Abstract
Introduction:
Due to their euphoric and sedative effects, opioid analgesics have high potential for abuse and dependence. In the last decade in the USA and many Western European Countries the prescription rates of opioid analgesics have steadily increased.
Objective:
This study describes 5-year trends in the prescription of opioid analgesics and risk indicators such as duration, dose and ‘doctor shopping’ in Northern Germany.
Methods:
The annual rates of opioid analgesic prescriptions have been analysed for patients with statutory health insurance processed by the North German Pharmacy Data Center for the years 2011–2015. A distinction was made between non-cancer patients and cancer patients, and different groups according to prescription duration and dose level.
Results:
Between 2011 and 2015, the annual number of patients with opioid analgesic prescriptions increased from 500,000 to 550,000. About half of non-cancer-patients (85% of the total sample) and cancer patients received opioid analgesics for 90 days or less. The rates for long-term prescriptions (⩾9 months) ranged between 6–7% for non-cancer patients and 7–8% for cancer patients. Between 1.2–1.8% received opioid analgesics in doses of more than two defined daily doses. The majority of non-cancer patients with opioid analgesic prescriptions were female. The average age of non-cancer patients was 66 years. About 80% of non-cancer patients with first opioid analgesic prescription received World Health Organization step II medication.
Conclusion:
For the first time, this study provides comprehensive patient-related analyses of opioid analgesic prescriptions in Germany over a 5-year period. Despite a slight increase in the overall number of opioid analgesic prescriptions, an epidemic spread of opioid analgesics cannot be observed.
Background
Opioids are used as highly effective pain killers, in anaesthesia (sedation) and as pharmacological options for maintenance therapy in the management of opioid dependence. Depending on their potency, the majority of opioids are subject to legal controls and regulations including special requirements with regard to prescriptions and documentation. Due to their euphoric and sedative effects and the related high potential for abuse and dependence opioids are often trafficked outside of regular prescription (so called ‘black market’). In the USA the unregulated proliferation of opioids has resulted in an opioid epidemic in the recent years, culminating in 47,600 opioid overdose deaths in 2017 (Lippold et al., 2019). In 24–35-year-olds, 20% of deaths were related to opioids in 2016 (Gomes et al., 2018). High amounts of overdose deaths were caused by fentanyl or fentanyl analogues (Hedegaard et al., 2018), mainly by illicitly manufactured fentanyl (Jones et al., 2018). The prevalence of prescribed opioid use among adults in the USA increased from 4.1% in 1999/2000 to 6.8% in 2013/2014 (Mojtabai, 2018), particularly among long-term users. In 1999/2000, 45.1% of all patients received prescribed opioids for prolonged periods of time, compared with 79.4% in 2013/2014 (Mojtabai, 2018). At 12.8% for the years 2017/2018, high rates of opioid analgesics (OAs) prescriptions have also been found in the UK. Half of these patients have been taking these drugs for at least 12 months (Marsden et al., 2019). Between 2015/2016 and 2017/2018, however, the prevalence of OA intake decreased slightly (Taylor et al., 2019). In Germany in 2014 the prevalence rate of taking prescribed opioids among patients with statutory health insurance (SHI) was 4.8% (without-codeine prescriptions), and the proportion of long-term users, with 28.8% among all patients with OA prescriptions, was much lower (Buth et al., 2019).
Germany ranks number two (after USA) among the countries reporting consumption of opioids for pain management in the period 2016–2018 (International Narcotics Control Board, 2020). Whereas the overall prescriptions of defined daily doses (DDDs) of OAs in Germany have continuously increased from 360 m in 2008 to 423 m in 2017 (Böger and Schmidt, 2018), there are currently no indications for an epidemic opioid use among patients (Marschall et al., 2016). This finding is in line with a recent review by Rosner et al. (2019), including 12 German studies for the years 1990–2018. According to a recent study by Kraus et al. (2019) the number of people with opioid addiction in Germany is about 166,000 and has hardly changed over the past 20 years. Cooper et al. (2017) reported comparable findings regarding opioid use in the UK. In the Netherlands Kalkman et al. (2019) found a substantial increase in opioid prescriptions from 4.1% to 7.5% between 2008 and 2017. Chenaf et al. (2019) stated for France that from 2004 to 2017, prescription opioid use at least doubled and oxycodone use increased particularly – although, without an indication for an ‘opioid epidemic’. Wertli et al. (2017) found a similar trend for Switzerland. In their sample, which represented one-sixth of the Swiss population, the number of potent OA prescriptions more than doubled between 2006 and 2013. Nevertheless, according to an estimate by Glaeske (2018), up to 400,000 people in Germany are abusing opioids or are dependent, and recent epidemiological data are missing as respective general population surveys did not distinguish between OA and other analgesics (Atzendorf et al., 2019).
A relevant problem in comparing findings of epidemiological studies are differences in the definition of ‘abusive’ or ‘dependent’ use of opioids. Especially among patients with chronic pain, estimates of prevalence of opioid misuse, abuse and addiction are difficult to determine (Ballantyne, 2015). Whereas some studies focus on long-term use of opioids (Buth et al., 2017) or on high-dose use (Dunn et al., 2010; Gomes et al., 2011) others are using a combination of dose and intake duration (Häuser et al., 2018; Kobus et al., 2012; Marschall et al., 2016).
Generally, the intake of opioids for more than 3 months and doses that exceed the recommendations suggested in the S3 guideline ‘Long-term opioid use in non-cancer pain’ (Häuser et al., 2014) (currently equivalents to 120 mg morphine) are regarded to be problematic. Other studies have used further parameters to determine potential opioid abuse, such as additional hospitalizations due to psychoactive substances use problems (Marschall et al., 2016), number of consulted doctors to obtain higher amounts of opioids (‘doctor shopping’) or regular concurrent use of drugs for anxiety or insomnia (Skurtveit et al., 2011). Depending on which indicator and/or cut-off has been chosen to define abusive or dependent opioid use, the respective prevalence rates can vary substantially.
By considering these methodological differences, the aim of this study was to describe 5-year trends in the prescription of OAs in Germany by defining the (problematic) intake of OAs on the basis of dose and duration of prescriptions. Prevalence rates and trends for the years between 2011 and 2015 have been calculated on the basis of prescription data for different patient groups from Northern Germany with a SHI, which is mandatory for German inhabitants. Especially, people with higher income or with specific professions (e.g. civil servants) have the opportunity to leave the SHI and switch to the private health insurance sector (in 2015, 11.1% of German inhabitants had private health insurance). These cases as well as private prescriptions for SHI insured patients (which can be made for individual reasons) are not part of the data set. Further study objectives included to identify specific medications of particular relevance for different groups of problem opioid users and whether this relevance has changed over time. These analyses can contribute to a better understanding on the prevalence and risk indicators such as duration, dose and so-called ‘doctor shopping’ in the prescription of OAs for non-cancer-patients (NCPs) and cancer patients (CPs).
Methods
Based on prescriptions for SHI patients processed by the North German Pharmacy Data Center (NARZ/AVN) for the years 2011–2015, patients with OAs prescriptions have been analysed for each following observation year within this period. For this purpose also prescription data from 2016 was available.
The NARZ/AVN processes SHI prescription data from all SHI companies for the vast majority of pharmacies in the federal states of Schleswig-Holstein (89.6% of all pharmacies), Hamburg (86.5%), Lower Saxony (76.8%) and Bremen (89.9%). For Northern Germany, the data provide, therefore, almost complete and representative insights into the prescription of all OA medications. For the statistical analyses OAs of the Anatomical Therapeutic Chemical (ATC) code group N02A have been included. In correspondence to Schubert et al. (2013), prescriptions containing codeine were excluded. Dihydrocodeine (ATC code N02AA08) has been included only if the main indication was pain treatment. In the observational period, between 11.2 m (in 2011) and 11.4 m SHI-insured citizens (in 2015) were living in the respective four federal states. The total population was 12.9 m in 2011 and 13.2 m in 2015.
An anonymous dataset was extracted from the NARZ/AVN database including data on patient code, age, place of residence and (indirectly) gender (for a complete description of the data protection concept, see Verthein et al., 2013). Besides this, information about the prescribing physician (including practice site and medical speciality) and the pharmacy were obtained. Further, data on the prescribed medication (i.e. formulation, concentration, single and total dose) were part of the dataset. All data were retrieved in text-tab format (ASCII) and processed and analysed by using IBM Statistical Package for the Social Sciences (SPSS) 25 (IBM, 2017).
The period of an OA prescription within an individual observation year was calculated as the sum of the time intervals between filled prescriptions plus a ‘probable intake duration’ of the last prescribed medication. The basis for the probable intake duration of the last prescribed medication was the average concentration per day of previous prescriptions. If a patient had received only one single prescription of a medication group within a year, the duration of intake was set as the number of DDDs of the medication (for detailed information see Buth et al., 2019).
As the dataset did not include patients’ diagnoses, a distinction was made between NCPs and CPs on the basis of further prescribed medications that indicate cancer treatment (for example cytostatic drugs). Patients with OA prescriptions between 2011–2015 were divided into five groups: short-term prescriptions up to 3 months (⩽90 days) according to the clinical practice guideline ‘Long-term opioid use in non-cancer pain’ (LONTS) (Häuser et al, 2014), prescriptions over prolonged periods of time between 3–9 months (91–274 days), long-term prescriptions over 9 months with a low dose (⩾275 days and ⩽1 DDD/day), long-term prescriptions over 9 months with a higher dose (⩾ 275 days and >1 to 2 DDD/day) and one group with long-term high-dose prescriptions (⩾275 days and >2 DDD/day). The majority of the evaluations are limited to the group of NCPs. The analyses of initial OA prescriptions included patients with an OA prescription between 2011–2015 and for the first time since 2006, the first year from which data was available.
Results
Between 2011 and 2015, the annual number of SHI patients with OA prescriptions was between 500,000 and 550,000 (Table 1). This correspondents to a prevalence rate of 4.5% in 2011 and 4.8% in 2015 (Buth et al., 2019). Based on co-medications, about 15% of these patients were identified as CPs. In the respective years about half of the NCPs and CPs received OA prescriptions for 90 days or less, with slightly higher prescription rates among NCPs (Table 1). The proportion of CPs with OA prescriptions for 3–9 months was higher than for NCPs (21% vs 16%). Long-term prescription rates of 9 months (or 275 days) or more ranged between 6–7% for NCPs and 7–8% for CPs. Only a small group of patients received prescribed OAs sin doses of more than two DDDs for 9 months or more. Although the small differences between 2011 and 2015 reach statistical significance (Chi2-test, NCP: χ2=326.3, p<0.001, CP: χ2=133.1, p<0.001), apart from an overall increase in the number of patients with OA prescriptions, neither a relevant upwards nor a downwards trend can be identified with regard to period and dose of OA prescriptions.
Groups of non-cancer patients (NCPs) and cancer patients (CPs) according period and dose of opioid analgesic (OA) prescription between 2011–2015.
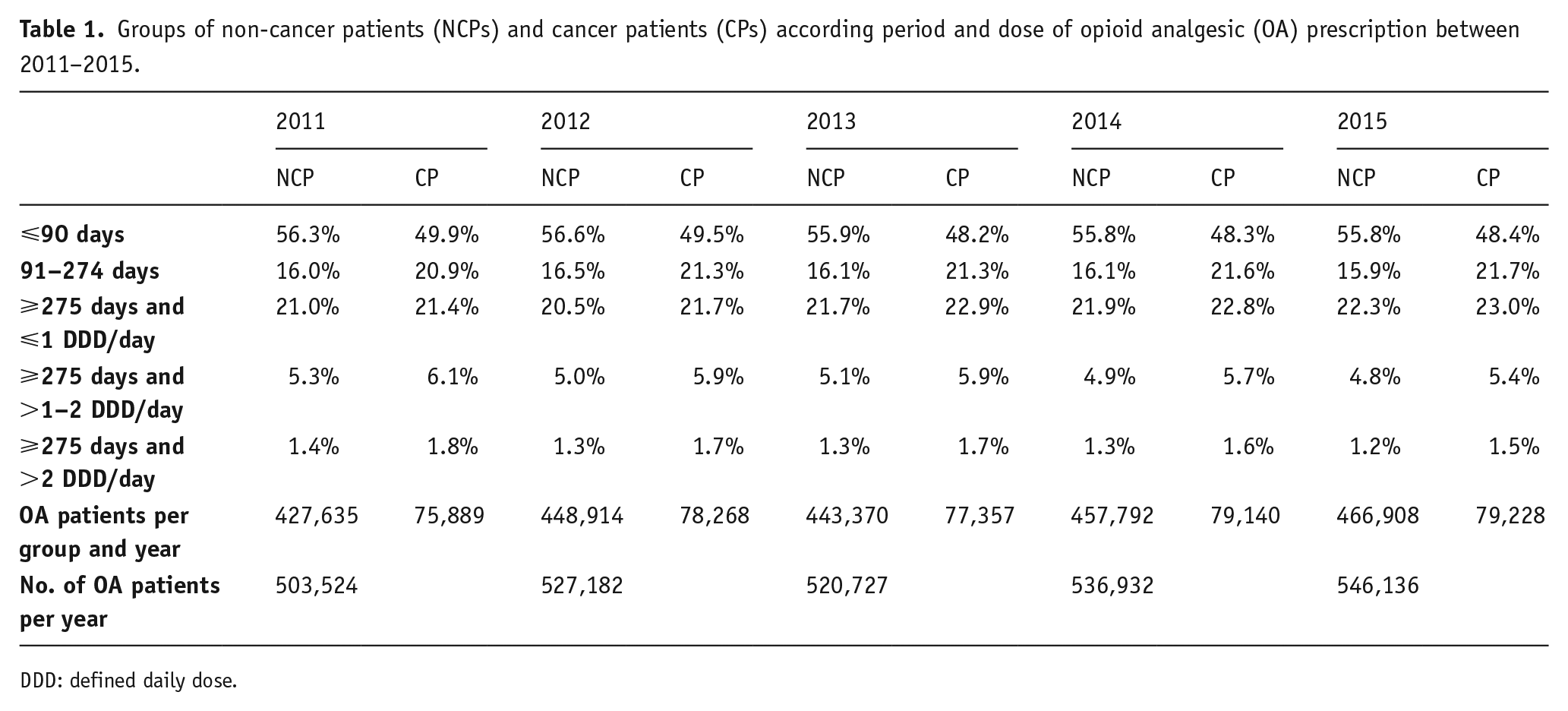
DDD: defined daily dose.
In 2015, the majority (63%; n=275,586) of the NCPs with OA prescriptions were female. Higher proportions of males were found in the group of patients with short-term prescriptions (⩽90 days) and particularly among those with long-term prescriptions for more than 9 months and doses higher than two DDDs (Table 2). The average age of NCP was 66 years. More than half of the NCPs (53%) were 60 years or older, 38% at least 75 years old. The number of long-term prescriptions increased with higher age of the NCPs, but only in the group of patients with low-dose prescriptions below one DDD. Long-term prescriptions of high-doses of OAs (>2 DDD) were most prevalent among the age groups between 45–74 years.
Gender and age of non-cancer patients (NCPs) (n=466,908) in groups according to duration and dose of opioid analgesic (OA) prescriptions in 2015 as well as number of respective prescribers including their medical specialties.
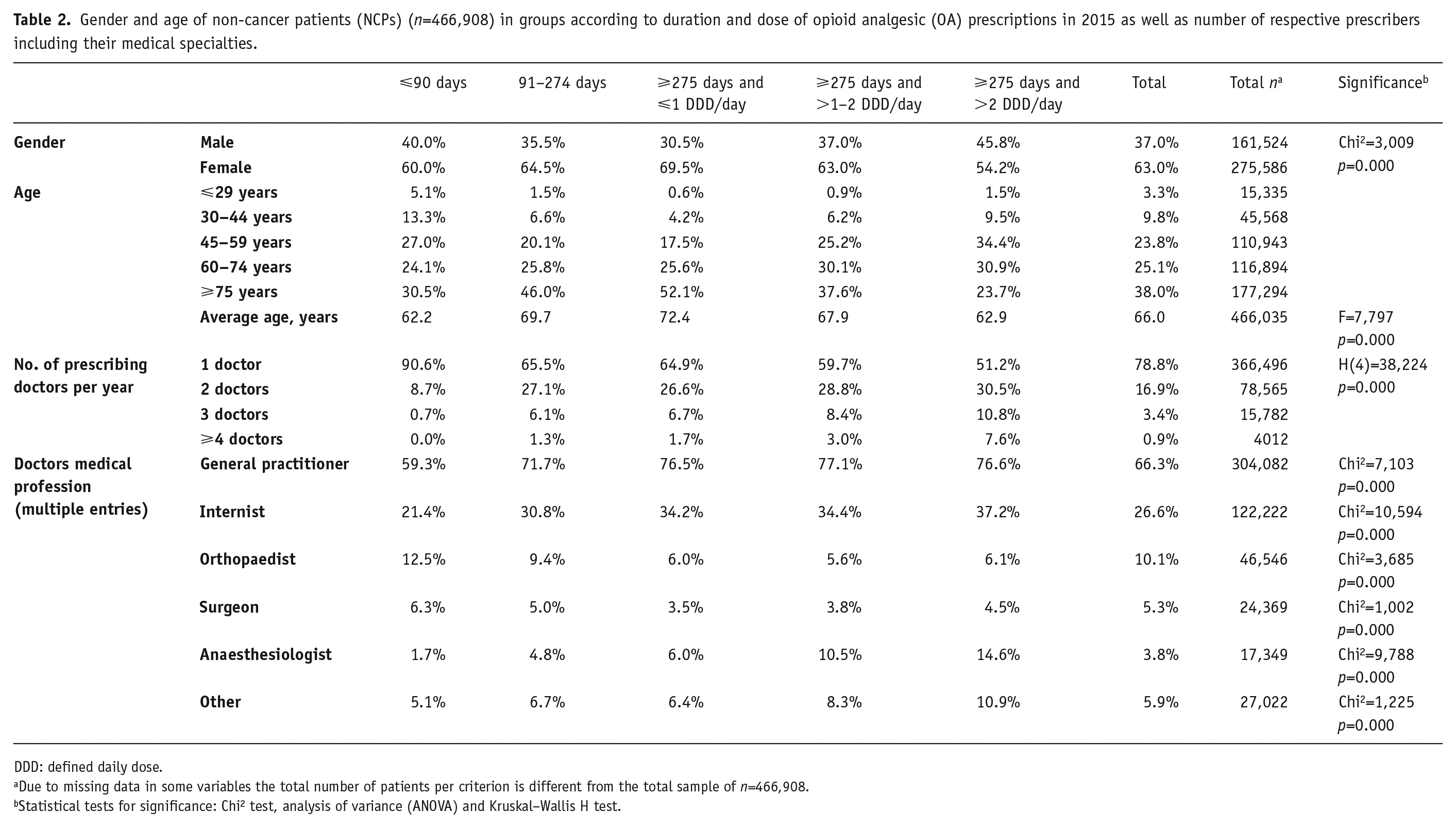
DDD: defined daily dose.
Due to missing data in some variables the total number of patients per criterion is different from the total sample of n=466,908.
Statistical tests for significance: Chi2 test, analysis of variance (ANOVA) and Kruskal–Wallis H test.
Within a year more than three out of four patients (78.8%) received an OA prescription from one physician, 17% from two and a minority of 4.3% from three or more physicians. There was an association between prolonged prescription periods with higher dose levels and an increased number of prescribing doctors. Among NCP patients with long-term OA prescriptions in high doses (>2 DDDs) in 2015, almost half received prescriptions from two or more physicians. The majority of prescribers were general practitioners (GPs) (66.3%) followed by internists (26.6%). The proportion of prescribing GPs and anaesthesiologists was comparatively low among patients with short-term OA prescriptions (⩽90 days), whereas the proportion of prescribing internists and anaesthesiologists was comparatively high in NCPs with long-term and higher OA dose (>1 DDD) prescriptions (Table 2).
It should be emphasized that for the years 2011–2014, patients’ characteristics show a very similar pattern. The distribution of gender (male: 36.2–37.0%), age (mean age: 65.7–66.0 years) and number of doctors (one doctor: 78.8–80.0%) remains stable for the total sample as well as between the specific groups.
The majority of NCPs with a first OA prescription between 2011–2015 received a WHO step II medication. In each of the 5 years, the proportion of WHO step II medications was at least 80% (Table 3). In each year, there was a tendency for the proportion of NCPs with WHO step III medications to increase in long-term and with higher doses. When OAs were prescribed for the first time and in long-term and higher doses they included medications with higher potency more often (WHO step III). However, between 2012 and 2015, the proportion of step II and III medications among NCP with long-term prescription of more than one DDD per day diverged towards less potent step II medications. This is not the case for NCPs with short-term (⩽90 days) OA prescriptions (related to their first prescription). For this group, the proportion of step II and III medication did not change significantly between 2011 and 2015.
Non-cancer patients (NCPs) with first opioid analgesic (OA) prescription according to World Health Organization (WHO) pain ladder (step II and III) in groups according to duration and dose of OA prescriptions from 2011–2015.
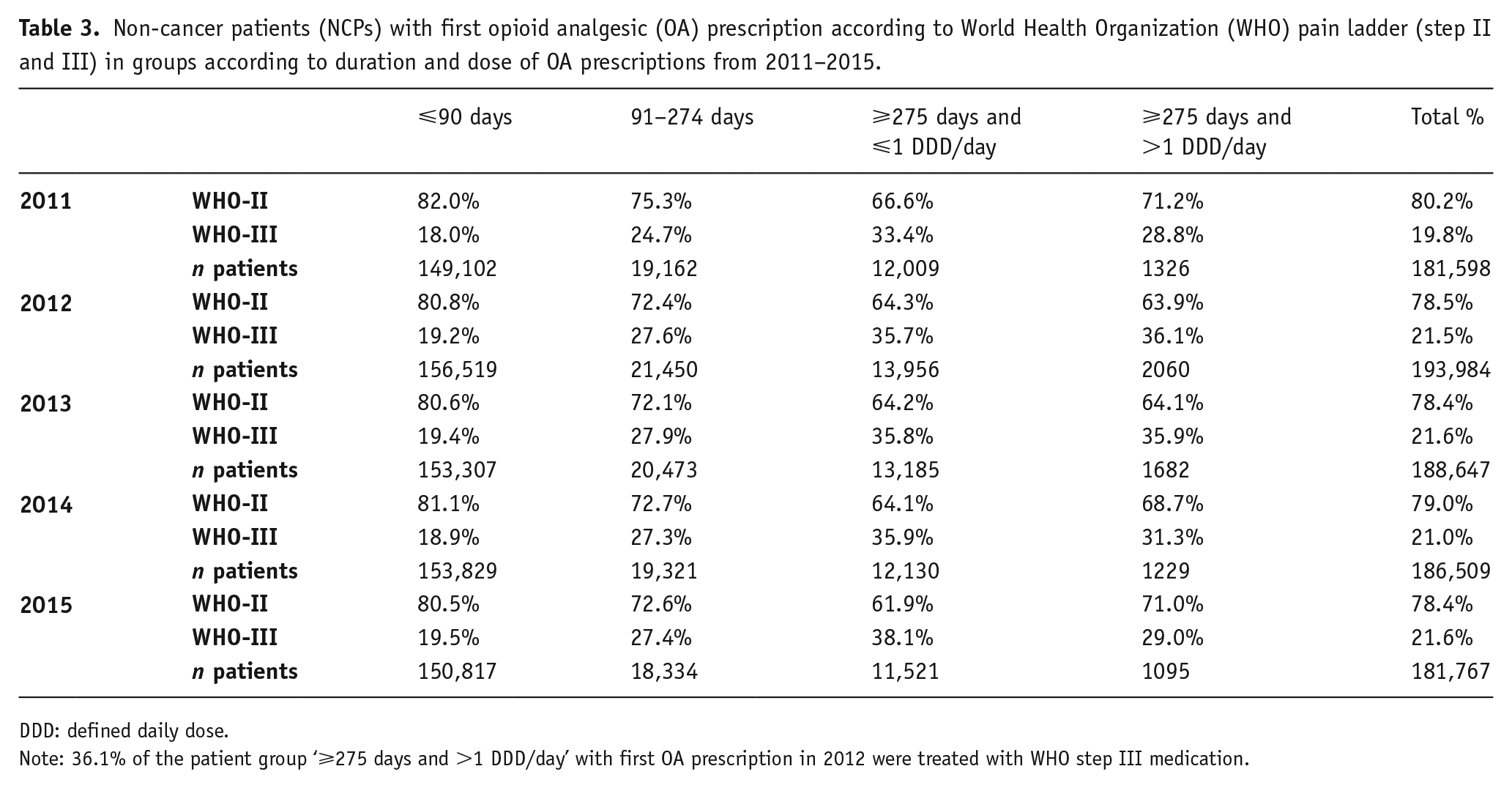
DDD: defined daily dose.
Note: 36.1% of the patient group ‘⩾275 days and >1 DDD/day’ with first OA prescription in 2012 were treated with WHO step III medication.
The WHO step II medications tramadol and tilidine were the most often used medications for OA first-time prescriptions (Table 4). The proportions of both medications changed only slightly over time, whereas the number of tilidine prescriptions increased while tramadol prescriptions decreased between 2011 and 2015. The proportion of oxycodone increased, fentanyl prescriptions slightly decreased in the same period of time. Over the years observed, NCPs with first-time prescriptions for more than 9 months received tilidine and fentanyl more often. In contrast, tramadol was much less frequently used as the first medication for NCPs with long-term prescriptions.
Type of opioid analgesic (OA) for non-cancer patients (NCPs) with first OA prescription by patient groups according to length of prescription and dose from 2011 to 2015.
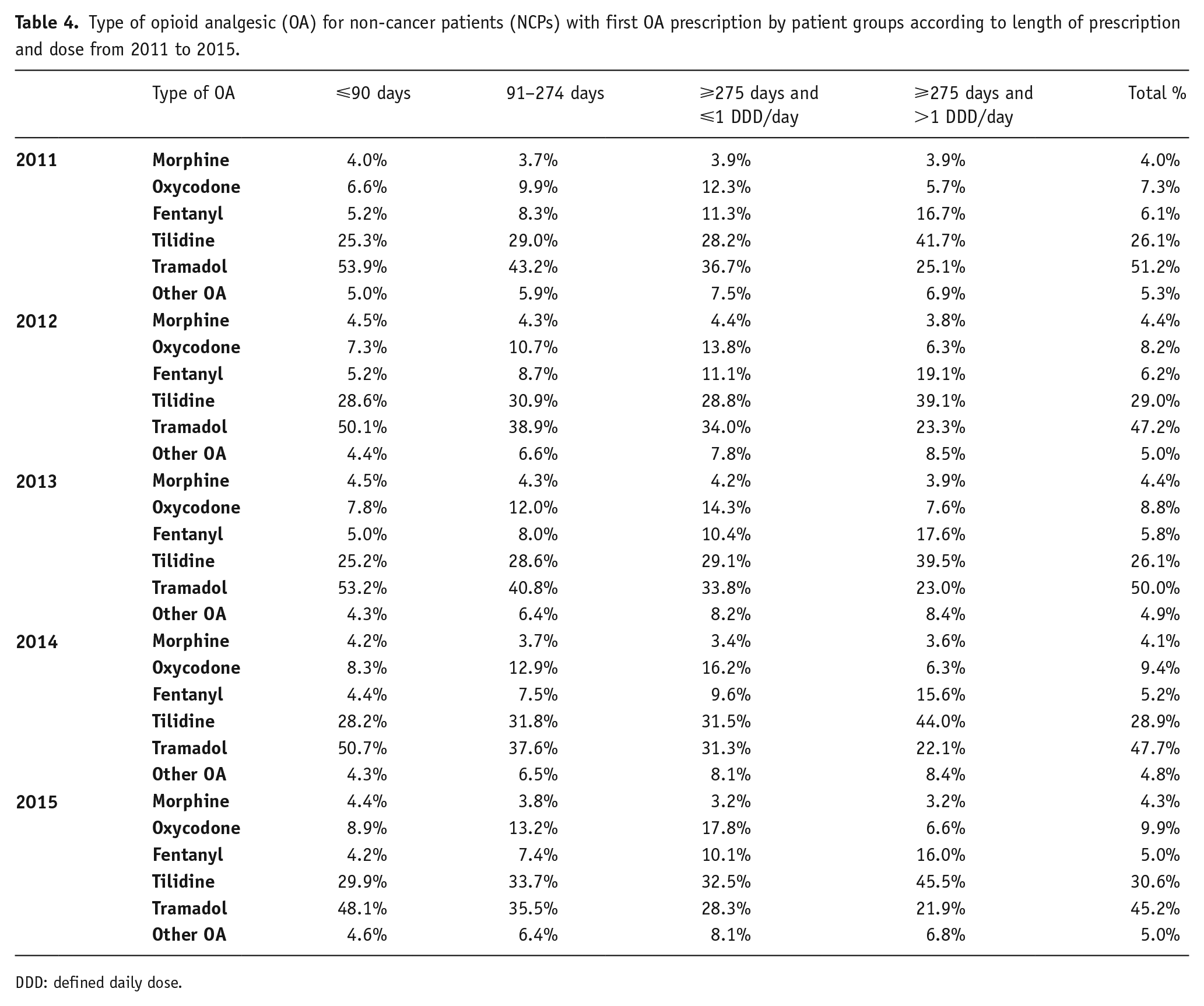
DDD: defined daily dose.
Discussion and conclusions
This study aimed to describe long-term trends in the prescription of OAs between 2011 and 2015 in Northern Germany. In order to identify possible risks of OA abuse, different groups of patients were compared according to duration and dose of OAs.
In line with previous research in the field, our findings confirm that despite a slight increase in the overall number of OA prescriptions, an epidemic spread of OAs cannot be observed for the recent years in Germany (Buth et al., 2019; Rosner et al. 2019; Seitz et al., 2019). Also, between different patient groups (NCPs/CPs, OA prescription periods and doses), we found no substantial changes in the proportions of long-term prescriptions between 2011 and 2015. The statistical significance of the small changes between 2011 and 2015 is due to the huge sample size; however, the relevance of these changes is negligible. Our findings are in contrast to reported increases (of about 4%) of long-term prescriptions among NCPs for the years 2001–2009 (Buth et al. 2019; Schubert et al. 2013). Although Germany is on the second rank after the USA among the countries with the highest average consumption of opioids for pain treatment (International Narcotics Control Board, 2020), the societal conditions and dynamics for an opioid epidemic and overdose death crisis differ between both countries. For instance, most OAs are highly regulated medications and controlled by the German Narcotics Act. In contrast in the USA, OAs (predominantly oxycodone, accompanied by aggressive marketing) were prescribed in high doses and for long periods for pain management (International Narcotics Control Board, 2020). Except for a short fentanyl overdose epidemic in Bavaria between 2011–2014 among persons of whom the majority were known to the police as problem drug users (Sinicina et al., 2017), OA medication does not contribute substantially to the number of drug-related deaths in Germany. In 2018, 7.1% of drug related deaths were caused by synthetic OAs (Drogenbeauftragte, 2019). The stable trend of long-term OA prescriptions in recent years in Germany indicates cautious prescribing behaviour by the treating doctors. This low-risk behaviour largely based on short-term prescriptions and low doses among NCPs and CPs leads to a lower risk of developing an opioid crisis in Germany.
For the assessment of potential OA abuse, the group of NCPs are of particular interest, as these patients potentially develop an OA intake that is unconnected with the basic diagnosis or indication of the OA prescription. With about 85% the majority of patients in this analysis were NCPs. We found differences between groups of NCPs with different periods of time and doses of OA prescriptions: For the year 2015, the proportion of men among NCPs with short-term OA prescriptions and among those with high dose long-term prescriptions was disproportionately high. Further, the average age was the lowest among these two groups. Since younger age and male gender are risk factors for OA abuse (Edlund et al., 2010; Marschall et al., 2016), it can be assumed that among these two prescription groups, the proportion of NCPs with abusive OA use might be increased.
The number of prescribing physicians can be a further indicator for potential abusive OA intake (Young et al., 2019). In the present study a higher number of prescribing physicians was found among long-term users with daily doses of more than one DDD in 2015. However, given the missing diagnoses, no final conclusions can be made on the basis of the data of this study, as the number of doctors prescribing OAs per se is not a precise direct indicator for so-called ‘doctor shopping’. Furthermore, the likelihood of multiple types of physicians being involved – especially anaesthesiologists who are the specialists in pain management – increases with the number of prescriptions. Indeed, receiving OA prescriptions from several doctors (for non-medical use) appears to play a less important role compared with benzodiazepines (BZD) or Z-drug prescriptions (Verthein et al., 2019). Also, Hulme et al. (2018) found in their meta-analysis that the main source of pharmaceutical opioids for non-medical use are friends and family and that illegitimate practices such as ‘doctor shopping’ are mainly uncommon. In contrast to this, Ponté et al. (2018) have calculated a doctor shopping indicator (DSI) to compare patients with OA and those with BZD prescriptions and concluded that OA and BZD have a comparable DSI. However, the highest ‘doctor shopping’ quota was found for the BZD flunitrazepam, and the overall prescription rates of analgesics appears to be higher in France than in Germany (Jacob and Kostev, 2018).
The vast majority of SHI-insured NCPs received OA prescriptions from GPs, the second largest group of physicians in Germany (11.1% of all physicians) after internists (14.0%) (Bundesärztekammer, 2018). Both, GPs and internists were disproportionately frequent long-term prescribers which might be related to the fact that the present study was not able to separate sufficiently NCPs from CPs (see in the following). Among patients with short-term OA prescriptions, orthopaedists account for a relatively high proportion (12.5%) of prescriptions, suggesting that they often prescribe OAs temporarily and as intended (e.g. for musculoskeletal pain). So far, little research has been done on different professional medical groups prescribing OAs (Marschall and L’hoest, 2011; Tölle et al., 2019). The data presented in this study give an almost representative insight for Northern Germany for the first time.
WHO step II medications are predominately used for first-time prescriptions to NCPs. Overall, the proportions of step II and step III medications changed very slightly over the period of 5 years. However, one-fifth of all NCPs treated with OAs received a highly potent step III medication as a first-time prescription. This rate was higher compared with German data for the year 2009 (16%) (Schubert et al., 2013). In particular with regard to fentanyl, which is primarily used in the form of patches, this prescription practice can be regarded as critical. First, as first-time admission to opioid-naïve patients it is only recommended if oral use of painkillers is not feasible (Garbe et al., 2012). Second, as we found in this study, among long-term users with more than one DDD per day the proportion of NCPs with first-time fentanyl prescription was substantially increased. With regard to the increased proportion of first-time tilidine prescriptions in this group, further research is needed to clarify whether the findings are related to the particular addictive potential or the medical treatment concept of the prescribing physicians.
For the first time, the present study provides comprehensive patient-related analyses of OA prescriptions in Germany over a period of 5 years. The strength of this study lies in the almost complete coverage of the prescription data from all SHIs across four German federal states. This also results in a relevant limitation: prescriptions on private recipes as well as all privately insured persons – about 11% of all insured persons in Germany – were not included in the study. However, the amount of SHI-insured persons switching to private OA prescriptions might be less relevant as for BZD and Z-drugs. Hoffmann et al. (2012) found lower OA prescription rates and smaller quantities (DDD) in the southern federal states of Germany compared with prescriptions in the northern and eastern regions. However, as this study is only based on data from one health insurance company and for the year 2011 it remains unclear whether these regional differences apply to all German SHI patients. In order not to overload the results we omitted stratified analyses by sex and age groups. Findings from studies by Schubert et al. (2013) and Jacob and Kostev (2018) as well as our own studies (Buth et al., 2019) indicate that women and older patients have higher rates and longer periods of OA (and pain medication) prescriptions.
Treede and Zenz (2015) pointed out that the proportion of opioid prescriptions for non-cancer pain based on data from 2010 is 77%, indicating the need to update the S3 clinical guideline ‘Long-term use of opioids for chronic non-tumour related pain’. A further limitation of our study is that the identification of CPs was only based on concomitant medication and/or on prescriptions by oncologists. Given this, it might be the case that the group of NCPs also included CPs.
Treede and Zenz (2015) also point out that the ‘magic’ 3-month limit was mainly defined on the requirements of regulatory authorities rather than on clinical effectiveness. Prolonged pain treatments with OAs can therefore have a therapeutic justification despite this 3-month rule.
Another limitation is the immanent assumption that the prescribed OA is actually taken. Physicians often recommend lower levels of intake, or patients stop prematurely or take lower doses (Knopf and Grams, 2013). The assumed OA intake included in this study could therefore be overestimated.
Footnotes
Acknowledgements
The authors would like to thank Ommo Meiners from NARZ/AVN for technical support and advice on the preparation and generation of the database. The author contributions were as follows: UV and MSM conceptualized and designed the study. BS, SB and AD made a substantial contribution to the concept, acquisition of data, the analyses and interpretation. UV drafted the manuscript. BS, MSM and SB commented and revised it critically for important intellectual content. All authors approved the version to be published.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: UV received speaker’s honoraria and travelling expenses from Mundipharma GmbH and received travelling expenses from CAMURUS GmbH. BS received an unrestricted educational grant and travelling expenses from CAMURUS GmbH. SB, AD and MSM declare no conflicts of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The underlying research project was funded by the Innovation Committee of the Federal Joint Committee (Gemeinsamer Bundesausschuss, G-BA) (project No. 01VSF16049).
