Abstract
Background:
Antipsychotic-induced weight gain (AIWG) is a major concern in psychiatry, where there is a mortality gap between those with mental illness, particularly schizophrenia, and the general population. One development proposed is using centrally-acting opioid receptor antagonists (CORAs) such as naltrexone and samidorphan.
Objective:
The systematic review and meta-analysis evaluated the available human clinical trial data on the effect of CORA on AIWG.
Methodology:
Four online databases (MEDLINE, EMBASE, PsycINFO, and Cochrane) were searched for randomized-controlled trials (RCTs) on the topic. The primary outcome was change in bodyweight. Secondary anthropometric outcomes included percentage bodyweight change, BMI change, and absolute risk of weight gain. Meta-analysis was conducted on primary outcome.
Results:
Nine RCT articles (samidorphan = 6, naltrexone = 3) and two extension studies from RCTs (both samidorphan) were identified. Meta-analysis of four RCTs (n = 1416) found olanzapine/samidorphan was associated with less weight gain than olanzapine alone (mean difference in bodyweight change: −1.18 kg; 95% CI: −1.67 to −0.68). Olanzapine/samidorphan was also superior to olanzapine for changes in BMI (−0.65 kg/m2; 95% CI: −1.1 to −0.28), waist circumference (−1.5 cm; 95% CI: −2.67 to −0.32), and risk reduction for gaining 7% body weight (−12.4%; 95% CI: −18.27 to −6.54) or 10% body weight (−10.8%; 95% CI: −16.21 to −5.45). Naltrexone did not separate from placebo for change in weight or BMI.
Conclusion:
CORA, specifically samidorphan, was effective at reducing weight gain in individuals prescribed olanzapine. The small effect sizes and discrepancy between samidorphan and naltrexone suggest effects may be timing dependent, not a class effect, or dependent on the antipsychotic combination.
Keywords
Introduction
Antipsychotic-induced weight gain (AIWG) and obesity in severe mental illness (SMI) are major concerns in psychiatry (Correll et al., 2011; NHS England, 2024; Woodall et al., 2024) and are often associated with second-generation antipsychotics (SGAs), such as olanzapine, clozapine, quetiapine, and risperidone (Huhn et al., 2019; Pillinger et al., 2020). SGAs are increasingly used in psychiatry, both in preference over first-generation antipsychotics and an increasing range of uses including off-label use (Højlund et al., 2019; Pillarella et al., 2012; Rhee et al., 2018, 2020). This is a concern as obesity is more prevalent in SMI, including schizophrenia (25.5%; Afzal et al., 2021), and it can lead to life-limiting diseases, including diabetes, heart disease, stroke, and cancer (Centers for Disease Control and Prevention, 2022). This may contribute to the mortality gap seen in SMI (reported estimates from 5 to 15 years) (Laursen et al., 2019; Lawrence et al., 2013; Plana-Ripoll et al., 2019), where reduction in life-years-lost from suicide has been offset by more life-years-lost from physical illnesses (Plana-Ripoll et al., 2020). In schizophrenia—where SGAs are most often used—this includes higher rates of obesity-related causes, including diabetes, heart disease, and stroke (Chen et al., 2022; Correll et al., 2022; Peritogiannis et al., 2022; Saha et al., 2007).
There have been calls for psychiatrists to play a more active role in addressing this health inequality by being more involved in supporting and/or managing their patients’ physical health care (Lawrence and Kisely, 2010; Lee et al., 2024; NHS England, 2024). SMI patients may form a vulnerable group, who may experience deprivation, marginalization, and have limited access to services. They may also lack insight and/or capacity or be otherwise hard to engage with due to the SMI. Hence, as doctors with regular contact with them, psychiatrists have a special duty of care to address their needs and are uniquely positioned to do so. This includes managing the adverse effects from SGAs that they prescribe such as weight gain and obesity (Lee et al., 2024; Saha et al., 2007).
Weight management typically involves making appropriate lifestyle adjustments (e.g., diet, exercise). Although there is meta-analytic evidence that behavioral interventions for AIWG are efficacious, the actual effect sizes are small and unlikely to translate into meaningful improvements in overall health (Álvarez-Jiménez et al., 2008). There has therefore been growing interest in the possible role of adjunctive pharmacotherapy to help combat antipsychotic-induced weight gain. Currently, antidiabetic medications are typically utilized for this purpose (Cooper et al., 2016; De Silva et al., 2016; Lee et al., 2022), though some of these medications (e.g., metformin) remain off-label. This can be limiting as not all patients may be suitable for these treatments. Additionally, psychiatrists do not typically prescribe or oversee such medications and may be thus less familiar with its management.
More recently, there have been developments in the use of centrally-acting opioid receptor antagonists (CORAs). This includes the approval of the combination medication naltrexone/bupropion for general weight management as well as the recent marketing of a olanzapine/samidorphan combination treatment, intended to mitigate the metabolic effects of olanzapine.
Background
Primarily indicated for the treatment of schizophrenia and bipolar affective disorder, antipsychotics are associated with weight gain (Huhn et al., 2019). A variety of mechanisms have been postulated to explain this propensity to increased appetite and subsequent weight gain. The traditionally understood mechanism of antipsychotic action—namely D2 dopamine receptor antagonism—is itself postulated to increase food consumption by diminishing the hedonic response to satiety mediated by dopamine transmission in the nucleus accumbens of the ventral striatum (Reynolds and Kirk, 2010).
The endogenous opioids, endorphin and enkephalin, have been implicated both in the homeostatic regulation of eating and the hedonic response to food consumption. Modulation of their effects at their primary receptors—mu-, kappa-, and delta-opioid receptors (MOR, KOR, and DOR)—has become an area of interest as a potential target for weight management pharmacotherapy (Mazereel et al., 2020; Nielsen et al., 2021; Reynolds and Kirk, 2010). Opioid receptor expression and stimulation in a variety of brain regions have been associated with increased food consumption and preference for high-calorie food in animal models (Murray et al., 2014; Nogueiras et al., 2012). Particularly, increased expression and binding of MOR in the nucleus accumbens is associated with binge eating and increased food consumption, as is direct administration of MOR agonists to this area, suggesting causal effects of MOR activation on behavioral drive to consume food (Murray et al., 2014). Furthermore, the administration of opioid antagonists, such as naloxone, nalmefene, and naltrexone, reduces both the amount of high-calorie food consumed in a single sitting and the rated pleasantness of high-calorie food in human trials, but without affecting reported levels of hunger (Yeomans and Gray, 2002). We can therefore speculate that MOR antagonists may redress the deficits in satiety signaling caused by D2 receptor antagonism, reducing overall food consumption.
The homeostatic regulatory effects of endogenous opioids on food consumption and energy expenditure are mediated by their actions on proopiomelanocortin (POMC) positive neurons found in the arcuate nucleus of the hypothalamus (ARC). POMC is a precursor peptide, the cleavage of which produces a range of neuropeptides and hormones, including adrenocorticotropic hormone, melanocyte-stimulating hormones, and the endogenous opiates beta-enkephalin and metenkephalin. Expressed widely during development, in adulthood, POMC is expressed in only in two specific locations; the POMC-positive neuronal population in ARC and the nucleus tractus solitarius of the brainstem (Zhan, 2018). These neuronal populations are sensitive to a range of neurotransmitters associated with appetite and feeding behaviors, including serotonin, endocannabinoids, and acetylcholine, as well as circulating hormone and nutrient molecules including insulin, leptin, ghrelin, glucose, and amino acids (Toda et al., 2017), making them a central node of convergent appetite regulation pathways. Activation of POMC neurons in ARC is associated with reduced food consumption and increased energy expenditure. This is evidenced by mice who have undergone selective ablation of POMC neurons in ARC, and in humans who have specific dysfunction of this neuronal population; both of whom exhibit hyperphagia and obesity (Toda et al., 2017; Zhan, 2018).
The Dopamine, Serotonin and Noradrenaline Receptor antagonists (DSNRAs), clozapine, and olanzapine are consistently found to have the greatest effect on bodyweight despite having low affinities for the D2 receptor (Dayabandara et al., 2017; Huhn et al., 2019). Instead, their obesogenic effects are thought to result from the high degree of antagonism they exert at the H1 histamine receptors and 5-HT2C serotonin receptors, effecting increased appetite and reducing satiety, respectively (Balt et al., 2011). H1 receptors are then further implicated, along with M1 muscarinic acetylcholine receptors by causing sedation, which causes reduced physical activity and therefore caloric expenditure (Green et al., 2000).
POMC neurons express serotonin 5-HTC2 receptors (Romanova et al., 2018), agonism of which results in depolarization of the neurons with anorectic consequences having demonstrated in both mice (Doslikova et al., 2013) and humans (Thomas et al., 2018). The obesogenic effects of medications used to treat psychosis with potent 5HT2C receptor antagonism, such as olanzapine and clozapine (Balt et al., 2011), are therefore mediated by directly reducing activity in this neuronal population and therefore its anorectic effects.
POMC neurons in ARC also express MOR, a G-coupled inhibitory receptor, on post-synaptic dendritic membranes. Agonism at this receptor reduces the activity of POMC, the behavioral consequence of which being increased food consumption (Nogueiras et al., 2012). The administration of MOR antagonists like naltrexone and samidorphan would therefore counter this action, reducing the inhibitory effects of endogenous opioids on POMC neurons, increasing their activity and so reducing caloric intake while increasing expenditure. By co-administering MOR antagonists with 5HT2C-antagonizing antipsychotic medications, imbalances in neurochemical regulation of POMC activity may be corrected, with a net normalization of homeostatic drive toward food consumption.
Taken together, this suggests a role for endogenous opioids in the control of feeding behaviors where release in ARC increases food consumption, and release in the nucleus accumbens reinforces feeding behaviors based on experienced pleasantness, possibly reinforcing a preference for high-calorie foods. Demonstration that opiate antagonists can attenuate these effects therefore suggests that this is a potentially helpful mechanism by which obesogenic effects of dopamine and serotonin receptor antagonists can be mitigated. As the existing evidence demonstrates that opioid antagonists reduce the drive to consume highly palatable foods, administration of these agents at the beginning of antipsychotic treatment may limit the degree of weight gain this treatment causes. It is important to recognize that the mechanism by which opioid antagonists may attenuate weight gain is distinct from those by which antipsychotics are thought to cause it. As the obesogenic effects of DSNRAs may not be dissociable from their therapeutic effects, a distinct mechanism acting at a different neurotransmitter system may be essential to avoid the loss of antipsychotic efficacy. As the endogenous opioid system appears to be central pathways on which other systems converge to regulate metabolism, this may represent an ideal candidate.
Objective
This systematic review and meta-analysis aimed to identify and analyze the available human clinical trial data for the effectiveness of prescribing CORA to manage AIWG.
Methodology
This systematic review and meta-analysis was conducted in accordance with guidelines set by the Preferred Reporting Items for Systematic Reviews and Meta-analysis (Page et al., 2021). The protocol was registered with PROSPERO (ID: CRD42023455663) (Lee et al., 2023).
Eligibility criteria
Only randomized-controlled studies (RCTs), and studies continuing from those trials, investigating the effect of CORA on weight in individuals receiving antipsychotic medication were included for this review. The primary outcome was mean bodyweight change, while secondary outcomes included other anthropometric parameters (e.g., body mass index (BMI), percentage achieving target weight change, etc), cardiometabolic parameters (e.g., lipid profile, glycated hemoglobin (HbA1c) levels, etc), and reported adverse outcomes. Further requirements for inclusion were that all participants were consenting adults (aged 16 and above) and received antipsychotics either shortly prior to or throughout the duration of the trial.
More specifically, the primary outcome examined was the difference in change in bodyweight in kilograms between antipsychotic plus CORA versus antipsychotic alone or antipsychotic plus placebo at the end of study follow-up. Secondary anthropometric outcomes examined were difference in changes in percentage bodyweight, BMI, and waist circumference, and the absolute risk difference (ARD) in gaining more than 7% bodyweight and gaining more than 10% bodyweight. The choice of ARD rather than odds ratio (the most common form of reporting in retrieved manuscripts) was made to improve interpretability and avoid overstating effect sizes (Chu et al., 2021). Also examined were secondary metabolic outcomes for the difference in change in total cholesterol (mg/dL), triglycerides (mg/dL), glucose (mg/dL), and HbA1c (%).
Other studies, such as observational studies and quasi-experimental studies, were not included in this review, to better facilitate meta-analysis of the data. Other exclusions were articles that were only available in abstract form and studies that lacked clear ethical approval. Trials involving children were not part of the review’s inclusion criteria, though no trials were excluded solely on this basis. Likewise, no language restrictions were placed to be included in the review.
Search strategy and selection process
Literature search was performed using MEDLINE, EMBASE, PsycINFO, and the Cochrane Library online databases. Search terms used included a combination of CORA medication names and terms relating to antipsychotics and weight or AIWG. A detailed list has been included in Appendix A.
An initial search was first performed on September 01, 2023, covering a period from the databases’ inception to the search date. A second, updated search was subsequently performed on August 07, 2024, covering from January 01, 2023 to the second search date to identify any articles published since the start of the review.
Initial screening was based on the articles’ titles and abstracts, with the search and screening completed independently by two authors. Differences in eligibility findings were discussed, and decision (screened in or out) was made through consensus.
Full-text evaluation for suitability based on the established inclusion and exclusion criteria was undertaken for all articles initially screened in, with all authors involved in this process. Each article was evaluated independently by at least two authors, and final decision for inclusion in the review was made through consensus agreement of all authors.
Data were extracted using a self-developed data extraction tool, which has been included in Appendix B.
Risk of bias assessment
Risk of bias assessment was undertaken for all studies included. RCTs were assessed using the Cochrane risk-of-bias tool for randomized trials, version 2 (RoB-2) (Sterne et al., 2019) while extension studies were assessed using the Risk Of Bias In Non-randomized Studies—of Interventions (Sterne et al., 2016); both developed by the Cochrane Bias Methods Group. Each study was assessed by at least two authors, with classifications determined through consensus.
Data analysis
Random effects meta-analyses for each outcome were performed using the metan command in Stata 13.0 (Fisher et al., 2024), applying the Restricted Maximum Likelihood ( Corbeil and Searle, 1976) method to estimate between-study variance. Continuous effect sizes were expressed as mean differences for changes in bodyweight (primary outcome), BMI, waist circumference, HbA1c, blood glucose, total cholesterol, and serum triglyceride. Dichotomous effect sizes were expressed as ARD for changes in probability of ⩾7% bodyweight gain and probability of ⩾10% bodyweight gain. All used confidence intervals (CIs) of 95%. Weighting was by the inverse variance method, ensuring that larger studies contributed more to the pooled estimate.
Due to incompatibility in controls (antipsychotic with placebo, placebo), samidorphan and naltrexone RCTs could not be combined for meta-analysis and have been analyzed separately. Insufficient studies were retrieved to perform meta-regression. The included studies were tested for heterogeneity using the I2 statistic and assessed subjectively.
Although too few studies permit sensitive assessment of publication bias, a funnel plot and trim-fill analysis were consulted using JASP (Version 0.18.3) (JASP Team, 2024) for the primary outcome and did not show evidence of publication bias. Egger’s test was not carried out due to the small number of trials (n < 10), limiting its reliability.
Results
Initial search of the online databases on September 01, 2023 yielded 450 records (Figure 1), with 321 records remaining for manual screening after duplicates were removed. Of these, 297 were screened out, and full-text assessment was undertaken for the remaining 24 records. Thirteen were excluded (abstract only = 9, unrelated outcome measures = 4), with 11 articles accepted for inclusion in the narrative synthesis. This consisted of nine RCT articles (Correll et al., 2020, 2023; Kahn et al., 2023; Lyu et al., 2018; Martin et al., 2019; Meyer et al., 2023; Silverman et al., 2018; Taveira et al., 2014; Tek et al., 2014) and two open-label study articles (Kahn et al., 2021; Yagoda et al., 2021) that were extensions of RCTs. Four of the articles (RCT analyses = 3 (Correll et al., 2023; Meyer et al., 2023); open-label extension = 1 (Kahn et al., 2021)) were from a single trial (ENLIGHTEN-2).
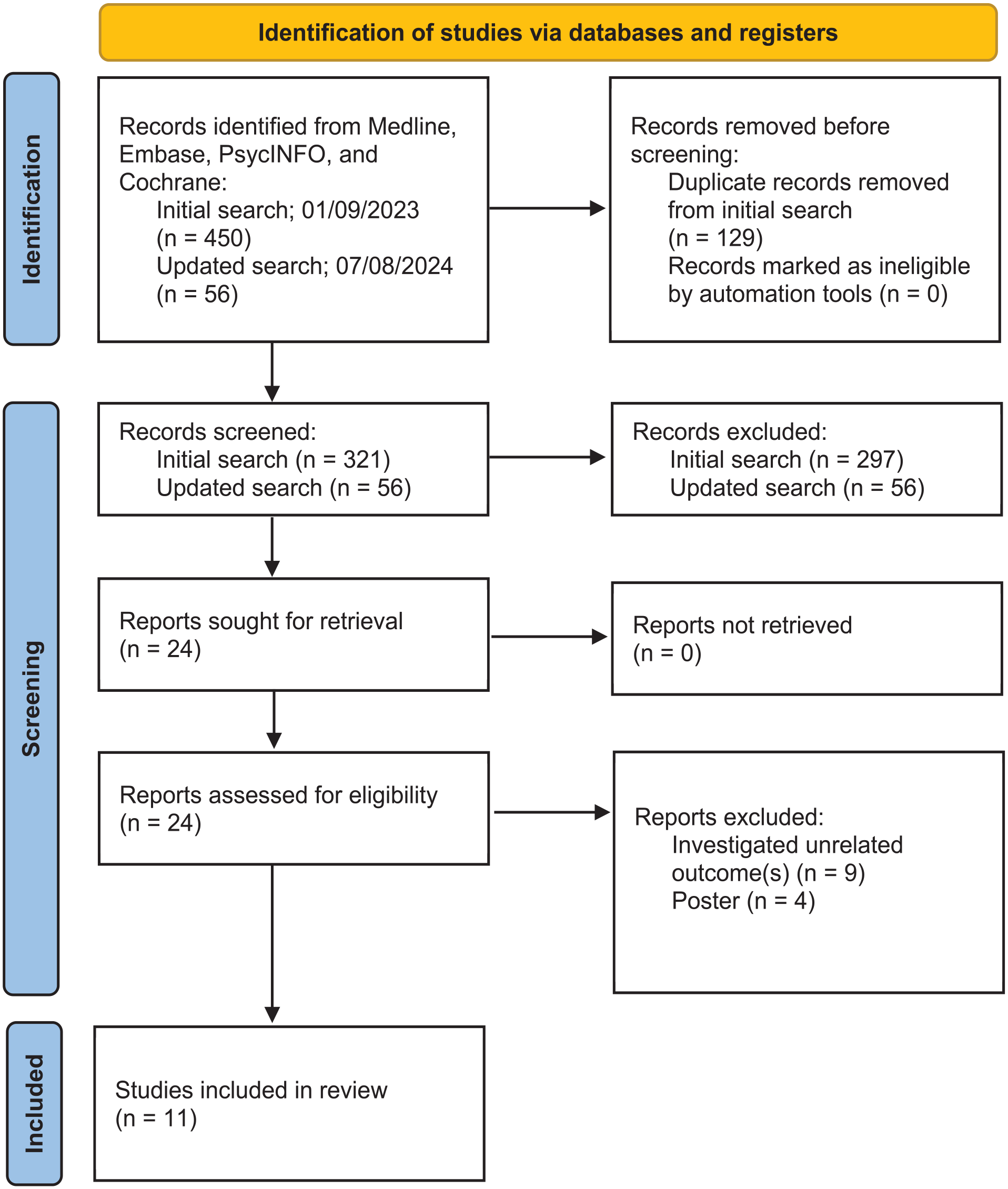
Article selection process.
One notable exclusion was Potkin et al. (2020), which was the original clinical trial (ENLIGHTEN-1) for its open-label extension study Yagoda et al. (2021). The aim of ENLIGHTEN-1 was to assess efficacy and safety with respect to psychiatric outcomes, demonstrating the non-inferiority of olanzapine/samidorphan to olanzapine alone for schizophrenia. A second search of the same online databases on August 07, 2024 yielded 56 records (Figure 1) but all were screened out either for lack of relevance or for already having been identified from the original search.
The descriptions and demographics of the accepted articles are described in Tables 1 and 2, and their results are in Table 3. In total, 9 separate trials were identified across 11 articles; totaling 1416, participants (intervention = 779; control = 637; extension studies only = 431). Of the included trial analyses, five investigated samidorphan across eight articles (Correll et al., 2020, 2023; Kahn et al., 2021, 2023; Martin et al., 2019; Meyer et al., 2023; Silverman et al., 2018; Yagoda et al., 2021) and three investigated naltrexone (naltrexone monotherapy = 2 (Taveira et al., 2014; Tek et al., 2014); naltrexone/bupropion = 1 (Lyu et al., 2018)). No other CORA (e.g., naloxone, nalmefene) was investigated, and there were no trials investigating multiple CORAs. No studies had a comparator arm that used a weight-managing agent/intervention (e.g., metformin, GLP-1 receptor agonist), and all blinded participants to treatment allocation. Naltrexone studies compared to placebo and combination olanzapine/samidorphan compared to olanzapine alone. Study periods ranged from 21 days to 52 weeks (RCTs only = 21 days to 24 weeks). All but two studies (Lyu et al., 2018; Silverman et al., 2018) were conducted in outpatient settings. One samidorphan study (Martin et al., 2019) conducted subgroup analysis based on dose (samidorphan 5–20 mg), though it should be noted that samidorphan 10 mg is the only approved dose (combined with variable doses of olanzapine) and that it is not approved for doses above 10 mg.
Article characteristics.
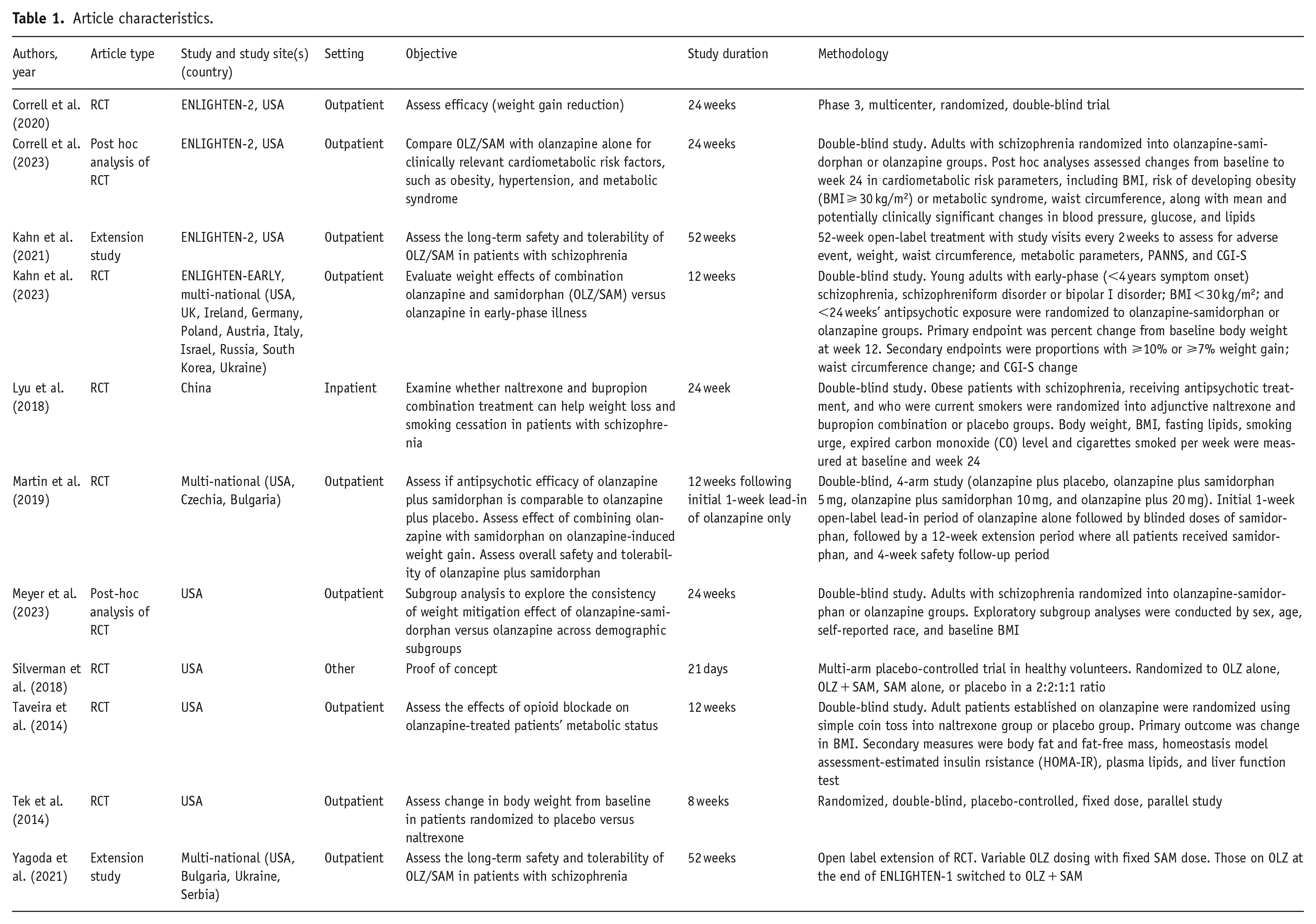
Participant demographics.
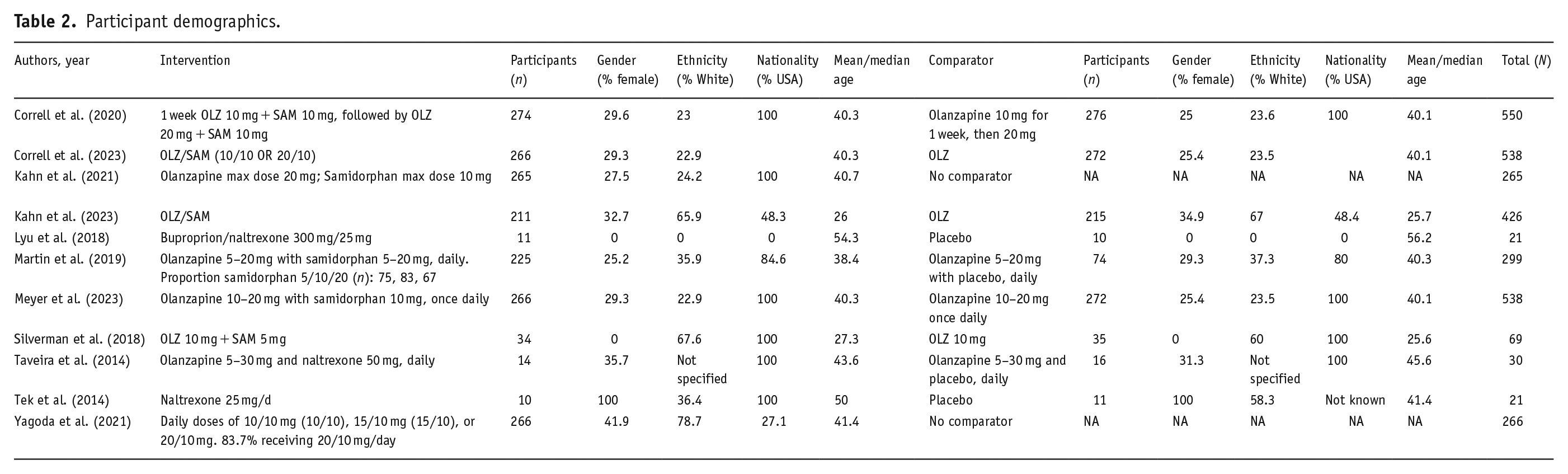
Article results and conclusions.
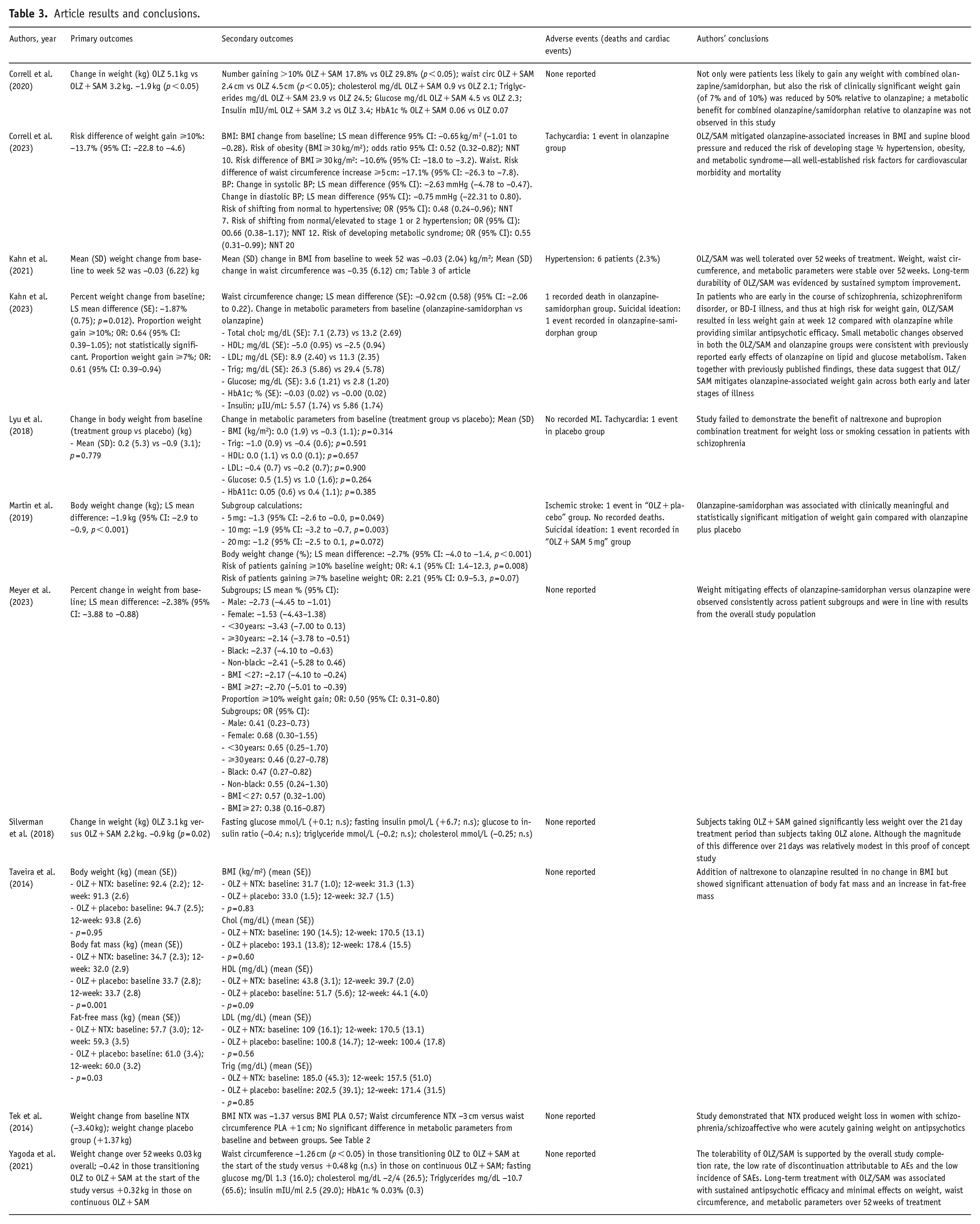
Risk-of-bias assessments for each study are described in Table 4. Gradings were varied among RCT articles (low risk = 3; some concerns = 5; high risk = 1) owing to heterogeneity in methodology. By contrast, the open-label trials fared more poorly; with both at high risk of bias, owing to risk of confounding (domain 1).
Risk-of-bias assessments.
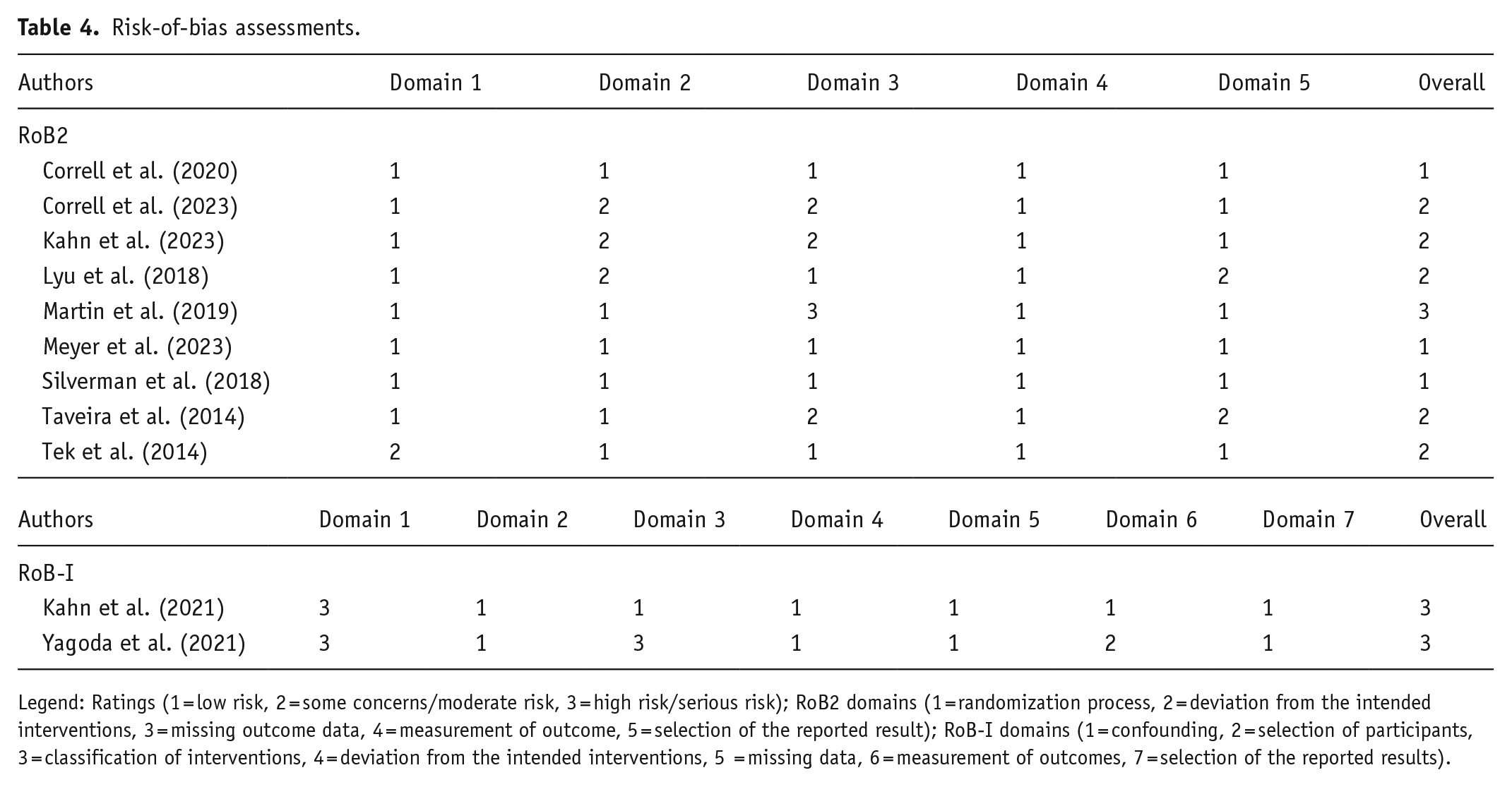
Legend: Ratings (1 = low risk, 2 = some concerns/moderate risk, 3 = high risk/serious risk); RoB2 domains (1 = randomization process, 2 = deviation from the intended interventions, 3 = missing outcome data, 4 = measurement of outcome, 5 = selection of the reported result); RoB-I domains (1 = confounding, 2 = selection of participants, 3 = classification of interventions, 4 = deviation from the intended interventions, 5 = missing data, 6 = measurement of outcomes, 7 = selection of the reported results).
For primary outcome, mean difference in bodyweight change was statistically significant for samidorphan when trialed as olanzapine/samidorphan against olanzapine (MD: −1.18 kg; 95% CI: −1.67 to −0.68; p < 0.001) while naltrexone was not when evaluated against placebo. Due to the small number of studies using naltrexone-containing interventions (n = 3), it was thought more meaningful for naltrexone/bupropion to be analyzed with naltrexone monotherapy studies in the naltrexone subgroup. However, neither naltrexone monotherapy nor naltrexone/bupropion was significant when analyzed separately. Primary outcome funnel plot and sensitivity analysis have been included in Appendix C. No publication bias was found for the primary outcome (Figure 2).
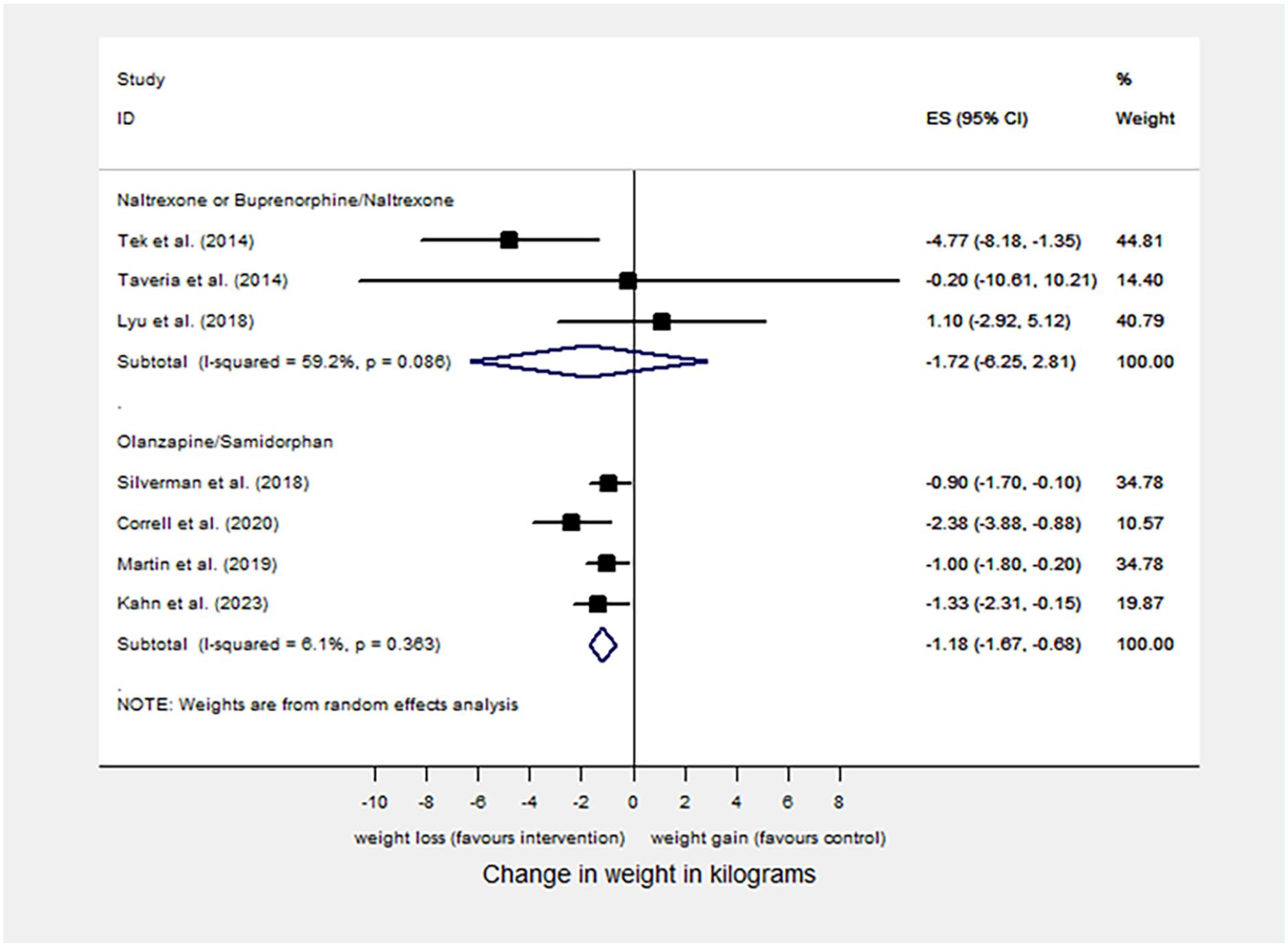
Forest plot for mean difference in bodyweight change (kg).
Secondary outcomes meta-analyzed included other weight and obesity-related outcomes (BMI, risk of weight gain) and cardiometabolic outcomes (cholesterol, triglyceride, glucose, glycated hemoglobin (HbA1c)), and tabulated in Tables 5 and 6, with their respective forest plots included in Appendix D. BMI change was found to be statistically significant, favoring intervention, for olanzapine/samidorphan but not significant in the naltrexone trials; though it is noted that the evidence for olanzapine/samidorphan around BMI consists of a single RCT (Correll et al., 2023) and has yet to be replicated. Three trials (Correll et al., 2023; Kahn et al., 2023; Martin et al., 2019) compared the risk of weight gain with olanzapine/samidorphan against standalone olanzapine and this was statistically significant for both, weight gain ⩾7% (risk difference: −12.40%; 95% CI: −18.27 to −6.54; p < 0.001) and ⩾10% (risk difference: −10.83%; 95% CI: −16.21 to −5.45; p < 0.001) on meta-analysis. However, neither were statistically significant in Martin et al. (2019) and only Correll et al. (2023) was statistically significant for ⩾10% weight gain when these results are expressed in risk difference.
Summary of meta-analyses of anthropometric outcome measures.
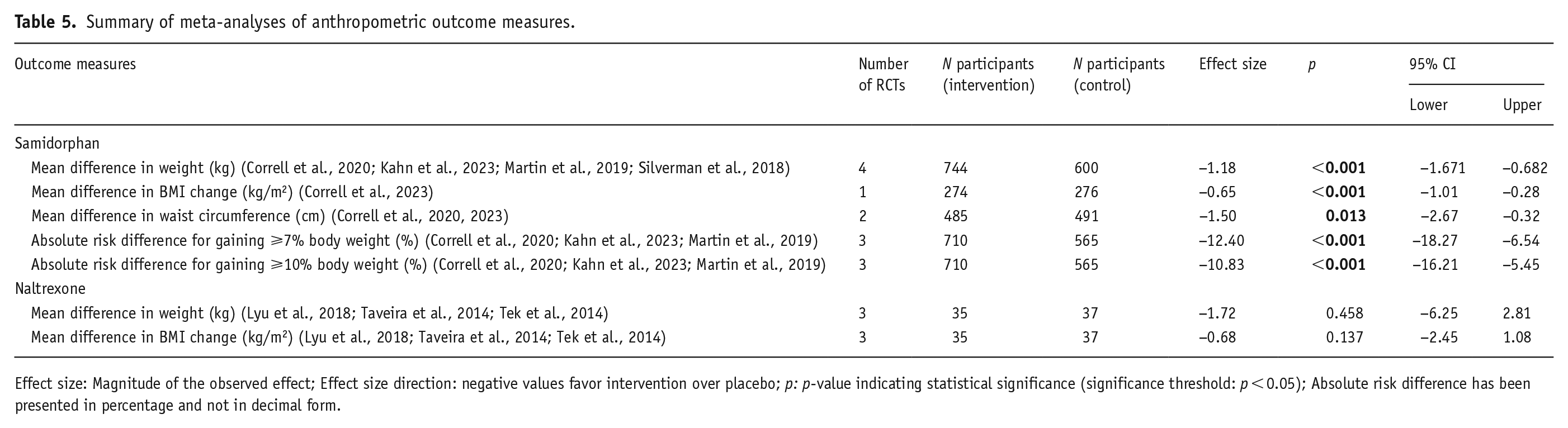
Effect size: Magnitude of the observed effect; Effect size direction: negative values favor intervention over placebo; p: p-value indicating statistical significance (significance threshold: p < 0.05); Absolute risk difference has been presented in percentage and not in decimal form.
Summary of meta-analyses of laboratory cardiometabolic outcome measures.
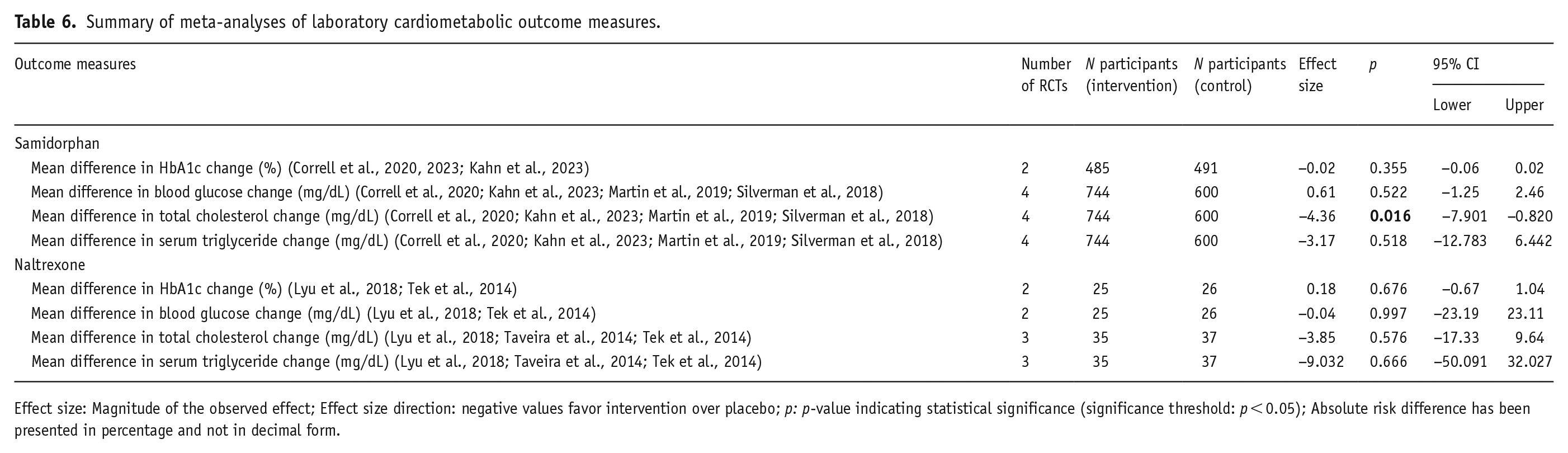
Effect size: Magnitude of the observed effect; Effect size direction: negative values favor intervention over placebo; p: p-value indicating statistical significance (significance threshold: p < 0.05); Absolute risk difference has been presented in percentage and not in decimal form.
Analysis by baseline demographical subgroup was conducted by two olanzapine/samidorphan trials (Martin et al., 2019; Meyer et al., 2023) around treatment dose, sex, age, ethnicity, and baseline BMI. Here, it was found that weight change was not statistically significant in the following subgroups—samidorphan 20 mg (mean: −1.3 kg, 95% CI: −2.5 to 0.1) (Martin et al., 2019), female (mean: −1.53%, 95% CI: −4.43 to 1.38) (Meyer et al., 2023), under 30-year-old (mean: −3.43%, 95% CI: −7.00 to 0.13) (Meyer et al., 2023), and non-black subgroups (mean: −2.41%, 95% CI: −5.28 to 0.46) (Meyer et al., 2023).
In the open-label extension studies, participants taking established olanzapine treatment were initiated on samidorphan alongside the treatment group; with all participants then followed-up for 52 weeks. The authors found that olanzapine/samidorphan was well tolerated, with low rate of discontinuation attributable to adverse effects, and antipsychotic efficacy was maintained long-term (Kahn et al., 2021; Yagoda et al., 2021). After 52 weeks, mean weight change was −0.03 kg (SD: 6.22) (Kahn et al., 2021); with little meaningful difference in terms of weight change between participants newly initiated on samidorphan at the beginning of the trial extension, and those previously established on samidorphan (−0.42 vs 0.32 kg) (Yagoda et al., 2021).
Discussion
Interpretation
Our findings indicate that olanzapine/samidorphan partially mitigates AIWG when compared against olanzapine. We did not find any effect of naltrexone alone or in combination with bupropion on AIWG.
The effect on reducing weight gain appears smaller than comparable treatment options, such as GLP-1 receptor agonists (Khaity et al., 2023) and metformin, at face value (De Silva et al., 2016). However, this difference may be related to study design factors and direct head-to-head trials would be beneficial.
Overall, our findings potentially point to CORA only having a narrow use case in AIWG, in the presence of other available options. While treatment with GLP-1 receptor agonists and metformin may be better established, the former is administered by injection, and both are medication groups not typically prescribed by psychiatrists. Instead, they typically fall into the domain of endocrinologists and general practitioners, who prescribe them for Type 2 diabetes mellitus. This may limit access to those medications without the involvement of the appropriate specialists. Furthermore, there is developing concern regarding development of suicidal ideation among patients prescribed GLP-1 receptor agonists (Guirguis et al., 2024; McIntyre et al., 2024; Tobaiqy and Elkout, 2024) though such links remain unproven with surveillance remaining ongoing (European Medicines Agency, 2024; Food and Drug Association, 2024; Wang et al., 2024). Aripiprazole—whether as adjunct or in place of the patient’s regular antipsychotic—is another option described in guidelines by the British Association of Psychopharmacology, Maudsley, and American Psychiatric Association (2020; Cooper et al., 2016; Taylor et al., 2020). However, this may involve either reducing/discontinuing the patient’s regular antipsychotic or exposing the patient to high combined doses of antipsychotic, and so may not always be suitable. In that respect, it may be that CORA is an option when psychiatrists are unable to access metformin and/or GLP-1 receptor agonists; there is concern around risk to self (e.g., suicidal ideation, self-harm); and/or introduction of aripiprazole is unsuitable.
Limitations
The main limitation is that trials evaluating samidorphan and naltrexone used different comparators; preventing their results from being combined for meta-analysis. There was also a lack of head-to-head comparison against other advocated treatments (e.g., metformin, GLP-1 receptor agonists, aripiprazole, nonpharmacological interventions) to investigate superiority.
Furthermore, there was heterogeneity in the included studies’ methodology. This included study duration (8–52 weeks); medication and dose (naltrexone 25–50 mg, naltrexone/bupropion 25 mg/300 mg, samidorphan 5–20 mg); sample size (21–550 participants); and target population (individuals with diagnosed SMI, healthy individuals). This limited the interpretation and what conclusions could be drawn from the results, due to the variables’ potential to influence effect size and significance.
For instance, Lyu et al. (2018) was conducted in an inpatient setting and sought to investigate both, the weight-reducing and smoking-cessating properties of naltrexone/bupropion simultaneously. These are potential confounders as smoking cessation is associated with weight gain (Filozof et al., 2004), whereas the inpatient setting may limit participants’ access and opportunity to engage in regular physical activity.
Additionally, RCTs investigating naltrexone (including naltrexone/bupropion) were small in scale (n ⩽ 30) and may have been underpowered to detect significant results. This contrasts with the studies using samidorphan, which were generally larger, with all but Silverman et al. (2018)—a phase 1 trial—involving over 200 participants each. In the trials of olanzapine/samidorphan, the CORA was initiated at the same time as the antipsychotic. The trials of naltrexone versus placebo involved initiating the CORA in patients who had already experienced weight gain.
Samidorphan studies solely included participants receiving olanzapine, which is recognized as one of the most obesogenic antipsychotics (Huhn et al., 2019), as opposed to other antipsychotics. The observed effect may not generalize to other antipsychotics. It is plausible that samidorphan may have a greater or lesser effect on AIWG from other, less obesogenic antipsychotics. This limitation in the literature may be a consequence of samidorphan being developed as, and currently only available in, the olanzapine/samidorphan combined preparation. We are however unaware of any mechanistic reason to anticipate samidorphan working differently if paired with other antipsychotics. Combined, these differences in methodology between samidorphan and naltrexone trials render it impossible to say whether there is a class effect of CORAs.
RCT length was limited to 24 weeks or less. Longer-term data—which was limited only to samidorphan—was instead based on open-label, extension studies (Kahn et al., 2021; Yagoda et al., 2021) where all participants received the intervention (specifically, samidorphan), with there being no long-term RCTs comparing CORA recipients and non-CORA recipients. It is possible that the relatively modest weight reduction obtained for CORA may be due to the duration of the trials and that RCTs of longer duration could produce more pronounced results.
Geographically, study sites were heavily concentrated in the United States and Europe, with only two studies including participants from outside this region—specifically, Israel (Kahn et al., 2023), South Korea (Kahn et al., 2023), and China (Lyu et al., 2018). Populations from Africa, Australasia, South America, and South Asia were completely absent from the available data, and we were thus unable to include them to inform our review and meta-analysis.
Future directions
Our findings identify early but conflicting results around the efficacy of CORA at managing AIWG. We observed that different medications within the CORA class potentially produce significantly different clinical effects. One possible explanation may be differences in their pharmacological properties; with Tan et al. (2022) describing samidorphan as having a higher affinity to MOR and DOR receptors, and more potent antagonism at MOR, when compared to naltrexone, and Cunningham et al. (2019) suggesting that it may be contributed by samidorphan having peripheral—in addition to central—effects. Further research in these dimensions could shed light on the pathophysiology of AIWG and development of more effective medications for it. The olanzapine/samidorphan trials reported their primary outcomes with an emphasis on the number of participants gaining 7% or 10% bodyweight. The results from our analysis demonstrate rather modest mean reductions in weight gain, waist circumference, and BMI which are considerably less flattering to the medications overall efficacy.
Currently, samidorphan shows the most promise amongst existing CORA. However, there is a need for further study and replication. It is notable that all samidorphan trials identified share the same commercial funder. Independent study would be desirable to see; as would further trials involving non-Western populations and different antipsychotics; using more standardized methodology and consistent outcome measures that include its clinical usefulness.
Other avenues of research would be more detailed exploration of its safety profile, including any potential effects on mental health symptoms and/or interactions with antipsychotics; economic studies and/or cost-effectiveness; as well as long-term outcomes. Ideally, there would also be head-to-head trials comparing its efficacy against other treatment options to more directly investigate superiority, as this may inform clinical practice and where CORA sits—if at all—within treatment guidelines for AIWG.
Conclusion
Overall, our systematic review and meta-analysis found that CORA was effective at reducing AIWG. However, the evidence for this is moderate at best, with a lack of studies that were low risk of bias, and limited to samidorphan only—which produced only a small effect size on subgroup analysis. Studies using naltrexone had mixed results, whilst no data were available for naloxone and nalmefene. With samidorphan, evidence was limited to individuals prescribed olanzapine, with no data for any other antipsychotic. All of this highlights a need for further research around CORA in AIWG.
Supplemental Material
sj-docx-1-jop-10.1177_02698811251337374 – Supplemental material for Centrally-acting opioid receptor antagonists as a treatment for antipsychotic-induced weight gain: A systematic review and meta-analysis of clinical trial data
Supplemental material, sj-docx-1-jop-10.1177_02698811251337374 for Centrally-acting opioid receptor antagonists as a treatment for antipsychotic-induced weight gain: A systematic review and meta-analysis of clinical trial data by Kenn Lee, Matthew Twohig, Nguemo Pauline Idoko and Benjamin David Williams in Journal of Psychopharmacology
Supplemental Material
sj-docx-2-jop-10.1177_02698811251337374 – Supplemental material for Centrally-acting opioid receptor antagonists as a treatment for antipsychotic-induced weight gain: A systematic review and meta-analysis of clinical trial data
Supplemental material, sj-docx-2-jop-10.1177_02698811251337374 for Centrally-acting opioid receptor antagonists as a treatment for antipsychotic-induced weight gain: A systematic review and meta-analysis of clinical trial data by Kenn Lee, Matthew Twohig, Nguemo Pauline Idoko and Benjamin David Williams in Journal of Psychopharmacology
Supplemental Material
sj-docx-3-jop-10.1177_02698811251337374 – Supplemental material for Centrally-acting opioid receptor antagonists as a treatment for antipsychotic-induced weight gain: A systematic review and meta-analysis of clinical trial data
Supplemental material, sj-docx-3-jop-10.1177_02698811251337374 for Centrally-acting opioid receptor antagonists as a treatment for antipsychotic-induced weight gain: A systematic review and meta-analysis of clinical trial data by Kenn Lee, Matthew Twohig, Nguemo Pauline Idoko and Benjamin David Williams in Journal of Psychopharmacology
Supplemental Material
sj-docx-4-jop-10.1177_02698811251337374 – Supplemental material for Centrally-acting opioid receptor antagonists as a treatment for antipsychotic-induced weight gain: A systematic review and meta-analysis of clinical trial data
Supplemental material, sj-docx-4-jop-10.1177_02698811251337374 for Centrally-acting opioid receptor antagonists as a treatment for antipsychotic-induced weight gain: A systematic review and meta-analysis of clinical trial data by Kenn Lee, Matthew Twohig, Nguemo Pauline Idoko and Benjamin David Williams in Journal of Psychopharmacology
Supplemental Material
sj-docx-5-jop-10.1177_02698811251337374 – Supplemental material for Centrally-acting opioid receptor antagonists as a treatment for antipsychotic-induced weight gain: A systematic review and meta-analysis of clinical trial data
Supplemental material, sj-docx-5-jop-10.1177_02698811251337374 for Centrally-acting opioid receptor antagonists as a treatment for antipsychotic-induced weight gain: A systematic review and meta-analysis of clinical trial data by Kenn Lee, Matthew Twohig, Nguemo Pauline Idoko and Benjamin David Williams in Journal of Psychopharmacology
Footnotes
Acknowledgements
None.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
