Abstract
3,4 Methylenedioxymethamphetamine (MDMA)-assisted therapy has been recently found to be highly effective for treatment of posttraumatic stress disorder (PTSD). Previous studies have been inconclusive in elucidating potential MDMA genotoxicity. We performed three regulatory compliant studies to investigate the potential of genotoxic effects of MDMA treatment in humans: (1) an
Introduction
3,4 Methylenedioxymethamphetamine (MDMA) or “ecstasy,” is an investigational new drug, currently in Phase 3 of human trials. MDMA has been found to have preliminary efficacy in treating post-traumatic stress disorder (PTSD) when delivered in an integrated multimodal intervention through MDMA-assisted therapy, allowing participants to create a stronger bond with their therapist and tap into emotionally upsetting or traumatic experiences (Burge, 2020). MDMA is a sympathomimetic drug that modifies the release, re-uptake, and longevity of dopamine, serotonin, and norepinephrine in the synaptic cleft. Although MDMA does share similar targets to currently approved antidepressants, its mechanism of action, time to onset, and durability of effects and dosing intervals differ greatly (Kalant, 2001; MDMA Investigator’s Brochure 12th Edition, 2020). Contrary to currently used psychiatric medications, MDMA is not administered daily for months or years on end, but in three divided dose exposures over the course of a few months (Kalant, 2001). Currently there is mixed reporting on the absence or presence of MDMA genotoxicity in various preclinical test systems (Barenys et al., 2009; Frenzilli et al., 2007; Hariri et al., 2010; Parolini et al., 2014; Yoshioka et al., 2007). In order to settle the lack of consensus in previous literature, we conducted three regulatory compliant studies investigating the genotoxic potential of MDMA in humans: (1) an
Methods
An
MDMA HCl was evaluated in the Ames assay (Hamel et al., 2016) in
MDMA HCl was evaluated in the
Results
All positive and negative control values in all assays were within acceptable ranges, and all criteria for a valid assay were met.
In the
In vitro chromosome aberration assay in CHO-WBL cells.
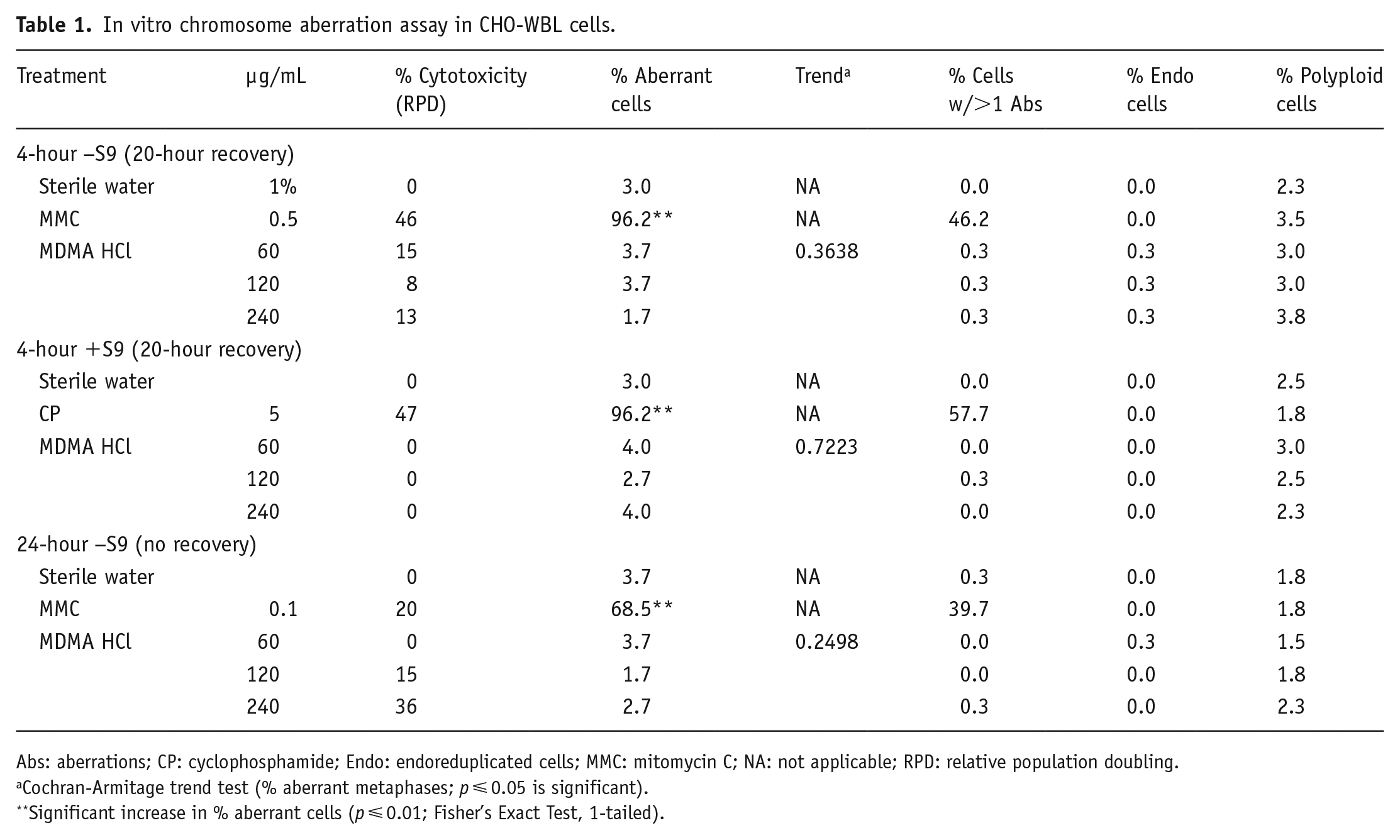
Abs: aberrations; CP: cyclophosphamide; Endo: endoreduplicated cells; MMC: mitomycin C; NA: not applicable; RPD: relative population doubling.
Cochran-Armitage trend test (% aberrant metaphases;
Significant increase in % aberrant cells (
In the Ames assay, no increases in mean revertant frequencies were observed in any tester strain and any dose of MDMA ±S9 (Table 2). Therefore, MDMA HCl was negative for inducing bacterial reverse gene mutations ±S9 under the conditions of this test.
Ames assay results – average revertant colonies per plate (SD).
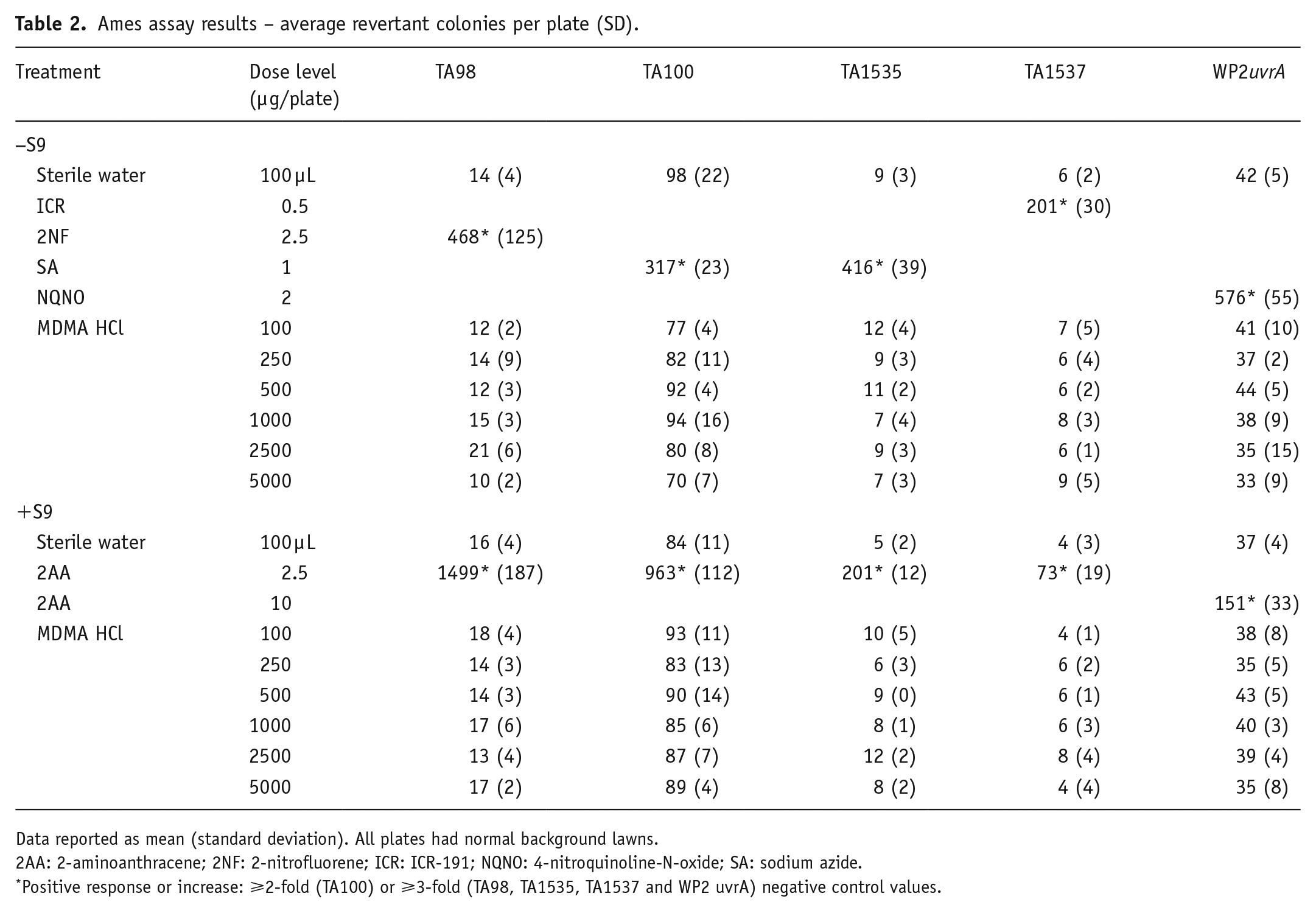
Data reported as mean (standard deviation). All plates had normal background lawns.
2AA: 2-aminoanthracene; 2NF: 2-nitrofluorene; ICR: ICR-191; NQNO: 4-nitroquinoline-N-oxide; SA: sodium azide.
Positive response or increase: ⩾2-fold (TA100) or ⩾3-fold (TA98, TA1535, TA1537 and WP2 uvrA) negative control values.
Adverse clinical observations, as well as reduced bodyweight and food consumption, were observed in the
Discussions
MDMA did not show any signs of genotoxicity across the three robust test systems analyzed. These results are consistent with past articles from Hariri (Hariri et al., 2010) and Yoshioka (Yoshioka et al., 2007). We highlight that these studies are the recommended studies to rule out genotoxicity potential by regulators internationally (ICH Expert Working Group, 2011). Additionally, all studies conducted here were carried out with test system exposures above the relevant clinical Cmax for MDMA (of 195–252 ng/mL) (MDMA Investigator’s Brochure 12th Edition, 2020; Vizeli and Liechti, 2017). We note that results from Barenys et al. (2009), did show signs of DNA damage in sperm and testes of male rats. However we caution that the experiments from Barenys and colleagues were performed with 36 drug exposure days in rapid succession prior to collection of tissue for the comet test, resulting in many fold higher dosages than ever expected or allowed clinically. Additionally, the Barenys study administered MDMA subcutaneously which would be resultant in increased exposure compared to the oral route (as MDMA has a relative oral bioavailability of 37–68% (MDMA Investigator’s Brochure 12th Edition, 2020)). Overall, these results indicate the apparent lack of genotoxicity for MDMA. We hope that these results can guide the safe use of MDMA in humans.
Supplemental Material
sj-docx-1-jop-10.1177_02698811211033603 – Supplemental material for Three regulatory compliant test systems show no signs of MDMA-related genotoxicity
Supplemental material, sj-docx-1-jop-10.1177_02698811211033603 for Three regulatory compliant test systems show no signs of MDMA-related genotoxicity by Isaac Victor Cohen, Laken Barber, Tyson Paul Dubnicka, Sara Beth Hurtado, Sarah Ann Tincher, Leon Frank Stankowski and Berra Yazar-Klosinski in Journal of Psychopharmacology
Footnotes
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: BYK is a paid employee of the 501(c)3 non-profit MAPS, the sponsor of this study, with a joint appointment at MAPS PBC, a wholly owned subsidiary of MAPS. LB is a paid intern at MAPS PBC. TPD, SBH, SAT, and LFS are paid employees of Charles River Labs where the experiments funded by MAPS were performed.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: BYK and LB received financial support for the preparation of the manuscript of this article during their course of employment for MAPS and MAPS PBC. The remaining author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
