Abstract
A constant challenge in experimental stroke is the use of appropriate tests to identify signs of recovery and adverse effects linked to a particular therapy. In this study, we used a long-term longitudinal approach to examine the functional brain changes associated with cortical infarction in a mouse model induced by permanent ligation of the middle cerebral artery (MCA). Sensorimotor function and somatosensory cortical activity were evaluated with fault-foot and forelimb asymmetry tests in combination with somatosensory evoked potentials. The stroke mice exhibited both long-term deficits in the functional tests and impaired responses in the infarcted and intact hemispheres after contralateral and ipsilateral forepaw stimulation. In the infarcted hemisphere, reductions in the amplitudes of evoked responses were detected after contralateral and ipsilateral stimulation. In the intact hemisphere, and similar to cortical stroke patients, a gradual hyperexcitability was observed after contralateral stimulation but no parallel evidence of a response was detected after ipsilateral stimulation. Our results suggest the existence of profound and persistent changes in the somatosensory cortex in this specific mouse cortical stroke model. The study of evoked potentials constitutes a feasible and excellent tool for evaluating the fitness of the somatosensory cortex in relation to functional recovery after preclinical therapeutic intervention.
Introduction
Stroke is a leading cause of death and a major cause of disability in the Western world. Embolisms of the cerebral arteries beyond the circle of Willis, account itself for 15–30% of strokes. 1 Currently, early revascularization by intravenous thrombolysis or intra-arterial mechanical thrombolysis is the only accepted treatment to promote early reperfusion to improve survival and prevent long-term disability in patients. Preclinical models are needed to investigate the pathogenesis of stroke and the development of neuroprotective or restorative therapies. Animal models of focal ischemia induced by the occlusion of the MCA have been developed to reproduce the most frequent form of stroke in humans. 2 The proximal occlusion of the MCA results in focal ischemia that causes an extensive damage in the cortex and striatum. By contrast, the occlusion of the distal part of the MCA (dMCAO) causes damage that is primarily restricted to the frontal and parietal cortices, which results in infarct size reproducibility across animals;3,4 however, the sensory deficits are relatively minor, which limits the evaluation of the effects of chronic treatments for stroke and the ability to examine possible long-term side effects.
In this context, the use of electroencephalography (EEG) and somatosensory evoked potentials (SSEPs) has been shown to be useful for exploring the integrity of the sensory pathways of the central nervous system. The value of SSEPs in the prediction of functional recovery after stroke has also been examined and found to correlating well with the subsequent level of disability and functional outcome. 5 In cortical stroke models that affect the somatosensory area, SSEPs constitute an advantageous methodology for examining spontaneous and therapy-induced functional recovery, specifically when conventional behavioral tests are not sensitive enough. In rats submitted to focal ischemia, SSEPs have served to evaluate both the efficiency of MCA occlusion during the surgery6,7 and the long-term sensory deficits that follow various treatments applied during the acute and chronic phases of stroke. 8 However, the use of SSEPs has been more restricted in mouse models, despite the fact that genetically modified animals permit the exploration of the contributions of different genes to the physiopathology of stroke and their implications in neuroprotection and brain repair mechanisms. In few recent studies, the activity of infarcted cortex has been examined in mice with techniques with good temporal resolution such as electrophysiology and voltage-sensitive dye (VSD) imaging.9,10 Chronic electrophysiological recordings enable examination of the short- and long-term progression of somatosensory function in the same animal even when the field potentials are relatively low in amplitude as occurs, for example, after ipsilateral stimulation. Therefore, the use of electrophysiological recordings together with brain imaging might be useful for exploring the mechanisms of functional recovery that are associated with neuroprotection, neurorestoration, and cortical reorganization after different pharmacologic or stem cell transplantation approaches.
The aim of this study was to examine the feasibility of the use of long-term longitudinal electrophysiological recordings in mice to analyze the dynamic progression of somatosensory function in a specific model of dMCAO. After stroke, the infarcted mice exhibited permanent failures in the fault-foot and forelimb asymmetry tests, and greater deficits in both of these behavioral tests were correlated with greater dysfunction in the somatosensory cortex. Our results highlight the sensitivity and reproducibility of SSEPs in the exploration of post-ischemic mechanisms of cortical functionality in stroke and after preclinical therapeutic interventions.
Materials and methods
Additional method details can be found in supplementary information (available on the JCBFM website; see the supplementary information link at the top of the online article).
Animals
Adult male C57BL/6 mice (20–28 g body weight; 8–10 weeks old) were bred and housed in the animal facility of the Center for Biomedical Technology. The animals were housed in individual cages with free access to food and water in an animal room with a controlled temperature and a natural light cycle. Daily routines were performed between 7 a.m. and 4 p.m. by authorized personnel. The experimental setup of this study is illustrated in Figure 1 and is also described in the supplementary information. All procedures were approved by the Technical University of Madrid (Ethical Committee for Animal Research) before the commencement of any of the studies. All procedures were performed under the Spanish Regulations for animal experimentation (Law 53/2013) and according to the ARRIVE (animal research: reporting in vivo experiments) guidelines.
Experimental approach for the examination of the temporal progression of somatosensory cortical activity and sensorimotor function in mice submitted to permanent distal MCA occlusion. Somatosensory evoked potentials (SSEPs) and two behavioral tests (grid-walking and cylinder tests) were used to evaluate the influence of cortical infarction on the progression of sensorimotor deficits and the subsequent recovery. The functional tests and the SSEPs recordings were performed two weeks after implantation of epidural electrodes (stereotaxic surgery) to obtain the baseline scores. One week later, we induced the cortical infarction in the same mice by occlusion of the distal part of the middle cerebral artery (dMCAO). After stroke, the behavioral tests were performed at 1, 3, 6, and 8 weeks post-stroke, while the SSEPs were performed every week during the first month after ischemia, and every two weeks during the last month of study.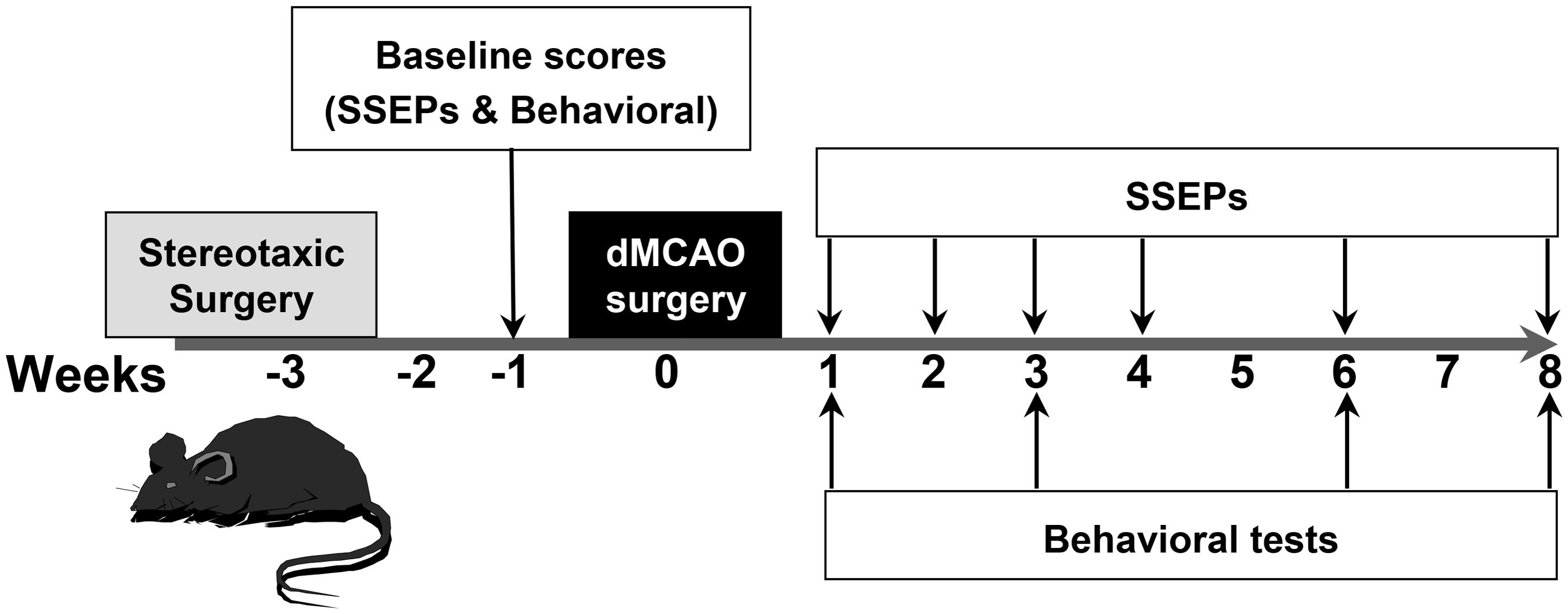
Stereotaxic surgery and SSEPs recordings
Animals were anesthetized with ketamine (100 mg/Kg) and xylazine (10 mg/kg) prior to the stereotaxic implantation of subdural electrodes for recording of SSEPs. The mice were secured in a stereotaxic frame (David Kopf Instruments, California, USA) and under aseptic conditions a skin incision was made and four small holes drilled through the skull. The electrodes (0.2 mm diameter) were made with polyimide-insulated stainless steel wires (PlasticsOne, Virginia, USA). Two electrodes were implanted in the right and left somatosensory cortices (±2.0 mm lateral and + 0.5 mm rostrocaudal from bregma, depth 0.5 mm). Two screws were placed over the visual cortex of both hemispheres and served as the indifferent and ground electrodes (±2.0 mm lateral and −3 mm rostrocaudal from bregma). The electrodes were secured in place with cyanoacrylate-based glue and dental acrylate (Duraly, Illinois, USA). The animals were allowed two weeks to recover from surgery before experiments started. The electrodes placement was assessed following the recording of SSEPs from both hemispheres after ipsi- and contra-lateral forelimb stimulation to obtain the baseline scores. One week later, focal cerebral ischemia was induced by direct occlusion of the distal MCA as described below. Brain histology was also performed in some mice to examine the relative position of the electrodes in the somatosensory cortex (Supplementary Figure S3).
Electrical stimulation and SSEPs recordings were performed under anesthesia with ketamine (100 mg/kg) and xylazine (10 mg/kg). The SSEPs were recorded during the first 10–25 min after i.p. injection of anesthesia, a time period of stable SSEPs amplitudes (Supplementary Figure S4). The depth of anesthesia was assessed as a function of the loss of reflex tail movement after pinch and the amplitude of the EEG signal. For peripheral stimulation, a tape was used to affix a pair of silver electrodes over the ventral surface of forelimb (distal to the elbow) along the median nerve pathway. The stimuli were applied with a frequency of 1 Hz. In all the cases, the magnitude of applied electrical stimuli was twice the current level necessary to obtain a supramaximal motor response (1–2 mA; 0.5 ms duration). This current level is defined as the value of intensity able to elicit a visually evident and specific movement in the forepaw (palmar flexion of wrist), which is not significantly modified after increasing the stimulation intensity. Signals were amplified (×103) and filtered (band-pass, 10–2.000 Hz) using a portable electromyography (EMG)-evoked potentials (EP) device (Micromed, Mogliano Veneto, Treviso, Italy); 30 to 60 responses were averaged for each trial, and the procedure was repeated three times for each forepaw.
Permanent MCA occlusion
Focal cerebral ischemia was induced by direct occlusion of the distal part of the middle cerebral artery (MCA) as we have described previously. 11 Briefly, mice were anesthetized with ketamine (100 mg/kg) and xylazine (10 mg/kg). A vertical skin incision (0.5 cm) was made between the right eye and ear under a dissection microscope (Amscope, California, USA), and the temporal muscle was cut and separated to expose the right lateral aspect of the skull. The MCA was identified through the semi-translucent skull, and a burr hole (0.8 mm diameter) was made using a microdrill. After identification, the dura mater was resected, and the artery was ligated distal to the lenticulostriate branches. Sham operated animals were subjected to an identical procedure with the exception that MCA was not ligated.
Infarct volume and area measurement
The infarcted area was evaluated with the 2, 3, 5-triphenyltetrazolium chloride (TTC; Sigma, St. Louis, MO, USA) staining. 12 Briefly, the mice were sacrificed by decapitation 24–72 h after MCA ligation. The brains were removed and sliced in 1-mm-thick coronal sections by using a brain matrix (WPI, Florida, USA). The brain slices were immersed in a solution containing 1% of TTC in phosphate-buffered saline (PBS) at room temperature for 10 min and fixed with 4% paraformaldehyde solution in PBS. All coronal sections were digitalized, and the area of cerebral damage was traced with a standard computer-assisted image analysis technique (Image J software; NIH). The volumes of the healthy and ischemic tissues in both hemispheres were calculated by measuring the infarcted and the undamaged areas on separate slices, multiplying the areas by the slice thickness, and summing the values for all slices. The infarct percentages were calculated as percentages of the contralateral (intact) hemisphere to correct for edema.
Behavioral tests
Cylinder test
The cylinder test has been used to demonstrate long-term functional impairments in rats and mice that have been subject to cortical ischemia affecting the somatosensory area.13,14 To evaluate forepaw asymmetry, the mice were placed in a clear Plexiglas cylinder (15 cm in height × 10 cm in diameter). During normal vertical exploration, the animal supports its weight on either or both of its forepaws. Twenty-five contacts were counted and recorded per trial and the paw or paws used for each contact were scored. Blinded evaluation was performed on video recordings by one investigator (L.P.). The mice were placed in the cylinder for a maximum of 5 min. The laterality index was calculated as (Contacts (Right)-Contacts (Left))/ (Contacts (Right) + Contacts (Left) + Contacts (Right and left)); positive scores denote preferential use of the right (unaffected) paw and negative scores indicate preferential use of the left paw. 14
Fault-Foot test
The grid test was performed to evaluate the abilities of the mice to place the forepaws on a wire while moving along a grid floor during spontaneous exploration. 15 We analyzed the frequencies of slips with both forepaws, which are related to the mice’s abilities to accurately grasp the grid wires. The mice were placed on a circular grid with 13 × 13 mm2 grid squares and allowed to freely explore for 5 min. The animal behaviors were videotaped and the total numbers of right and left forepaws steps during locomotion (typical range of 200–400 total steps) and the total number of foot faults for each forelimb were scored. Blinded evaluation was performed on video recordings by L.P. A foot fault was considered positive when the respective forepaw fell or slipped between the rungs causing the animal to lose balance.
Statistical analysis
The R package and SigmaPlot (Systat, Germany) were used for the statistical analyses. All values are expressed as mean ± the standard error of the mean (S.E.M.). To examine significant differences in the volume of infarct at different time points after surgery, we performed a one-way analysis of variance (ANOVA) test. The temporal pattern of cortical activity (SSEPs) and sensorimotor function (behavioral tests) was analyzed using a two-way ANOVA, followed in case of significant differences by Tukey’s post hoc test. The ANOVA analysis was performed using deficits, amplitudes or inter-hemispheric ratios as dependent variables, and time and groups (sham & stroke) as independent variables. For SSEPs responses after contralateral stimulation, multiple comparisons for differences between different weeks and the baseline values were determined by Tukey’s test. For behavioral testing and SSEPs responses after ipsilateral simulation, differences between groups (for behavioral tests) or between hemispheres (for ipsilateral potentials) were analyzed for each temporal point by Tukey’s post hoc test. Correlations between different variables were determined using the Pearson product-moment correlation coefficient. p-values below 0.05 were considered statistically significant.
Results
Distal permanent MCAO
Permanent occlusions of the MCA were performed at the distal level and resulted in relatively variable infarct sizes between different animals that involved the cortical areas of the distal territory of the MCA without affecting the subcortical regions as indicated by TTC staining (Figure 2(a)). The analyses of the coronal brain sections indicated that the infarcted areas were greater in the sections that primarily contained the primary somatosensory cortex (Figure 2(b)). The calculations of infarct volumes corrected for swelling due to edema that were used to examine the temporal progression of the lesions induced by dMCAO revealed no significant changes in the interval from 24 to 72 h (Figure 2(c)).
Evaluation of brain damage after middle cerebral artery occlusion (MCA). (a) Representative TTC staining images of coronal sections 24 hours after MCA occlusion. The infarct area (in white) is mainly localized to cortical regions (scale bar represents 0.5 cm). (b) Regional distribution of the infarcted areas in coronal sections along the rostrocaudal axis as measured from bregma. (c) Evolution of the infarct volume across time after focal ischemia induced by MCA occlusion. The infarct volumes are represented as percentages of the total volume of the contralateral intact hemisphere. The data are shown as the means ± the standard error of the mean (SEM) of one experiment for each temporal point (24, 48, and 72 h) with a minimum of 5–7 mice for experiment. No significant differences were found in the volume of infarct across the time (one-way ANOVA test).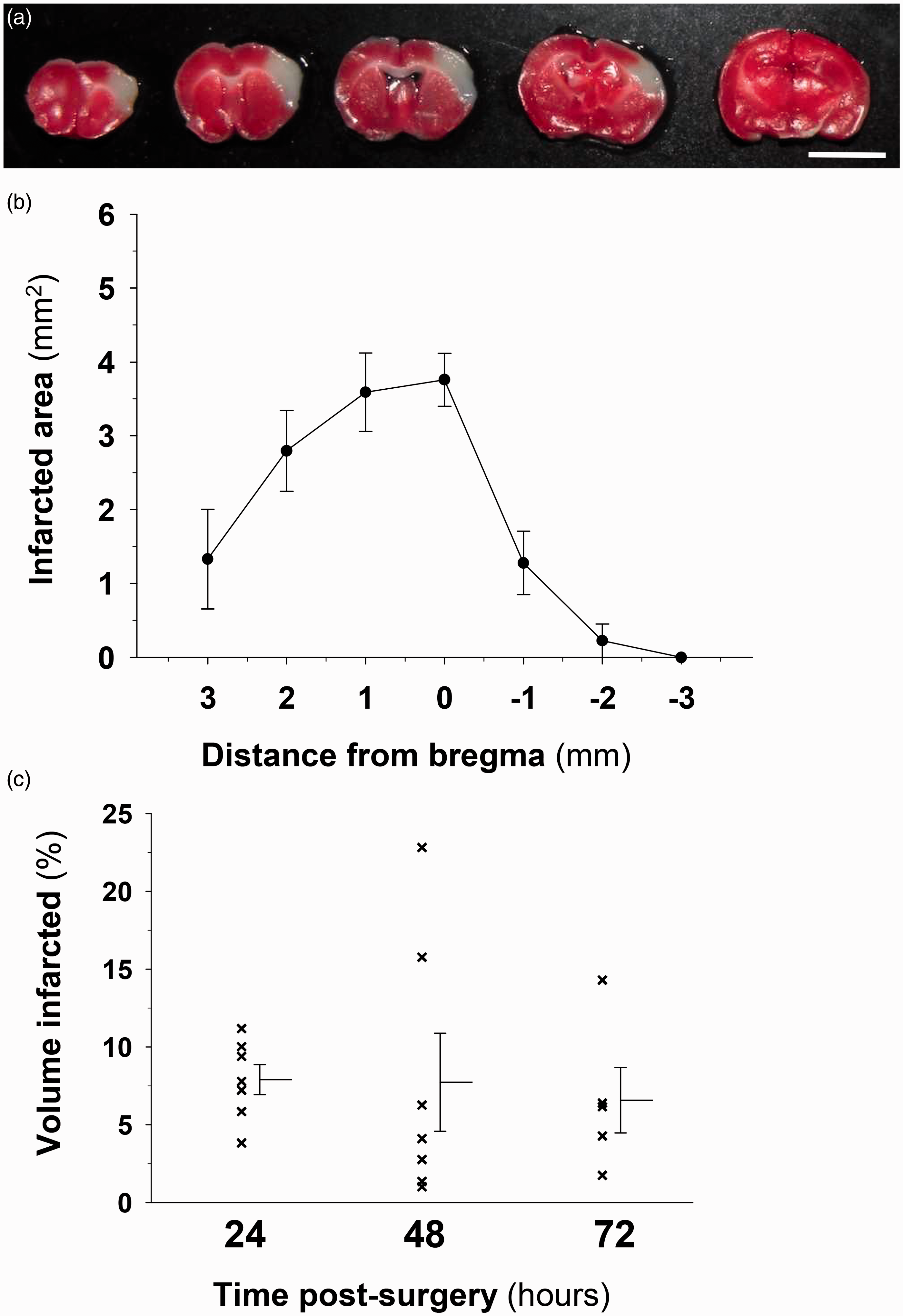
Behavioral tests of somatosensory and motor function
In the initial 6–12 h following surgery, the ischemic mice exhibited contralateral hemiparesis, but 24 h after focal ischemia, we did not detect obvious functional differences between the ischemic and sham animals. We performed a re-evaluation of different functional tests that have previously used in the models of focal and global ischemia, specifically the cylinder and the grid tests. To avoid a training effect of testing itself especially in the case of the grid test (supplementary Figures S1 and S2), both behavioral tests were performed in sham and ischemic mice in spaced weeks apart (Figure 1). Under these conditions, no habituation was observed in the sham mice across the time after surgery (Figure 3(c) and 3(d)).
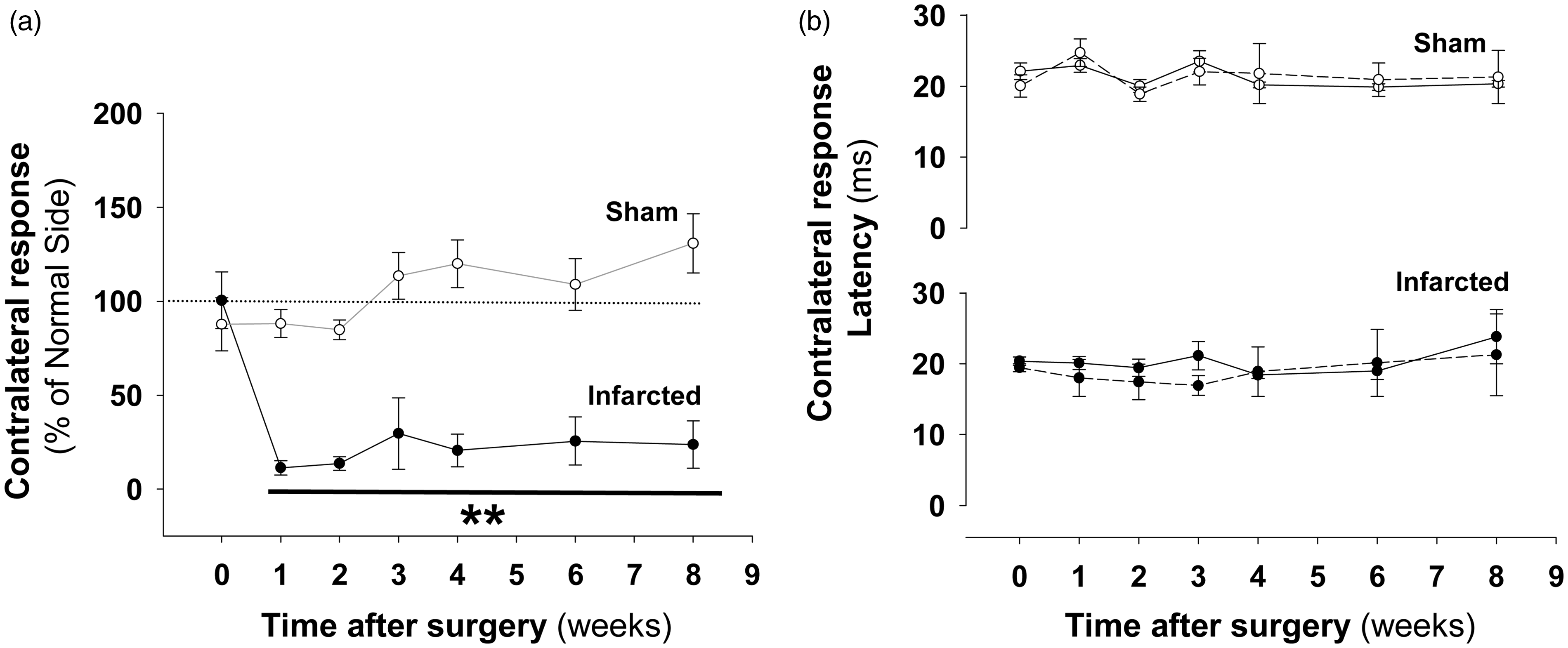
Focal ischemia induced by distal MCA occlusion causes long-term functional deficits in mice. (a) Cylinder test. Use of the contralateral (left paw), ipsilateral (right paw) ,and both forelimbs in the cylinder test after MCA occlusion expressed as percentages of total paw contacts. (b) Asymmetry in forelimb use in the cylinder test was scored as (Right – Left)/(Right + Left + Both). Higher positive values of the laterality index indicate preferential use of the right, unaffected limb and therefore sensorimotor impairment. The empty bars represent the sham group, and the solid bars represent the ischemic mice. For the grid-walking test, (c) and (d) show the percentages of footslips for the left and right forepaws, respectively. The empty bars represent the sham group, and the solid bars represent the stroke mice. The data are shown as the means ± the SEM of two independent experiments with a minimum of 6–10 mice per group; sham or stroke; and temporal point. The asterisks denote significant differences between groups for each temporal point (two-way ANOVA followed by Tukey’s test; *p < 0.05; **p < 0.01).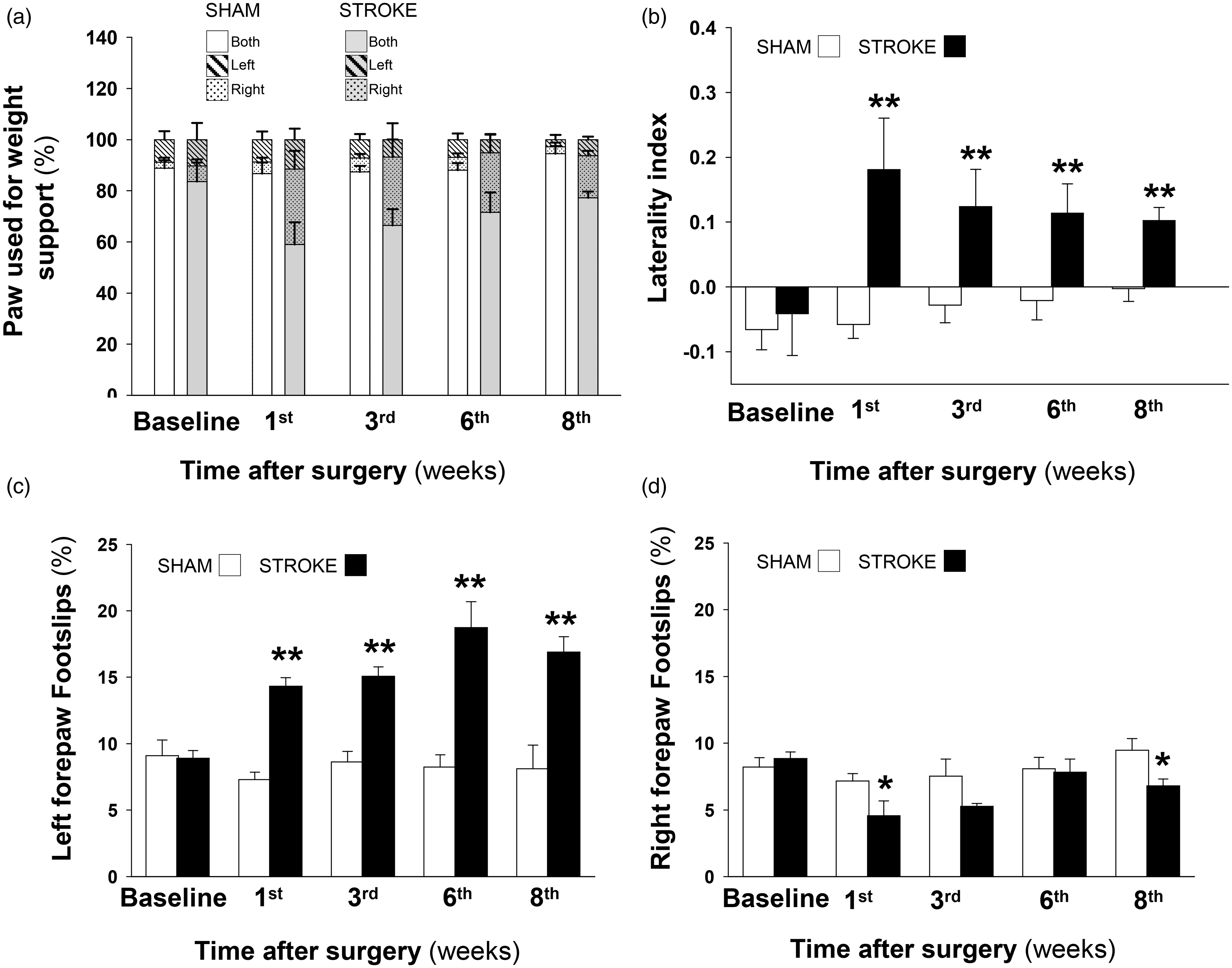
In our study, the cylinder test revealed the preferential use of the paw ipsilateral to the infarcted hemisphere, while the sham animals exhibited no preference regarding the use of a particular paw across time after the craniotomies (Figure 3(a) and (b)). In the ischemic mice, laterality was maximal one week after stroke and gradually recovered and returned to subnormal levels in the subsequent eight weeks (Figure 3(b)). In the walking grid test, which allows for the discrimination of ischemic from control mice based on different levels of motor coordination and forepaw asymmetries, the percentage of footslips using the left forepaw (contralateral to the damaged hemisphere) was significantly higher and remained elevated at eight weeks in the stroke compared to the sham animals (Figure 3(c)). In the stroke group, the number of footslips with the right forepaw was initially reduced at one week and slowly returned to the basal values; in the sham animals, the right footslips did not vary with time in this study (Figure 3(d)).
Contralateral and ipsilateral EPs
The activity of the somatosensory cortex, including the forepaw representation area, was examined with SSEPs that were firstly recorded at baseline (two weeks after the implantation of the electrodes). In response to electrical stimulation in each forepaw, several components were present in the respective contralateral hemisphere, but a large negative wave (N1) was particularly obvious (Figure 4(a), left traces). This prominent wave reached its maximal amplitude (peak) 20 ms after the constant-current stimulus and likely reflected the activities of different cell populations in the somatosensory cortex.
16
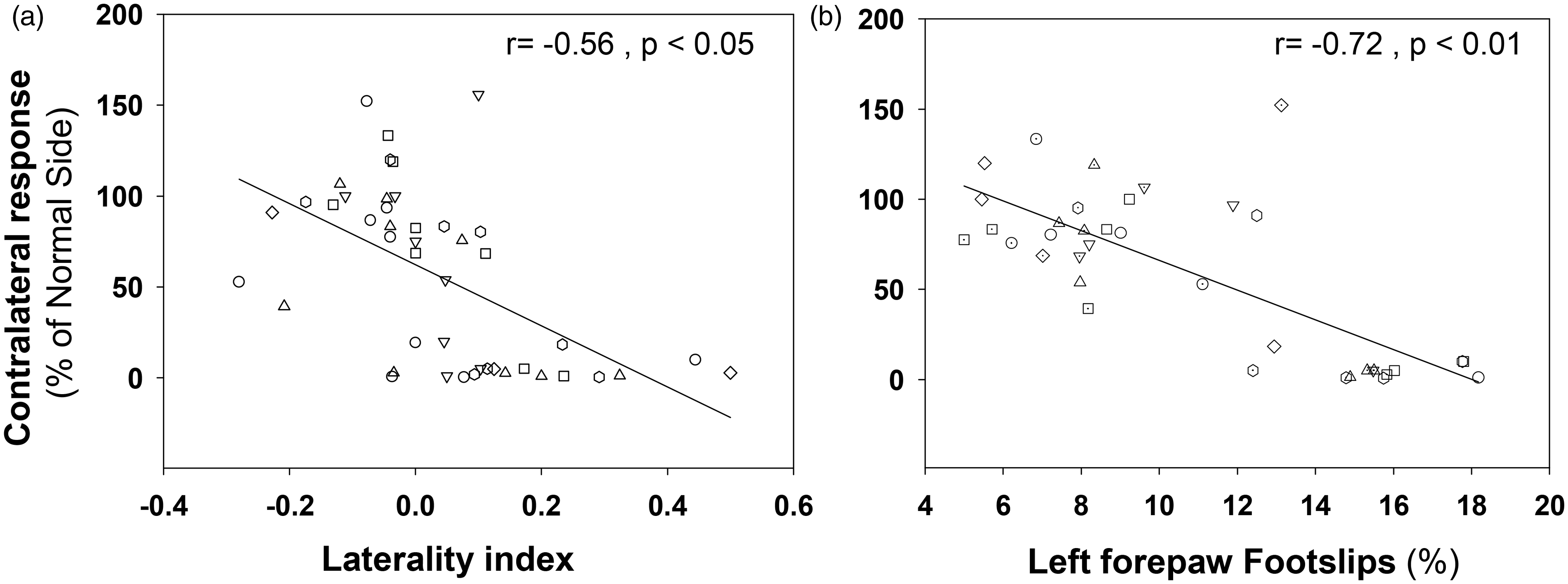
We analyzed the evolution of this somatosensory response (N1 peak) across eight weeks post-infarction and relative to the baseline condition. The sham mice exhibited similar potential amplitudes in both hemispheres in response to contralateral stimulation both at baseline and after craniotomies (Figure 4(a), traces in panel i, and Figure 4(c)). In the ischemic mice, the response of the damaged hemisphere to contralateral stimulation involved a prolonged and marked reduction of the N1 peak amplitude (Figure 4(a); black traces in panel ii and Figure 4(b); solid circles) compared to the corresponding contralateral response in the healthy hemisphere, which exhibited a progressive and substantially greater response compared to the baseline condition (Figure 4(a); blue lines in panels ii and Figure 4(b); empty circles). The majority of the infarcted mice exhibited a complete abolition of the EPs in the injured hemisphere that was irreversible across time (Figure 4(a), black traces in panel ii). Exceptionally, 2 of 15 mice (N = 3 experiments) exhibited a progressively increasing response that reached amplitude values that were still considerably below those recorded in the baseline condition (Figure 4(a), black traces in panel iii), responses that were associated with progressive better performance in both behavioral tests (data do not shown). Interestingly, in the ischemic mice, a positive correlation was found between the amplitudes of SSEPs in the intact and infarcted hemispheres (Pearson correlation; r = 0.52, p < 0.01). In summary, after infarction and in response to contralateral stimulation, a progressive increment in the N1 peak amplitude was observed in the intact hemisphere, and a marked amplitude reduction was detected in the damaged hemisphere (Figure 4(b)); in the sham animals, the amplitudes of the responses to contralateral stimulation were similar between hemispheres and no significant changes were observed with the post-craniotomy time (Figure 4(c)). These results were translated into ratios (SSEPdamaged/SSEPintact) close to 100% in the sham animals; i.e. the values of the N1 peak amplitudes were similar among hemispheres, and substantial reductions in these ratios were observed in the group of ischemic animals (Figure 5(a)). The reduction in the N1 peak with post-ischemia time was not accompanied by changes in the latency relative to the pre-ischemic condition (the latency values were calculated from the amplitude peaks > 10 µV; Figure 5(b)). To examine the relationship between the sensorimotor function evaluated by the cylinder and grid tests and the somatosensory cortical activity examined by SSEPs, we performed a Pearson correlation analysis including data from sham and stroke mice. The greater deficits in the two tests were associated with the lower ratios of SSEPs responses (Figure 6(a) and (b)). When this analysis was performed exclusively in the stroke group, we found a significant correlation only in the case of the grid test (grid test r = −0.89; p < 0.01, cylinder test r = −0.39; p = 0.17). In agreement with these results, the activity of the intact hemisphere was related with the degree of sensorimotor impairment, finding that the higher amplitudes of SSEPs responses correlated with the lower number of footslips of the affected paw in the grid test (r = −0.55, p < 0.01). These results support the sensitivity of the grid test to identify signs of sensorimotor deficits that were associated with significant changes of evoked activity in the somatosensory cortex of both hemispheres.
Somatosensory evoked potentials (SSEPs) were impaired after distal MCA occlusion. (a) Examples SSEPs recorded from the left and right cortices in response to contralateral forepaw stimulation. The temporal evolution of the EP amplitudes in the sham (panel i) and ischemic mice (panel ii & iii) in the baseline condition and two, four, and six weeks after surgery are shown. The black and (Continued) Figure 4. (Continued) blue lines represent records from the affected and intact hemispheres, respectively. Normalized EP response amplitudes (N1 voltage peak) after contralateral stimulation of the infarcted (b) and sham mice (c) across time post-surgery. The blue and black lines represent values from the intact and affected hemispheres, respectively. The data are shown as the means ± the SEM of three independent experiments with a minimum of 10–15 mice per group; sham or stroke; and temporal point. The asterisks denote significant differences with respect the corresponding baseline values (two-way ANOVA followed by Tukey’s test; *p < 0.05). Analysis of somatosensory responses after contralateral stimulation in mice with cortical infarctions. (a) Ratio of the contralateral responses in the sham (empty circles) and stroke (solid circles) mice across time post-surgery. The ratios are expressed as EP amplitude in the damaged hemisphere with respect the normal hemisphere (SEPdamaged/SEPintact). (b) The latencies of the N1 peaks in response to contralateral stimulation across time in the sham (empty circles) and infarcted (solid circles) mice. The solid and dashed lines represent the latencies for the operated and non-operated hemispheres, respectively. The data are shown as the means ± the SEM of three independent experiments with a minimum of 10–15 mice per group; sham or stroke; and temporal point. For every group, the asterisks denote significant differences with respect the corresponding baseline values (two-way ANOVA followed by Tukey’s test; **p < 0.01). Sensorimotor functional deficits correlate with reduced activity in the somatosensory cortex. Pearson correlation analyses of the ratio values (SSEP response) obtained after contralateral forepaw stimulation and the scores in the cylinder (a) and grid-walking (b) tests. The laterality index and percentage of left forepaw footslips were the parameters from the respective tests that were used and are plotted against the ratio values (different symbols are used to unmask similar values). Equivalent numbers of mice from the sham and ischemic groups, at one, three, six, and eight weeks after the craniotomies were included in these analyses. The correlation values are shown at the top of each plot. Significant correlation values were observed in both functional tests but were particularly strong in the grid-walking test.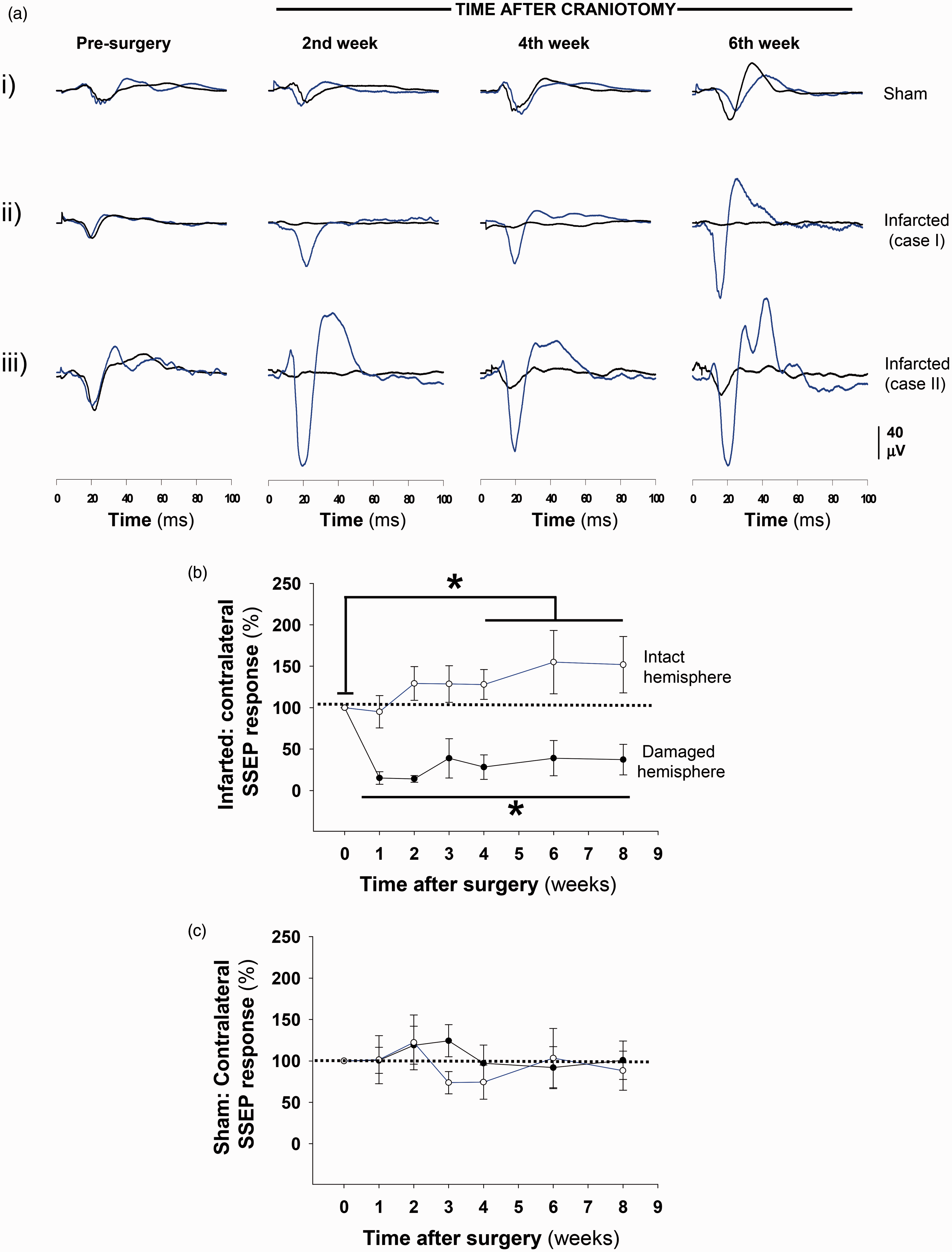
Amplitudes and latencies of N1 wave of contralateral and ipsilateral SSEPs in sham mice.
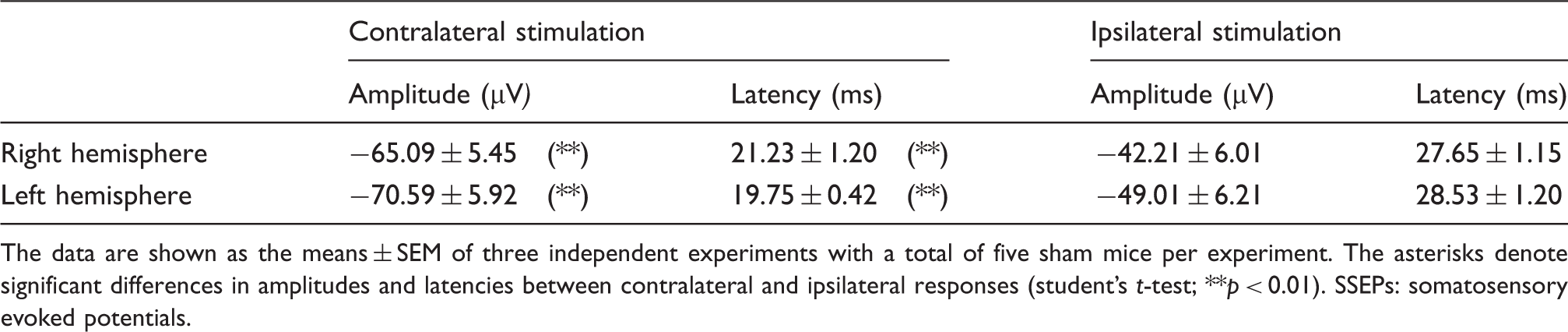
The data are shown as the means ± SEM of three independent experiments with a total of five sham mice per experiment. The asterisks denote significant differences in amplitudes and latencies between contralateral and ipsilateral responses (student’s t-test; **p < 0.01).
SSEPs: somatosensory evoked potentials.
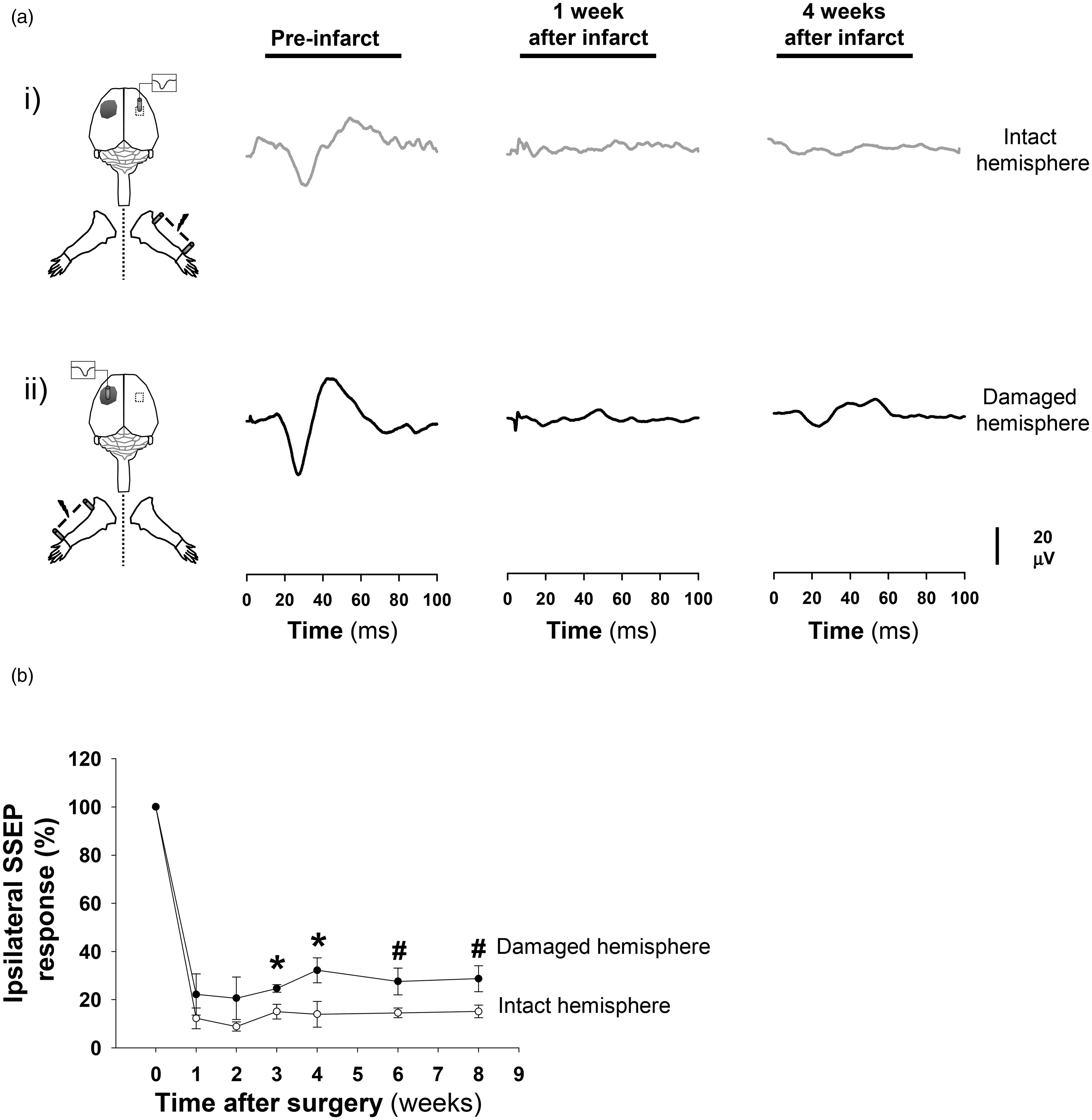
Potentials evoked by ipsilateral stimulation are impaired in mice after dMCAO. On the left side, cartoon showing the experimental setup with the position of electrodes for ipsilateral recordings in relation with the infarcted area (gray spot) and the stimulated forepaw. (a) Examples SSEPs recorded from both hemispheres after ipsilateral stimulation showing the temporal progression of the ipsilateral EP amplitude in the intact (panel i, gray line) and affected (panel ii, black line) hemispheres at baseline and at one and four weeks after dMCAO. Note that, after stroke, all responses in both hemispheres after ipsilateral stimulation were nearly completely abolished. (b) Normalized amplitude of the EP response (N1 voltage peak) after ipsilateral stimulation in the infarcted mice across time post-ischemia. The empty and solid circles represent the values from the intact and damaged hemispheres, respectively. The data are shown as the means ± the SEM of three independent experiments with a minimum of 10–15 mice per temporal point. The asterisks denote significant differences between the hemispheres for each temporal point (two-way ANOVA followed by Tukey’s test; *p < 0.05; #p < 0.08).
Discussion
Our results show the existence of profound functional changes in the two hemispheres after unilateral cortical stroke induced by a dMCAO approach. In the cylinder and grid tests, the infarcted mice exhibited long-term deficits of varying degree, although some functional recovery was observed in the case of the cylinder test. A strong correlation was determined between the electrophysiological responses of the intact and infarcted somatosensory cortices and the degree of functional impairment.
In agreement with previous work, the resulting infarcts were small and localized to the sensorimotor cortical region, and brain swelling was not obvious.3,4,11 The infarct areas were consistently restricted to regions anterior to bregma and concentrated primarily in the somatosensory cortex in the barrel and forelimb receptive fields; the primary motor cortex was less affected. A similar regional distribution has previously been demonstrated in the mouse models of the distal occlusion by electrical coagulation. 4
We have studied the short- and long-term effects of focal ischemia on sensorial and motor skills by combining functional tests and SSEPs. In the cylinder test, the laterality index in the ischemic mice was maximal at the first week after MCA occlusion and subsequently decreased over time towards values of sham mice, which was most likely associated with partial and slow recovery; however, this tendency did not reach statistical significance. Thus, in our hands, the cylinder test seemed to be appropriate for the examination of functional impairments as has been previously reported with this dMCAO model14,17 and in photothrombotic stroke models.9,13 In the foot-fault test, with respect to the sham group, we detect an increased percentage of slips in the paw contralateral to the infarcted hemisphere for at least eight weeks post-infarct, as has been previously reported in rats with cortical infarction over four weeks after surgery 18 and more recently in mice over eight weeks in a cortical photothrombotic stroke model. 13 In our study, the SSEPs of the infarcted hemisphere were essentially impaired in the majority of animals, and there was no statistical evidence of functional recovery; these observations might justify the existence of the persistent deficits in sensorimotor function exhibited in both tests, especially in the grid test. The stroke mice did not recover when they performed the grid test, but in the cylinder test, a trend of progressive recovery to a subnormal level was observed and might have been related to the fact that the cylinder test involves an easier task than that of the grid test for which most likely required greater sensory and motor coordination. Alternatively, the cylinder test might not discriminate between ischemic animals with different degree of sensorimotor impairment after the dMCAO approach.
Before ischemia, negative deflections (N1 wave) were detected in both hemispheres in response to forepaw contralateral stimulation. We believe that the N1 peak primarily represents the activity of the forepaw representation area in the pre-stroke condition, because this region is primarily activated 20 ms after contralateral forepaw stimulation. 9 The main entrance of sensory information from the thalamus is the border of cortical layer Vb/VI, the granular layer IV and the lower part of layer III. In layer IV, axons of stellate cells and axon-collaterals of pyramidal cells distribute the somatosensory signals within cortical columns establishing synapses on the apical dendrites of pyramidal cells in the supragranular layers II/III and the infragranular layer Vb. 19 In studies performed in rodents with epidural electrodes, it has been proposed that the N1 wave mostly reflects the activity of pyramidal cells in layer Vb resulting in depolarization of apical dendrites of supragranular layer II/III pyramidal cells. 16 Similar to our results, nearly complete and durable abolitions of forelimb contralateral evoked potential responses in the infarcted hemisphere after cortical ischemia are well known and have been previously described in several species including humans.8,20 Imaging studies also support the absence of responses in the damaged hemisphere after contralateral stimulation.21,22 In our study, only 2 of 15 mice showed progressive increases of the responses in the infarcted hemisphere associated with possible functional recovery. At this point, it remains unclear to us whether these small potentials in the damaged hemisphere (Figure 4(a); panel iii) represent portions of residual activity within the forepaw cortical area 8 or are associated with new activity in the peri-infarct tissue, specifically in portions of the motor area as has been previously reported at longer times after forepaw ischemia, when spontaneous functional recovery takes place. 9
In the ischemic mice, progressively increasing amplitudes of responses were observed in the intact hemisphere after contralateral stimulation. In stroke patients and in rodents, an enhanced activity in the non-affected hemisphere has been described after contralateral stimulation of the non-affected member,22–24 which has also been related to a larger motor response from the non-affected hand.25,26 This hyperexcitability in the intact hemisphere (diaschisis) has been commonly associated with better motor recovery of the affected member after hemiparetic stroke. 27 The hyperexcitability in the intact hemisphere might be associated with a lack of inhibition between homologous regions of the somatosensory cortex through the corpus callosum (CC).21,22 Whether this hyperexcitability has a positive or a negative effect on recovery is an actual point of debate.24,27,28 In our model of cortical infarction by MCAO, the increasing excitability in the intact hemisphere persisted for longer durations as has been observed in cortical stroke patients.
We also examined the ipsilateral activations of the somatosensory cortices in the intact and infarcted hemispheres. Ipsilateral activation is a mechanism that is poorly understood; it has been proposed that an ipsilateral potential can be generated by inter-hemispheric projections through the CC after contralateral hemisphere activation, 29 via direct projections of ascending non-decussating fibers to the ipsilateral hemisphere 30 or via volume conduction from the contralateral hemisphere. 31 Several studies have demonstrated that the primary somatosensory cortex (S1) projects extensively at the border between S1 and the secondary somatosensory cortex in the opposite hemisphere. 32 Callosal connections are probably required for integrating lateralized somatosensory input. In our study, in the pre-stroke condition, the short delay between the ipsilateral and contralateral potentials (∼5−10 ms; table 1) was compatible with inter-hemispheric CC conduction.33,34 However, after stroke, the ipsilateral activation of the intact hemisphere was severely inhibited with respect to the pre-infarct condition. This effect was observed in the ischemic but not in the sham animals, which indicates that this reduced response was dependant on the changes in the infarcted hemisphere and might not be related to the direct projections of ascending uncrossed fibers from the affected paw. Thus, the reduced response in the intact hemisphere after ipsilateral stimulation might be related with the abolition of SSEPs potentials in the damaged hemisphere (contralateral stimulation). In a recent study in mice, no ipsilateral response was detected in the intact hemisphere after a distal MCAO approach. 23 By contrast, the ipsilateral potentials of the damaged hemisphere were also severely reduced after stroke. Although high evoked responses were detected in the intact hemisphere after contralateral stimulation, responses that were probably transmitted through the CC to the opposite hemisphere (damaged), a small conductivity of the extracellular medium or the destruction of a considerable number of cortical synapses, restricted to the infarcted region, might attenuate the amplitude of the ipsilateral potentials in the affected hemisphere. By contrast, transcallosal degeneration has been reported to occur in the cortex opposite to the lesion after stroke 35 and also might explain the reduction of these ipsilateral SSEPs. This initial and severe reduction of ipsilateral potentials in the damaged hemisphere were followed by a progressive and slight increase in the response at later time points, which might be related with the phenomena of hyperexcitability that occurs in the intact hemisphere at longer times after ischemia. In this context, the results of the correlation analysis suggest a close relationship between the amplitudes of SSEPs responses generated in the intact and damaged hemispheres after contralateral and ipsilateral stimulation, respectively.
Several limitations of our study should be mentioned. First, housing the mice individually in cages might be related with the poor functional recovery that we have observed in our work, because social interaction in an enrichment environment have been shown to improve neurobehavioral function after stroke in rat models of photothrombosis 36 or ligation of the MCA. 37 The effects of enrichment environment on functional outcome can be explained by depression of the inflammatory response in the ischemic hemisphere, recovery of neuronal functions and connectivity, and post-ischemia brain plasticity. Second, to minimize the suffering of animals and to avoid movement artifacts, the electrophysiological recordings were performed under anesthesia with ketamine/xylazine. In humans, it is a common practice to monitor EPs in patients anesthetized during spinal cord surgery. 38 In monkeys and rats, the somatosensory cortex has been mapped under anesthesia with ketamine and many similarities to the awake state as well as certain differences have been reported.39,40 Although it is commonly accepted that different anesthetics as ketamine or urethane produce consistent alterations in the cerebral evoked response to forepaw stimulation, similar patterns of activation have been found for example in the sensorimotor cortex between urethane-anesthetized and awake mice. 41 In addition, SSEPs have been performed in rodents under anesthesia to evaluate the effectiveness of different treatments or to examine the activity of the somatosensory cortex after ischemia.10,42,43 In our study, we chose ketamine/xylazine because it has been largely documented that volatile and barbiturate anesthetics including isoflurane depress and/or abolish EPs in humans and different species of animals including rodents.38,44 The electrophysiological recordings were started 10 min after ketamine/xylazine injection and we never used more than 10–15 min to complete a full recording session of contra and ipsi-lateral potentials; in this time frame, EP-amplitudes during deep anesthesia are relatively stable. 45 Although ketamine might modify the responsiveness of the cortex itself and the sensory message along the afferent pathway to the cerebral cortex, the same procedure was applied to all mice, and in these specific conditions, the SSEPs were mostly modified in the stroke mice while preserved in the sham animals. Another point that deserves consideration relates to the influence of ketamine in the performance of the behavioral tests. Ketamine produces profound behavioral deficits, and this effect is mediated in part by the interruption of neurotransmission in different structures of the limbic system as the amygdala and hippocampus. At the same doses as used in the present study, the chronic daily administration of ketamine induces significant behavioral deficits, especially locomotor hyperactivity, that persisted several days after the withdrawal of the drug. 46 We always left 5 days between the application of a dose of ketamine/xylazine during the SSEPs recordings and the subsequent functional test the next week. Under this experimental paradigm, no significantly fluctuations in the scores for both functional tests were observed over time in the group of sham mice, which suggests a reduced influence of the anesthesia in the performance of both behavioral tests across the study.
Although genetically modified mice are now becoming increasingly used, the employ of SSEPs recordings has been more restricted in this specie, particularly in this type of cortical infarction due to permanent distal MCAO. The small size of the animals might constitute a difficulty, but we have corroborated the feasibility of making long-term chronic recordings in the same animal using two separate surgeries. Our results demonstrate the reliability of SSEPs to examine functional outcome in mice with damage in the somatosensorial cortex, which is characteristic of this specific stroke model, widely used to mimic human ischemic stroke. The combined use of functional tests and SSEPs across time post-ischemia can serve to examine the effectiveness of neuroprotective drugs and the contributions of different genes in the evolution of cortical damage after stroke by using genetically modified mice, helping us identify new potential targets for medication development. We also have shown the usefulness of the SSEPs to examine phenomena of brain plasticity in the intact hemisphere after unilateral cortical stroke. In our study, the correlation analysis indicates that the hyperexcitability was mostly observed in the mice with less degree of impairment after the dMCAO approach. This hyperexcitability in the intact hemisphere and the partial recovery observed exceptionally in some mice might be linked with the phenomena of plasticity at the spinal cord level. After stroke, the recovery of skilled forelimb movements has been associated with the rewiring of midline-crossing corticospinal fibers from the intact hemisphere to the denervated side of the spinal cord in rats submitted to unilateral pyramidotomy, 47 and recently in a therapeutic model of stroke in rats using antibodies against anti neurite-growth-inhibitory protein (Nogo-A) and subsequent rehabilitative training. 48
Collectively, here we have identified the existence of long-term deficits in mice after cortical infarction induced by ligation of the distal part of the MCA. Sensorimotor skills were substantially affected in parallel with impairments in the somatosensory cortices of the both the infarcted and non-infarcted hemispheres. Long-term examinations of somatosensory and motor cortical function enable the study of the mechanisms of plasticity in peri-infarct areas 9 or, alternatively, the long-term reactivation of damaged representations. 8 In the preclinical scenario, the sensitivity and reproducibility of SSEPs make them appropriate for a short- and long-term examination of cortical function after stroke and for the evaluation of the effectiveness of drugs in neuroprotection and cell-therapy-based neurorestoration approaches.
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Community of Madrid Grant Neurotec-S2010/BMD-2460 (to D.G.-N.), from Mexican National Committee of Science and Technology (CONACyT to L.P) and funds from the Spanish Ministry of Economy and Competitiveness (Consolider CSD2008-00005 and BFU2013-33821 to L.C.B.). We would like to thank Soledad Martinez for the excellent technical assistance. We also express our gratitude to Carlos Paino for his assistance with the histological images.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Authors’ contributions
JB and LP contributed equally to this work. JB and D G-N designed the research; LP and JB performed the majority of the experiments with the help of L F-G, R M-M and D G-N; R M-M, LCB and MR provided crucial reagents; LP, JB and D G-N analyzed the data; JB and D G-N interpreted the results; JB, LCB, MR and R M-M helped write and revise the manuscript and D G-N wrote the paper.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
