Abstract
Background:
Medicinal cannabis products containing Δ9-tetrahydrocannabinol (THC) are increasingly accessible. Yet, policy guidelines regarding fitness to drive are lacking, and cannabinoid-specific indexations of impairment are underdeveloped.
Aims:
To determine the impact of a standardised 1 mL sublingual dose of CannEpil®, a medicinal cannabis oil containing 100 mg cannabidiol (CBD) and 5 mg THC on simulated driving performance, relative to placebo and whether variations in vehicle control can be indexed by ocular activity.
Methods:
A double-blind, within-subjects, randomised, placebo-controlled, crossover trial assessed 31 healthy fully licensed drivers (15 male, 16 female) aged between 21 and 58 years (M = 38.0, SD = 10.78). Standard deviation of lateral position (SDLP), standard deviation of speed (SDS) and steering variability were assessed over time and as a function of treatment during a 40 min simulated drive, with oculomotor parameters assessed simultaneously. Oral fluid and plasma were collected at 30 min and 2.5 h.
Results:
CannEpil did not significantly alter SDLP across the full drive, although increased SDLP was observed between 20 and 30 min (p < 0.05). CannEpil increased SDS across the full drive (p < 0.05), with variance greatest at 20–30 min (p < 0.001). CannEpil increased fixation duration (p < 0.05), blink rate (trend p = 0.051) and decreased blink duration (p < 0.001) during driving. No significant correlations were observed between biological matrices and performance outcomes.
Conclusions:
CannEpil impairs select aspects of vehicle control (speed and weaving) over time. Alterations to ocular behaviour suggest that eye tracking may assist in determining cannabis-related driver impairment or intoxication. Australian and New Zealand Clinician Trials Registry, https://anzctr.org.au(ACTRN12619000932167).
Introduction
Recent and ongoing amendments to international laws have resulted in greater access to a range of cannabis products through legalisation for recreational and therapeutic purposes (Ruheel et al., 2021). Cannabidiol (CBD) and Δ9-tetrahydrocannabinol (THC) are the two most well-known compounds found in the cannabis plant, with THC responsible for the psychoactive effects associated with cannabis use, while CBD is known for its potential therapeutic properties (Kalaba and Ware, 2022). Products containing CBD and THC are increasingly utilised for their proposed medicinal properties and are commonly used in combination to manage chronic pain and conditions such as epilepsy, insomnia and anxiety (Busse et al., 2021; Whiting et al., 2015). Despite increased availability, there remains a lack of controlled research on their effects across a wide range of safety-critical outcomes such as driving, and methods to discern impairment from drug presence remain underdeveloped (McCartney et al., 2021). In some jurisdictions, therapeutic use may result in a positive roadside saliva test for THC with potential penalties for driving under the influence, irrespective of the degree of impairment (Perkins et al., 2021). Consequently, it is critical to index the specific effect of varying cannabinoid product permutations on key driving performance and visual attention outcomes.
Expert consensus shows that cannabis use is associated with at least a modest increase in crash risk (International Council on Alcohol, Drugs, and Traffic Safety, 2022), and recent meta-analytic research has further revealed that drivers who test positive for THC or associated metabolites are approximately 30%–40% more likely to be involved in a collision, relative to drivers who test negative for cannabis (Rogeberg et al., 2016). Acute cannabis consumption has been linked to slowed reaction time (Hunault et al., 2009; Liguori et al., 1998), reduced working memory capacities (Curran et al., 2002; Lamers et al., 2006; Ploner et al., 2002; Ramaekers et al., 2021; Solowij et al., 2002), alertness and sustained attention abilities (Crean et al., 2011; Wadsworth et al., 2006), translating to a higher overall collision risk compared to unimpaired drivers (Sagberg et al., 2015; Tement et al., 2020). Research utilising on-road and simulated driving tasks following cannabis use has shown varying degrees of impairment on more direct measures of driving ability, including weaving of the vehicle (Hartman et al., 2015; Ramaekers et al., 2000), maintenance of speed (Tement et al., 2020) and variability in steering (Alvarez et al., 2021; Downey et al., 2013; Ronen et al., 2008). Impairments in driving performance associated with cannabis use are primarily attributed to THC, its main psychoactive component (Solowij et al., 2002). However, extant research indicates that cannabis-induced driving impairment varies by route of administration (i.e. oral, inhaled combusted smoke, inhaled vapour), dose and sample demographics (i.e. current and past cannabis use), with such variations having important implications for current medical cannabis users. Yet, a direct causative relationship between therapeutic cannabinoid products including THC and driving behaviour is still lacking, with robust and reliable objective methods to determine driver state due to cannabis usage only in the early stages of development (e.g. Shahidi Zandi et al., 2021).
Both inhaled and oral-based acute cannabis consumption has been implicated in the regulation of oculomotor processes essential to the selection and uptake of visual stimuli, with THC selectively impacting several aspects of oculomotor and visual attention processes critical to driving (Arkell et al., 2022a; Ploner et al., 2002). THC-associated deficits in the ability to control and suppress eye movements in a precise and deliberate manner have been shown to increase saccadic latency and inaccuracy among both acute and ongoing cannabis users (Kleinloog et al., 2012; Yoon et al., 2019; Zuurman et al., 2008). Monitoring of ocular markers linked to driving impairment, such as saccadic activity (Di Stasi et al., 2012), gaze behaviour (Khan and Lee, 2019) and eyelid/blink characteristics (Dong et al., 2011), has been increasingly utilised within next-generation vehicle safety systems (Hayley et al., 2021). These systems presently lack sufficient specificity to distinguish cannabis use from other intoxication profiles, resulting in a critical need to index cannabinoid-specific alterations to oculomotor control to further define behavioural impairment relevant to driving (Arkell et al., 2021).
A significant number of individuals who use cannabinoid preparations, particularly those containing THC, believe that their medical cannabis use does not impact their ability to drive and subsequently perceive the risks associated with driving under the influence of cannabis as relatively low (Arkell et al., 2020a). According to Arkell et al. (2020a), medicinal cannabis users express a high level of confidence in their ability to accurately assess their driving performance following cannabis consumption. However, limited research exists on whether individuals’ self-reported perceived driving quality reflects actual cannabis-related alterations to driving performance, particularly at lower THC dosages.
Given the multidimensional nature of driving, and the relevance of co-monitoring of ocular activity as a novel means to determine driver state due to cannabis use, the present study aims to assess whether a standard 1 mL sublingual dose of CannEpil®, a 20:1 CBD to THC medicinal cannabis oil, impacts driving performance and eye-movement behaviour during driving. An absolute level of THC present in oral fluid and plasma, as well as CBD and THC metabolites 11-OH-THC and THC-COOH in plasma, were quantified following CannEpil administration to determine potential intoxication–impairment interactions. Based on the reviewed evidence, we hypothesise that the administration of CannEpil will not significantly impair driving ability, but may produce variations in eye-movement outcomes relative to placebo.
Methods
Participants
This double-blind, within-subjects, randomised, placebo-controlled, crossover trial comprised a final sample of 31 healthy adults (16 males, 15 females) aged between 21 and 58 years old (M = 38.13, SD = ±10.78) who weighed between 50 and 98 kg (M = 73.10, SD = ±12.42). Recruitment was conducted via convenience sampling methods utilising print and online advertisements, and an email campaign distributed to a database of prior participants who had consented to be contacted for future studies. All participants had previous experience with cannabinoid products without any negative effects (subjective report) and were required to abstain from any illicit drug use in the 2 weeks prior to and for the duration of the study. Inclusion criteria specified that all participants are fluent in written and spoken English, have a full and unregulated driver’s licence and are all regular drivers (>4000 km/year). Participants were excluded if they self-reported misuse of illicit drugs or if they had a history of substance abuse or dependence on any drugs, were taking prescription medication, had a significant medical condition, were pregnant or breastfeeding or had participated in any other investigational study within the 30 days prior to enrolment. All participants provided written informed consent. This project was approved by the Swinburne University of Technology Human Research Ethics Committee (2019/20220392-9708), and the study protocol was prospectively registered with the Australian and New Zealand Clinician Trials Registry (ACTRN12619000932167). The trial was conducted in accordance with Good Clinical Practice guidelines and the ethical standards of the Declaration of Helsinki.
Measures
Driving simulator
Driving performance was assessed using the Forum8 driving simulator consisting of a car with adjustable seats, dashboard, steering wheel, indicators, break and accelerator pedals. Three integrated monitors were positioned in front of the steering wheel, displaying a realistic scenario and current speed. A driving simulation developed by Forum8 and tailored to Australian traffic scenarios was utilised for its previously shown sensitivity to drug and alcohol-associated driving impairment (Shiferaw et al., 2019). Participants were required to drive with a steady lateral position in the left lane while maintaining a steady speed of 100 km/h for the duration of 40 min. Driving performance was assessed 90 min post-dosing. Primary outcomes were standard deviation of lateral position (SDLP), standard deviation of speed (SDS) and steering variability (SV).
Ocular monitoring
Oculomotor activity including gaze and blink behaviour was simultaneously recorded using a cap-mounted research-grade eye tracker by SensoMotoric Instruments (SMI, Teltow, Germany) which captures pupil and corneal reflections (with 50 Hz sampling rate) alongside a scene camera. Gaze parameters include fixation rate and duration, gaze transition entropy (GTE) and stationary gaze entropy (SGE), as outlined by Shiferaw et al. (2019). Blink parameters include rate, duration and number of microsleeps per minute. Microsleeps were defined as a blink duration of
Driving Simulator Sickness Questionnaire
Completion of driving simulations can lead to mild motion-induced sickness for some individuals. To monitor this, in combination with treatment effects, the Driving Simulator Sickness Questionnaire (DSSQ) was administered following the driving simulation task. The DSSQ is a self-report questionnaire consisting of 16 symptoms which are answered on a four-point Likert-type scale ranging from 0 = ‘none’ to 3 = ‘severe’. Self-reports were completed during both testing visits immediately following the driving task.
Perceived Driving Quality Scale
Perceived driving ability was measured using the Perceived Driving Quality Scale (PDQS) developed by Verster and Roth (2012), which consists of a visual analogue scale ranging from 0 = ‘I drove exceptionally poorly’ to 100 = ‘I drove exceptionally well’, with a midpoint of 50 = ‘I drove normally’. Subjective driving assessments were collected immediately following the driving task at 135 min post-dosing.
Acute-dosing and biological sampling procedures
The active treatment CannEpil® and matched placebo were provided by MCG Pharmaceuticals. CannEpil is a phytocannabinoid-based product containing a ratio of 20:1 CBD to THC delivered in an oil vehicle and is indicated for the treatment of epilepsy and insomnia. The given dose of 1 mL CannEpil contains 100 mg of CBD and 5 mg of THC. CannEpil and placebo were centrally randomised and counterbalanced by the study sponsor (Cannvalate) with un-blinding information held by a disinterested third party (Clinical Trials Coordinator). All treatments were individually packaged and were identical in labelling, appearance and taste. At each testing session, 1 mL of palatable bearer oil containing either CannEpil or placebo was administered via a syringe sublingually. Peak effect was expected at approximately 120 min with acute effects beginning to be felt from 60 min after ingestion and potentially lasting for 4–6 h (Badowski, 2017). Two experimental sessions were scheduled a minimum of 7 days apart to avoid any carryover effects.
Prior to administration of CannEpil or placebo, oral fluid was screened for the presence of THC, benzodiazepines, cocaine, amphetamines and opiates using a Securetec 6S DrugWipe to ensure drug abstinence. Oral fluid and plasma samples were collected at two time-points during each testing session, both prior to (30 min post-dosing) and following the driving task (~2.5 h post-dosing). Following collection and aliquot, oral fluid (primary and duplicate) samples were stored at −20°C for shipment to Racing Analytical Services laboratory, Flemington, Victoria. All oral fluid samples were analysed in duplicate according the Australian/New Zealand Drug testing standard (AS/NZS 4308:2008). A registered research nurse collected 15 mL of venous blood via a single blood draw using an ethylenediamine tetraacetic vacutainer. Venous whole blood samples were centrifuged at 3000 rev/min for 10 min, prior to the surfaced plasma being pipetted into individual specimen vials. All collected plasma samples were stored at −80°C until collection by a courier for shipment to the Victorian Institute of Forensic Medicine for biochemical analysis of THC, 11-OH-THC, THC-COOH and CBD concentrations.
Procedure
Figure 1 displays an overview of the study schedule on testing days.
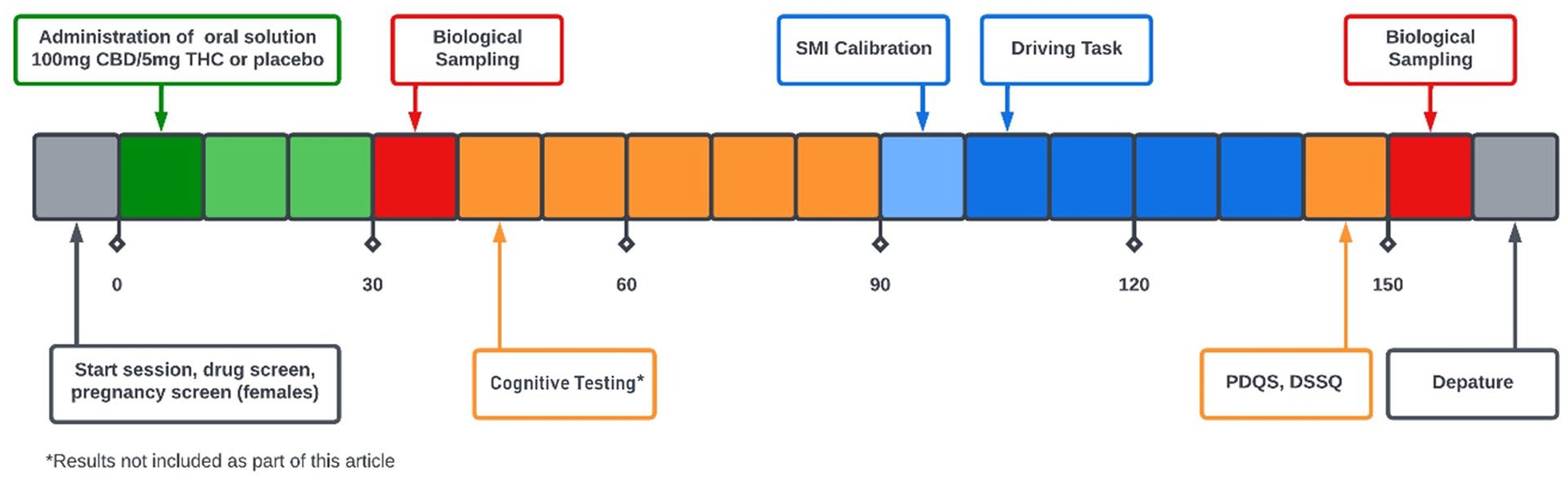
Overview of study session procedures.
Participants were screened over the phone prior to attending a screening visit and two experimental sessions at the Centre for Human Psychopharmacology. During screening, participants discussed their medical and drug history with a nurse, completed the Beck Depression Inventory II and Beck Anxiety Inventory (Beck et al., 1961, 1988) for mental health screening and completed a 10 min practice session on the driving simulator. Participants were asked to have a light breakfast of something that they could replicate across both testing sessions and to abstain from alcohol for 24 h and caffeine for 12 h prior to testing sessions. Female participants were asked to provide a urine sample to test for pregnancy. Participants were informed through a departure information sheet of the potential for the study drug to remain detectable in their system for up to 48 h and of restrictions from operating a vehicle or heavy machinery after testing visits. Participants were reimbursed for their time and were provided with a transportation voucher upon leaving the testing site.
Statistical analyses
To account for variability in performance upon commencing the driving task, outcome data were analysed from when participants reached 90 km/h, as performed previously by Hayley et al. (2018). Driving and ocular outcomes were examined over time across four 10 min interval time-bins (T1, T2, T3 and T4) during the 40 min total drive. All statistical analyses were performed using SPSS (version 26). All analytical procedures were two-tailed, and statistical significance was defined as p < 0.05. Linear fixed effects models with restricted maximum likelihood estimation were used to investigate driving performance and oculomotor outcomes. The likelihood ratio statistic was used to determine the best-fitting variance structure with compound symmetry, or in the case of unequal variances, diagonal covariance matrices determined as best fit. For driving performance and oculomotor outcomes, both treatment condition and time were entered as repeated factors and fixed effects, with separate models built to investigate each outcome. Post hoc paired t-tests with planned Bonferroni adjustments for multiple comparisons were conducted to further explore differences in condition and time. Paired-samples t-tests were performed to investigate differences in perceived driving quality between treatment conditions. Linear regressions were used to investigate whether oral fluid and plasma THC concentrations or plasma levels of 11-THC-OH, THC-COOH and CBD were significantly correlated with driving performance or ocular outcomes.
Noting recent findings revealing sex differences in acute cannabis effects (Arkell et al., 2022b), we examined covariates including sex, cannabis use history and driving experience prior to the planned analysis. Examined covariates did not significantly contribute to the explanation of results and thus were not retained in the final model. Prior to analyses, data were also evaluated for completeness, and the analysis of standard residuals was performed to identify potential outliers. Outliers were identified for outcomes including SV, blinks per minute, microsleeps, fixation duration and 30 min oral fluid THC and plasma CBD concentrations. Sensitivity analyses were conducted with and without these outliers included in the dataset, with results not altered enough for different conclusions to be drawn (i.e. no change in significance) for all outcomes. All outliers were considered genuinely unusual (i.e. not due to error) and thus were retained for analysis (Aguinis et al., 2013). No other outliers were identified (Std Residual Min = −2.93, Std Residual Max = 2.94).
Results
Cannabis use history
Participants reported their cannabis use frequency as ⩽ once per week (9.7%), ⩽ once per fortnight (22.6%), or ⩽ once per month (16.1%). Participants who reported using cannabis more frequently than once weekly during initial screening were excluded as they did not agree to abstain from cannabis use prior to participation. The remainder of the sample (51.6%) reported having abstained from cannabis use in the 12 months prior to enrolment in the study. Figure 2 displays a Consolidated Standards of Reporting Table (CONSORT) diagram depicting recruitment flow.
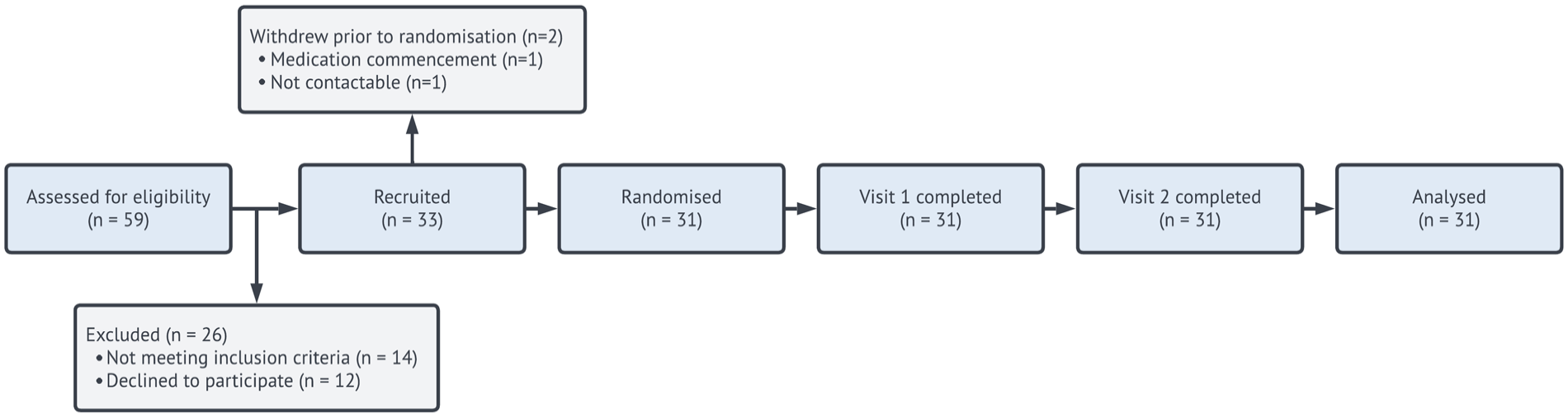
CONSORT/flow diagram.
Driving performance
Summary data including means and standard deviations (±) for driving outcome measures are presented in Table 1. There was no main effect for condition, time or its interaction for SDLP; however, post hoc analyses revealed increased SDLP in the CannEpil condition (M = 28.92, SD = ±5.73) compared to placebo (M = 27.08, SD = ±5.52) at T3 (F(1,209) = 4.60, p < 0.05). There was a significant main effect for both condition (F(1,209) = 5.12, p < 0.05) and time (F(3,209) = 3.67, p < 0.05) for SDS, but not its interaction (F(3,209) = 1.16, p = 0.325). Post hoc analyses revealed increased SDS in the CannEpil condition (M = 2.52, SD = ±1.36) compared to placebo (M = 2.01, SD = ±1.23) at T3 only (F(1,209) = 7.33, p < 0.01). There was a main effect for time within treatment conditions for SV (F(3,304) = 14.73, p < 0.001), with increased SV at T2 in comparison to T3 and 4 in the CannEpil condition (p < 0.01). Similarly, SV was significantly increased at T2 in comparison to T1, T3 and T4, in the placebo condition (p < 0.001). No other condition or interaction effect for SV was noted. Differences in driving performance outcomes SDLP, SDS and SV between CannEpil and placebo across time are displayed in Figure 3.
Driving performance (mean (SD)) overall and across time after administration of CannEpil or placebo.
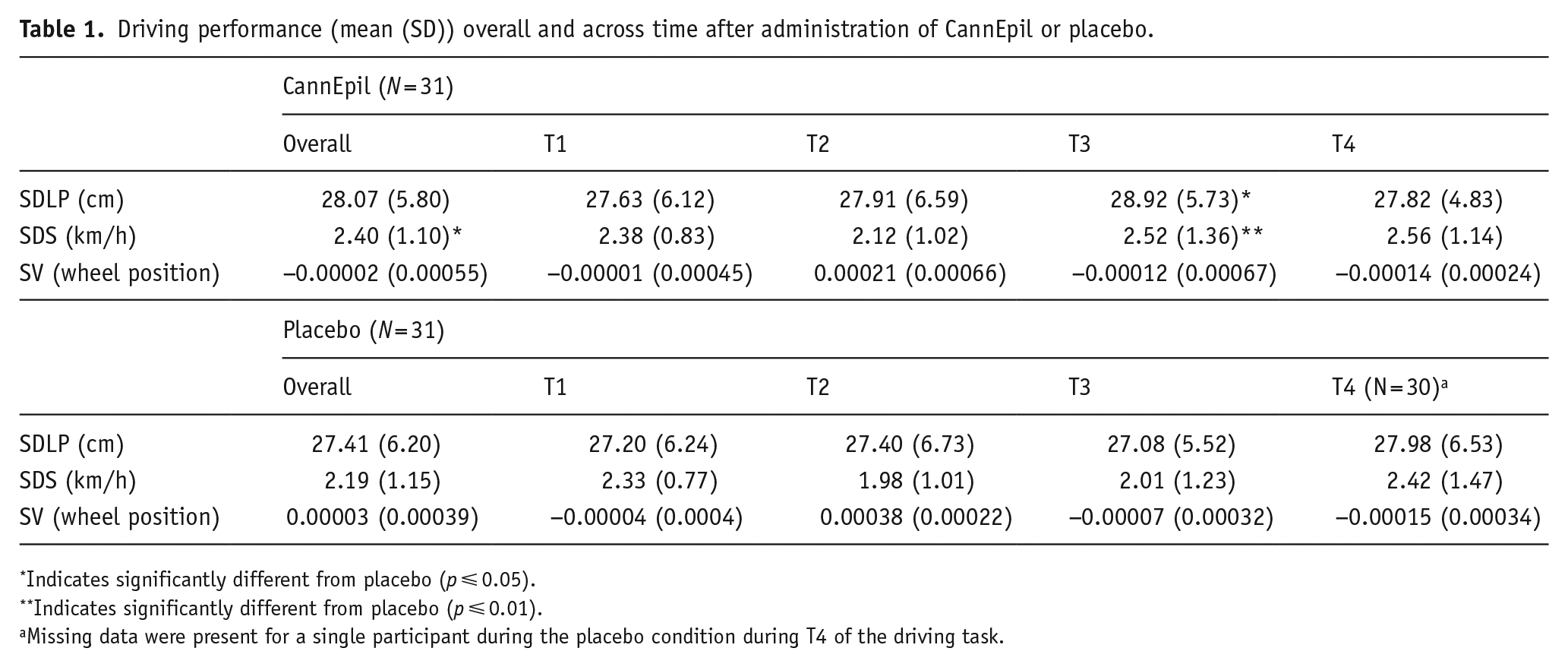
Indicates significantly different from placebo (p ⩽ 0.05).
Indicates significantly different from placebo (p ⩽ 0.01).
Missing data were present for a single participant during the placebo condition during T4 of the driving task.
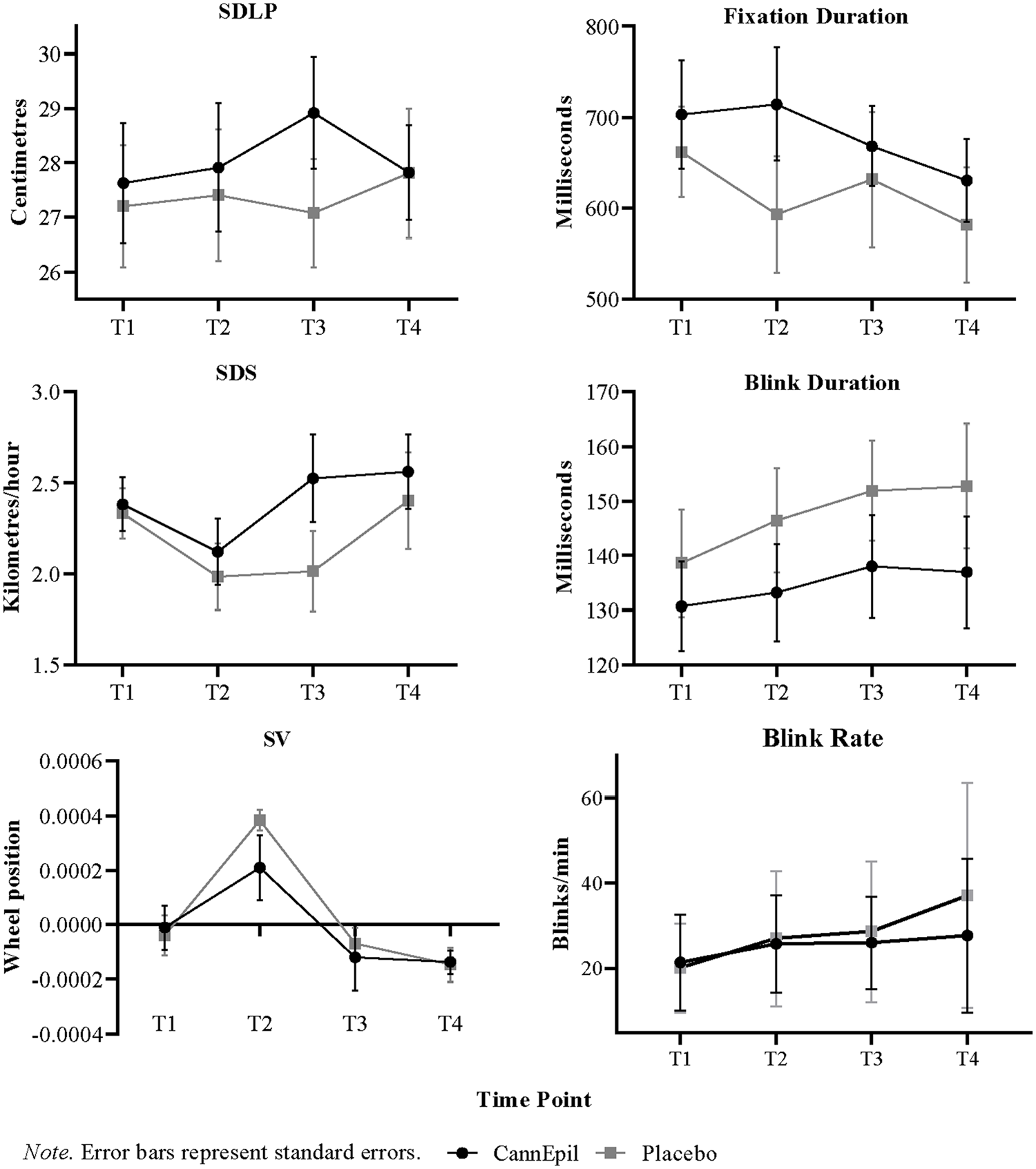
Variations in driving and oculomotor outcomes between CannEpil and placebo across time.
Oculomotor outcomes
Summary data including means and standard deviations (±) for oculomotor outcomes are presented in Table 2. Differences in fixation and blink duration (ms) and blink rate between CannEpil and placebo across time are displayed in Figure 3. There was a main effect of condition for fixation duration (F(1,91) = 5.697, p < 0.05) with significantly longer saccadic fixation in the CannEpil condition (M = 714.47, SD = ±232.16) compared to placebo (M = 593.06, SD = ±239.78) at T2 (F(1,91) = 5.461, p < .05). A significant main effect of condition for blink duration was shown (F(1,91) = 11.020, p ⩽ 0.001), with shorter duration in the CannEpil condition (M = 136.97, SD = ±38.34) relative to placebo (M = 152.72, SD = ±42.87) at T4 (F(1,91) = 4.236, p < 0.05). A trend was also observed across time for blink rate, with number of blinks per minute increasing over the duration of the drive, across both CannEpil and placebo conditions (F(3,40) = 2.81, p = 0.051). There were no significant main effects of condition, time or their interaction for number of fixations per minute, GTE, GSE or microsleeps (all p > 0.05).
Oculomotor parameters (mean (SD)) overall and across time after administration of CannEpil or placebo.
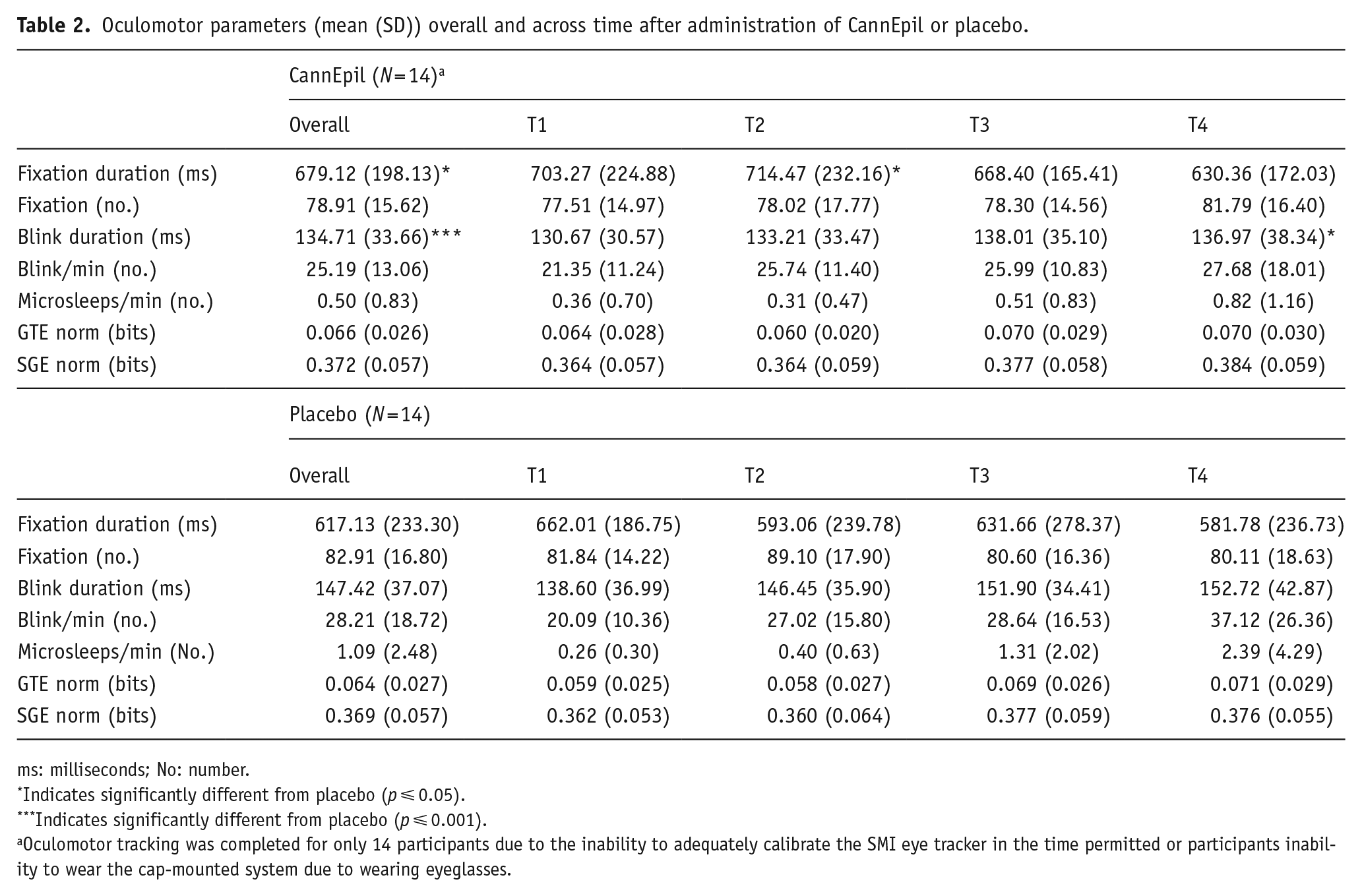
ms: milliseconds; No: number.
Indicates significantly different from placebo (p ⩽ 0.05).
Indicates significantly different from placebo (p ⩽ 0.001).
Oculomotor tracking was completed for only 14 participants due to the inability to adequately calibrate the SMI eye tracker in the time permitted or participants inability to wear the cap-mounted system due to wearing eyeglasses.
Perceived driving quality and simulator sickness
During the CannEpil condition, 39% of participants reported experiencing moderate driving simulator sickness symptom severity, compared with 27% of participants in the placebo condition. A further 6% of participants reported experiencing severe driving simulator sickness symptom severity during the CannEpil condition, including difficulty concentrating, pressure in the head and fatigue, compared with only a single participant (3%) in the placebo condition reporting severe fatigue. There were no significant differences between CannEpil (M = 45.70, SD = ±26.44) and placebo (M = 50.03, SD = ±16.77) in reported PDQS (t(29) = 1.094, p = 0.283).
Biological sampling
Summary data including means and standard deviations for oral fluid THC levels and plasma concentrations of THC, 11-OH-THC, THC-COOH and CBD are presented in Table 3.
Oral fluid THC levels and plasma concentrations of THC, 11-OH-THC, THC-COOH and CBD at 30 min (T1), 2.5 h (T2) and overall.
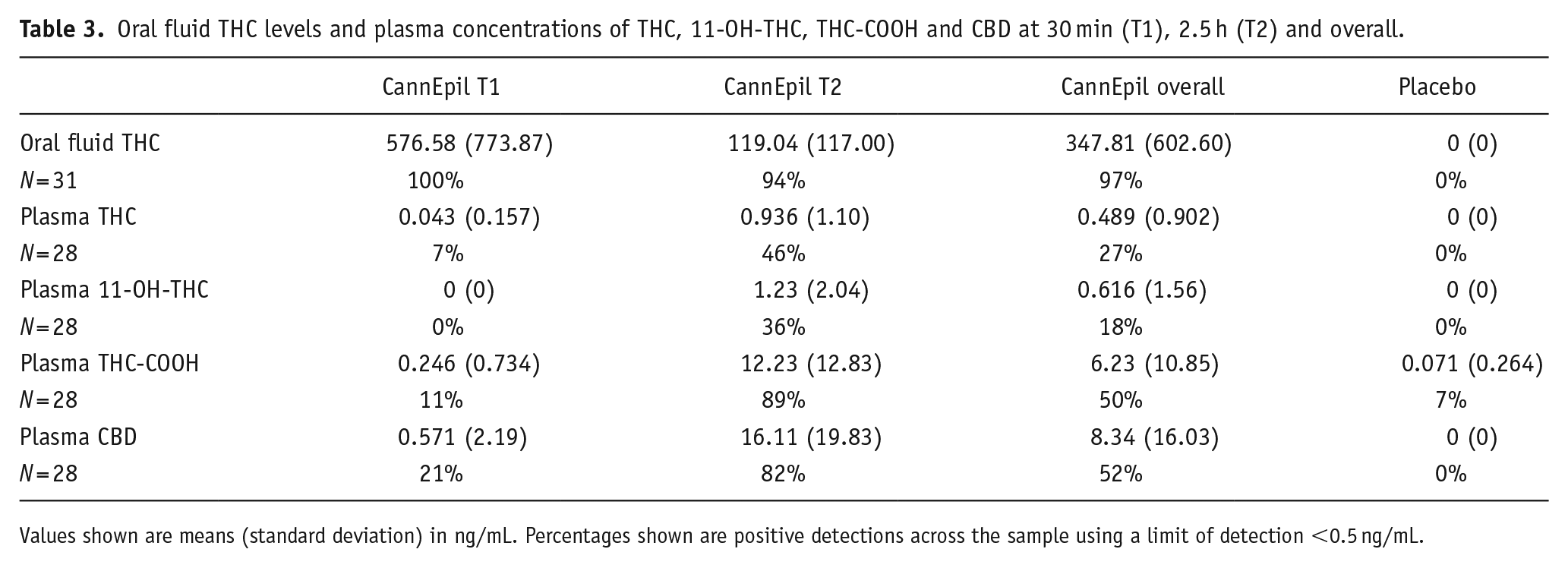
Values shown are means (standard deviation) in ng/mL. Percentages shown are positive detections across the sample using a limit of detection <0.5 ng/mL.
Concentrations of THC in oral fluid are greatest at T1 (M = 576.58 ng/mL, SD = ±773.87), sampled at 30 min post-treatment consumption and significantly decline at T2 (M = 119.04 ng/mL, SD = ±177), sampled at 2.5 h post-treatment consumption (F(1,30) = 14.51, p ⩽ 0.001). Concentrations of THC, 11-OH-THC and THC-COOH in plasma are greatest at T2, sampled at ~2.5 h post-treatment consumption. Oral fluid THC concentrations and plasma levels of THC and 11-OH-THC during the placebo condition were all confirmed at 0 ng/mL. THC-COOH plasma concentrations were detected in two participants (0.9 and 1.1 ng/mL) during the placebo condition. Differences across time in oral fluid THC concentrations and levels of THC, 11-OH-THC and THC-COOH in plasma during the CannEpil condition are displayed in Figure 4. Oral fluid THC concentrations at T1 are significantly correlated with blink rate (F(1,12) = 14.780, p < 0.01) accounting for 55.2% of the variation of the sample, with a large size effect (adjusted R2 = 0.515) according to Cohen (1988). Oral fluid THC concentrations correlation with blink rate is depicted in Figure 5. No other significant correlations were observed between biological matrices and performance outcomes.
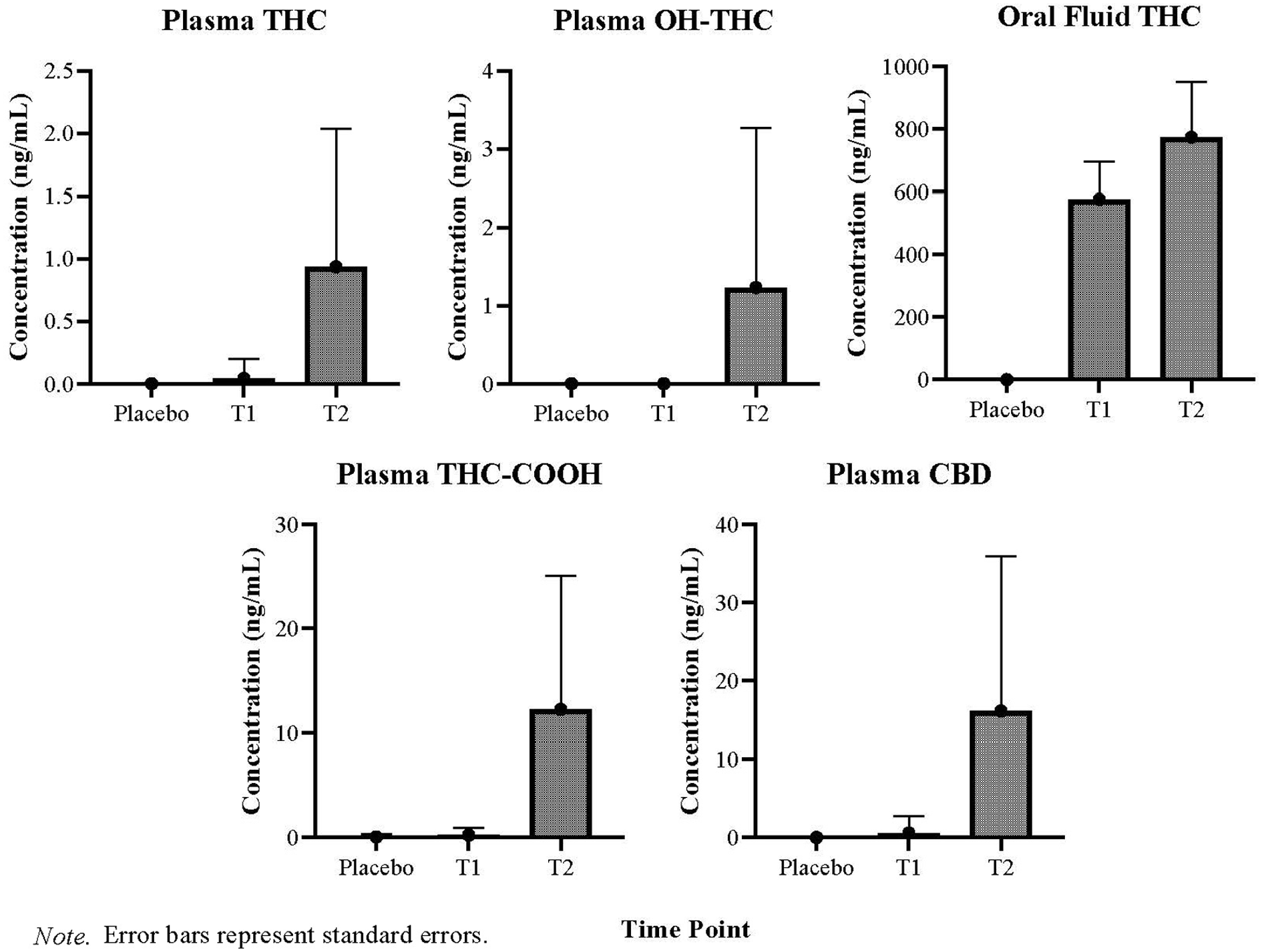
Means and standard deviations of oral fluid and plasma concentrations at 30 min (T1) and 2.5 h (T2) after CannEpil administration.
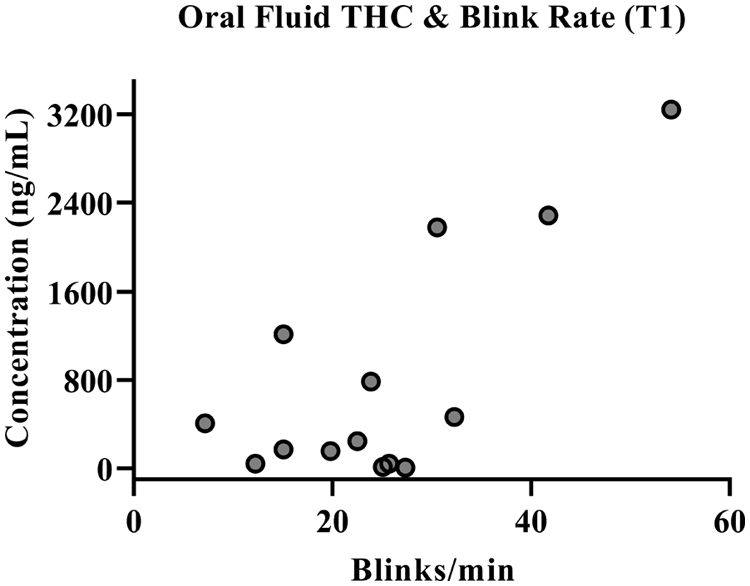
Oral fluid THC concentration correlation with blink rate after CannEpil administration at T1.
Discussion
This randomised, within-subjects, double-blind, placebo-controlled trial demonstrates that in healthy adults an acute sublingual dose of CannEpil® impairs select aspects of vehicle control (speed and weaving) over time and alters oculomotor activity during driving relative to placebo. Increased fixation duration while monotonous highway driving under the influence of even low doses of THC may, in part, reflect time-on-task behavioural effects and increased compensatory attentional/cognitive mechanisms to counteract intoxicant sedating effects while under the influence of these substances. Standardised assessment of performance decrements at therapeutic doses will inform policy guidelines concerning responsible use of medicinal cannabis products and assist both providers and consumers in identifying potential indicators of risk during tasks such as driving.
Administration of CannEpil did not significantly alter SDLP (i.e. weaving of the vehicle) across the full length of the driving task; however, increased SDLP was observed in the CannEpil condition between 20-30 min, relative to placebo. These findings are partially consistent with previous research that links cannabis, and THC specifically, with impairments in vehicle control (Moskowitz, 1985) and increased SDLP (Bosker et al., 2012; Hartman et al., 2015; Ramaekers et al., 2000). Interestingly, Arkell et al. (2019) observed that vaporisation of a combined CBD/THC preparation impaired SDLP in a more complex car following task, but not during a monotonous highway drive. Similarly, during an on-road 100 km long highway drive, a CBD-dominant vaporised cannabis preparation did not produce variations in SDLP relative to placebo, while increased SDLP was observed for THC-dominant and THC/CBD-equivalent preparations (Arkell et al., 2020b). The lack of robust impairment across lateral vehicle control in the present study may be attributed to the relatively simple and monotonous driving task, which may mask the true magnitude of effects. Moreover, the high CBD to THC ratio and comparatively low THC dose of the cannabis preparation administered in the present study may have mitigated some of the effects of THC impairment (Englund et al., 2022). It is also important to note that extant clinical research has primarily focused on smoked or vaporised cannabis, rather than sublingual or oil preparations. Given that acute intoxication may be prolonged with oral THC products (Vandrey et al., 2017), differences in impairment profiles may be partially attributed to a more gradual or lesser onset of peak effects.
SDLP remains a highly sensitive index of THC-related impairment and is among the most consistently used objective measure of continuous behaviour whilst driving (Charlton and Starkey, 2013; Lococo and Staplin, 2006; McCartney et al., 2021; Ramaekers et al., 2006; Verster and Roth, 2011). While SDLP may not directly predict crash risk, increases in lane weaving may predict increased lane departure occurrences (Downey et al., 2013; Papafotiou et al., 2005) therefore increasing the likelihood of collision (Hartman et al., 2015). Many jurisdictions hold driving laws designating 0.05% or 0.08% BAC as a minimum limit of impairment, equating to a SDLP increase of approximately 2.4 cm, with this value often utilised as a comparable indicator of cannabis-related driving impairment (Hartman et al., 2015; Lococo and Staplin, 2006). Given the potential of greater inter-individual variability in simulated driving tasks compared to on-road tasks (Veldstra et al., 2015), the observed greatest SDLP increase of 1.84 cm at only a single time point is likely not substantial enough to translate to a clinically relevant change nor of concern in the context of on-road driving.
Driving speed increased after CannEpil administration across the full length of the driving task and varied as a function of time, coinciding with increases of SDLP. Extant research has more often associated medicinal cannabis consumption with overall reductions in speed (Lenné et al., 2010; Rafaelsen et al., 1973; Ronen et al., 2008), largely as a result of driver’s efforts to mitigate lapses in their attention (Neavyn et al., 2014). Speed increases observed in the present study may reflect drivers’ reduced motivations to employ compensatory driving behaviours, potentially due to an absence of task complexity or realistic driving risk associated with more simplistic simulated highway scenarios (Arkell et al., 2019). The observed impairments in driving performance, which were most evident at 20–30 min, may additionally be attributed to the monotonous nature of the driving task. Participants anecdotally reported experiencing increased boredom and fatigue around this particular timeframe, providing some insight into the potential cause of the observed effects. Contrary to previous research with higher administered THC dosages (Lenné et al., 2010; Ronen et al., 2010), CannEpil consumption with comparatively low THC content did not alter variability in steering behaviour. Nonetheless, evidence for time-on-task effects suggests deterioration of performance during driving and some compensatory steering over-correction, which may be exacerbated during on-road conditions or during poor traffic conditions.
CannEpil increased fixation duration during driving relative to placebo, which was most pronounced between 10-20 min of the driving task. These results partially support limited previous work showing an increase in saccadic latencies following THC administration (Huestegge et al., 2009; Zuurman et al., 2008), particularly during immediate response tasks (Ploner et al., 2002). Here, increased duration of fixation is detected between presentation of a target and commencement of the target saccade. Increased fixation duration is similarly observed among chronic ongoing cannabis users (Huestegge et al., 2010) providing additional evidence implicating the cannabinergic system in temporal aspects of saccadic control during dynamic and demanding tasks such as driving.
Reductions in blink duration were additionally observed after CannEpil consumption, relative to placebo, which was most pronounced towards the final 10 min of the driving task. Decreased blink duration in the CannEpil condition coincides with the observed increase in blink rate, with less time taken to complete a blink, thereby allowing more blinks to occur. A trend was observed for number of blinks per minute across time, with blink rate gradually increasing over the duration of the drive, irrespective of treatment condition. Oral fluid THC concentrations at 30 min also showed a strong positive relationship with blink rate, suggesting a potentially shared mechanism in acute-dosing conditions. Decreased blink duration following CannEpil consumption may be linked to increased occurrences of cannabis-induced eyelid tremors (Hartman et al., 2016; Porath and Beirness, 2019; Porath-Waller and Beirness, 2013) or increased ocular irritation given the proposed involvement of cannabinoid receptors in the modulation of ocular pain and inflammation that can trigger dry eye (Nguyen and Wu, 2019; Vučković et al., 2018). While blink rate may provide a useful indicator for recency of use, its contribution to predicting behavioural effects relevant to traffic safety are likely peripheral at best. Developments in objective impairment monitoring utilising eye-movement behaviour may help advance methods to determine driver impairment or intoxication due to cannabis consumption, particularly following recent/acute use.
THC concentrations in oral fluid were not significantly correlated with driving performance outcomes or oculomotor outcomes revealing of functional impairment. This finding is supported by extant research demonstrating that THC in oral fluid is generally detectable in its highest concentration immediately after cannabis consumption (Vandrey et al., 2017), but highlights the inter and intra-individual variability in THC elimination over time (Niedbala et al., 2001). Thus, it is likely a relatively poor indicator of cannabis/THC induced impairment (McCartney et al., 2022). Plasma concentrations of THC, 11-OH-THC, THC-COOH and CBD were also not significantly correlated with driving or ocular outcomes, underscoring the weak association between detected levels of THC or its metabolites and behavioural effects, particularly at lower THC dosages (Ginsburg, 2019). Despite this, the presence of THC in oral fluid concentrations during the present study may produce a positive result for THC on roadside saliva drug tests within 2 h of using this treatment, potentially exposing drivers to legal penalties despite a lack of objective behavioural impairment.
Limitations and future research
The findings from the present study are to be considered in light of some methodological limitations. As the study was limited to healthy volunteers who were primarily non-regular users of cannabis, our ability to generalise these results to those who consume medicinal cannabis products at higher doses or frequency is not clearly established. As CannEpil was administered as a single acute dose in the present study, the potential impact of repeated dosages also remains unclear. Indeed, more regular cannabis use may facilitate partial tolerance to the effects of CBD and/or THC (Arkell et al., 2020b; Hartman and Huestis, 2013) and further diminish observable driving performance impairments such as those reported in the present study. Secondly, the use of a simulated driving task involving a simplistic highway drive with less traffic and perceptual cues than would be typically encountered during on-road scenarios may protect against sensitivity to THC’s impairing effects that are associated with greater neurocognitive demand (Hartman and Huestis, 2013). While this type of highway driving task has previously shown sensitivity to drug and alcohol-related impairment, it is likely that the intricacies of real-world driving are still not fully represented. Despite this, simulated driving tasks remain essential in providing objective and accurate representations of driving ability in a time-sensitive manner.
We acknowledge that our study did not gather information on abuse liability, as we utilised an acute-dosing paradigm. Although we recognise that subjective intoxicant effects are an important consideration, we did not include these measures in the present work, and this is a noteworthy limitation. Lastly, oculomotor results were not available for the entire sample, due to technical issues with device calibration. To preserve overall data integrity, some cases were discarded for our analysis (e.g. where only one of two ocular measures was captured). Nonetheless, this is the largest and most robust acute-dosing paradigm which examines ocular behaviour during driving under the influence of a representative cannabis medication in applied settings to date, and thus overcomes much of the issues with previous work in this emerging space (Fant et al., 1997; Ploner et al., 2002; Shahidi Zandi et al., 2021).
Future research is encouraged to evaluate the impact of repeated doses over extended periods of time, to assess whether observed behavioural changes are mitigated or altered by partial tolerance effects. In addition, incorporating measures to assess the impact of subjective intoxication on performance would provide valuable insight. Future research would further benefit from inclusion of diverse groups of medical cannabis consuming patient populations to expand our understanding of how ocular parameters may be altered during driving tasks, and whether this is contingent on experience, setting or treatment modality. This will serve to better distinguish the extent to which specific performance domains are sensitive to cannabis-related impairment and under what degree of task demand such impairment arises.
Conclusion
An acute dose of CannEpil produces objective impairment in vehicle control characterised by deficits in maintenance of vehicle speed, and to a lesser extent, maintenance of lateral positioning. Concurrent increases in fixation duration and reductions in blink duration after cannabis consumption provide additional evidence for the role of cannabis in the regulation of oculomotor control and provide possible points for objective intoxication monitoring. Biological sampling data suggest that cannabinoid and metabolite concentrations in oral fluid and plasma are not highly indicative of impairment nor reflect key performance indicators.
Supplemental Material
sj-docx-1-jop-10.1177_02698811231170360 – Supplemental material for Effect of CannEpil® on simulated driving performance and co-monitoring of ocular activity: A randomised controlled trial
Supplemental material, sj-docx-1-jop-10.1177_02698811231170360 for Effect of CannEpil® on simulated driving performance and co-monitoring of ocular activity: A randomised controlled trial by Brooke Manning, Amie C Hayley, Sarah Catchlove, Brook Shiferaw, Con Stough and Luke A Downey in Journal of Psychopharmacology
Footnotes
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: ACH is supported by a Rebecca L. Cooper Al and Val Rosenstrauss Fellowship (GNT: F2021894). All other authors declare that there is no conflict of interest.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The research project findings discussed here were supported by a research grant from Cannvalate (Australia), Toorak, Australia to the Principal Investigator and study author, Prof. Luke Downey. The authors confirm that the study sponsor did not have any influence on the decision to publish the manuscript nor the information contained within.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
