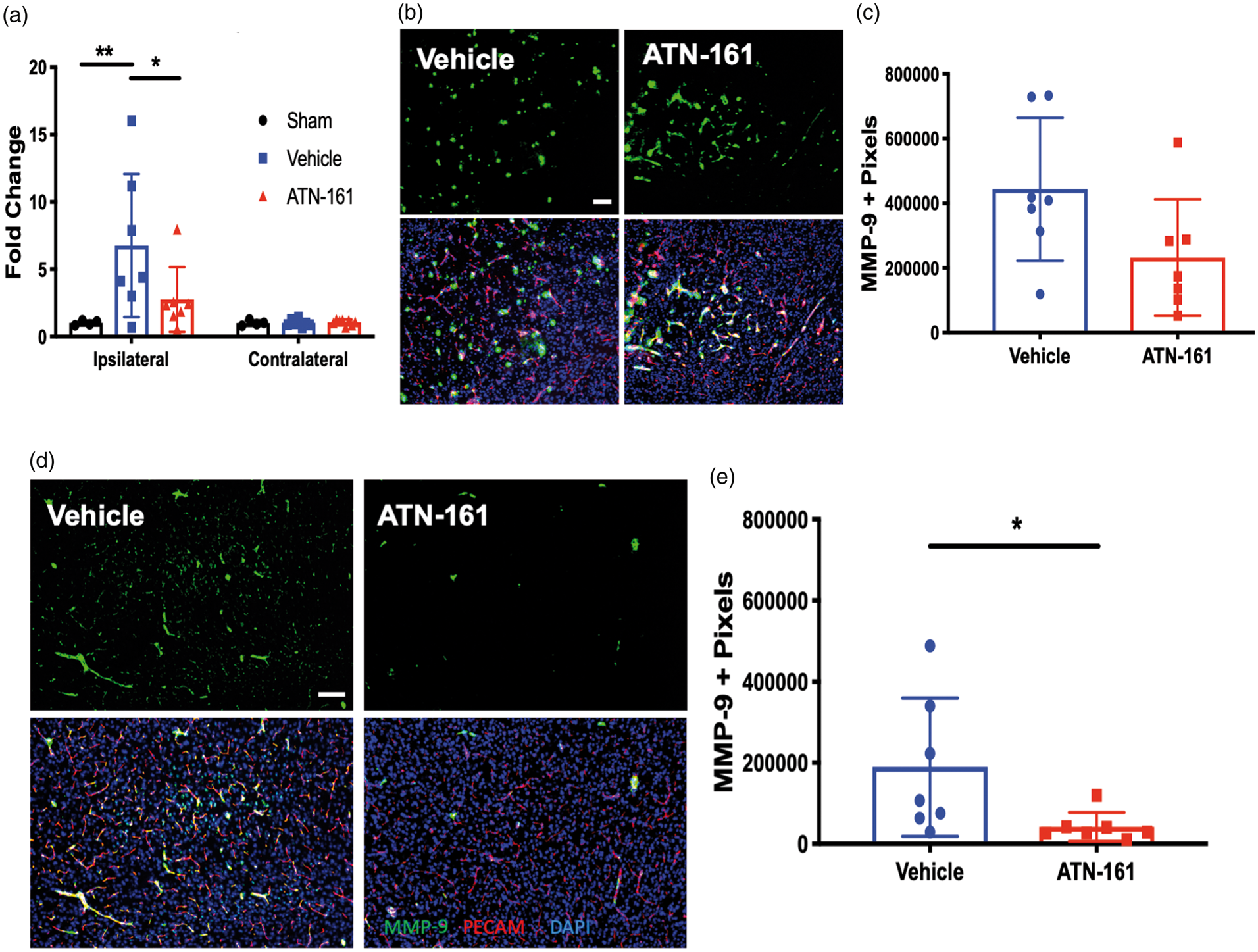
ATN-161 reduces the inflammatory response in the infarcted region
Inflammatory cascades occur acutely and last for up to a week after stroke. This involves an increase in inflammatory cytokines and neutrophil infiltration, all localized to the core site of injury.
5
Furthermore, integrins have been implicated in many inflammatory processes.
9
To determine ATN-161’s potential effects on post-stroke inflammation, we first determined its effects on the expression of the master inflammatory cytokine, IL-1 ATN-161 reduces inflammatory cells after experimental ischemic stroke. qPCR analysis of ipsilateral cortex at PSD3 for (a) IL-1β (p = 0.0006; Vehicle 27.04 ± 19.96, n = 8; Sham 1.023 ± 0.1666, n = 4; p = 0.0192, ATN-161 11.89 ± 9.235, n = 7 95% Cl 4.209 to 18.30) and (b) CXCL12 (p = 0.0022; Vehicle 0.5271 ± 0.1779, n-7; Sham 1.015 ± 0.1964, n = 4; ATN-161 0.8488 ± 0.2846, n = 8). (c) Representative images of (d) 4 × magnification, scale bar = 250 µm. 20 × images of representative core areas are outlined in red boxes. (d) Analysis for CD45 positive pixels at PSD3 (p = 0.0225; Vehicle 499674 ± 290485, n = 7; ATN-161 215451 ± 106062, n = 8). (e) A visual overview of integrin α5β1’s effects on the cerebrovascular unit following ischemic stroke. (f) A visual overview of ATN-161’s effects on integrin α5β1 and the post-stroke cerebrovasculature.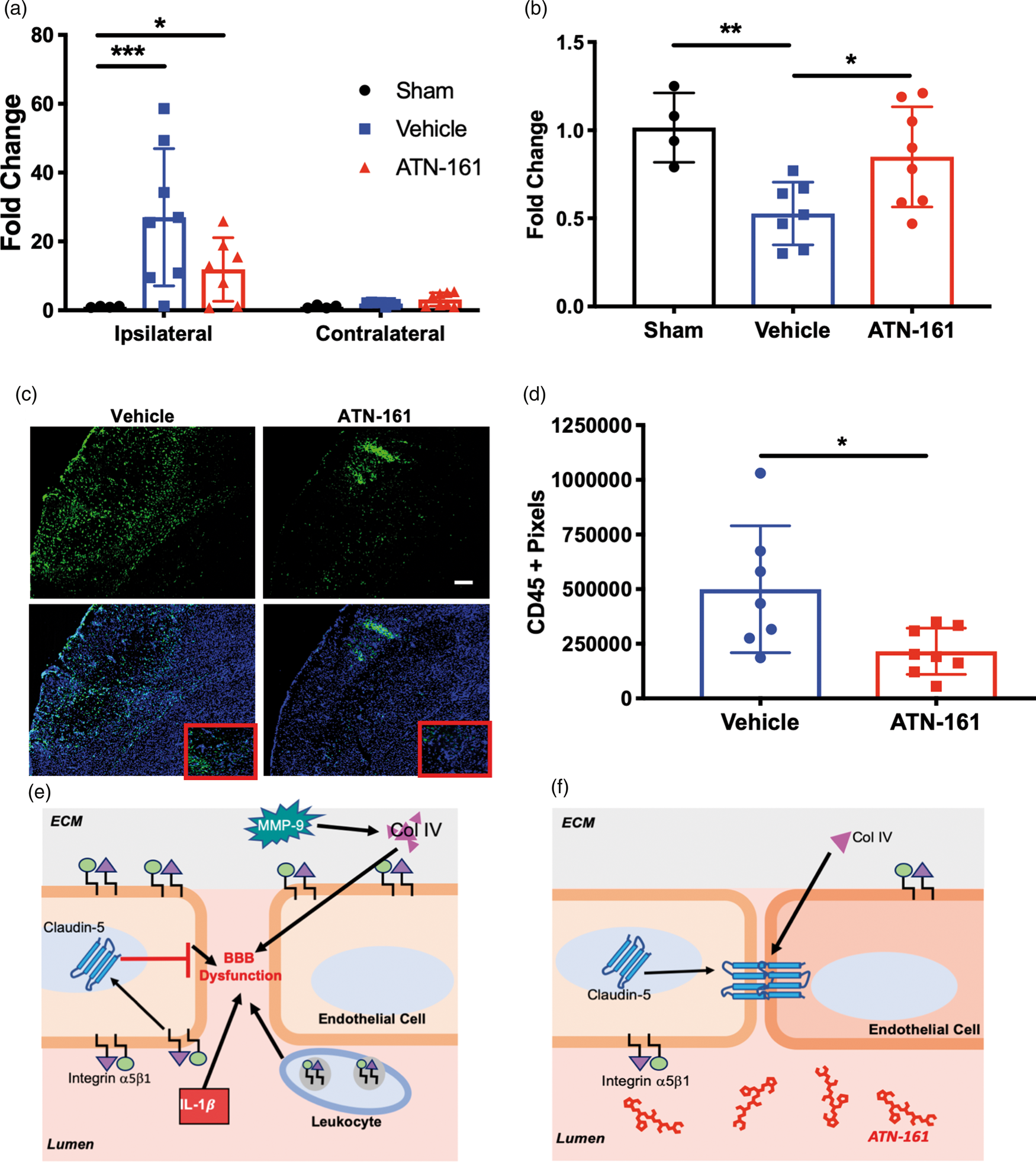
Additionally, integrins have not only been implicated in cytokine and chemokine expression, but also in migration of inflammatory neutrophils to the site of injury, a mechanism highly implicated in edema and expansion of the core.5,6 Even though neutrophils have the highest significance of all leukocytes trafficking to the site of injury, all categories of leukocytes have a role. 21 Because of this, we determined the effect of ATN-161 on CD45, a pan leukocyte marker, at PSD3 via immunofluorescence. Again, fewer infiltrating cells were found in the ipsilateral stroked brain parenchyma with ATN-161 treatment (Figure 7(c) and (d); p = 0.0225; Vehicle 499674 ± 290485, n = 7; ATN-161 215451 ± 106062, n = 8). Taken together, inhibition of integrin α5β1 with ATN-161 reduces the inflammatory load following ischemic stroke.
Discussion
Our goal was to determine the therapeutic potential of blocking the α5β1 integrin with ATN-161 in experimental ischemic stroke. ATN-161 has been previously described as an anti-angiogenic therapeutic, where the safety and efficacy of ATN-161 have been described in cancer pre-clinical research and clinical trials. 17 Here, we demonstrated that ATN-161 treatment led to decreases in infarct volume, edema, and functional deficit through inhibition of the integrin α5β1 following experimental ischemic stroke. Furthermore, we found that post-stroke ATN-161 administration stabilizes the BBB as evidenced by decreased IgG extravasation, stabilization of collagen IV and claudin-5 expression, as well as significantly fewer infiltrated leukocytes, and decreases in both MMP-9, CXCL12, and IL-1β mRNA expression through inhibition of integrin α5β1. The effects of integrin α5β1 on cerebrovasculature following ischemic stroke are summarized in Figure 7(e) and (f). Additionally, these results are consistent with our previous studies using endothelial selective α5KO mice that demonstrated that these mice were profoundly resistant to ischemic injury and BBB disruption. 14 To our knowledge, the current study is the first to demonstrate therapeutic efficacy of inhibiting integrin α5β1 in ischemic stroke.
Importantly, ATN-161-mediated effects on function only occurred after all three injections were administered. This suggests a need for continuous acute and subacute inhibition of integrin α5β1 following ischemic stroke to inhibit any intermittent vascular remodeling that may occur, a major role of integrin α5β1. 22 In fact, previous studies have shown that the acute increase of integrin α5β1 following ischemic stroke continues to increase through PSD7, 12 suggesting a possible positive feedback loop on expression and function of integrin α5β1. Thus, acute and subacute post-stroke doses of ATN-161 both inactivate and reduce the expression of integrin α5β1 in the context of this potential positive feedback loop.
“Flooding the system” with ATN-161 is clinically possible as previous clinical trials (Phase I and Phase II for tumorigenesis) showed limited side effects (dry mouth, cellulitis, and paresthesia at 1 mg/kg 10-min infusions for one out of four patients).17,23,24 Our findings further support the reported safety of ATN-161 with our treated mice exhibiting no physiological differences (Figure 1(a) to (d)) compared to their controls. ATN-161 has been shown to bind not only integrin α5β1 but also integrins αvβ3 and α6β1, though preference is for integrin α5β1 when highly expressed. 23 Furthermore, integrin αvβ3 and α5β1 expression after stroke is more acute and very brief, potentially limiting the non-specificity of ATN-161. 25
In this study, we based our three-dose administration strategy on the acute upregulation of integrin α5β1. 10 In addition, we used only one concentration of ATN-161 (1 mg/kg) based on previous research by Donate et al. which showed that ATN-161 exhibits a U-shaped dose response curve in vitro, where doses equivalent to 1–5 mg/kg produced the most significant reduction in activity (angiogenesis and tumor growth), with 1 mg/kg being the most effective. 23 Therefore, additional stroke dose–response studies should be performed. Likewise, additional studies addressing ATN-161’s therapeutic window, and potential long-term effects, need to be conducted.
Upon ATN-161 administration, we found reduced edema volume and less IgG extravasation in ATN-161 that was corroborated with strong correlations on ATN-161-treated mice that was lost with Vehicle-treated mice. The decreased IgG extravasation is indicative of reduced BBB permeability as normal paracellular transport allows free passage of 80 Da proteins, while IgG is size limited at 150 kDa.5,25,26 Collectively, these results, in addition to previous results in α5KO mice showing reduced IgG extravasation in the brain parenchyma, exposes the importance of integrin α5β1’s role in determining infarct evolution following reperfusion. By removing integrin α5β1, the amount of BBB breakdown may be predictable based on infarct volume, though the exact nature of this relationship (i.e. causative vs. correlative) requires further investigation.
It is also important to note that IgG is one of the largest probes of BBB integrity used and, therefore, does not take into account smaller BBB leaks. However, our analysis of the BBB components at the basement membrane and TJ level, areas severely compromised following ischemic stroke,
27
suggests a highly stable BBB with ATN-161 treatment. This is through a significant increase in claudin-5 (tight junction) and collagen IV (ECM) protein, while ECM degradation protein, MMP-9, decreased. These findings are in agreement with findings from Osada et al. where they determined that
Inhibition of integrin α5β1 via ATN-161 also resulted in reduced IL-1β expression and fewer infiltrating CD45 + leukocytes. Leukocytes (neutrophils, monocytes, macrophages, etc.) are highly implicated in increasing edema through adhesion of
As integrin α5β1 is pro-angiogenic, long-term inhibition may prove to be deleterious as previous studies have shown that patients with increased vascular density tend to have better stroke outcomes.26,36,37 Furthermore, studies using angiogenic promotors, particularly VEGF, resulted in increased, leaky vessels with no impact on infarct volume when induced post-stroke.9,37 Collectively, this confirms that timing is everything regarding post-stroke angiogenesis. As vascular remodeling and pruning involves disassembly of brain vessels, it leaves those vessels vulnerable to the influx of inflammatory cells, cytokines, and reactive oxygen species, which are all known to exacerbate damage. Thus, early inhibition of endothelial cell activation/angiogenesis via ATN-161 may be beneficial in prohibiting the inflammatory cascade and BBB breakdown, while the short half-life (3–4.5 h) could allow for future angiogenesis to proceed. 14 This may also allow for the intriguing possibility of combining early/acute inhibition of α5β1 integrin with its later activation to collectively limit early injury and subsequently promote angiogenic neurorepair.
Intriguingly, our results are seemingly in direct contrast to a recent study demonstrating that administration of a single IP dose of the small peptide α5β1 integrin
These seemingly contrasting results may be reconciled by pointing out several differences between the two studies. Besides the obvious differences (rat vs. mouse transient MCAO model, single vs. repeated dosing, etc.), ATN-161 is known to preferentially target and interact with the activated form of α5β1 integrin, i.e. that which is present in the brain after stroke, while PHSRN shows no predilection for any particular α5β1 integrin state.
23
Therefore, it cannot be ruled out that some, or even a significant extent of PHSRN’s post-stroke effects seen by Wu et al. could be due to off-target non-cerebral α5β1 or integrin αvβ324 integrin interactions. Indeed, Wu et al. hint at a potential PHSRN interaction in the liver as a confounding factor, but failed to show any systemic effects after PHSRN administration. A therapeutic benefit of agonizing α5β1 integrin after stroke is also inconsistent with our findings that α5β1 integrin endothelial-cell specific KO mice are profoundly resistant to ischemic brain injury.
14
Finally, previous studies have shown PHSRN to have
One limitation of our study is the use of IP administration of ATN-161 vs. the more clinically relevant IV administration. We performed IP ATN-161 administration because of technical concerns; repeated IV dosing through the tail vein in initial experiments resulted in scarring and clotting at the area of the vessel, with subsequent neurobehavioral effects and high animal attrition. This is in addition to the known neuroprotection produced from administered anestetics. 40 Another limitation of our study is that bioavailability of ATN-161, regardless of the route of administration, is currently unknown. However, it has been shown that different routes of administration of ATN-161 does experience vascular uptake. Previous studies using IV, 17 IP, 41 and intravitreous 42 injection have resulted in specific vascular effects due to changes in integrin α5β1. Furthermore, these changes are more apparent where integrin α5β1 is the most highly expressed at the target area, rather than off target effects systemically. For these reasons, we pursued an IP administration route in the preclinical setting.
Other limitations of our study include our transient MCA occlusion model which produces reproducible, but small and cortically limited infarcts which could minimize ATN-161’s therapeutic effects (stroke volumes cannot be determined as smaller than zero). Furthermore, these small infarcts show limited motor and sensory effects, thus hampering our ability to test behavioral deficits past the maximum expansion at PSD3. As previously discussed, integrin α5β1 is expressed on both endothelial cells and leukocytes; thus, further investigations into the direct role of ATN-161 on both are needed to further elucidate ATN-161’s mechanisms of action. This is in addition to investigation of ATN-161 in additional stroke models as well as in aged, female, and co-morbid animals as per the STAIR recommendations.
Conclusion
Our studies show that inhibition of α5β1 integrin with ATN-161 is beneficial following experimental ischemic stroke through reduction of infarct volume and edema, while also transiently increasing functional outcomes. Furthermore, ATN-161 stabilizes the BBB, and reduces inflammation/immune cell infiltration into the brain. Collectively, our results suggest that ATN-161 may be a promising, novel therapeutic for ischemic stroke.
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: NIH R01NS065842 to DE and GB and TL1TR001997 to GB.
Acknowledgements
The authors would like to acknowledge Joseph Goodman for his assistance with the Neuroscore.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Authors’ contribution
DE, JF, and GB conceived the experiments. DE and KS performed surgeries and behavior. DE, KS, and AT performed experiments. DL performed MRI analysis. All authors contributed to writing and editing the manuscript.
