Abstract
Background:
Social behaviour is the expression of one of the most generally accepted independent dimensions of personality. Serotonergic neurotransmission has been implicated in typical social response and drugs that promote serotonin (5-hydroxytryptamine (5-HT)) release have prosocial effects. By using the social interaction test, we have previously demonstrated sociability as a temperamental trait in male Wistar rats.
Aims:
To assess sociability in male rats of the Sprague–Dawley strain and in female rats of both Wistar and Sprague–Dawley strain, and extracellular levels of 5-HT in rats with high and low sociability (high sociability (HS)- and low sociability (LS)-rats).
Methods:
Social interaction test conducted with different weight-matched partners was used to assess sociability, and in vivo, microdialysis was performed before and after administration of a low dose (2 mg/kg) of parachloroamphetamine (PCA) in the prefrontal cortex, dorsamedial striatum and ventral tegmental area.
Results:
Similarly to male Wistar rats, female Wistars and Sprague–Dawley rats of both sexes displayed trait-wise sociability. Male Wistar HS-rats had lower extracellular levels of 5-HT in prefrontal cortex at baseline and after administration of PCA, and higher PCA-induced increase of extracellular 5-HT in ventral tegmental area. In dorsomedial striatum, PCA elicited a comparable increase in extracellular dopamine in HS- and LS-rats, but higher release of 5-HT in HS-rats. Comparison of PCA-induced 5-HT release in prefrontal cortex of male and female Sprague–Dawley rats revealed a larger 5-HT response in female HS-rats.
Conclusions:
5-HT release potential is higher in rats with high expression of sociability trait, whereas some regionally variable differences may be related to relative contributions of social motivation and anxiety in shaping social behaviour.
Keywords
Introduction
Sociability, an important dimension in the structure of temperament/personality in humans and animals, is an individual’s tendency or propensity to associate with other individuals, the association not being driven by reproductory or aggressive urges (Gartland et al., 2022), which defines the ability of the individual to function in a social environment and has a major impact on health (Seebacher and Krause, 2019). The roots of social behaviour are deep in evolutionary history but its modern neurobiological underpinnings appeared with mammals (MacLean, 1985; Panksepp, 1998). Many different neurotransmitter systems and neuromodulators are implicated in social behaviour (Battivelli et al., 2024; Jurek and Neumann, 2018; Kask et al., 1998; Poshivalov, 1981; Robinson and Gradinaru, 2018; Yizhar and Levy, 2021), but 5-hydroxytryptamine (serotonin; 5-HT) appears as the most universal neurotransmitter in the expression of sociability-related personality dimensions (Glover et al., 2022; Knutson et al., 1998; Tse and Bond, 2002). A variety of studies have revealed that traits negatively associated with sociability, such as high impulsivity, aggressiveness and anxiety in humans, non-human primates and other species are linked to low 5-HT neurotransmission (da Cunha-Bang and Knudsen, 2021; Fahlke et al., 2002; Grimmett and Sillence, 2005; Suomi, 2005; Turecki, 2005). 5-HT-ergic function-enhancing compounds reduce the negative affective experience and increase affiliative behaviour in healthy humans and other primates (Knutson et al., 1998; Nichols, 2022; Shively et al., 2017). Action of recreationally used drugs, such as 3,4-methylenedioxymethamphetamine (MDMA), that release 5-HT is perceived as prosocial, and enhances positive affective response to social feedback (Bershad et al., 2024). However, a negative association between 5-HT turnover and social competence has also been described in a study on monkey dominance hierarchy (Yodyingyuad et al., 1985), but this appears to be changeable as social groups take form.
Expression of social behaviour in real-time depends on several factors. Nevertheless, research on human temperament has demonstrated that sociability is a persistent trait (Martin et al., 2023). By applying the social interaction test (File and Hyde, 1978) that had mostly been used to measure the effects of anxiogenic and anxiolytic drugs, we have demonstrated that male Wistar rats possess an individually characteristic level of social activity, which is in correlation with ex vivo levels of 5-hydroxyindoleacetic acid (5-HIAA), the major stable metabolite of 5-HT, in the frontal cortex (Tõnissaar et al., 2004). While social behaviour is dependent on situational factors, including the social activity of the partner, the average behaviour across tests with variable novel partners predicted performance in each test, and the vulnerability to anhedonia induced by chronic variable mild stress (Tõnissaar et al., 2008). Oxidative metabolism mapping by cytochrome oxidase histochemistry revealed an association of the sociability trait with the activity in brain regions controlling motivation, stress reactivity and anxiety (Kanarik and Harro, 2018).
Remarkable differences have been reported between common rat strains in some tests of social behaviour (Bagosi et al., 2023; Netser et al., 2020), and these may relate to sex differences in serotonergic function (da Silva et al., 2022; Poceviciute et al., 2023). The behaviour of male and female rats in anxiety tests may also remarkably differ (Johntson and File, 1991). The present study therefore extended the assessment of sociability to female Wistar rats and to another rat strain, the Sprague–Dawley rats. The main focus was however on the comparison of in vivo extracellular 5-HT levels and 5-HT release potential in animals with low and high sociability (HS) by administration of a low dose of parachloroamphetamine (PCA), a drug that fully substitutes for MDMA in the drug discrimination test (Baker et al., 1995). Both PCA and MDMA increase extracellular levels of monoamines owing to binding to vesicular and neuronal membrane monoamine transporters. Because PCA and MDMA also release, to a smaller extent, dopamine (Iyer et al., 1994), and the combined action of 5-HT and dopamine may be responsible for their unique psychopharmacological properties (Bankson and Cummingham, 2001; Schenk and Highgate, 2021), this was also measured in some experiments.
Material and methods
Animals and general procedure
Overview of methods in each of the four experiments (and their main outcome) is presented in Supplemental Table 1, and order of procedures in Supplemental Figure 1. Male Wistar rats for microdialysis studies in Experiment I (n = 34) and Experiment II (n = 28) at the University of Tartu animal house were obtained from Scanbur BK AB, Sweden. Wistar rats (male n = 30, female n = 20) used in sex comparisons in Experiment III were bred locally at the Tallinn University of Technology animal house from parent rats obtained from Charles River Laboratories. Male (n = 20) and female (n = 20) Sprague–Dawley rats in Experiment IV were obtained from the breeding centre of the Institute of Biomedicine and Translational Medicine of the University of Tartu and tested at the Tallinn University of Technology animal house. Animals were treated according to European Directive 2010/63/EU and National Animal Welfare legislation. They were accommodated in conventional transparent polycarbonate or H-TEMP polysulfone cages with a proper floor area, covered with aspen bedding (with enrichment in the form of nesting material, chew sticks, shelter in Experiments III and IV) under controlled light cycle (lights on 08:00–20:00), temperature (22°C–24°C) and ad libitum access to clean (filtered by reverse osmosis and UV treated) water and food pellets (V1534-000 Rat/Mouse Maintenance complete diet, ssniff Spezialdiäten GmbH, Germany). Before the experiments, animals were group-housed.
All procedures were carried out in compliance with the Animal Research Reporting of In Vivo Experiments (ARRIVE) guidelines and protocols were approved by the Ethical Committee for Animal Experiments of the Estonian Ministry of Rural Affairs (permissions nr 127, 134, 149) in accordance with Directive 2010/63/EU of the European Union.
Measurement of social behaviour and sociability
Ten days before the beginning of the experiments animals were housed individually. The social interaction test (File et al., 1993) was carried out in three (Experiments I–III) or four (Experiment IV) separate sessions with 7–10-day intervals as previously described (Kanarik and Harro, 2018; Tõnissaar et al., 2004). Two unfamiliar, weight-matched rats of the same sex were placed for 10 min into a brightly lit chamber (30 cm × 30 cm × 60 cm) with a floor covered with wood shavings. Rats were paired on the basis of their body weight before each test and always tested with a novel, unfamiliar partner. The total time spent in active social behaviour (allogrooming, sniffing the partner, crawling under and over, following) was recorded manually by one or two experienced researchers. Inter-rater reliability was always >0.90. The mean score of time spent in social interaction was normally distributed in all behavioural experiments. The rats were classified as low sociability (LS) or high sociability (HS) animals based on the median split of their average social activity over all social interaction tests (Supplemental Figure 2).
Microdialysis
This was carried out in freely moving animals with microdialysis probes implanted on the previous day. Microdialysis on male Wistars (Experiments I and II) was performed more than 10 years ago and then the animals were anaesthetized with chloral hydrate (350 mg/kg, IP; Orion); in the recent Experiment IV on Sprague–Dawley rats anaesthesia was achieved with a mixture of ketamine (60 mg/kg, IP; Bioketan, Vetoquinol SA) and medetomidine (0.5 mg/kg, IP; Dorbene Vet, Syva SA). Y-shaped home-made microdialysis probes were used in all experiments. The dialysis membrane used was polyacrylonitrile/sodium methylsulphonate copolymer (Filtral 12; inner diameter 0.22 mm; outer diameter 0.31 mm; AN 69, Hospal, Bologna, Italy). In Experiment I, one probe with 5 mm shaft length and 4 mm active tip was implanted into the medial prefrontal cortex and the second with an 8.5 mm shaft length and 1 mm active tip into the ventral tegmental area according to coordinates from Paxinos and Watson (1986). The coordinates for implantation were as follows; prefrontal cortex: AP 3.3 mm, ML −0.8 mm, DV −5.0 mm; ventral tegmental area: AP −5.3 mm, ML −2.5 mm, DV −8.4 mm, implanted at an angle of 12°, from bregma and dura. In Experiment II, a microdialysis probe with 5.5 mm shaft length and 2.5 mm active tip was implanted into the dorsomedial striatum with the coordinates: AP +0.2 mm, ML: +3.0 mm (left), DV: −5.3 mm, implanted at an angle of 11.3°, from bregma and dura. In Experiment IV, the probe with 5 mm shaft length and 4 mm active tip was implanted into the medial prefrontal cortex (AP 3.3 mm, ML −0.8 mm, DV −5.0 mm). Two stainless steel screws and dental cement were used to fix the probes to the scull. Lidocaine 1% solution (Lidocaine, Grindeks) was used locally prior to surgical procedures and meloxicam (Meloxidyl, 1 mg/kg SC; Ceva Sante Animale) for alleviating postoperative pain. After the surgery, rats were placed in individual cages in which they remained throughout the experiment. Atipamezole (1 mg/kg SC; Alzane, Syva SA) was administered to facilitate the recovery from general anaesthesia. Animals were placed in 21 cm × 36 cm × 18 cm individual cages in which they remained throughout the microdialysis experiment. Rats were given about 24 h for recovery and microdialysis procedure was conducted in awake freely moving animals. After microdialysis, rats were decapitated and brains were dissected to verify the location of the probe visually.
Microdialysis probes were connected to syringe pumps (World Precision Instruments, Inc, Sarasota, FL, USA) and sample collectors with flexible FEB-tubing (i.d. 0.12 mm; AgnTho’s AB, Sweden) and perfused with the Ringer solution (147 mM NaCl, 4 mM KCl, 1.2 mM CaCl2, 1.0 mM MgCl2, 1.0 mM Na2HPO4, 0.2 mM NaH2PO4; pH 7.20–7.22) at a constant rate of 1.5 μl/min. After connecting the animal to the microdialysis system, the perfusate was discarded during the first 120 min to allow for stabilization. Then six baseline samples were collected, followed by the administration of PCA (2 mg/kg, IP); thereafter further samples were collected. All rats received PCA since we had observed that under similar conditions vehicle treatment does not lead to monoamine release (Kanarik et al., 2024, unpublished). In all experiments, samples were collected in 15-min periods either into vials prefilled with 7.5 μl of 0.02 M acetic acid or injected automatically into the column for online measurement. Upon completion of the experiment, the animals were deeply anaesthetized and, the brains were removed.
Microdialysates were assayed for 5-HT (and dopamine (DA) in Experiments II and IV) using high-pressure liquid chromatography (HPLC) with electrochemical detection, as previously described (O’Leary et al., 2016), with some variation between experiments that did not appear to affect the results. In all experiments, Luna C18(2) columns (150 × 2 mm, 5 µm), kept at 30°C, were used, and the mobile phase was pumped through the column at a rate of 0.2 ml/min. The mobile phase contained: 0.05 M sodium citrate buffer at pH 5.3; 0.02 mM ethylenediaminetetraacetic acid, 2 mM KCl (in all experiments); 4.1 mM (Experiment I), 4.9 mM (Experiment II) or 3.5 mM (Experiment IV) sodium octantesulphonate; 18% (Experiment I), 18.5% (Experiment II) or 14% (Experiment IV) acetonitrile. In Experiment I, the chromatography system consisted of the Agilent series 1100 pump and thermostatted autosampler, column thermostat and ESA Coulochem II detector with ESA 5011 analytical cell. The potential of the electrode used for measurements was +250 mV. In Experiments II and IV the HPLC system consisted of a LC-10AD or 20AD pump, CBM-10A or 20A controller (Shimadzu Corporation, Kyoto, Japan), Decade II digital electrochemical amperometric detector (Antec Leyden BV, Zoeterwoude, The Netherlands) with electrochemical flow cell VT-03 (2 mm GC WE, ISAAC reference electrode, Antec Leyden BV). 5-HT and DA eluted from the column were measured with a glassy carbon working electrode maintained at a potential of +0.4 V versus Ag/AgCl reference electrode; data were acquired using a Shimadzu LC Solution system. In all experiments, the concentrations of monoamines were estimated by comparing peak heights from the microdialysates with those of external standards of known concentration of 5-HT and DA.
Data analysis
Statistical analysis of the behavioural data was performed using the GraphPad Prism 8 software (GraphPad Software, Boston, MA, USA). t-Test comparisons were between high and LS groups. Normality of the distribution was assessed with the Shapiro–Wilk’s test. Biochemical data were analysed using StatView 4.5 software (Abacus Concepts, Cary, NC, USA). Group differences (independent variables Sociability and/or Sex) after significant repeated measures (Time) analysis of variances (ANOVAs) were examined by post hoc Fisher’s Protected Least Significance Difference test. Baseline release was calculated based on the last six fractions collected before the administration of PCA of each animal; their average value was compared at each time point after drug treatment. The correlations shown are Pearson correlation coefficients. All other data are expressed as means ± SEM. Statistical significance was set at p < 0.05.
Results
Experiment I: Extracellular 5-HT in the prefrontal cortex and ventral tegmental area in male Wistar rats
We hypothesized that in these two brain regions baseline 5-HT levels would be lower in HS-rats, but higher after administration of PCA. In this experiment, the mean social activity time in the low and HS groups was 78.3 ± 4.8 s and 119.6 ± 3.2 s, respectively. Significant effects of Sociability (F(1,24) = 4.88, p < 0.05) and Time (F(14,336) = 29.9, p < 0.0001) on the levels of 5-HT in the prefrontal cortex emerged. LS-rats had higher extracellular levels of 5-HT in the prefrontal cortex than HS-rats (Figure 1). As expected, PCA significantly increased 5-HT levels 30 min after administration until the end of the microdialysis experiment.
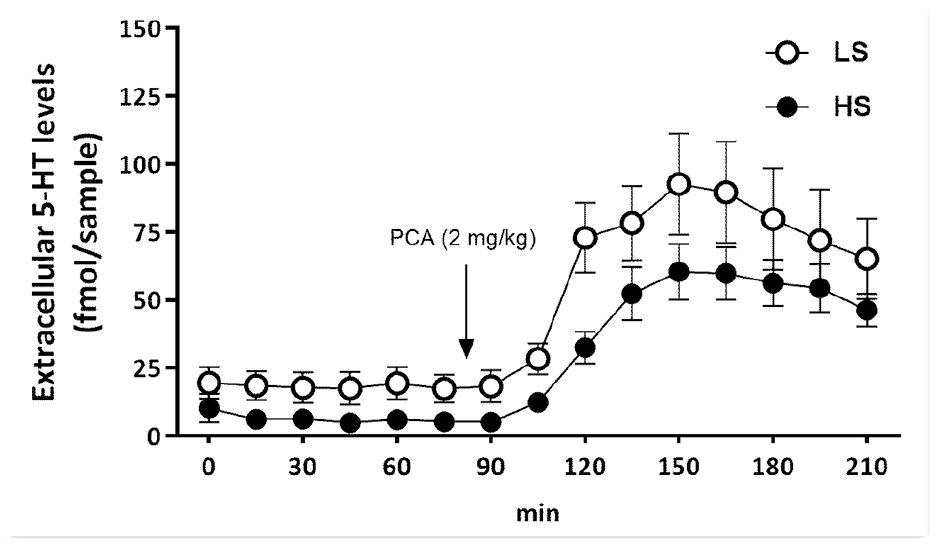
Extracellular 5-HT levels in prefrontal cortex, measured in freely moving conscious animals. PCA (2 mg/kg) was administered 75 min after beginning of probe collections.
In the ventral tegmental area, there was no main effect of Sociability, but an effect of Time emerged (F(14,366) = 36.6, p < 0.0001), as PCA increased 5-HT levels 30–45 min after administration. PCA increased 5-HT release, but an interaction between sociability and time emerged for this brain region (F(14,366) = 2.41, p < 0.05). PCA had a stronger effect on 5-HT release in the HS group during the early period after administration (Figure 2). There was no significant correlation of 5-HT release between prefrontal cortex and ventral tegmental area either at baseline or after PCA administration (data not shown).
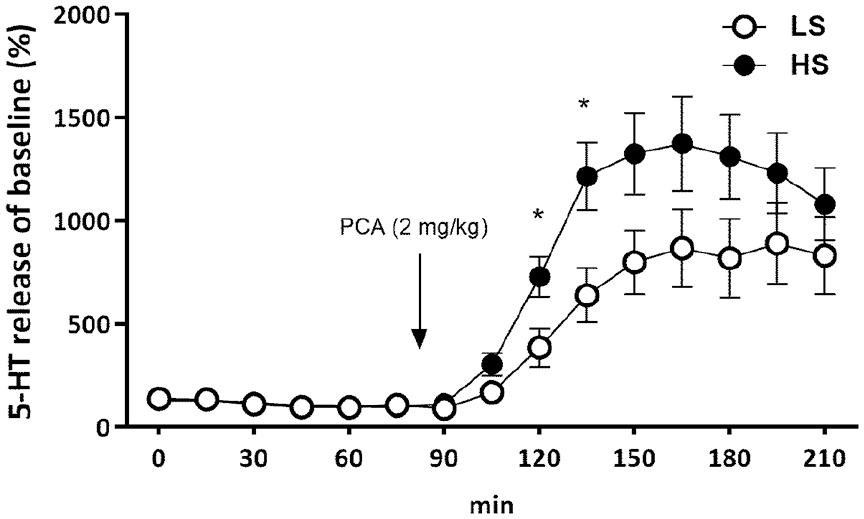
Serotonin release of baseline (%) in ventral tegmental area after administration of PCA (2 mg/kg).
Experiment II: Sociability and extracellular dopamine and 5-HT in the dorsomedial striatum in male Wistar rats
We expected to observe an increase in both 5-HT and dopamine levels after administration of PCA, and higher 5-HT but not dopamine release in HS-rats. In this experiment, time spent in social interaction was 73 ± 3 s for HS- and 40 ± 3 s for LS-rats. Baseline levels of dopamine did not differ between HS- and LS-phenotypes. PCA expectedly induced an increase in extracellular dopamine, but this was independent of phenotype (Figure 3(a)). Rats with higher levels of sociability tended to have higher baseline levels of 5-HT (3.89 ± 0.81 and 2.32 ± 0.34 fmol/sample for HS- and LS-rats, respectively; Sociability F(1,24) = 3.20; p = 0.09). PCA-induced serotonin release was more pronounced in rats with higher sociability levels (time F(23,577) = 20.2; p < 0.001; Time × Sociability F(1,577) = 2.54; p < 0.01; Figure 3(b)).
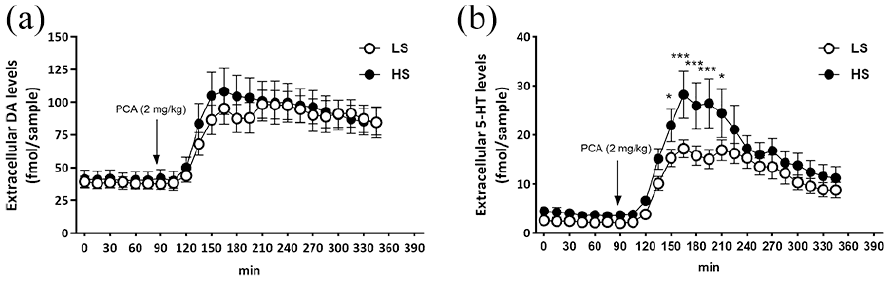
Sociability and extracellular dopamine and 5-HT in the dorsomedial striatum in male Wistar rats after administration of PCA (2 mg/kg). (a) Dopamine; (b) 5-HT.
Experiment III: Sociability in male and female Wistar rats
This experiment was conducted in a different animal house and aimed at the comparison of male and female rats. We hypothesized that, similarly to male rats, sociability as a predictable level of social activity in different tests would also be possible to establish in female animals. Mean social interaction time was 84 ± 5 s and 139 ± 5 s for male LS- and HS-rats, respectively, and 42 ± 4 s and 83 ± 6 s for female LS- and HS-rats, respectively. Similarly to previous studies in male Wistars, correlations of performance in single social interaction tests were variable, but the average social activity correlated similarly to behaviour in each test (Table 1).
Correlations between the SI tests in Wistar rats.

SI: social interaction.
p = 0.072. *p < 0.05. **p < 0.01. ***p < 0.001.
Experiment IV: Sociability and extracellular dopamine and 5-HT levels in male and female Sprague–Dawley rats
The hypothesis was that sociability is possible to assess in Sprague–Dawley rats as well as in Wistars, and that PCA-induced increase in 5-HT but not dopamine would be higher in HS-rats. Mean social interaction time was 112 ± 8 s and 192 ± 10 s for male LS- and HS-rats, respectively, and 108 ± 12 s and 216 ± 13 s for female LS- and HS-rats, respectively. Again, as in Wistar rats, correlations of social activity in single tests were variable, but the average social activity correlated similarly to behaviour in each test. A deviation from that pattern was the behaviour of male Sprague–Dawley rats in their first social interaction test (Table 2).
Correlations between the SI tests in Sprague–Dawley rats.
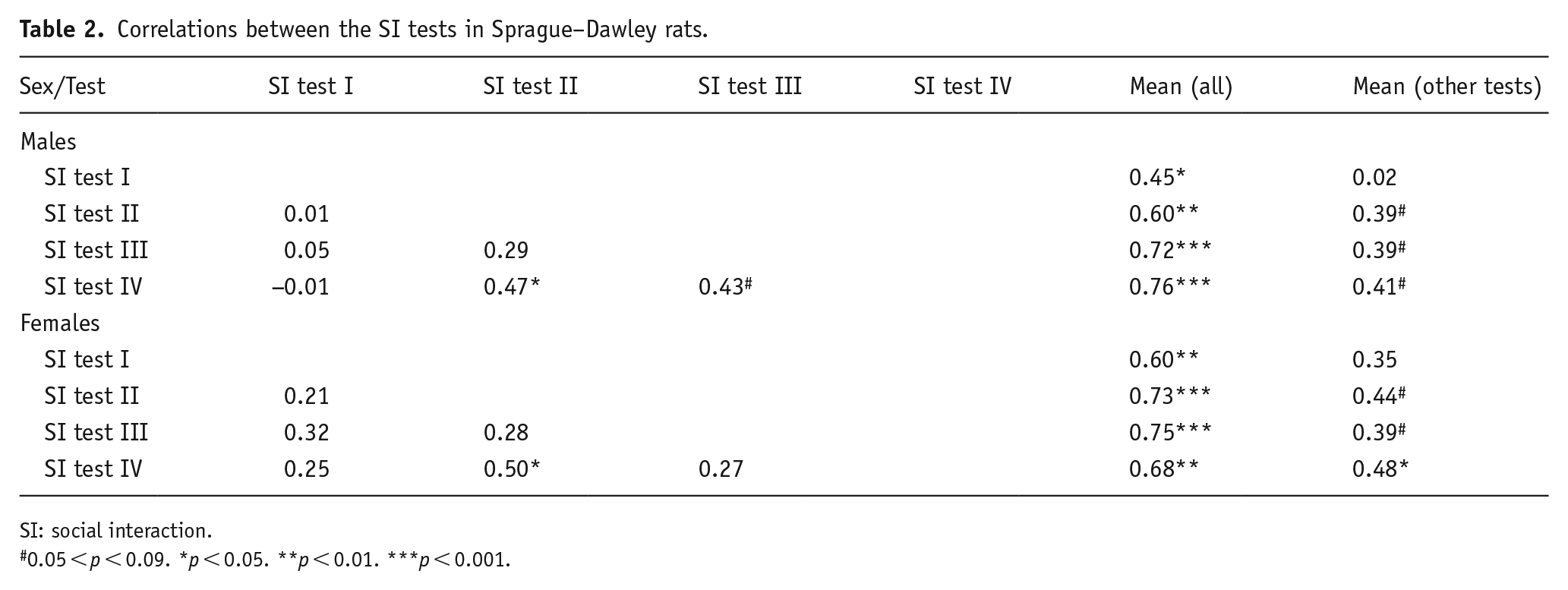
SI: social interaction.
0.05 < p < 0.09. *p < 0.05. **p < 0.01. ***p < 0.001.
Baseline extracellular levels of dopamine in the PFC were not related to Sociability but were higher in female rats (18.4 ± 3.2 fmol/sample vs 10.7 ± 0.8 fmol/sample, respectively; Sex F(1,35) = 5.57; p < 0.05). In response to PCA (2 mg/kg), dopamine levels increased comparably in all groups, while return to baseline levels was somewhat hindered in female rats, especially amongst the highly social animals, as compared to males (Sex F(1,35) = 7.36; p < 0.05); Time F(25,875) = 11.8; p < 0.001; Figure 4). Baseline extracellular levels of serotonin in the prefrontal cortex in male rats had a statistically non-significant tendency of being higher than in females (21.5 ± 3.3 fmol/sample vs 12.8 ± 3.3 fmol/sample, respectively; main effect of Sex F(1,33) = 3.51; p = 0.07) and neither was there any statistically significant effect of Sociability, while in females there was a twofold difference between HS- and LS-rats (16.6 ± 5.9 fmol/sample vs 8.6 ± 3.2 fmol/sample, respectively). Administration of PCA (2 mg/kg) elicited a marked increase in extracellular 5-HT levels (Figure 5). The proportional increase from baseline levels was related to Sociability and sex (Sociability × Sex × Time F(25,825) = 2.32; p < 0.001). Across the experiment, female rats had an overall higher release of 5-HT than males (main effect of Sex F(1,33) = 8.19; p < 0.01) but this was particularly clear after administration of PCA (Sex × Time F(25,825) = 7.04; p < 0.001). In turn, the 5-HT release elicited by PCA was by far the highest in female HS-rats (Figure 5). 5-HT release elicited by PCA in male rats was smaller than in females and it was overall lower in LS-rats (Sociability × Time F(25,825) = 3.36; p < 0.001) but group-wise, LS females and high and LS males did not differ from each other significantly (Figure 5).
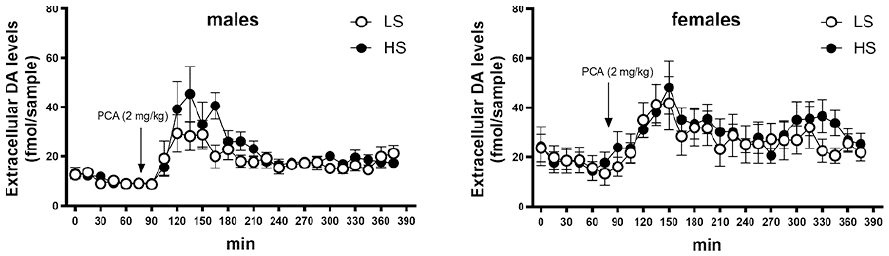
Extracellular dopamine levels in prefrontal cortex in male and female Sprague–Dawley rats before and after administration of PCA (2 mg/kg).
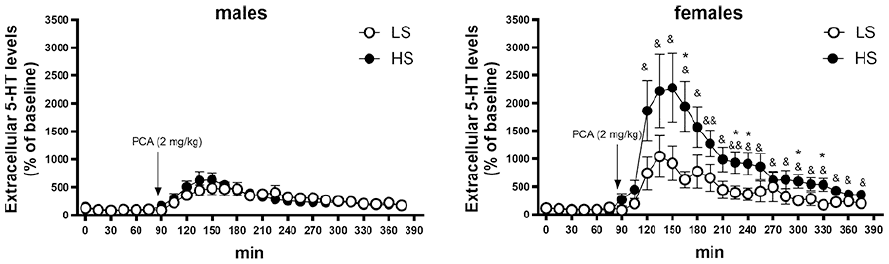
Serotonin release of baseline (%) in prefrontal cortex in male and female Sprague–Dawley rats after administration of PCA (2 mg/kg).
Discussion
Results of the present study in rat support the notion that social behaviour has a trait-like basis, this being in a positive association with serotonin release capacity. Of particular interest is the finding of a high serotonin release potential in the prefrontal cortex of female rats that are highly sociable.
The social interaction test that was exploited in the assessment of sociability was originally developed and validated for the detection of anxiolytic and anxiogenic drug effects by File et al. (File and Hyde, 1978; File et al., 1993, 1996; Kenny et al., 2000). While the test was found to classify drugs with effect on anxiety properly, it was also noticed that the drug-free performance of rats in the social interaction test did not correlate well with their behaviour in other types of anxiety tests (Ramos et al., 1997). These facts made some of us hypothesize that rat behaviour in the social interaction test is shaped, besides general anxiety, by the individual level of sociability trait (Tõnissaar et al., 2004), one of the most fundamental temperamental dimensions (Depue and Collins, 1999). In several independent experiments on male Wistar rats, it was found that a moderate positive correlation existed between the activities of partners in a test, that the social activity of individual animals in different tests was not stable, but a much more consistent correlation was present for activities in every single test with the average performance in all tests (Kanarik et al., 2018, unpublished; Tõnissaar et al., 2004, 2008). Similar findings emerged for the Sprague–Dawley strain and female rats in the present study. Behaviour in the very first social interaction test was different in Sprague–Dawley male rats; this resembles male Sprague–Dawley rat behaviour in a study of trait-wise exploration of novel physical environment where the performance of both Wistar and Sprague–Dawley rats in the first test was less correlated with all subsequent tests, whereas this deviation was much larger in Sprague–Dawleys (Mällo et al., 2007). It should be noted that at the first testing of social behaviour the arena has been unfamiliar to animals in all our studies, something that has appeared to be of minor importance for experiments with Wistar rats but is necessary to consider for other strains.
Higher sociability in male Wistar rats was associated with lower tissue levels of 5-HIAA, the main metabolite of 5-HT, in frontal cortex, indirectly suggestive of lower 5-HT release/overflow in HS- rats (Tõnissaar et al., 2004). For the interpretation, it may be important to point out that throughout the sociability studies, animals remain single-housed, and in this experiment rats were sacrificed several days after any social contact. In contrast, no association between frontal cortical 5-HIAA levels and sociability was found in a second experiment where animals had been sacrificed immediately after a social interaction test (Tõnissaar et al., 2004). As lower 5-HIAA suggests lower 5-HT release, it was hypothesized that sociability may be related to lower basal 5-HT release in some brain regions but this becomes masked by neurochemical alterations during social stimulation. In the present study, male Wistar HS-rats had at baseline indeed lower extracellular 5-HT levels in the prefrontal cortex. Tonically elevated serotonergic function in the rat prefrontal cortex has been found to relate to deficits in impulse control (Dalley et al., 2002). The prefrontal cortex provides the highly conserved sub-cortical circuits, from where social behaviours emerge, with dynamic, context-dependent modulation that integrates current conditions, past experiences and goals (Yizhar and Levy, 2021). Both in rats and primates, 5-HT neuronal activity is implicated in anxious, impulsive, defensive, and aggressive behaviours, which reduce social interactions and social affiliations (Evans et al., 2006; Krakowski, 2003; Niederkofler et al., 2016).
Examination of 5-HT release potential by the PCA treatment revealed a comparable prefrontal increase in the extracellular 5-HT levels in HS- and LS-rats; in fact, the proportional increase of extracellular 5-HT from baseline levels tended to be higher in HS-rats, but this difference was not statistically significant. PCA, similarly to the widely used MDMA, causes massive depolarization-independent release of 5-HT and, to a smaller extent, dopamine. MDMA is abused for its prosocial effects and acutely administered PCA, similarly to MDMA, reduces aggressive tendencies (Ögren et al., 1980). In drug discrimination tests, PCA fully substitutes for MDMA (Baker et al., 1995), but not for amphetamine (Marona-Levicka et al., 1995). Worldwide, PCA generally is not a controlled substance, because owing to its much higher acute neurotoxicity it is not commonly abused; nevertheless, trace amounts of PCA have been detected in illicit street drug samples in China where it has become a controlled substance (Chen et al., 2016). We have selected the dose (2 mg/kg) in dose-response experiments (Häidkind et al., 2004) with the aim of having a robust and reliable, yet sub-maximal effect. That the 2 mg/kg dose of PCA is suitable for in vivo microdialysis experiments was previously established in another experiment (Mällo et al., 2008). The fact that highly social animals have lower baseline 5-HT overflow and high release capacity in prefrontal cortex may suggest a common mechanism, for example, more efficient transport across cell and/or storage vesicle membranes. It might also be that low basal 5-HT overflow in highly social animals is the result of single housing that is more stressful for them. Indeed single-housed highly social rats were more susceptible to chronic mild stress (Tõnissaar et al., 2008). Whatever the mechanism behind it, given that PCA shares the mechanism of action with MDMA, and that in animal experiments it fully substitutes for the latter in the drug discrimination task, its effects may be indicative of the neurochemistry of prosocial action.
In the first experiment, we inserted another microdialysis probe into the ventral tegmental area (VTA). VTA is a part of the circuitry supporting social behaviours (Sato et al., 2023); in VTA, serotonin turnover is affected by social versus isolation housing (Broadfoot et al., 2023) and is correlated with expression of appetitive ultrasonic vocalizations during social interaction (Hamed et al., 2015). We had a hypothesis that the low 5-HIAA frontal cortical tissue content in HS-rats (Tõnissaar et al., 2004) could be related to the serotonergic activity in VTA: It has been described that the activation of 5-HT2C receptors by acutely increased 5-HT levels causes a significant decrease in the basal firing rate of VTA neurons and reduce mesocorticolimbic dopaminergic neurotransmission (Di Matteo et al., 2002). As dopamine release may divert activity from social to environmental stimuli (Panksepp, 1998) it could be speculated that, while activation of 5-HT2C receptors in most of the brain areas may cause an anxiogenic effect in the social interaction test (Kantor et al., 2006; Merali et al., 2006), the higher 5-HT release potential in VTA suppresses dopamine release during social interaction through activation of 5-HT2C receptors, and promotes social activity more effectively in HS-rats. Indeed, the huge variability of the results in literature on the associations between 5-HT-ergic function and social behaviour in different tests (Ando et al., 2006; Bagdy et al., 2001; Duxon et al., 2000; File et al., 1996; Hamon et al., 1999; Harro, 2010; Kennedy et al., 1993; Lightowler et al., 1994) is likely resulting from actions through different receptor subtypes in distinct brain regions that form the sociability network or impact on its activity. It was observed that 5-HT release potential in VTA, as revealed by the effect of PCA on extracellular 5-HT levels, was higher in HS-rats. This led to the question of whether the 5-HT release potential could be higher in HS-rats in other brain regions. Dorsomedial striatum was selected for the next microdialysis experiment owing to the significance of the mesotelencephalic dopamine projections for promoting active behaviours and because this was the brain region found in cytochrome oxidase histochemical mapping to have a linear correlation between the activity of oxidative metabolism and sociability, whereas LS-rats had the highest neural activity (Kanarik and Harro, 2018). Although the release of dopamine is pivotal in psychomotor stimulation, social behaviour can rather be reduced by preferentially dopamine-releasing drugs, such as amphetamine (Panksepp, 1998). Baseline and PCA-induced dopamine release were not different between HS- and LS-rats in the striatum, but PCA elicited a higher release of 5-HT in HS-rats similar to what was observed in VTA. Thus, sociability was selectively associated with 5-HT, not with dopamine release.
Social activity of male Wistar rats in the presented experiments, while variable, was in the same range as in previous studies (Kanarik and Harro, 2018, unpublished; Tõnissaar et al., 2004, 2008), as was the correlation of social activity between partners. Female Wistar rats in the present study had lower social activity than male Wistar rats, but this was a single experiment not designed for the purpose of sex comparison. However, higher social anxiety has previously been reported for female rats of other strains (Johnston and File, 1991; Stack et al., 2010). This is in contrast with the higher exploration of the physical environment by females (see Liiver et al., 2023 and references within). In Sprague–Dawley rats, no such difference was observed in the present study: Both male and female Sprague–Dawley rats had higher levels of social activity in the social interaction test than we have ever observed with Wistars, and the activity was similar in males and females. While one study suggestive of higher social anxiety in female rats was conducted in the Sprague–Dawley strain (Stack et al., 2010), it should be noted that this study rather assessed the behaviour of a resident rat towards an intruder.
In Sprague–Dawley rats, administration of PCA also expectedly led to an increase of extracellular levels of dopamine and 5-HT in the prefrontal cortex. No difference in dopamine levels, either at baseline or after PCA treatment, was observed between HS- and LS-rats. Male HS- and LS-rats did not differ significantly either in 5-HT levels. Female rats had proportionally much higher 5-HT release compared to males after administration of PCA, and this was largely owing to the very strong response in female HS-rats that had lower baseline levels of 5-HT. As was the case in the experiment on male Wistar rats and in dorsamedial striatum, sociability was associated with PCA-induced 5-HT but not dopamine release.
We have previously found that HS- and LS-rats, at least in the case of the frontal cortex in Wistars, do not differ in the tissue levels of 5-HT and 5-HT transporters (Tõnissaar et al., 2008). Thus, the baseline differences in extracellular 5-HT between LS- and HS-rats should not be owing to a higher uptake of 5-HT in HS-rats, which has been demonstrated in rats with low exploratory activity and higher anxiety (Mällo et al., 2008). LS-rats, which had higher extracellular 5-HT levels, may have more 5-HT in the releasable pool, which may be result of higher 5-HT synthesis (Hasegawa et al., 2006) because the releasable pool is preferentially replenished with newly synthesized 5-HT (Kleven et al., 1983).
Interestingly, PCA at a similar dose used in the present study was reported not to increase hippocampal 5-HT release in isolation-reared rats, in contrast to its effect in rats reared as usual (Muchimapura et al., 2002). While we did not study the hippocampus, the effect of PCA was robust in all brain regions that we examined. Our animals were reared group-housed and one could thus speculate that the social deficits observed in isolation-reared animals (Fone and Porkess, 2008; Powell and Swerdlow, 2023) are based on neurochemical mechanisms that also control the capacity of 5-HT release.
Overall, in vivo microdialysis experiments revealed either lower baseline extracellular 5-HT levels, higher 5-HT release capacity by PCA, or both. Particularly prominent was the effect of PCA in the prefrontal cortex of highly sociable female rats. In the case of rats, several studies have suggested higher 5-HT synthesis rate in female rats (Carlsson and Carlsson, 1988a, 1988b; Haleem et al., 1990). In humans, a variety of differences in the expression of molecular markers of the 5-HT system have been reported between sexes, including higher 5-HT transporter density in females, with the possible consequence of higher extracellular 5-HT levels (Cosgrove et al., 2009), but in contrast to rat studies, 5-HT synthesis has been assessed as higher in male subjects (Nishizawa et al., 1997). On the other hand, 5-HIAA levels in CSF, known to correlate inversely with impulsivity, are higher in females (Markianos et al., 2010), suggestive of higher release. It may be noteworthy that 5-HT synthesis has also been reported as more readily suppressed by 5-HT1A receptor stimulation in females (Haleem et al., 1990). Detailed comparative studies of individual differences in the regulation of serotonergic neurotransmission would be desirable, in order to ascertain whether the differences observed relate to sex or the sociability trait, or both.
Limitations of the presented experiments include use of variable methods of anaesthesia in older and recent experiments at microdialysis probe implantation, but even if these had any effect on some aspects of results, the main conclusions would remain untouched. The social interaction test sums up all social activity so we can not specify whether the differences in extracellular 5-HT levels correspond to some aspects of social behaviour more than to others. Neither have we assessed whether sociability measured during the dark phase of the diurnal cycle would have produced identical results, but have no reason to expect otherwise. We did not assess estrous cycle in female rats, given that sociability was calculated from social interaction time in different tests with probably different stages of the cycle. Previously we have reported that in our conditions estrous cycle was unrelated to the expression of exploratory behaviour (Liiver et al., 2023), but this does not exclude its relationship with social behaviour. Further studies should address the possible effect of estrous cycle on the expression of the sociability trait.
Conclusively, rats with HS have lower baseline extracellular 5-HT levels in the prefrontal cortex, but higher 5-HT release capacity in several brain regions from brainstem to cortex. The role of the specific aspects of 5-HT release in social behaviour may, however, vary between males and females.
Supplemental Material
sj-docx-1-jop-10.1177_02698811241283710 – Supplemental material for Serotonin release by parachloroamphetamine in rats with high and low sociability: High prefrontal release capacity in sociable females
Supplemental material, sj-docx-1-jop-10.1177_02698811241283710 for Serotonin release by parachloroamphetamine in rats with high and low sociability: High prefrontal release capacity in sociable females by Marianna Norden, Margus Kanarik, Karita Laugus, Aet O’Leary, Kristi Liiver, Margus Tõnissaar, Ruth Shimmo and Jaanus Harro in Journal of Psychopharmacology
Supplemental Material
sj-tif-2-jop-10.1177_02698811241283710 – Supplemental material for Serotonin release by parachloroamphetamine in rats with high and low sociability: High prefrontal release capacity in sociable females
Supplemental material, sj-tif-2-jop-10.1177_02698811241283710 for Serotonin release by parachloroamphetamine in rats with high and low sociability: High prefrontal release capacity in sociable females by Marianna Norden, Margus Kanarik, Karita Laugus, Aet O’Leary, Kristi Liiver, Margus Tõnissaar, Ruth Shimmo and Jaanus Harro in Journal of Psychopharmacology
Supplemental Material
sj-tif-3-jop-10.1177_02698811241283710 – Supplemental material for Serotonin release by parachloroamphetamine in rats with high and low sociability: High prefrontal release capacity in sociable females
Supplemental material, sj-tif-3-jop-10.1177_02698811241283710 for Serotonin release by parachloroamphetamine in rats with high and low sociability: High prefrontal release capacity in sociable females by Marianna Norden, Margus Kanarik, Karita Laugus, Aet O’Leary, Kristi Liiver, Margus Tõnissaar, Ruth Shimmo and Jaanus Harro in Journal of Psychopharmacology
Footnotes
Acknowledgements
Assistance of Dr Marika Eller, Ms Anni Simon and Ms Kelly Haapsal in conducting some of the experiments is gratefully acknowledged.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the Estonian Research Council (PRG1213) to JH and the Tallinn University ASTRA project TU TEE financed by the European Union European Regional Development Fund (2014-202040116-0033).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
