Abstract
Background:
Ketamine, a rapid-acting N-methyl-D-aspartate receptor antagonist used as a therapeutic for treatment-resistant depression (TRD), is usually administered intravenously or intranasally.
Aims:
This randomized, double-blind, placebo-controlled, Phase 1 study investigated safety and tolerability (primary endpoint), pharmacokinetics (PK) and pharmacodynamics (PD) of an immediate-release oral ketamine.
Methods:
Healthy volunteers (18–55 years) were randomized to each receive two single doses of oral ketamine (40–240 mg) and one oral placebo dose. Treatment-emergent adverse events (TEAEs) and PK and PD assessments (e.g., Bond and Lader visual analogue scale, Modified Observer’s Assessment of Alertness/Sedation Scale) were assessed up to 24 h after dosing. Descriptive statistics were used.
Results:
Nineteen participants were randomized (mean age: 31 years; male, 68%); 18 completed the study. Eighty mild or moderate TEAEs were reported following oral ketamine (40–240 mg) and five following placebo. There were no TEAE-related discontinuations. Most TEAEs (86%) were considered probably related to study drug. The most common TEAEs with oral ketamine were dissociation (26 events), dizziness (nine events) and headache (nine events). A positive relationship between increasing ketamine doses and dissociation events was observed. PK parameters (Cmax, AUCinf) of oral ketamine and its primary metabolites (2S,6S;2R,6R-hydroxynorketamine, R/S-norketamine) were dose proportional. Transient changes in mood and dissociation were detected 1 h postdose with a return to predose values after ~4 h.
Conclusions:
There were no unexpected safety signals with oral ketamine. PK properties were consistent with those reported for other rapid-acting formulations. These findings warrant further investigation of oral ketamine capsules in TRD (EudraCT No. 2019-001019-22).
Introduction
Major depressive disorder (MDD) is a leading burden of disease worldwide, with cases surging dramatically in 2020 to 246 million, mainly due to the impact of COVID-19 (Covid-19 Mental Disorders Collaborators, 2021). MDD is diagnosed by symptomatic criteria (American Psychiatric Association: DSM-5 Task Force, 2013), which include severely diminished psychosocial functioning and increased suicidal risk (Filatova et al., 2021; World Health Organization, 2023).
The aetiology of MDD is multifactorial (Filatova et al., 2021), with the cornerstone of current treatments targeting monoamine neurotransmitter systems. However, up to one-third of patients with MDD fail to respond to traditional first-line antidepressants and adjunctive medications, resulting in a large cohort of patients with treatment-resistant depression (TRD; Kennedy and Giacobbe, 2007; McLachlan, 2018; Voineskos et al., 2020). These patients present with high rates of relapse, morbidity and mortality and incur substantial healthcare costs (McIntyre et al., 2023).
Management approaches for TRD are rapidly evolving and either include brain stimulation modalities (e.g. electroconvulsive therapy) or less-invasive treatments, including ketamine (McLachlan, 2018; Voineskos et al., 2020). Ketamine has been shown to provide rapid reduction (<24 h) of suicidal ideation in patients with TRD versus other treatments (Ahmed et al., 2023; Grunebaum et al., 2018).
Ketamine is a rapid-acting N-methyl-D-aspartate (NMDA) antagonist that primarily exerts anaesthetic and analgesic effects via NMDA receptors. Its antidepressant effects are mediated at NMDA receptors predominantly located on GABAergic interneurons, enhancing glutamate neurotransmission and impacting other mood-relevant cortico-limbic brain regions (Derkach et al., 2007; Zanos and Gould, 2018). Attenuation of dopaminergic, serotonin, opioid, sigma and cholinergic receptors may also contribute to ketamine’s dissociative or antidepressant effects (Jelen et al., 2023; Kokkinou et al., 2018; Williams et al., 2018; Zanos et al., 2018). The contribution of these different pathways to the behavioural effects of ketamine is not fully known, and they may also be attributed to its different metabolites. Ketamine is an enantiomeric mix of (S)-ketamine (esketamine) and (R)-ketamine (arketamine) and is metabolized by hepatic microsomal cytochrome P450 enzymes to several metabolites including norketamine, dehydronorketamine, hydroxyketamine and (2S,6S;2R,6R)-hydroxynorketamine (HNK; Kamp et al., 2020).
(R)-ketamine has demonstrated more potent and longer-lasting antidepressant effects with fewer side effects and abuse liability than racemic ketamine and esketamine in preclinical models (Chang et al., 2019; Jelen et al., 2021). Similar effects were observed when intravenous (IV) (R)-ketamine was used in an open-label pilot study of individuals with TRD (Leal et al., 2021). R-ketamine also has a lower clearance rate than S-ketamine, which may explain its prolonged activity (Kamp et al., 2020). Furthermore, 2R,6R-HNK (solely produced via R-ketamine metabolism) is also a more potent antidepressant than 2S,6S-HNK (Dutton et al., 2023; Zanos and Gould, 2018). In comparison, esketamine has a three- to four-fold greater binding affinity for NMDA receptors than (R)-ketamine (Jelen et al., 2021). Studies have shown that administration of esketamine via nasal spray resulted in a faster onset of dissociative symptoms than IV administration, and it was approved by the US Food and Drug Administration in 2019 for use with an oral antidepressant in TRD (US Food and Drug Administration, 2019). However, regulatory bodies note the abuse potential (National Institute for Health and Care Excellence, 2020) of ketamine, and that safety must be considered and assessed (Rivas-Grajales et al., 2021; US Food and Drug Administration, 2019). Other considerations include cost, variable bioavailability and nasal side effects (Rivas-Grajales et al., 2021; US Food and Drug Administration, 2019). Oral ketamine formulations offer a viable alternative to parenteral or nasal formulations as they have similar efficacy with potential benefits for home use (Hull et al., 2022; Swainson et al., 2022), and there is an extensive body of work illustrating the clinical benefits of orally administered ketamine in TRD (Al Shirawi et al., 2017; Glue et al., 2020, 2024). Non-IV racemic ketamine is also recommended by the Canadian Network for Mood and Anxiety Treatments guidelines as an adjunctive treatment for TRD based on the available evidence (Lam et al., 2024). In light of this, we conducted a Phase 1 study to evaluate the safety, tolerability and pharmacokinetic (PK) and pharmacodynamic (PD) profiles of an oral racemic ketamine with an immediate-release formulation in healthy volunteers.
Methods
Study design
This was a randomized, double-blind, placebo-controlled, partial crossover Phase 1 study (EudraCT No. 2019-001019-22) designed to investigate the safety and tolerability (primary objective) and PK/PD profiles (secondary objectives) of single ascending doses of an immediate-release oral ketamine capsule in healthy volunteers. The study was conducted between 13 June 2022 (first participant, first visit) and 28 April 2023 (last participant, last visit) at NIHR King’s Clinical Research Facility, embedded within King’s College Hospital NHS Foundation Trust, London, UK. The study was conducted following the Declaration of Helsinki, the International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use Guideline for Good Clinical Practice (ICH E6(R3)), applicable Medicines for Human Use (Clinical Trials) Regulations (2004) and Good Manufacturing Practice (International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use, 2023; The National Archives, 2004). The study (IRAS project ID: 1004055) received ethical approval from the NHS Health Research Authority, London – Chelsea Research Ethics Committee, London, UK.
Participants
Healthy adults aged 18–55 years were included with a body mass index of 18–30 kg/m2. Participants were excluded if they had anxiety, bipolar disorder or moderate to severe depression diagnosed using medical history and screening criteria from the DSM-5, Generalized Anxiety Disorder 7-Item assessment (score > 5), Mood Disorder Questionnaire (Q1 (yes to >7 events), Q2 (yes), Q3 (moderate or serious problem)) and Patient Health Questionnaire v9 (score > 5) (American Psychiatric Association: DSM-5 Task Force, 2013; Hirschfeld et al., 2000; Kroenke et al., 2001; Spitzer et al., 2006). This assessment was performed at screening with the provision to contact the general practitioner if necessary. Participants were also excluded if they had any history of suicide attempt(s) or suicidal ideation indicated by the Columbia-Suicide Severity Rating Scale (C-SSRS; Posner et al., 2011), or if they had any acute or chronic illness (e.g., diabetes mellitus, coronary heart disease, COVID-19), drug abuse or surgery that would affect study participation. Known hypersensitivity to ketamine or any of its components, any abnormal vital signs or liver tests, or a positive pregnancy test were also exclusion criteria. Patients with systolic/diastolic blood pressure outside the range 90–140/40–90 mmHg or heart rate outside the range 40–100 bpm taken in the supine position at screening were excluded. Smoking (<2 months), inclusion in another study (<3 months) and prescribed medication (<30 days) before study start were not permitted. Participants were required to use adequate contraception if sexually active and of child-bearing potential. All participants provided written informed consent to participate.
Randomization and drug schedule
Participants were split into two fixed groups via a generated randomization code run in SAS® v9.4 (SAS Institute, Cary, NC, USA). The doses for this Phase 1 study were chosen based on the lower average bioavailability of oral ketamine (around 20%) compared with IV ketamine (100%) (Glue et al., 2021; Zanos et al., 2018). All participants were randomized to one of six treatment cohorts shown in Figure 1; each participant attended three treatment sessions according to a dose escalation plan and received one placebo and two ketamine doses each. Group 1 received 40–120 mg, and Group 2 received 160–240 mg single escalating doses of oral ketamine, respectively. The ketamine dose range of 40–240 mg was selected, considering the variability in absorption. Dose escalation was employed to incrementally assess the safety of higher doses. A maximum tolerated dose was not explicitly determined, as this was a dose-ranging study aimed primarily at examining safety. However, doses were escalated, with predefined monitoring and dose adjustment protocols for safety. The decision to escalate doses was based on the absence of significant adverse events (AEs) in prior groups and on real-time PK and PD data.
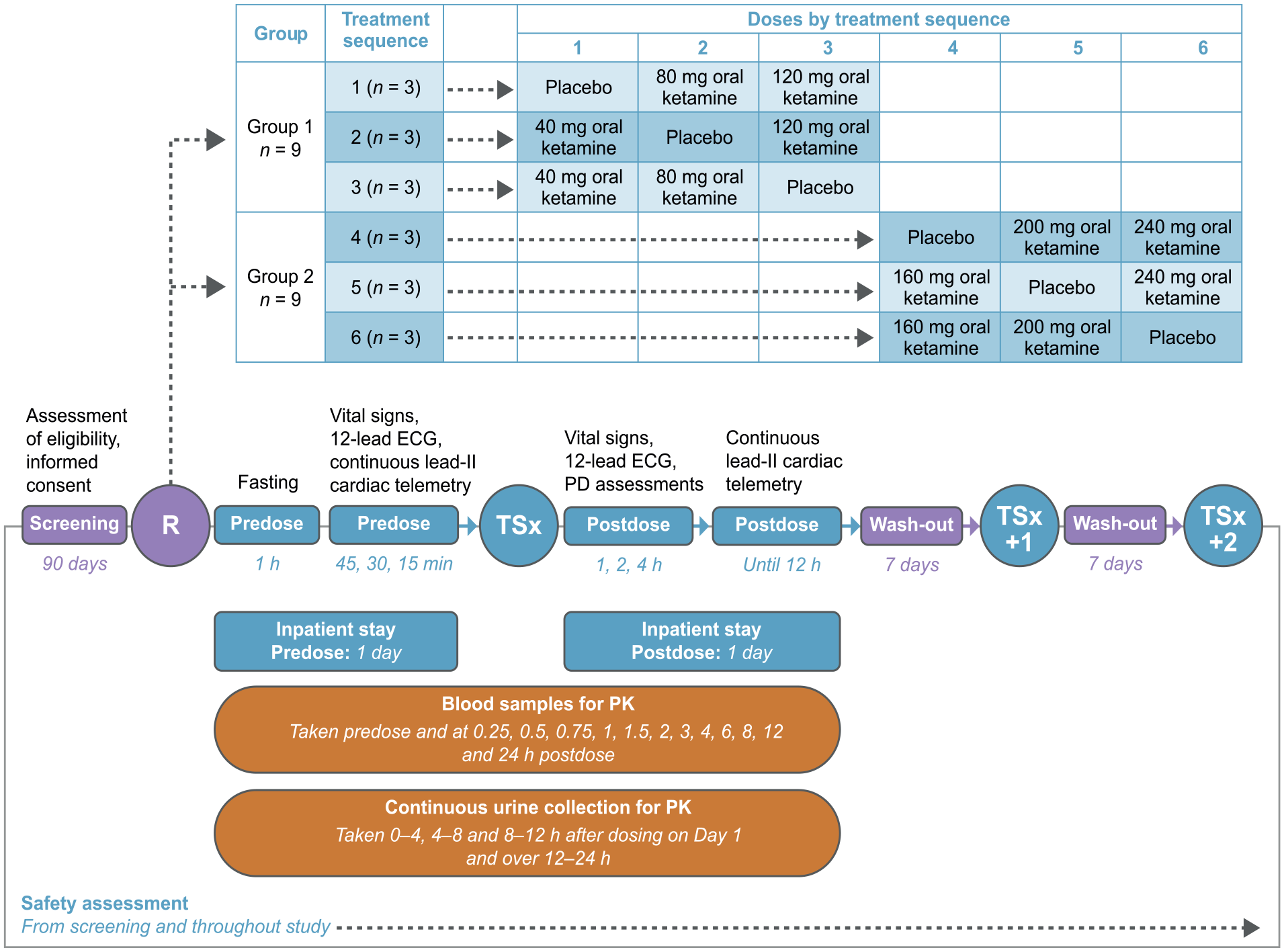
Study design and key assessments. Denominator was 36 participants (18 healthy volunteers (Across 6 Dose Levels) × 2 Active Treatment Sessions).
Participants were resident in the clinic from the day before dosing (Day −1) until 24 h after dosing (Day 2), with drug administration given under supervision using a 7-day washout period between doses. The last visit occurred 5–10 days after the final dose (Day 6–11). Oral capsules were taken under fasted conditions with 240 mL water at 08:00–11:00 h. Blinding was maintained using an identical placebo and active capsules supplied by the Sponsor (Neurocentrx Pharma, Ltd., Edinburgh, Scotland, UK). Study drugs were manufactured by Upperton Ltd. (Nottingham, UK) and packaged by Eramol Ltd. (Horley, UK). Drug purity was maintained by using temperature-controlled conditions during transportation and monitoring storage within the study site pharmacy. The Principal Investigator could access the randomization code if unblinding was required for any reason. Rescue medication was permitted and included lorazepam (distress or intolerable psychomimetic experience), metoclopramide (antinausea), paracetamol (analgesia) and amlodipine, bisoprolol or labetalol (antihypertensives).
Endpoints
Safety and tolerability
The primary endpoint was the safety and tolerability of oral ketamine assessed via vital signs, 12-lead electrocardiogram (ECG), cardiac telemetry, physical examination and laboratory tests (biochemistry, haematology and urinalysis) performed frequently until the last visit. C-SSRS was documented at screening and last visit. AEs were recorded from screening until the last visit and documented in electronic case report forms. AEs were coded according to the Medical Dictionary for Regulatory Activities v25.0, and their relationship to study drug was determined by the Principal Investigator.
Pharmacokinetics and pharmacodynamics
Secondary PK and PD endpoints for ketamine and its metabolites were measured throughout the study. Blood samples for PK analysis were collected predose and at multiple time points up to 24 h postdose. The assay employed for plasma ketamine and metabolite quantification had a lower limit of quantification (LLOQ) of 0.5 ng/mL. Ketamine and its major metabolites (2R,6R-HNK, 2S,6S-HNK, norketamine and dehydroketamine) were measured using a validated liquid chromatography-tandem mass spectrometry method. Details on the assay and LLOQ can be found in the Supplemental materials. Continuous urine samples were also collected after dosing at every session: 0–4, 4–8, 8–12 and 12–24 h, respectively (Figure 1).
Key PK endpoints included area under the concentration-time curve from time zero extrapolated to infinite time (AUCinf), clearance/fraction of dose absorbed, maximum plasma concentration (Cmax), terminal elimination half-life (t1/2) and time to maximum plasma concentration (Tmax).
For PD assessments of oral ketamine, participants completed the following: Bond and Lader – visual analogue scale (BL-VAS; total score for overall mood changes), Modified Observer’s Assessment of Alertness/Sedation scale (MOAA/S; for sedation), Cambridge Neuropsychological Test Automated Battery (CANTAB; for cognitive state using computerized testing for attention, episodic memory and working memory), Clinician-Administered Dissociative States Scale 27 (CADSS 27; for dissociation) and the Psychotomimetic States Inventory (PSI; for psychotomimetic effects). These assessments are well-validated and have been described elsewhere (Bremner et al., 1998; Bond and Lader, 1974; Chernik et al., 1990; Mason et al., 2008; Sharma, 2013). PD assessments were taken predose (Day −1; baseline), and at 1, 4 and 24 h after dosing on Day 1 of each session.
Statistical analysis
As this was a pilot study, no formal sample size calculation was undertaken. No inferential statistical testing was planned. Descriptive statistics were used for PK parameters. The baseline was defined as the last value before drug administration (predose on Day 1 of each session), or Day −1 if not recorded predose, or screening if not recorded predose or Day −1 (e.g. weight).
PK parameters for oral ketamine (the parent drug) and its metabolites were derived from individual concentration–time profiles using noncompartmental analysis. AUCinf was calculated using the linear-up/log-down trapezoidal method, and t1/2 was estimated using logarithmic regression of the terminal phase of the concentration–time curve. Dose proportionality was assessed by comparing the Cmax and AUCinf across the different doses. The relationship between dose and exposure was tested using correlation analysis. A power-law analysis was also conducted to explore the relationship between dose and the key PK parameters (Cmax and AUCinf). The power-law model (C = a * Doseb) was applied to evaluate proportionality. For calculation of all PK parameters, plasma concentrations below the limit of quantification (BLQ) prior to Tmax were taken as zero; all other values were taken as missing. It was not possible to impute a value for urine concentrations reported as BLQ. The amount excreted was set to zero when the concentration was BLQ.
An exploratory analysis was performed to determine the dose–exposure relationship between R-ketamine 40–240 mg and CADSS or BL-VAS total score. The maximal plasma concentration (Cmax, ng/mL) of R-ketamine at 1 h was used, and linearity was measured using Pearson’s correlation coefficient (r).
The primary endpoints were analysed using the Safety Analysis Set, defined as all participants who received at least 1 dose of study drug. PK endpoints were analysed using the PK Parameter Set (all participants who received at least 1 dose of study drug and had a PK sample taken and analysed). PD endpoints were analysed using the PD Analysis Set (all participants who received at least 1 dose of study drug and had at least one PD measurement).
All statistical analysis was performed using SAS® v9.4.
Results
Participants
Thirty-nine participants were screened, and 20 were enrolled. One participant discontinued prior to randomization due to clinically significant cardiology findings, and 19 participants were subsequently dosed (Group 1: n = 10; Group 2: n = 9). One participant was withdrawn from Group 1 after the first dose of study drug (placebo) due to noncompliance (positive cotinine test), and 18 participants completed the study (see Figure S1 in the Supplemental Material). There were no protocol deviations. Baseline demographics are summarized in Table 1. The mean (standard deviation (SD)) age was 31 (7) years, most participants were male (68%) and White (58%). Concomitant illness was reported in two participants (shigella infection (n = 1) and essential tremor (n = 1)).
Baseline and demographic characteristics.
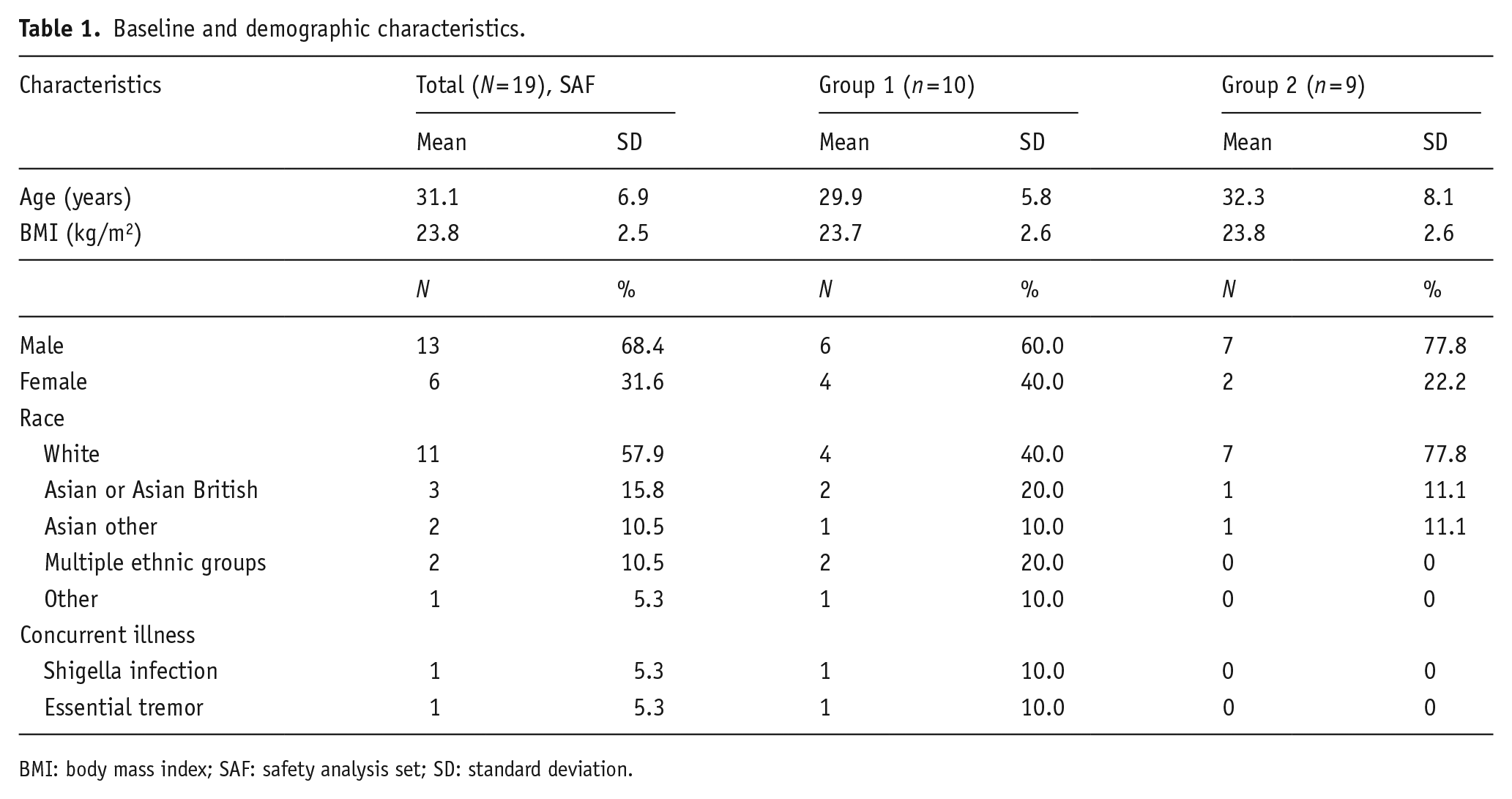
BMI: body mass index; SAF: safety analysis set; SD: standard deviation.
Safety and tolerability
Eighty-five treatment-emergent adverse events (TEAEs) were reported across all participants: 80 (94%) following oral ketamine (40–240 mg) and 5 (6%) following placebo (Table 2). All TEAEs were mild or moderate in severity, with rescue medication (metoclopramide) only required for one participant for intolerable nausea/vomiting. There were no severe TEAEs, serious AEs, discontinuations or dose alterations due to TEAEs or deaths. Most TEAEs (86%; n = 73) were considered probably related to study drug. The most common TEAEs with oral ketamine were dissociation (26 events), dizziness (nine events (one event with placebo)), headache (nine events) and nausea (seven events); there were two vomiting events with oral ketamine 200 and 240 mg; these were moderate and only occurred once. A positive relationship between increasing oral ketamine doses and dissociation events was observed, with four dissociation events graded as moderate in the 120 mg or greater oral ketamine dosing sessions.
Overview of TEAEs.
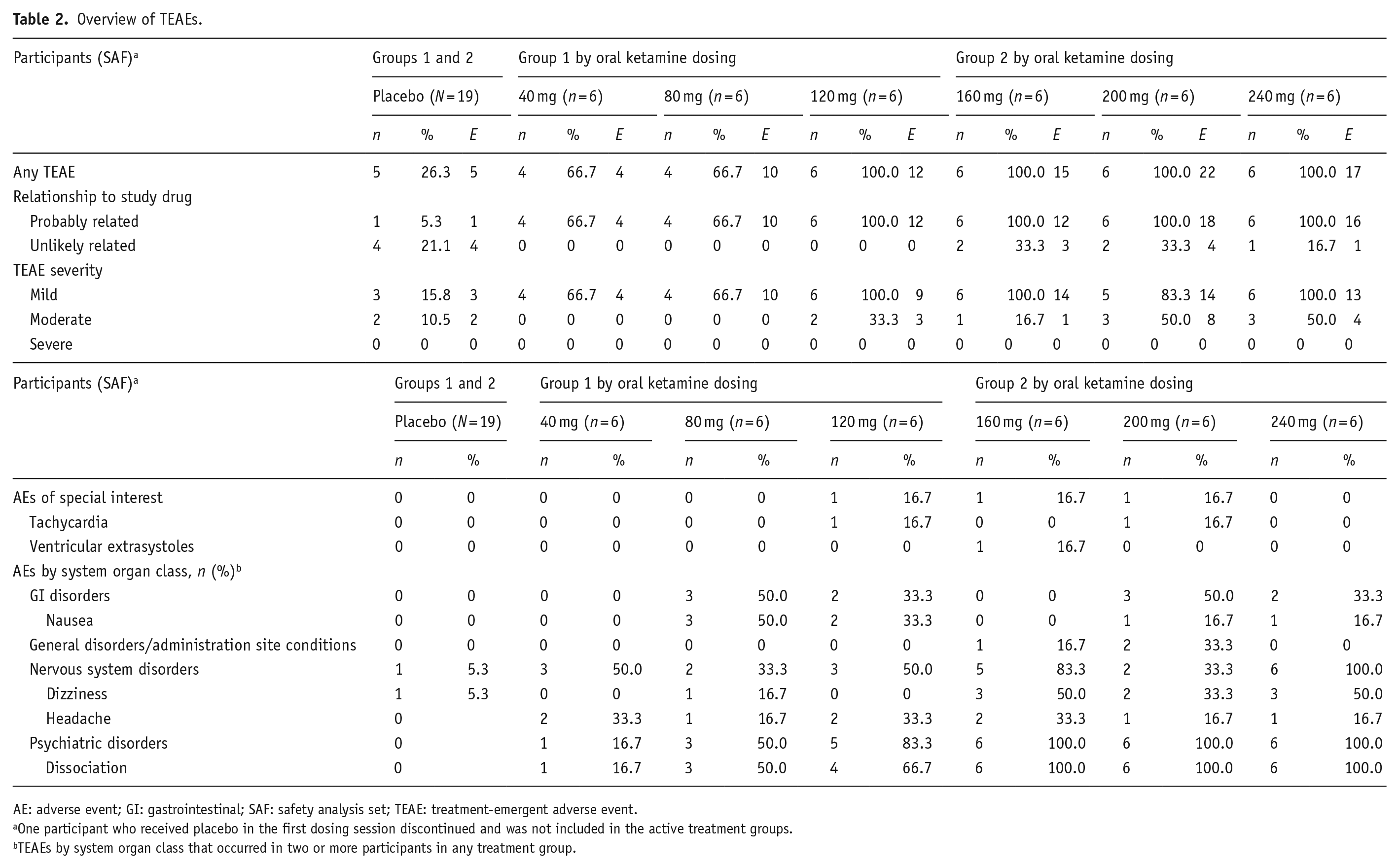
AE: adverse event; GI: gastrointestinal; SAF: safety analysis set; TEAE: treatment-emergent adverse event.
One participant who received placebo in the first dosing session discontinued and was not included in the active treatment groups.
TEAEs by system organ class that occurred in two or more participants in any treatment group.
Transient increases in diastolic and systolic blood pressure were observed 30 min postdose during the higher oral ketamine dosing sessions; mean change from baseline in systolic/diastolic blood pressure was +18/+11 mmHg (160–240 mg) versus +7/+2 mmHg (40–120 mg), respectively. These values returned to baseline after ~4 h, and none required clinical intervention. Three cardiac events of interest were reported with oral ketamine. These were mild tachycardia (120 mg), moderate tachycardia (200 mg) (heart rate up to 116 bpm) and mild ventricular extrasystoles (160 mg); the two tachycardia events were assessed as probably related to study drug. There were no clinically significant changes in ECG, including corrected QT interval (QTc), telemetry, vital signs or laboratory assessments throughout the study. No evidence of suicidal behaviour or ideation or neurological changes were reported based on the C-SSRS and physical examination.
Pharmacokinetics
The mean Cmax and AUCinf were dose proportional for R/S-ketamine and its metabolites (2R,6R-HNK, 2S,6S-HNK, Rac-dehydroketamine and R/S-norketamine) (Figure 2 and Table 3). In the power-law analysis, the exponent b was ~1.0 across the dose range studied for ketamine and its primary metabolites, which further supports dose proportionality.
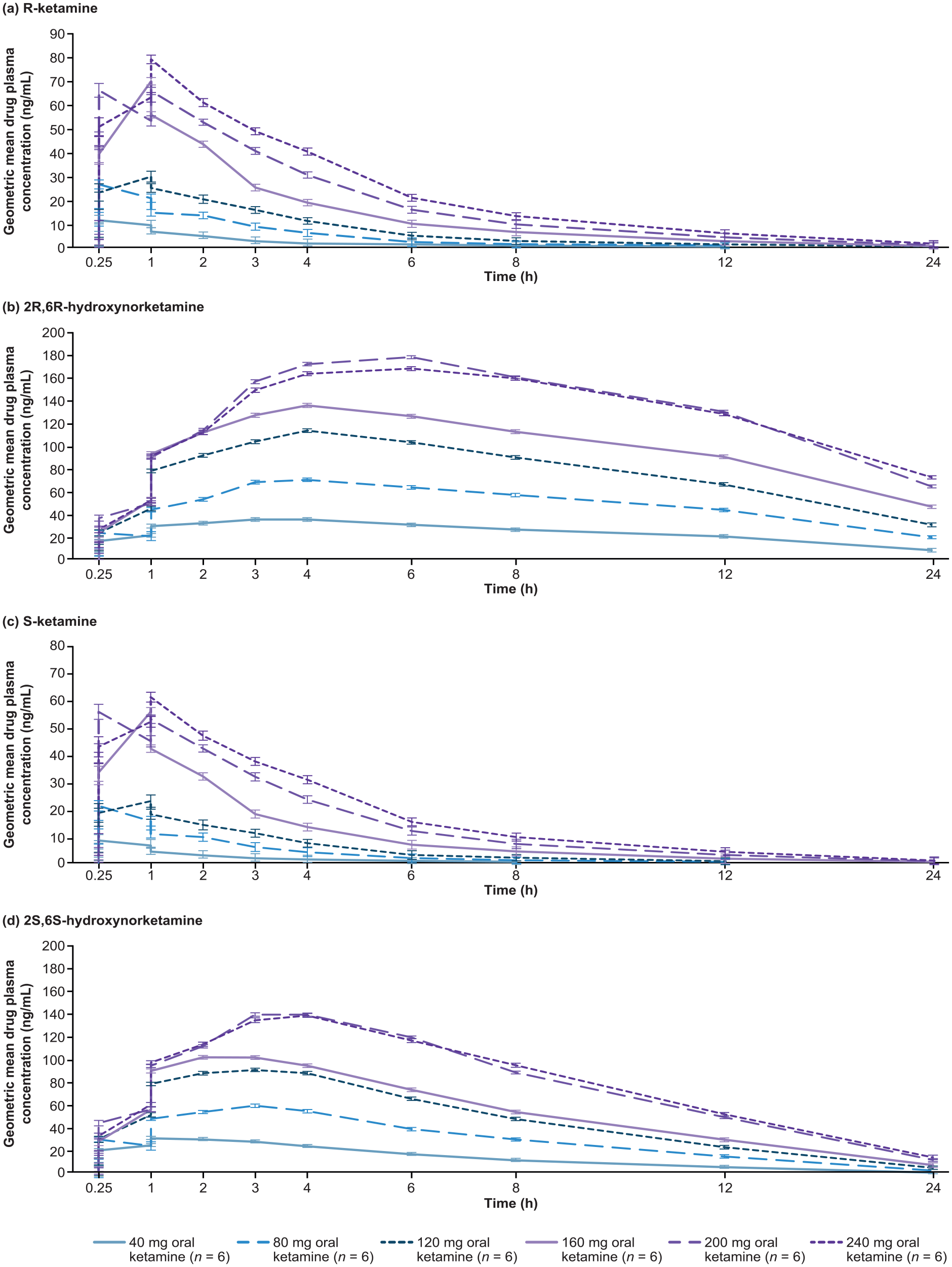
Geometric mean (SD) plasma concentration over 24 h for (a) R-ketamine, (b) 2R,6R-hydroxynorketamine, (c) S-ketamine and (d) 2S,6S-hydroxynorketamine (PKS; n = 18).
PK parameters (geometric mean, SD) of oral ketamine and its key metabolites (PKS; n = 18).
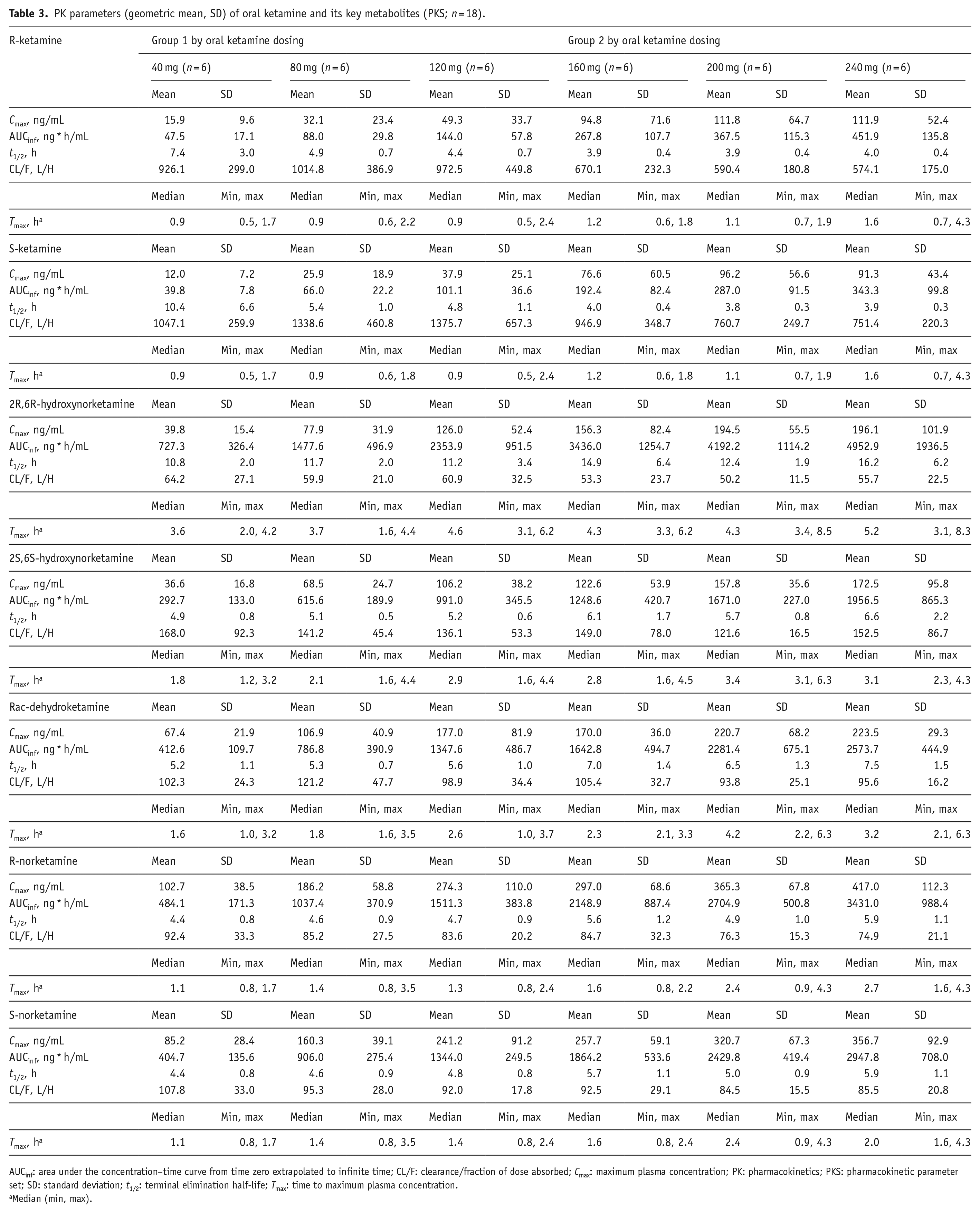
AUCinf: area under the concentration–time curve from time zero extrapolated to infinite time; CL/F: clearance/fraction of dose absorbed; Cmax: maximum plasma concentration; PK: pharmacokinetics; PKS: pharmacokinetic parameter set; SD: standard deviation; t1/2: terminal elimination half-life; Tmax: time to maximum plasma concentration.
Median (min, max).
The median Tmax of R/S-ketamine ranged from 0.9 h (40 mg) to 1.6 h (240 mg) and this is reflected by spikes in plasma concentration around this time point (Figure 2 and Table 3). Elimination of R-ketamine was rapid; urine excretion occurred within 12 h, and plasma concentration became negligible at this time point. Similar effects were observed with S-ketamine (Figure 2). Clearance was longer with increasing ketamine doses, but excretion occurred within 24 h for all metabolites. The highest exposure and slowest elimination occurred with 2R,6R-HNK (Figure 2).
Pharmacodynamics
Transient changes in dissociation (CADSS) were detected at 1 h across all oral ketamine doses; these persisted at 4 h particularly for higher doses (CADSS 27 score: 3 (200 mg), 6 (240 mg)) and returned to predose values after ~4 h. BL-VAS decreases were more significant at oral ketamine doses of 160–240 mg than at doses of 40–120 mg and were driven mainly by the alertness factor. At 1 h, BL-VAS decreased dose proportionally from oral ketamine 40–120 mg and plateaued at 160 mg. At 4 h, BL-VAS was still reduced (⩽0), with scores following oral ketamine 160–240 mg >40–120 mg (Figure 3; see also Table S1 in the Supplemental Material). Mean (95% confidence interval (CI)) CADSS total score increased across several doses (1.2 (−0.5, 2.8) with 40 mg to 21.2 (6.1, 36.3) with 240 mg), respectively, with a suggestion of a modest dose-response plateau above 160 mg (see Table S1 in the Supplemental Material). A positive linear relationship was observed between increasing ketamine doses (expressed as plasma Cmax) and CADSS total scores at 1 h (see Figure 4(a)). There was evidence of a negative linear relationship between oral ketamine dose and BL-VAS score (see Figure 4(b)). Both figures showed higher intra-participant variability.
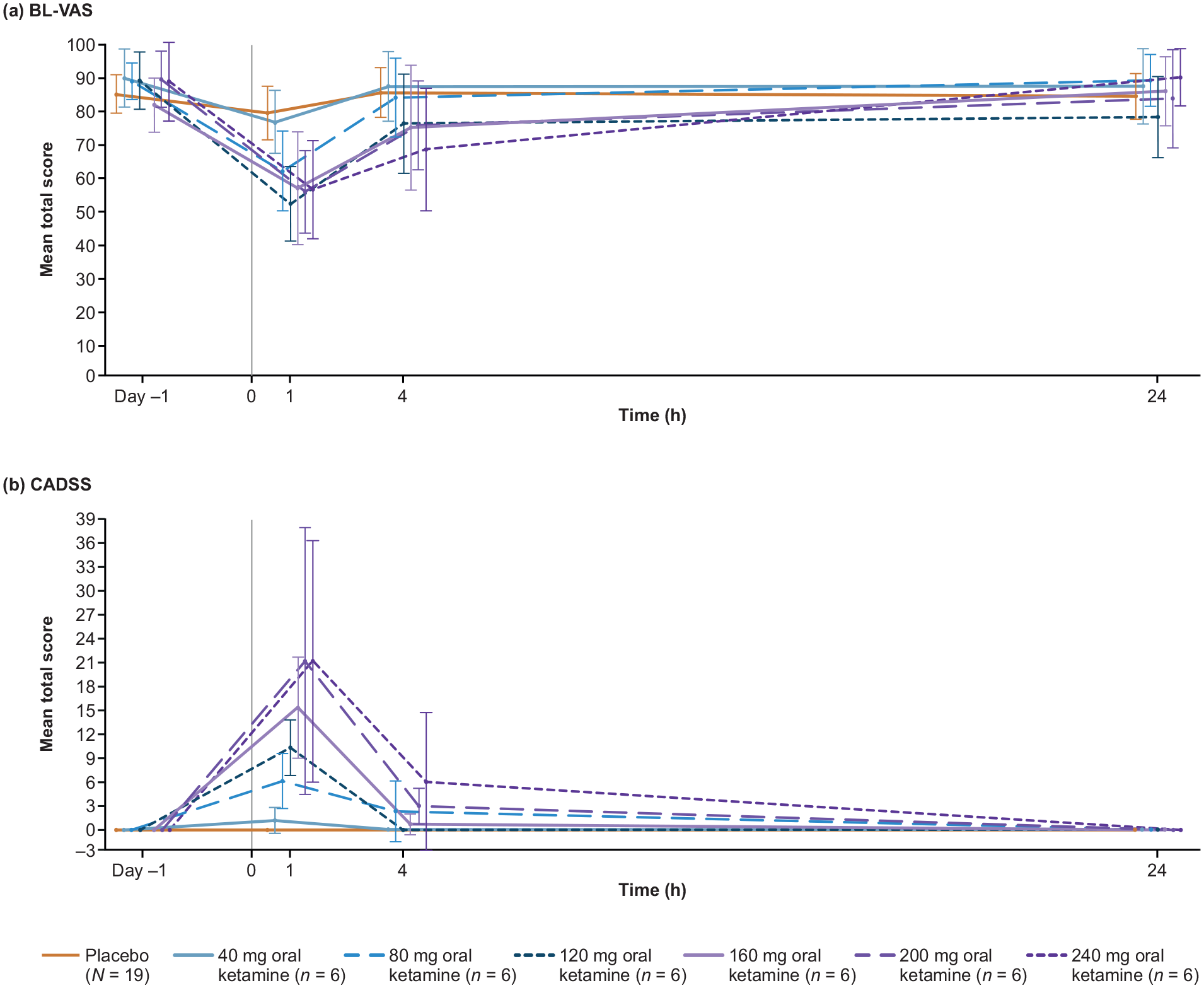
Total mean (95% CI) scores for (a) BL-VAS and (b) CADSS over 24 h following oral ketamine dosing (PDS; n = 18).
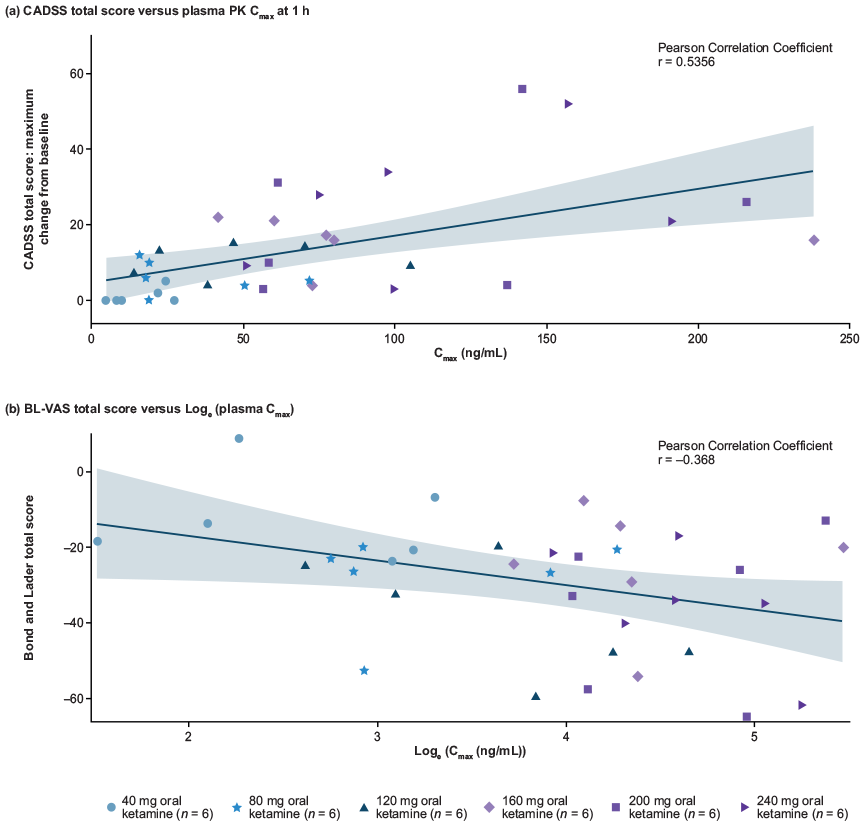
Exploratory analysis showing the exposure–response relationship between R-ketamine and (a) CADSS total score or (b) BL-VAS total score.
There were no changes in sedation score (MOAA/S); it remained at a maximum of five for all participants at every time point. There was evidence of a drug effect on cognitive performance (CANTAB tasks) and on psychotomimetic effects (PSI) (Table S1), but these changes were heterogeneous, with no evidence of a relationship between dose, time or magnitude of changes.
Discussion
This study provided data on the safety, tolerability and PK/PD profiles of an immediate-release oral ketamine formulation in development for patients with TRD and other major mental health conditions. In this study, oral ketamine was administered to 18 healthy volunteers enrolled in 6 double-blind, placebo-controlled cohorts over a ~30-day treatment period. Dose escalation proceeded in the context of prespecified dose escalation stopping criteria. A maximum tolerated dose was not established.
The primary endpoint was safety, and immediate-release oral ketamine was generally well tolerated at the dose range studied (40–240 mg). All TEAEs were mild to moderate and resolved within hours without study interruption, or medical intervention apart from administration of antiemetic medication for a single participant. The most common TEAE was dissociation (72%; n = 26/36).
Most dissociative events (61%; n = 22/36) were mild (i.e. transient, manageable effects) and improved within 4 h, as supported by reductions in the CADSS total score. However, we do note that at higher doses, particularly the 240-mg dose, CADSS scores remained elevated at the 4 h mark and the level of dissociation observed raises important considerations regarding appropriate safety monitoring postdose. An assessment of patients with TRD in future studies will be needed to inform the nature and duration of appropriate safety monitoring and the care setting for administration of oral dosage forms of ketamine in a therapeutic context (Ingrosso et al., 2024).
Of note, favourable findings regarding dissociation and oral dosing have emerged from a recent study of 231 patients with TRD and Montgomery-Asberg Depression Rating Scale (MADRS) scores ⩾ 20 who received open-label, extended-release ketamine tablets (R-107) 120 mg per day. Responders (MADRS score ⩽ 12 and reduction ⩾ 50% after 8 days) were then randomized to receive R-107 doses of 30, 60, 120 or 180 mg or placebo twice weekly for a further 12 weeks. Overall, 72.7% (n = 168/231) were classified as responders with low rates of dissociation, ranging from 2.9% (30 mg) to 15.6% (180 mg), respectively (Glue et al., 2024). In comparison, dissociation was experienced by 61%–75% of patients with TRD who received intranasal ketamine at therapeutic doses (US Food and Drug Administration, 2019). The relationship between dissociation and antidepressant response is not fully understood and remains a topic of investigation (Ballard and Zarate, 2020; Grabski et al., 2020). With respect to suicidal behaviour or ideation, we did not observe any change on the C-SSRS measure with oral ketamine over the dose range studied; however, this was expected given that the participants were healthy volunteers.
The observed dose-related transient effects of oral ketamine on PD measures of mood and cognition were expected and have been demonstrated in previous studies of healthy participants (Pomarol-Clotet et al., 2006). They may be indicative of drug action, but symptomatic effects are not directly transferable to patients with TRD or other mental health disorders.
We also found that 19% (n = 7/36) of participants reported mild to moderate nausea in the active groups, and two participants experienced vomiting – one of whom required rescue treatment with the antiemetic metoclopramide; this did not lead to treatment discontinuation. With respect to cardiac safety, there were two tachycardia events at higher oral ketamine doses (heart rate up to 116 bpm), no clinically significant changes in QTc and sympathomimetic blood pressure changes were transient; returning to baseline values within ~4 h without the need for clinical intervention. These findings are consistent with the known profile of ketamine and provide important information for dose planning in subsequent studies.
The PK results showed that via the oral route, the Tmax of R/S-ketamine occurred ~1 h after dosing across the 40–200 mg dose range. In comparison, the Tmax for intranasal esketamine is ~20 to 40 min after nasal administration (US Food and Drug Administration, 2019). The Tmax observed in our study is longer than the ~30 min observed for an oral ketamine solution consistent with expectations that liquid solutions often demonstrate faster absorption rates than solid oral formulations (Clements et al., 1982). Additionally, Glue et al. (2020, 2021, 2024) provide data on two immediate-release oral tablets with varying Tmax values, which reflect formulation-specific absorption kinetics. However, as this study was not designed as a comparative investigation, direct head-to-head comparisons with other formulations were not performed. Our findings also show that conversion to 2R,6R-HNK was rapid, with Tmax occurring within ~3 to 5 h. The Cmax and AUCinf demonstrated an approximately dose-proportional relationship for most ketamine metabolites following oral dosing, supporting the potential for oral administration to achieve a favourable drug profile.
Plasma concentrations achieved with this immediate-release oral ketamine and its metabolites were indirectly comparable with PK exposure data from two studies using therapeutic IV racemic (R/S)-ketamine or intranasal esketamine administration in patients with TRD (Kurosawa et al., 2023; Singh et al., 2016). The first indirect comparison with IV R/S-ketamine 0.5 mg/kg (administered over 40 min, 2–3 times weekly, up to 4 weeks; Singh et al., 2016) showed that the oral ketamine 120–240 mg PK exposure in our study was comparable to the peak: trough PK exposure (mean plasma Cmax) of IV R/S-ketamine observed during the first 6 h after dosing (see Figure S2(a) and (b) in the Supplemental Material). The second indirect comparison showed that oral ketamine 120–240 mg had similar peak: trough exposure (mean plasma Cmax) to intranasal esketamine 56 mg (self-administered twice weekly for 4 weeks; Kurosawa et al., 2023) in the first 6 h after dosing in patients with TRD (see Figure S2(c) and (d) in the Supplemental Material).
While the peak R,S-ketamine concentrations at 240 mg (~150 ng/mL) in this study fall on the lower end of the reported therapeutic range for IV ketamine (0.5 mg/kg), dose proportionality as shown by Cmax and AUCinf (illustrating the relationship between dose and systemic exposure) supports individualized dosing. We do note that the therapeutic range for IV ketamine appears to be narrow, with doses of ~0.5 mg/kg (corresponding to plasma concentrations of ~175 ng/mL) associated with clinical effect in patients with TRD. There is evidence to the effectiveness of the 0.5 and 1.0 mg/kg subanaesthetic doses of IV ketamine and no clear or consistent evidence for clinically meaningful efficacy of lower doses of IV ketamine (Fava et al., 2020).
Oral routes involve first-pass metabolism, leading to greater variability of the parent compound (Clements et al., 1982). We did observe evidence of intra-participant variability (Figure 4). This variability underscores the challenge of achieving consistent therapeutic concentrations using fixed oral dosing. Importantly, the higher doses tested (e.g. 240 mg) were associated with increased treatment-limiting side effects, including dissociation, dizziness and headache. Additionally, some participants at these doses had plasma concentrations >200 ng/mL, which are above the targeted range and may pose safety concerns. We also need to consider the relative contribution of 2R,6R-HNK; this metabolite may contribute to the observed PD effects, even in individuals whose R/S-ketamine plasma concentrations are subtherapeutic.
Drug clearance remained efficient at all doses tested with rapid elimination within 12 h. There was an increase in clearance at the higher doses, which may reflect a dose-dependent modulation of elimination kinetics and saturation of enzymatic pathways.
It is possible that dosing strategy (e.g. weight-based dosing) may address some of the issues described above, especially for higher doses. Additionally, incorporating mg/kg as a covariate in the analysis could enhance the interpretation of PK exposure data, particularly when comparing oral and IV routes of administration, as shown in Figure S2. This would facilitate a more robust understanding of the relationship between dose, plasma exposure, and therapeutic response in patients with TRD.
Limitations
The study had several limitations. This was a partial crossover study in healthy volunteers, and we cannot exclude the possibility of confounding between placebo and active drug effects on safety and PD measures; however, there was a 7-day washout period between doses and clearance was fast. We report the total score for BL-VAS assessments for simplicity and note that future studies in patients would benefit from analysing BL-VAS subscale parameters.
Healthy volunteers lack the comorbidities and concomitant medications often present in patients with depression. These factors can significantly influence both PK and PD parameters, including drug metabolism/sensitivity, systemic exposure, and therapeutic response.
Future studies in patients with TRD will inform dosing strategies to achieve optimal therapeutic effects, and allow an assessment of best practice for oral-route administration, considering care setting and appropriate safety monitoring, as well as the use of abuse-deterrent formulations. Finally, we did not address any differences related to age, sex or other clinical characteristics and we did not make direct comparisons with other oral formulations, nor did we compare with other oral routes, such as sublingual troches (lozenges), which lack data.
Although higher doses (e.g. 300–450 mg) have been used in sublingual studies (Hassan et al., 2022), the objectives of this study guided the use of doses up to 240 mg. The maximum tolerated dose was not assessed. Future studies could explore higher doses, particularly in patients with TRD, if warranted by clinical data.
In the current study, medication was administered under fasting conditions in a controlled environment. The potential food–drug interaction warrants further investigation.
Conclusions
There were no unexpected safety signals observed in this Phase 1 study of an oral ketamine immediate-release formulation at doses of 40–240 mg.
Transient mood changes and dissociation effects were detected approximately 1 h postdose, returning towards predose values within ~4 h. The PK parameters (Cmax, AUCinf) of oral ketamine and its primary metabolites (2S,6S;2R,6R-HNK, R/S-norketamine) demonstrated dose proportionality across the dose range studied, which further supports the feasibility of oral administration.
The findings from this Phase 1 study are encouraging; safety, PK exposure and clearance data were consistent with those reported for intravenous or intranasal formulations, but with a more convenient route of administration. Future studies of an abuse-deterrent, immediate-release, oral ketamine in patients with TRD and other major affective disorders are warranted and may be invaluable for optimizing dosing regimens and providing data to inform the nature and duration of monitoring needed across care settings for an oral dosage form.
Supplemental Material
sj-docx-1-jop-10.1177_02698811251340925 – Supplemental material for A randomized, double-blind, placebo-controlled, Phase 1 study to investigate the safety, tolerability, pharmacokinetics and pharmacodynamics of an immediate-release oral ketamine capsule in healthy volunteers
Supplemental material, sj-docx-1-jop-10.1177_02698811251340925 for A randomized, double-blind, placebo-controlled, Phase 1 study to investigate the safety, tolerability, pharmacokinetics and pharmacodynamics of an immediate-release oral ketamine capsule in healthy volunteers by Mutahira Qureshi, Daniel Silman, Romayne Gadelrab, Hans-Christian Stein, Pietro Carmellini, Graeme Duncan, Mitul A Mehta, Allan H Young, Carmel Reilly and Mario F Juruena in Journal of Psychopharmacology
Supplemental Material
sj-eps-2-jop-10.1177_02698811251340925 – Supplemental material for A randomized, double-blind, placebo-controlled, Phase 1 study to investigate the safety, tolerability, pharmacokinetics and pharmacodynamics of an immediate-release oral ketamine capsule in healthy volunteers
Supplemental material, sj-eps-2-jop-10.1177_02698811251340925 for A randomized, double-blind, placebo-controlled, Phase 1 study to investigate the safety, tolerability, pharmacokinetics and pharmacodynamics of an immediate-release oral ketamine capsule in healthy volunteers by Mutahira Qureshi, Daniel Silman, Romayne Gadelrab, Hans-Christian Stein, Pietro Carmellini, Graeme Duncan, Mitul A Mehta, Allan H Young, Carmel Reilly and Mario F Juruena in Journal of Psychopharmacology
Supplemental Material
sj-eps-3-jop-10.1177_02698811251340925 – Supplemental material for A randomized, double-blind, placebo-controlled, Phase 1 study to investigate the safety, tolerability, pharmacokinetics and pharmacodynamics of an immediate-release oral ketamine capsule in healthy volunteers
Supplemental material, sj-eps-3-jop-10.1177_02698811251340925 for A randomized, double-blind, placebo-controlled, Phase 1 study to investigate the safety, tolerability, pharmacokinetics and pharmacodynamics of an immediate-release oral ketamine capsule in healthy volunteers by Mutahira Qureshi, Daniel Silman, Romayne Gadelrab, Hans-Christian Stein, Pietro Carmellini, Graeme Duncan, Mitul A Mehta, Allan H Young, Carmel Reilly and Mario F Juruena in Journal of Psychopharmacology
Footnotes
Acknowledgements
Medical writing and editorial assistance were provided by Sam Phillips, PhD, of Parexel, and were funded by Neurocentrx Pharma, Ltd.
Author contributions
Mutahira Qureshi, Daniel Silman, Romayne Gadelrab, Hans-Christian Stein, Pietro Carmellini were involved in data collection. Mario F Juruena was involved in study design, data analysis and data interpretation. Mitul A Mehta and Allan H Young were involved in study design and data interpretation. Carmel Reilly was involved in study design. Mario F Juruena and Graeme Dunca contributed to the writing, reviewing and all authors confirm approval of the final manuscript.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Mitul A Mehta reports being a consultant for Boehringer Ingelheim, Sosei Heptares and Neurocrine and sits on the advisory board for Neurocentrx and Quolet. He has received speaker fees from Takeda. He has grant funding from the National Institutes of Health Research, Alzheimer’s Research UK, the Wellcome Trust, UKRI, Lundbeck and Sosei Heptares. Allan H Young was the Principal Investigator on multiple studies and has received grants for these. He has received funding for participation in advisory boards and lectures from Allergan, AstraZeneca, Bionomics, Boehringer Ingelheim, Compass, Eli Lilly, Flow Neuroscience, Janssen, LivaNova, Lundbeck, Neurocentrx, Noema Pharma, Novartis, Roche, Sage, Servier, Sumitomo Dainippon Pharma, Sunovion and Takeda. Young’s independent research is funded by the NIHR Maudsley Biomedical Research Centre at South London and Maudsley NHS Foundation Trust and King’s College London. The views expressed are those of the author(s) and not necessarily those of the NIHR or the Department of Health and Social Care. For the purposes of open access, the author has applied a Creative Commons Attribution (CC BY) license to any Accepted Author Manuscript version arising from this submission. Graeme Duncan is an employee of Neurocentrx Pharma, Ltd. Carmel Reilly was an employee of Neurocentrx Pharma, Ltd. at the time the study was conducted. Mario F Juruena is a Principal Investigator in several studies and has recently secured grants from the Academy of Medical Sciences/Royal Society, Wellcome, King’s College London, the National Institute for Health and Care Research (NIHR) Biomedical Research Centre (BRC) at the South London and Maudsley (SLaM) NHS Foundation Trust and the European Commission (EU). Juruena has received honoraria as an advisory board and speaker by leading pharmaceutical companies specializing in drugs used to treat affective and related disorders. These companies include Artmed, Bial, Daiichi Sankyo, EMS, Janssen (J&J), Lundbeck, Neurocentrx, Novartis and Takeda. Furthermore, Juruena has served on advisory boards for organizations such as MRC, NICE, NIHR, the Dutch Foundation, the Swiss NC, UKRI, UNESCO and WHO. Juruena’s contributions also hold copyrights for publications including Nature/Springer Books, Cambridge Press and Elsevier Press. It is essential to note that the views expressed in this article reflect the authors’ and do not necessarily represent the views of the aforementioned institutions or organizations. Mutahira Qureshi, Daniel Silman, Romayne Gadelrab, Hans-Christian Stein and Pietro Carmellini have no conflicts of interest to declare.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Neurocentrx Pharma, Ltd. and the Wellcome Innovator Award.
Data availability statement
Data are available on reasonable request to the corresponding author.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
