Abstract
Background:
Expert consensus-based clinically equivalent dose estimates and dosing recommendations can provide valuable support for the use of drugs for psychosis in clinical practice and research.
Aims:
This second International Consensus Study of Antipsychotic Dosing provides dosing equivalencies and recommendations for newer drugs for psychosis and previously reported drugs with low consensus.
Methods:
We used a two-step Delphi survey process to establish and update consensus with a broad, international sample of clinical and research experts regarding 26 drug formulations to obtain dosing recommendations (start, target range, and maximum) and estimates of clinically equivalent doses for the treatment of schizophrenia. Reference agents for equivalent dose estimates were oral olanzapine 20 mg/day for 15 oral and 7 long-acting injectable (LAI) agents and intramuscular haloperidol 5 mg for 4 short-acting injectable (SAI) agents. We also provide a contemporary list of equivalency estimates and dosing recommendations for a total of 44 oral, 16 LAI, and 14 SAI drugs for psychosis.
Results:
Survey participants (N = 72) from 24 countries provided equivalency estimates and dosing recommendations for oral, LAI, and SAI formulations. Consensus improved from survey stages I to II. The final consensus was highest for LAI formulations, intermediate for oral agents, and lowest for SAI formulations of drugs for psychosis.
Conclusions:
As randomized, controlled, fixed, multiple-dose trials to optimize the dosing of drugs for psychosis remain rare, expert consensus remains a useful alternative for estimating clinical dosing equivalents. The present findings can support clinical practice, guideline development, and research design and interpretation involving drugs for psychosis.
Introduction
Since the introduction of chlorpromazine and haloperidol seven decades ago, the number of drugs for psychosis has steadily increased with new drug entities and formulations resulting in dozens of treatment options for schizophrenia and other major psychiatric disorders (Rubio and Kane, 2022; Taylor et al., 2021). Competent use of drugs for psychosis requires clinical experience and knowledge of sound dosing practices. Such practice routinely includes dose increases to achieve symptomatic and functional improvement, gradual dose reduction to find tolerable and effective maintenance doses, and switching among agents and formulations as occurs in 15%–20% of patients treated with drugs for psychosis each year (Correll et al., 2021; Khandker et al., 2022; Nyhuis et al., 2010; Taylor et al., 2021). Remarkably, however, consensus remains elusive on how to dose individual drugs for psychosis, including changing from one to an approximately equivalent dose of another, especially with newer agents. Establishing clinically equivalent dose estimates and dosing recommendations contributes to optimizing the use of drugs for psychosis in clinical practice, informing practice guideline development, and designing and interpreting clinical research (Højlund et al., 2021; Leucht et al., 2020, 2021; Ostuzzi et al., 2022).
Methods of estimating dosing equivalencies and establishing dosing recommendations have limitations and can yield values that vary substantially (Leucht et al., 2015, 2016; Patel et al., 2013). Equivalent doses for many older drugs for psychosis, such as chlorpromazine and haloperidol, were supported by dose–effect relationships based on laboratory pharmacodynamic data, such as in vitro affinity at cerebral dopamine receptors. This preclinical approach does not account for important inter-species and individual differences in the disposition and pharmacokinetics of drugs for psychosis. Moreover, this approach assumes that laboratory pharmacodynamic dose–response relationships are directly related to clinical dose effects. Manufacturers may tend toward conservative dosing using small doses necessary to distinguish their product from placebo while limiting the risk of adverse effects. Attempts to estimate dosing equivalencies based on flexible dosing trials have been criticized for overestimating effective doses (Davis and Chen, 2004). Estimates based on trials involving multiple fixed-dose arms are informative but rare and often lack head-to-head comparisons with products of different manufacturers, thereby resulting in major gaps in data and equivalency estimates with wide confidence intervals (Leucht et al., 2020).
An alternative method for estimating drug dosing is to establish consensus among clinical research experts and experienced clinicians by employing survey techniques such as the Delphi method of consensus building. This method is particularly useful when unanimity of opinion does not exist owing to a lack of scientific evidence and varied clinical experiences (Jones and Hunter, 1995). In our 2010 International Consensus Study of Antipsychotic Dosing (ICSAD-1), we applied a two-stage Delphi method to survey experts on the use of drugs for psychosis to estimate consensus-based clinically equivalent doses and dosing recommendations (Gardner et al., 2010). That study involved 43 participants from 18 countries and provided recommendations for 61 drugs for psychosis, including equivalence estimates and initial, target range, and maximum doses of orally administered and injected agents then available, as well as dose-adjustment recommendations for specific clinical circumstances. Consensus on equivalency and dosing estimates was high among many, but not all drugs for psychosis. Since then, new drugs for psychosis and formulations have received regulatory approval from the U.S. Food and Drug Administration (FDA) and other regulators and entered clinical practice globally. Moreover, randomized controlled trials that directly compare a range of fixed doses of drugs for psychosis remain rare, precluding the availability of such data to develop dosing comparisons in an ideally objective and quantitative manner. In the present study, we again used a two-stage Delphi survey method for a second International Consensus Study of Antipsychotic Dosing (ICSAD-2) to address equivalency and dosing of newer drugs for psychosis, as well as older agents still lacking secure consensus about dosing.
Methods
Participants
We identified experts in clinical research and clinical use of drugs for psychosis from three sources: the ICSAD-1 participants database, searches of bibliographic databases, and recommendations from study participants. All previous participants from ICSAD-1 were invited to participate. To increase the number of participants and geographic representation, we conducted a literature search for prominent investigators of drugs for psychosis using the Scopus® database. Combinations of the following search terms were used: antipsychotic, schizophrenia, psychosis, randomized clinical trial, clinical trial, trial, and study (see details in the Supplemental Material). We reviewed titles and abstracts of lead authors and co-authors with five or more peer-reviewed publications involving clinical trials or secondary data analyses, including systematic reviews and meta-analyses relevant to drugs for psychosis. Using a snowball sampling approach, each participant was asked to recommend a colleague they considered to be an “expert in the clinical use of drugs for patients with schizophrenia and other psychotic disorders.” Individuals primarily affiliated with a pharmaceutical corporation were not eligible to participate.
Prospective participants were invited by e-mail. Nonresponders received up to three email invitations over 6 weeks. Participants provided electronically signed informed consent for both study inclusion and, separately, for public acknowledgment of having participated as per the approval of the Dalhousie University Ethics Review Board. Contact the authors for inquiries pertaining to data access.
Selection of drugs for psychosis
ICSAD-2 builds on the methods and results of the original consensus study, ICSAD-1 (Gardner et al., 2010). ICSAD-2 includes 26 drugs for psychosis: 15 orally administered agents, 7 long-acting injectables (LAIs), and 4 short-acting injectables (SAIs). Included in both ICSAD-1 and ICSAD-2 were 18 drugs for psychosis (eight oral agents: aripiprazole, haloperidol, olanzapine, paliperidone, quetiapine, risperidone, sertindole, and ziprasidone; four SAIs: haloperidol, triflupromazine, ziprasidone, and zuclopenthixol; and one LAI: risperidone microspheres). Drugs for psychosis were included if they received FDA regulatory approval in 2000 or later (as a proxy for global marketing), or if they failed to achieve dosing consensus in ICSAD-1. Some agents were included again in ICSAD-2 if a specific dosing discrepancy was identified, including (1) the 95% effective dose (ED95) based on dose–response analyses reported by Leucht et al. (2020) was less than half of the ICSAD-1 maximum dose (e.g., oral quetiapine); (2) the World Health Organization (WHO) daily defined dose (DDD) (WHO, 2023) was higher than the ICSAD-1 maximum dose (e.g., SAI triflupromazine HCl); or (3) the DDD was lower than the ICSAD-1 initial dose (e.g., SAI zuclopenthixol acetate). Oral risperidone, an agent with well-established and widespread international use, was included in ICSAD-2 as a control to assess the comparability of ICSAD-1 and ICSAD-2 survey processes and results. Reference agents for estimating clinically equivalent doses included olanzapine 20 mg/day for orally administered agents and LAIs and injectable haloperidol 5 mg for SAIs.
The survey’s design and development were based on ICSAD-1 (Gardner et al., 2010). It was divided into sections 1 and 2, each with three parts: (A) oral drugs, (B) LAIs, and (C) SAIs. REDCap® was used for survey entry, deployment, data collection, and data management (Harris et al., 2009, 2019). In each survey part, standardized vignettes provided clinical contexts for survey responses (see Supplemental Material for details). Vignettes were based on treating adult males with Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition (DSM-5) schizophrenia with ⩾2 years of exposure to drug treatment and not considered intolerant or treatment-refractory. Participants were instructed to give priority to efficacy over tolerability in their dose estimates.
In section 1, participants estimated doses that were clinically equivalent to the stated reference drugs for oral agents, LAIs, and SAIs. They rated their confidence in each estimate as follows: high (extensive experience, applies to most patients), medium (moderate experience, applies to most patients), or low (limited experience, frequent exceptions). In section 2, participants recommended initial (starting) doses, target dose ranges, and maximum doses for each drug. Participants also recommended dosing intervals for LAIs and were asked to indicate their practice regarding the use of a short-acting oral formulation to establish tolerability before initiating a course of LAIs.
We used a two-stage Delphi method to deploy the survey. All participants completing stage I were invited to complete the survey a second time. Stage II included an aggregated summary of responses for each question (including respondent count and dose range, median, mean, and standard deviation) as well as the participant’s individual responses to remind them of how they responded to each question during stage I. The contents of both survey stages were identical, except for adding oral haloperidol to stage II to address a specific concern raised by a participant about dosing recommendations for oral haloperidol from ICSAD-1. The time between the start of each stage was approximately 6 months.
Data analysis
Data reported are from participants who completed both stages of the survey. Analyses from ICSAD-1 had shown that participant factors (sex, age, years of experience, relevant publications/person, country, major diagnostic type of patients treated, and main career interest [research/clinical]) did not have significant associations with dosing recommendations (Gardner et al., 2010) and were not repeated for the present study.
We initially summarized estimates of clinical dosing equivalents and recommended dosing (initial, target range, and maximum) using means, standard deviations, medians, and interquartile ranges (IQRs). Correlations between median and mean dosage equivalency estimates were high for oral drugs (r = 0.999), SAI agents (r = 0.998), and LAI preparations (r = 0.992). We report only median and IQR per equivalency and dosing recommendation for practicality and to limit the impact of outliers. When possible, we contacted respondents to request adjustments to apparent data entry errors (e.g., “300-day” LAI dosing interval). Any remaining data quality issues were resolved by consensus at team meetings involving all authors. The coefficient of variation (CV = [SD/mean] × 100) was used to indicate the level of participant consensus per stage for each equivalency and dosing recommendation. Levels of consensus were rated as high (CV ⩽ 25%), moderate (CV 26%–33%), or low (CV > 33%). For oral agents, equivalency ratios were calculated relative to olanzapine as the reference agent. We added the equivalency ratio with chlorpromazine as a historical reference using data from ICSAD-1, which estimated 600 mg of chlorpromazine to be equivalent to 20 mg of olanzapine (Gardner et al., 2010).
We compared ICSAD-1 with ICSAD-2 dosing recommendations for drugs included in both surveys to identify changes in expert recommendations between the original and current surveys (Gardner et al., 2010). We also compared ICSAD dosing recommendations with existing empirically derived dosing estimates. The recommended upper bounds of the target dose ranges of 18 drugs for psychosis were compared to the near-maximum effective dose (ED95) estimates from meta-analyses of available fixed-dose studies (Leucht et al., 2020). The ED95 represents the estimated mean dose that produces 95% of the maximum observed reduction in symptoms estimated from randomized trials comparing multiple fixed doses of a drug. For the comparison, all upper-bound target dose range recommendations were taken from ICSAD-2 except amisulpride, for which we used data from ICSAD-1. Disparity in dosing was calculated as the absolute percentage difference between ED95 estimates and the recommended upper bound of the target range.
We report two sets of clinical equivalency estimates and dosing recommendations for the 1-month and 3-month formulations of paliperidone palmitate. The first set is based on the molecular weight (MW) of the active drug (paliperidone, MW 426.5) and the second used the MW of the ester (paliperidone palmitate, MW 664.89). This was done to reflect the two dosing regimens used internationally, one based on mg of active drug and the other based on mg of LAI paliperidone ester (Janssen Inc., 2020; Janssen Pharmaceuticals Inc., 2022a).
Statistical analyses employed commercial software (IBM SPSS Statistics for Mac, v. 28.0, Armonk, NY and Microsoft Excel, v. 16; Microsoft, Redmond, WA).
Results
Participants
This two-stage Delphi survey was deployed between June and December 2021 with 268 experts invited to participate. Stage I was completed by 88 (32.8%) participants and 72 (26.9%) completed both stages I and II. One participant who did not follow instructions in stage I was not invited to complete stage II. Participants represented a wide geographic distribution involving 24 countries or regions: Spain (n = 9), Japan (n = 8), the United States (n = 8), Italy (n = 6), Canada (n = 5), Germany (n = 5), Taiwan (n = 5), Austria (n = 3), Indonesia (n = 3), Brazil (n = 2), France (n = 2), Singapore (n = 2), Thailand (n = 2), Turkey (n = 2), Chile (n = 1), Czech Republic (n = 1), Hungary (n = 1), Ireland (n = 1), South Korea (n = 1), Portugal (n = 1), Slovakia (n = 1), South Africa (n = 1), Switzerland (n = 1), and UK (n = 1). The number of participants and countries represented increased by 67% (72 vs. 41) and 33% (24 vs. 18), respectively, between ICSAD-1 and ICSAD-2.
Most of the 72 participants were men (92%) with an average age of 55 years (SD = 11) and 27 years (SD = 10) of experience treating patients with drugs for psychosis. Respondents had a median of 40 (IQR 82.5) publications related to drugs for psychosis. The primary role related to the use of drugs for psychosis was clinical research for 22 participants (30.6%) and patient care for 50 (69.4%). The main areas of clinical research and practice were schizophrenia and other psychotic disorders (73.6%), bipolar disorder (22.2%), and other disorders (4.2%). Of 72 participants, 9 (12.5%) were returning participants from ICSAD-1, 24 (33.3%) were recruited upon being identified by our literature search for clinical research experts, and 39 (54.2%) were invited based on participant referrals.
Delphi effect and consensus
Consensus among participants improved from stage 1 to stage 2 of the Delphi survey. For clinical equivalency estimates and dosing recommendations overall, the average CV decreased from 40.1% in stage I to 32.0% in stage II. Also, consensus (based on moderate or high levels of agreement (CV < 33%)) rose from 61% in stage I to 87% in stage II for clinical equivalency estimates. Per dosage formulation, consensus rates were 14/15 (93%) for oral agents, 0/4 (0%) for SAIs, and 7/7 (100%) for LAIs. Levels of consensus also rose across all dosing recommendations (initial, target range, and maximum), from an average of 45% in stage I to 57% in stage II (61%, 0%, and 84% for oral, SAI, and LAI preparations, respectively). Consensus for dosing interval recommendations for LAI products increased from 61% to 86%, with an associated overall decrease in CV from 40% in stage I to 20% in stage II (see Supplemental Tables 6S, 7S, and 8S for CV values for each drug).
Clinically equivalent dose estimates and dosing recommendations
Participants reported clinical dose equivalencies and dosing recommendations for 26 drugs for psychosis (see details in Table 1 for oral, Table 2 for SAI, and Tables 3 and 4 for LAI formulations). For oral agents, the overall average median initial dose in olanzapine equivalents was 6.3 mg/day (SD = 4.4); the overall target dose range (average median of lower and upper bound ranges) was 10.3 to 20.3 mg/day; and the overall average median maximum dose for oral agents in olanzapine equivalents was 22.7 mg/day (SD = 4.4).
Clinical dosing equivalencies and dosing recommendations for orally administered drugs for psychosis.
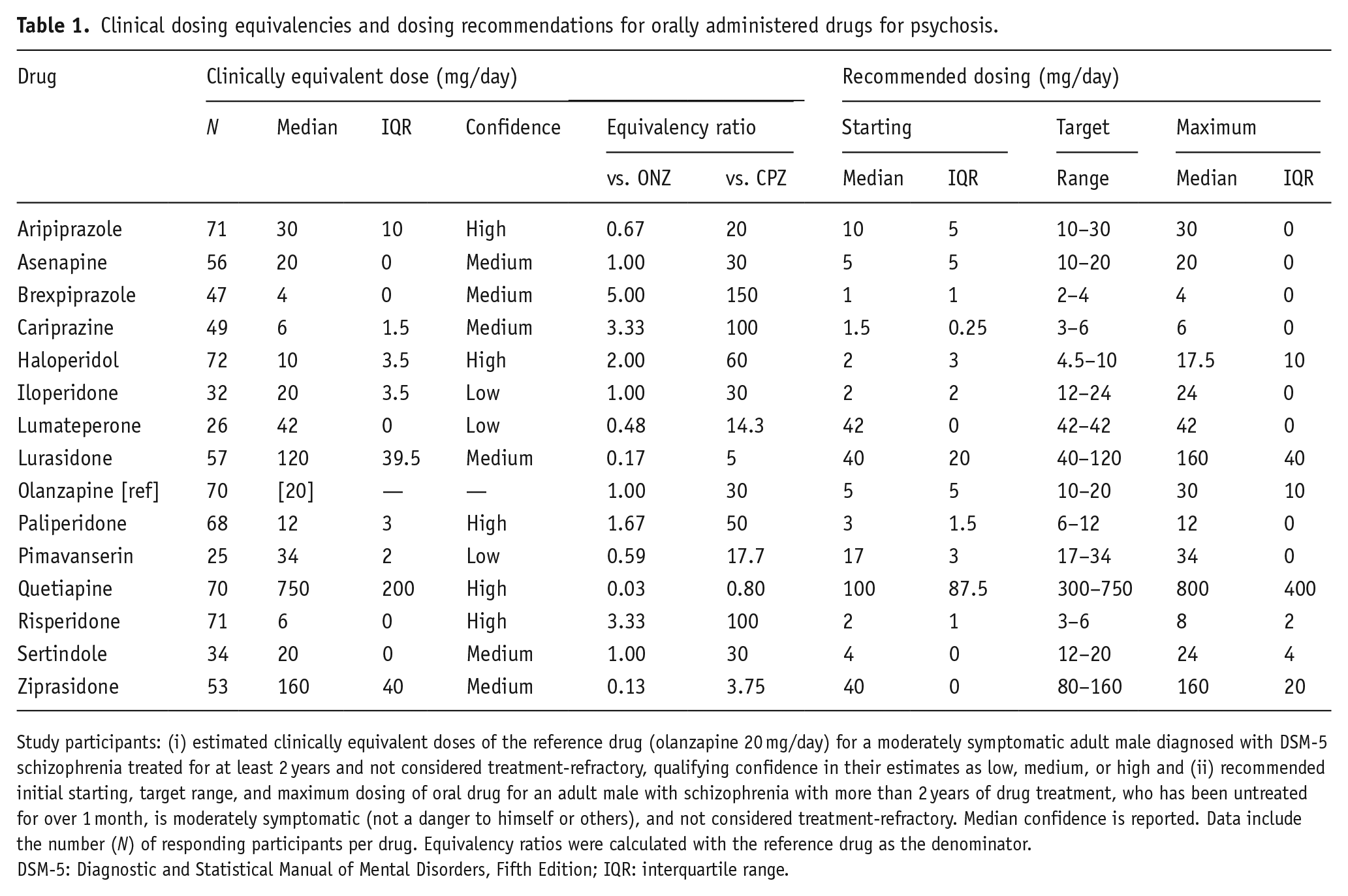
Study participants: (i) estimated clinically equivalent doses of the reference drug (olanzapine 20 mg/day) for a moderately symptomatic adult male diagnosed with DSM-5 schizophrenia treated for at least 2 years and not considered treatment-refractory, qualifying confidence in their estimates as low, medium, or high and (ii) recommended initial starting, target range, and maximum dosing of oral drug for an adult male with schizophrenia with more than 2 years of drug treatment, who has been untreated for over 1 month, is moderately symptomatic (not a danger to himself or others), and not considered treatment-refractory. Median confidence is reported. Data include the number (N) of responding participants per drug. Equivalency ratios were calculated with the reference drug as the denominator.
DSM-5: Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition; IQR: interquartile range.
Clinical dosing equivalencies and dosing recommendations for short-acting injected drugs for psychosis.
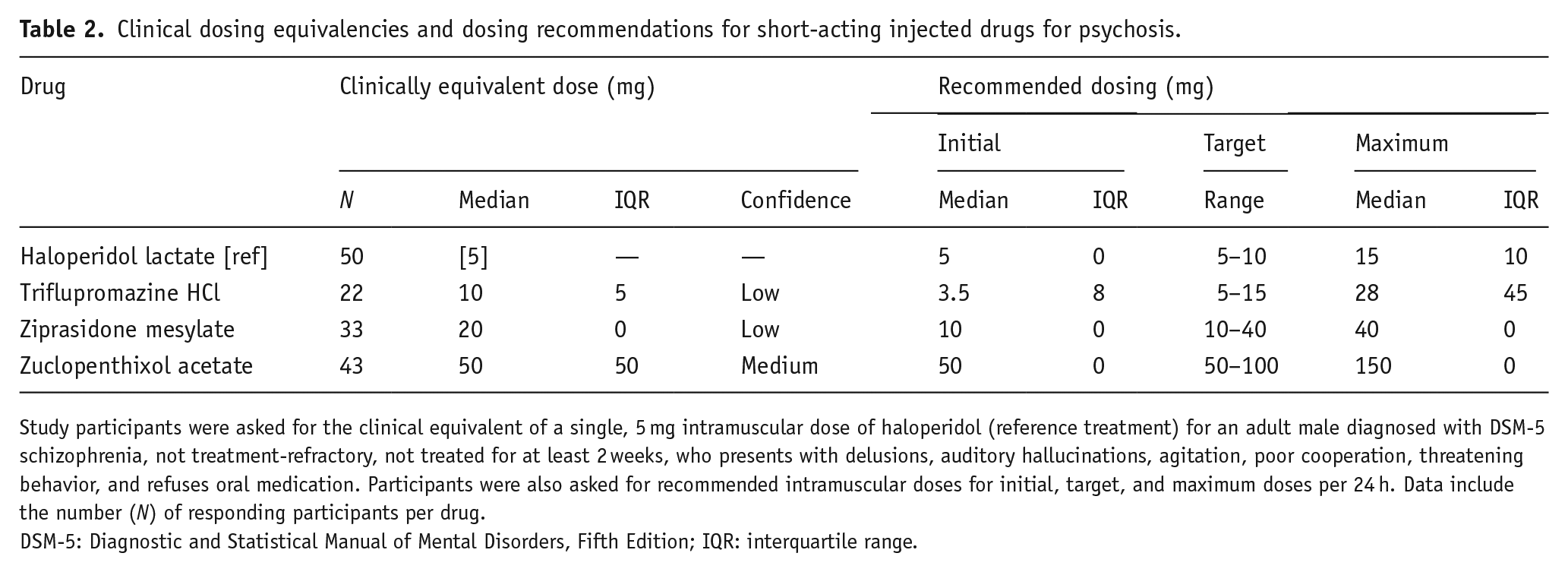
Study participants were asked for the clinical equivalent of a single, 5 mg intramuscular dose of haloperidol (reference treatment) for an adult male diagnosed with DSM-5 schizophrenia, not treatment-refractory, not treated for at least 2 weeks, who presents with delusions, auditory hallucinations, agitation, poor cooperation, threatening behavior, and refuses oral medication. Participants were also asked for recommended intramuscular doses for initial, target, and maximum doses per 24 h. Data include the number (N) of responding participants per drug.
DSM-5: Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition; IQR: interquartile range.
Clinical dosing equivalents for long-acting injected drugs for psychosis.
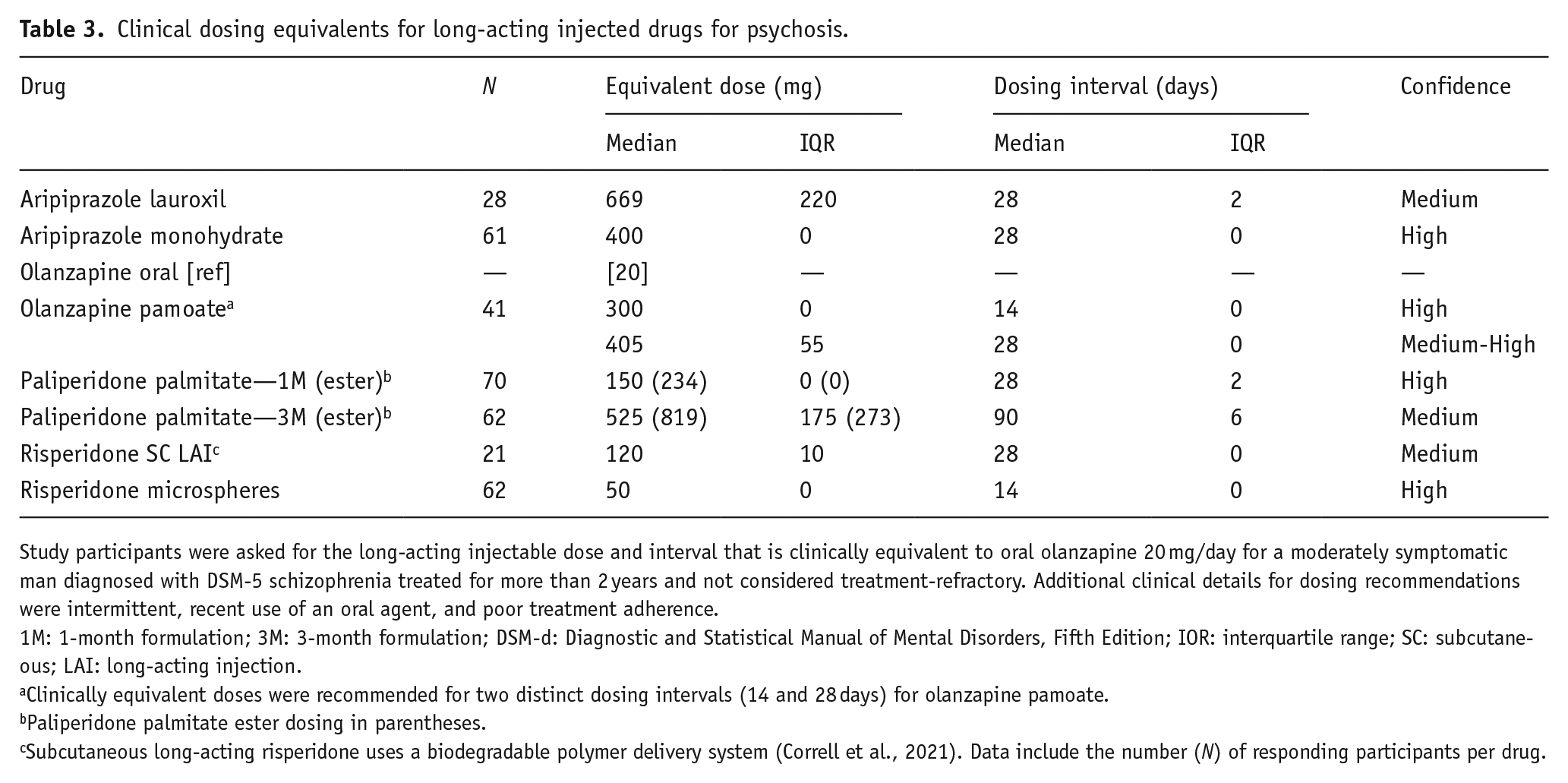
Study participants were asked for the long-acting injectable dose and interval that is clinically equivalent to oral olanzapine 20 mg/day for a moderately symptomatic man diagnosed with DSM-5 schizophrenia treated for more than 2 years and not considered treatment-refractory. Additional clinical details for dosing recommendations were intermittent, recent use of an oral agent, and poor treatment adherence.
1M: 1-month formulation; 3M: 3-month formulation; DSM-d: Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition; IOR: interquartile range; SC: subcutaneous; LAI: long-acting injection.
Clinically equivalent doses were recommended for two distinct dosing intervals (14 and 28 days) for olanzapine pamoate.
Paliperidone palmitate ester dosing in parentheses.
Subcutaneous long-acting risperidone uses a biodegradable polymer delivery system (Correll et al., 2021). Data include the number (N) of responding participants per drug.
Dosing recommendations for long-acting injected drugs for psychosis.
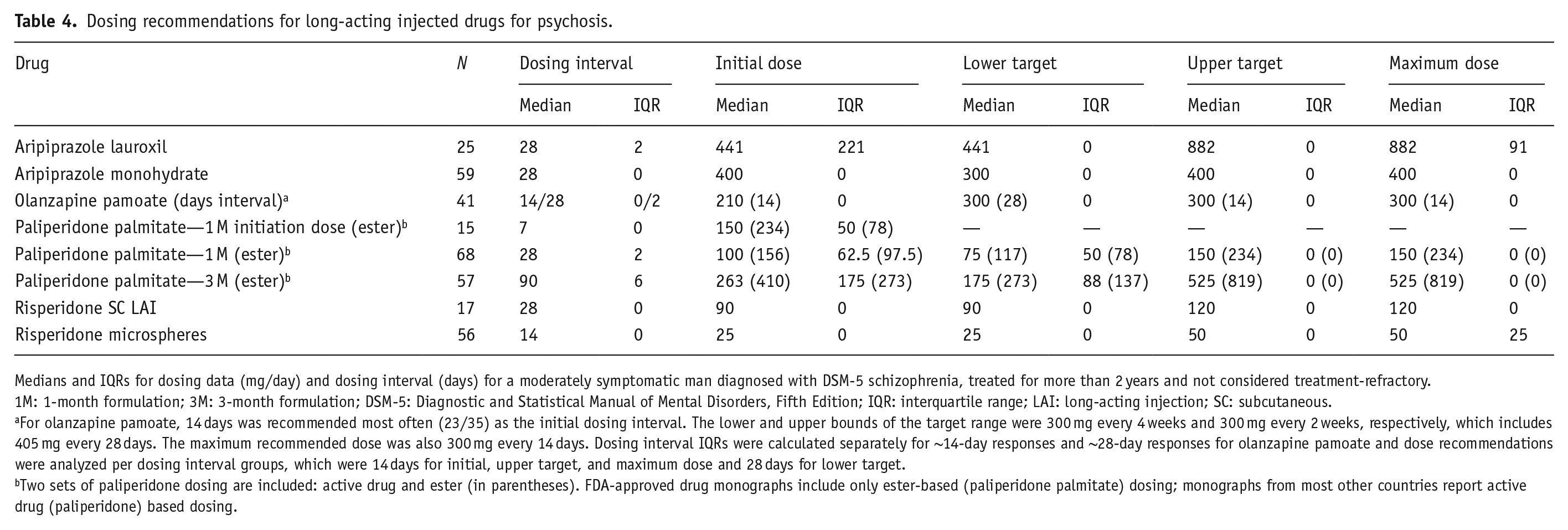
Medians and IQRs for dosing data (mg/day) and dosing interval (days) for a moderately symptomatic man diagnosed with DSM-5 schizophrenia, treated for more than 2 years and not considered treatment-refractory.
1M: 1-month formulation; 3M: 3-month formulation; DSM-5: Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition; IQR: interquartile range; LAI: long-acting injection; SC: subcutaneous.
For olanzapine pamoate, 14 days was recommended most often (23/35) as the initial dosing interval. The lower and upper bounds of the target range were 300 mg every 4 weeks and 300 mg every 2 weeks, respectively, which includes 405 mg every 28 days. The maximum recommended dose was also 300 mg every 14 days. Dosing interval IQRs were calculated separately for ~14-day responses and ~28-day responses for olanzapine pamoate and dose recommendations were analyzed per dosing interval groups, which were 14 days for initial, upper target, and maximum dose and 28 days for lower target.
Two sets of paliperidone dosing are included: active drug and ester (in parentheses). FDA-approved drug monographs include only ester-based (paliperidone palmitate) dosing; monographs from most other countries report active drug (paliperidone) based dosing.
Most study participants recommended establishing drug tolerability using an oral formulation, or the 1-month LAI formulation before using the 3-month LAI formulation of paliperidone palmitate, for each LAI included in ICSAD-2. Overall, 86% indicated that they always (71%) or usually (15%) test for tolerability before starting a treatment course with LAIs (see Supplemental Table 9S).
Dose changes between ICSAD-1 and ICSAD-2 surveys
The average number of participants responding to the 13 agents previously included in ICSAD-1 increased significantly between the two surveys (31.2 (SD = 12.7) vs. 54.8 (SD = 17.3), p = 0.001). There were changes to the equivalent dose for one oral agent (paliperidone), the initial dose for one oral agent (haloperidol), dosing ranges for six oral agents (all but olanzapine and sertindole), and the maximum dose for five oral agents (haloperidol, quetiapine, risperidone, sertindole, and ziprasidone). Overall, dosing trended modestly lower in ICSAD-2. Average changes to the oral initial dose, dosing range, and maximum dose were −4%, −16% to +3%, and −6%, respectively. Oral risperidone clinical equivalency and dosing recommendations were unchanged other than a small difference in dosing range (reduced from 4–6 mg/day to 3–6 mg/day) and maximum dose (reduced from 8.5 to 8.0 mg/day) indicating consistency between the two survey results. There were small changes to the dosing recommendations for three SAIs. For the fourth, triflupromazine HCl, there were several fold reductions in equivalency and dosing recommendations between surveys. For all SAIs, the level of consensus remained very low, with an average CV of 78% (see Supplemental Table 7S). There were no differences in clinical equivalency and dosing recommendations for risperidone microspheres between surveys. A complete set of updated clinical equivalencies and dosing recommendations for all drugs for psychosis included in ICSAD-1 and ICSAD-2 are provided in Supplemental Tables 10S–13S.
Comparison with near-maximum effective dose estimates
There was a 24.5% (SD 14.3%) and 25.5% (SD 16.7%) absolute dosing disparity for oral agents and LAIs, respectively, when comparing the upper bound of the target range with the estimated ED95 near-maximum effective dose (Table 5). The ED95 dose estimate was below the expert consensus-based upper bound dose recommendation for 8 of 14 oral agents and above for 6 of 14 agents, with an average dose disparity of −31.0% and +15.7%, respectively. The ED95 dose was lower than the upper bound of the target range for all four LAIs. The largest disparities were found for aripiprazole (oral −61.7%, lauroxil LAI −47.5%). Only five agents had a disparity of <20% (risperidone 5.2%, olanzapine pamoate 7.6%, sertindole 13.3%, ziprasidone 15.5%, and lurasidone 16.1%).
Comparability of fixed-dose modeling and consensus target dosing range for orally administered drugs for psychosis.
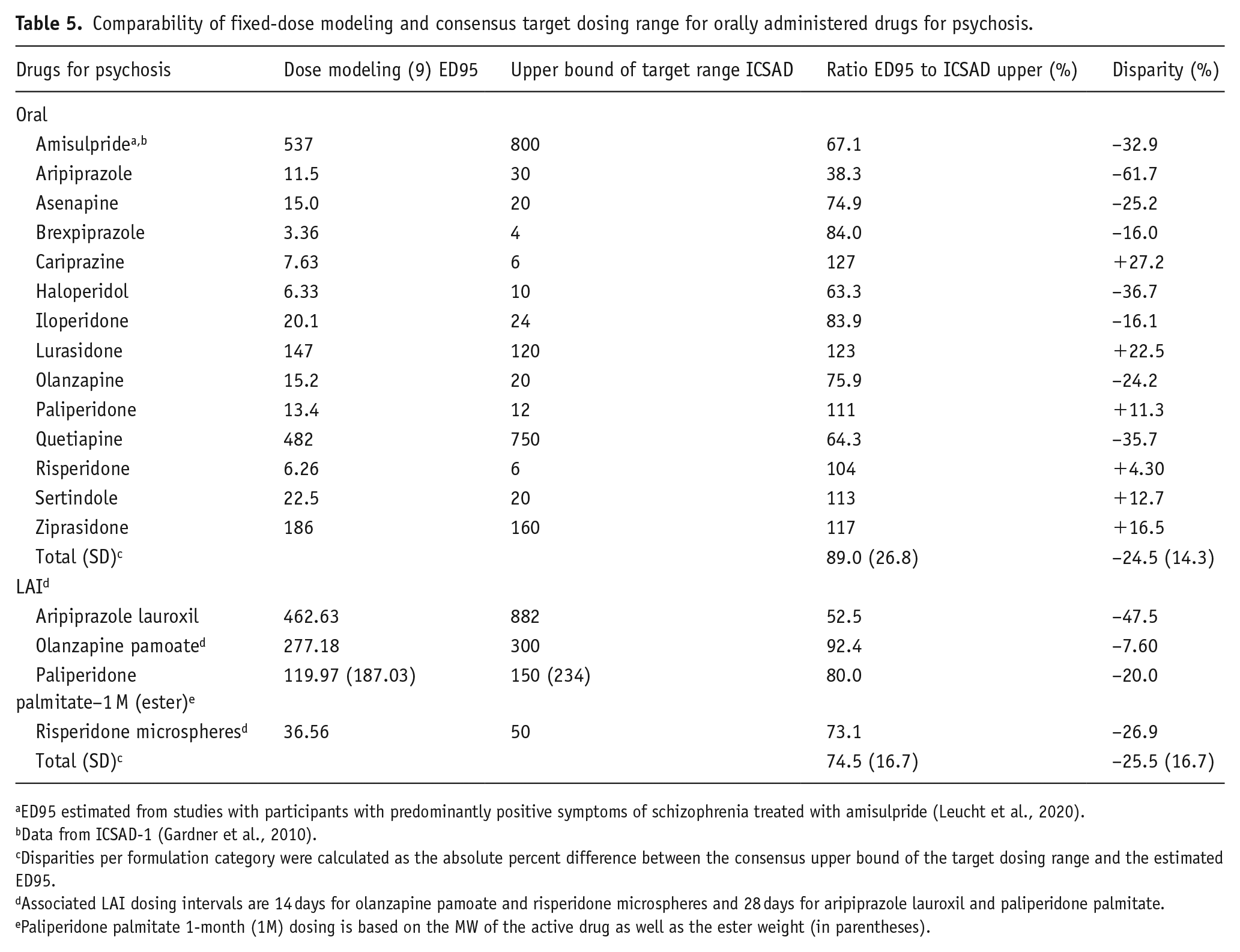
ED95 estimated from studies with participants with predominantly positive symptoms of schizophrenia treated with amisulpride (Leucht et al., 2020).
Data from ICSAD-1 (Gardner et al., 2010).
Disparities per formulation category were calculated as the absolute percent difference between the consensus upper bound of the target dosing range and the estimated ED95.
Associated LAI dosing intervals are 14 days for olanzapine pamoate and risperidone microspheres and 28 days for aripiprazole lauroxil and paliperidone palmitate.
Paliperidone palmitate 1-month (1M) dosing is based on the MW of the active drug as well as the ester weight (in parentheses).
Discussion
The present study, ICSAD-2, is an update to our original two-stage Delphi international consensus study on dosing of drugs for psychosis reported in 2010, ICSAD-1 (Gardner et al., 2010). ICSAD-2 involved a larger number of experts (72 vs. 41) and an expanded geographic sample (24 vs. 18 countries or regions). It includes 13 new expert-based equivalency estimates and dosing recommendations, including for seven orally administered drugs for psychosis (asenapine, brexpiprazole, cariprazine, iloperidone, lumateperone, lurasidone, and pimavanserin) and six LAI formulations (aripiprazole lauroxil, aripiprazole monohydrate, olanzapine pamoate, paliperidone palmitate (1-month and 3-month preparations), and risperidone SC LAI). Equivalency estimates and dosing recommendations for 13 other drugs for psychosis with low dosing consensus from ICSAD-1 or other identified dosing issues also were updated. The Delphi method was effective in improving consensus for both dosing equivalencies and dosing recommendations as evidenced by the reductions in CV from survey stages I to II. Confidence in clinically equivalent dosing estimates, using oral olanzapine 20 mg/day as the reference agent, was medium or high for most oral (11/14) and all LAI (7/7) formulations. For SAI formulations, confidence in equivalency estimates, using haloperidol 5 mg SAI as the reference agent, remained medium or low for the three formulations included.
Dosing guidance for the 13 newer drugs for psychosis agreed closely with manufacturers’ product monograph dosing recommendations. There were no differences between the median findings of the consensus dosing and manufacturer’s recommendations for three oral agents (brexpiprazole, iloperidone, lumateperone) and five LAI products (aripiprazole lauroxil, aripiprazole monohydrate, olanzapine pamoate, paliperidone pamoate 3-month dosing, and risperidone SC LAI) (Alkermes Inc, USA, 2021; Indivor Inc, USA, 2022; Intra-Cellular Therapies, USA, 2022; Janssen Pharmaceuticals Inc., USA, 2021b; Otsuka America Pharmaceutical, Inc, 2020, 2021; Vanda Pharmaceuticals Inc., 2017). Compared to manufacturers’ recommendations, our consensus findings recommended somewhat lower starting doses for asenapine and pimavanserin and lower target range bounds (upper and lower) for lurasidone and pimavanserin (Acadia Pharmaceuticals Inc, USA, 2020; Allergan, USA Inc, 2017; Sunovion Pharmaceuticals Inc, USA, 2019). Higher doses were recommended for the lower bound of the target range for cariprazine and paliperidone pamoate 1-month dosing (Allergan, USA Inc, 2022; Janssen Pharmaceuticals Inc., USA, 2022a). There were no differences in maximum recommended doses for any agent.
This comparison is important as the clinical use of drugs for psychosis does not always align with manufacturer’s recommendations, and manufacturer’s recommendations may not always reflect what will eventually be identified as optimal dosing for newer products. The history of dosing of drugs for psychosis is marked by notable changes in practice trends over time among all such drugs as well as for specific agents, with evidence of the persistence of controversial “high-dose” regimens exceeding typical maximum dosing guidance of ⩾1000 mg chlorpromazine equivalents per day (Malandain et al., 2022; Sim et al., 2009). The discovery that expert opinion is thus far aligned with product monograph dosing for newer drugs for psychosis may offer reassurance that a refined understanding of dosing of drugs for psychosis is emerging. The high level of agreement also may reflect, to some extent, limited clinical experience with newer agents and the corresponding influence of manufacturers’ recommendations on clinical practice.
Among eight oral agents included in both ICSAD-1 and ICSAD-2, dosing equivalencies remained stable except for paliperidone (Gardner et al., 2010). Its initial equivalency estimate of 9.0 mg (19 responses) was increased to 12 mg (68 responses). Dosing recommendations in ICSAD-2 trended lower than in ICSAD-1 overall, except for paliperidone’s dosing range (from 6–9 to 6–12 mg/day) and sertindole’s maximum dose (from 22 to 24 mg/day). Aripiprazole’s dosing range was modified from 15–30 to 10–30 mg/day. For haloperidol, the initial, target range, and maximum doses were reduced from 3 mg, 5–10 mg, and 20 mg, respectively, to 2 mg, 4.5–10 mg, and 17.5 mg/day. Quetiapine, risperidone, and ziprasidone dosing recommendations were reduced for the target range (quetiapine: from 400–800 to 300–750 mg/day; risperidone: from 4–6 to 3–6 mg/day; ziprasidone: from 120–160 mg/day to 80–160 mg/day) and maximum dose (quetiapine, from 1000 to 800 mg/day; risperidone, from 8.5 to 8.0 mg/day; ziprasidone, from 200 to 160 mg/day). These modest changes likely reflect experience-based changes in the clinical use of these agents over the 13-year interval between data collection for ICSAD-1 and ICSAD-2 and possibly relatively low numbers of responses for some agents in ICSAD-1. There was greater variance when comparing the ICSAD-2 dosing of these oral agents with manufacturers’ product monograph-recommended dosing, again possibly reflecting trends informed by more extensive clinical experience. Higher target range or maximum doses (33–100% higher) were recommended for aripiprazole, olanzapine, paliperidone, and ziprasidone compared to current monograph dosing recommendations (Janssen Pharmaceuticals Inc., USA, 2021a; Lilly USA, LLC, 2019; Otsuka America Pharmaceutical, Inc, 2020; Pfizer Inc, 2022). Lower start, target, or maximum doses (25–50% lower) also were recommended for paliperidone and risperidone (Janssen Pharmaceuticals Inc., USA, 2021a, 2022b).
SAI dosing remains the least well-established of the major types of formulations of drugs for psychosis. The average CV for the four SAIs was 78% compared to 34% for oral agents and 20% for LAIs. The overall CV for injected triflupromazine hydrochloride was 121% indicating that its dosing estimates are imprecise and unreliable. Triflupromazine is an aliphatic phenothiazine, similar to chlorpromazine, first used clinically in the 1950s (Rudy et al., 1958). It has a WHO DDD of 100 mg for both its oral and SAI formulations (WHO, 2023). It was included in ICSAD-2 since its ICSAD-1 maximum dose (68 mg/day) was well below its DDD; nevertheless, in the present survey, it received an even lower recommended maximum dose of 28 mg/day with an IQR of 36 mg. The DDD of zuclopenthixol acetate (30 mg) also was incongruent with ICSAD-1 dosing. However, including it in ICSAD-2 resulted in the same dosing recommendations other than an increase from 100 to 150 mg as the maximum 24-h parenteral dose.
Collectively, ICSAD-1 and ICSAD-2 offer clinical dose equivalences and dosing recommendations for 74 drugs for psychosis, including 44 oral, 14 SAI, and 16 LAI formulations. A direct comparison of dosing guidance was possible for 18 drugs (14 oral, 4 LAI) for which dose estimates for near-maximum symptom reduction are available (Leucht et al., 2020). Our analysis yielded a low to intermediate level of agreement in dosing when comparing ED95 with the upper bound of the target dose range, which is arguably the appropriate consensus dosing parameter expected to approximate the ED95. The average dosing disparity for oral agents was 24.5% (31.0% when the ED95 was below the consensus dose and 15.7% when above) and 25.5% for LAIs (all below). Both approaches offer valuable information, albeit not always consistent. ED95 requires fixed-dose randomized trials to derive a modeled dose–response curve that allows for a reasonably precise estimate of the dose associated with maximum clinical benefit. For most drugs for psychosis, such studies are few or absent. Consensus-based dosing can compensate for such research gaps but requires input from a wide range of clinical experts with extensive experience with each agent and formulation. For newer drugs, experience is relatively limited, and dosing recommendations will therefore tend to default to manufacturer’s recommendations with the possibility of changing as clinical experience increases over time. The direction of dose changes, if any, from the original manufacturer’s recommendations is difficult to predict as there are examples of both upward (e.g., olanzapine, ziprasidone) and downward (e.g., haloperidol, risperidone) dosing trends over time for the treatment of schizophrenia (Citrome et al., 2009a, 2009b; Citrome and Kantrowitz, 2009; Højlund et al., 2019). Dosing equivalency information also can be used to inform dose adjustments especially related to gradual dose reduction in stable psychotic disorder patients by identifying dose reductions that would represent unacceptable risks (Tani et al., 2020). In a synthesis of clinical trial data involving 10 drugs for psychosis, Leucht et al. (2021) estimated relapse risk at different doses presented as risperidone equivalents. Their findings of a nonlinear increase in relapse risk, especially evident at doses of ⩽3.5 mg/day of risperidone equivalents, may be used to inform and quantify estimates of relapse risk in clinical practice for a wide range of drugs for psychosis at different doses assuming the transferability of dosing equivalents and an approximately consistent shape of the relapse risk versus dose function across drugs of this type.
Clinical practice is moving increasingly to the use of newer LAI formulations of second-generation drugs for psychosis supported by findings of reduced risk for hospitalization and death (Kishimoto et al., 2021; Nasrallah, 2018; Taipale et al., 2018). The present survey expands the number of such newer formulations reviewed from one to eight. Dosing of risperidone microspheres remained stable between the two surveys and the level of dosing consensus was highest overall for LAIs in ICSAD-2. There also was general agreement with the need to confirm drug tolerability prior to initiating treatment with an LAI agent; only 15% of participants indicated establishing tolerability “sometimes”, “rarely”, or “never” before giving an LAI drug, indicating that special circumstances may sometimes justify bypassing tolerability testing and can be explored in future research.
Limitations
This study shares many of the limitations of ICSAD-1. Experience-based dosing recommendations can be influenced by individual idiosyncrasies and local dosing practices that can change over time. Participants also may have unidentified conflicts of interest, though individuals primarily affiliated with a pharmaceutical corporation were excluded from survey participation. Furthermore, our survey sample mostly included male researchers and clinicians from Africa, South America, and the Middle East were underrepresented despite efforts to achieve a broad sample base based on both gender and geography.
The reported clinical equivalency findings are based on specific patient and dosing vignettes and may not apply in all clinical circumstances. Prioritization of efficacy over tolerability may not always align with patient preferences. Male patients were described in the present clinical vignettes, although drugs for psychosis may produce larger effect sizes in females (Rabinowitz et al., 2014; Storosum et al., 2023), with previous suggestions that doses of drugs for psychosis be decreased by about 10% for female patients (Gardner et al., 2010). There are also sex differences in adverse effect profiles of drugs for psychosis (Aichhorn et al., 2007; Smith, 2010) and in the pharmacokinetics of individual agents (Aichhorn et al., 2007). The impact of 17 patient factors on dosing was reported in ICSAD-1 (Gardner et al., 2010). The largest recommended dose changes were associated with age (pre-puberty, adolescence, and >65 years of age), hepatic and renal impairment, first-episode psychosis, neurological syndromes, illness severity, and non-psychotic disorder diagnoses. Furthermore, the findings from ICSAD-1 and ICSAD-2 do not preclude the use of therapeutic drug monitoring as a tool to support the dosing of drugs for psychosis, especially for challenging clinical situations as elucidated in a recent joint consensus statement (Schoretsanitis et al., 2020).
Linearity of equivalency at lower or higher doses cannot be assumed. Therefore, a fixed equivalency ratio between two drugs for psychosis may not accurately reflect their therapeutic equivalencies and tolerability at different doses requiring an overall cautious approach involving medication switches. In addition, conditions other than schizophrenia for which such drugs are commonly used, including major mood disorders, various acute psychotic illnesses, schizoaffective disorders, and certain neuropsychiatric conditions, have dosing requirements that differ from those recommended here. Moreover, substantial variation and subtlety often arise in clinical practice at the level of individual patients. Finally, the recommendations and equivalencies in this study are estimates based on consensus summaries of individual expert opinions. They should be used in conjunction with findings from fixed-dose randomized trials assessing clinical response to drugs for psychosis.
Conclusions
Despite its limitations, the present survey of a broad international sample of participants with substantial expertise, and the successful application of the Delphi method to improve consensus, provides rigor and confidence in the reported findings for oral and LAI drugs for psychosis, though findings involving SAI products remain far less secure. ICSAD-2 greatly extends the findings of its predecessor ICSAD-1 and should contribute further to guiding clinical treatment, at least for the primary indication of drugs for schizophrenia, as well as to the design and interpretation of clinical research involving drugs for psychosis. Further randomized, controlled, multiple fixed-dose efficacy and tolerability studies, including head-to-head comparisons of products of different manufacturers, would help in further elucidating dosing equivalencies and optimizing dosing recommendations.
Supplemental Material
sj-docx-1-jop-10.1177_02698811231205688 – Supplemental material for Second International Consensus Study of Antipsychotic Dosing (ICSAD-2)
Supplemental material, sj-docx-1-jop-10.1177_02698811231205688 for Second International Consensus Study of Antipsychotic Dosing (ICSAD-2) by Matthew KT McAdam, Ross J Baldessarini, Andrea L Murphy and David M Gardner in Journal of Psychopharmacology
Footnotes
Acknowledgements
The authors thank all study participants; those who consented to be named included: Drs. Martin Alda (Canada), Mustafa Amin (Indonesia), Ross Baldessarini (USA), Miguel Bernardo (Spain), Michel Bioque (Spain), Istvan Bitter (Hungary), Chilis Bonginkosi (South Africa), Roberto Brugnoli (Italy), Tom Bschor (Germany), Ching-Jui Chang (Taiwan), Leslie Citrome (USA), Bonaventura Handoko Daeng (Indonesia), Guido Di Sciascia (Italy), Helio Elkis (Brazil), Andreas Erfurth (Austria), Clemente Garcia-Rizo (Spain), Perugi Giulio (Italy), Gerhard Gründer (Germany), Alkomiet Hasan (Germany), Yoji Hirano (Japan), Philip Janicak (USA), Yasuhiro Kaneda (Japan), Azimatul Karimah (Indonesia), Taro Kishi (Japan), Goergios Kotzalidis (Italy), Hsien-Yuan Lane (Taiwan), Jimmy Lee (Singapore), Stefan Leucht (Germany), Shih-Ku Lin (Taiwan), Chen-Chung Liu (Taiwan), Pierre Michel Llorca (France), Mario Louza (Brazil), Giovanni Manfredi (Italy), Massimo Carlo Mauri (Italy), Jonathan Meyer (USA), Roumen Milev (Canada), Richard Million (Canada), Pavel Mohr (Czech Republic), Vicente Molina (Spain), Andrea Murru (Spain), Shinichiro Nakajima (Japan), Isabella Pachiarotti (Spain), Eduard Parellada (Spain), Pornjira Pariwatcharakul (Thailand), Seon-Cheol Park (Korea), Jan Pecenak (Slovakia), Ana Lúcia Rodrigues Moreira (Ireland), Gabriele Sachs (Austria), Ludovic Samalin (France), Pedro Sanchez (Spain), Spyridon Siafis (Germany), Kasper Siegfried (Austria), Amílcar Silva-dos-Santos (Portugal), Georgios Schoretsanitis (Switzerland), Kang Sim (Singapore), Amarendra N. Singh (Canada), Manit Srisurapanont (Thailand), Martin Strassnig (USA), Scott Stroup (USA), Takefumi Suzuki (Japan), Hiroyoshi Takeuchi (Japan), David Taylor (U.K.), Hiroyuki Uchida (Japan), Juan Undurraga (Chile), Gustavo Vázquez (Canada), Javier Vázquez-Bourgon (Spain), Peter Weiden (USA), Yen Kuang Yang (Taiwan), Norio Yasui-Furukori (Japan), Ayşegül Yildiz (Turkey) and Nevzat Yuksel (Turkey). Evidence Synthesis Librarian Robin Parker supported the search for eligible participants.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Supported in part by a grant from the Bruce J. Anderson Foundation and by the McLean Private Donors Psychiatry Research Fund (to RJB). Otherwise, this research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
