Abstract
Objective:
To assess whether concomitant antipsychotic treatment has an influence on the antidepressive effects of repetitive transcranial magnetic stimulation (rTMS).
Methods:
We analyzed severity of depression before and after treatment with rTMS in a sample of 299 depressed in- and outpatients in retrospect in relation to treatment with drugs for psychosis. The sample consisted of real-life patients in a tertiary hospital. We ran group contrasts between the group taking and the group not taking drugs for psychosis, testing for differences in Hamilton Depression Rating Scale (HDRS). Effect sizes for HDRS group contrasts were reported as Cohen’s d and number needed to treat (NNT) calculated from d. To control for group differences we repeated the Student t-tests for the change in the HDRS using analysis of covariance including confounding variables.
Results:
Depressed patients taking drugs for psychosis showed significantly less amelioration of depressive symptoms after rTMS treatment as measured by absolute and relative change in HDRS with small effect size or NNT of 4.5 to 8.4, respectively. Controlling significant group contrasts revealed that the effect of taking drugs for psychosis does not depend on age, number of applied TMS pulses, type or severity of depression at baseline, comorbidities or differences in the intake of additional medication.
Conclusion:
Drugs for psychosis attenuate antidepressive effects of rTMS independently of confounding factors. The effect is statistically significant and of potentially great clinical importance. The exact mechanism remains to be elucidated and further studies are warranted.
Introduction
Repetitive transcranial magnetic stimulation (rTMS) is now well established as an effective and generally safe treatment for depressive illness as demonstrated by multiple clinical trials (Berlim et al., 2014; Lefaucheur et al., 2014; McClintock et al., 2018; Rossi et al., 2009) and is one of the main treatment options next to medication and psychotherapy.
The question of how concomitant medication may influence the effects of rTMS is of great clinical significance. Most depressed patients treated with rTMS are on psychiatric medication, as rTMS is currently rarely used as first-line or monotherapy.
Drugs for psychosis can be used in depression with psychotic features, but are not infrequently also used when such features are not clearly present as an augmentation strategy (Nelson and Papakostas, 2009). Therefore, being prescribed these drugs in addition to “classical” antidepressive treatment might be seen as indication of more severe illness or perhaps a genuinely different type of condition. Whether psychotic depression is simply a more severe and advanced form of non-psychotic mild, moderate and severe depression on a continuum or whether it represents a different entity has been and is a matter of debate (Rothschild, 2013). There is evidence that rTMS is less effective in depression with psychotic features (Grunhaus et al., 2000). Another group of depressed patients commonly taking drugs for psychosis are those with known bipolar disorder who are taking the drug as a drug for relapse prevention and in whom the physician has decided to continue the treatment during the depressed phase.
Several preclinical studies indicate that there are mechanisms by which drugs for psychosis might interfere with certain effects of rTMS treatment. These substances have complex, not yet fully understood effects on cortical excitability and specific neurophysiological, TMS-derived parameters, such as intracortical inhibition and facilitation (Ziemann et al., 1997) and, among others, the cortical silent period (CSP) (Frank et al., 2014; Hasan et al., 2013) possibly modulating cortical “receptivity” for rTMS stimuli and long-term neuroplastic effects.
Cho and Strafella (2009) and Strafella et al. (2001) reported that rTMS of prefrontal cortical areas can modulate dopamine release in specific brain areas in healthy volunteers. In the cited paper of Cho et al., left-, but not right-sided stimulation of the dorsolateral prefrontal cortex (DLPFC) modulated dopamine release in the cingulate and orbitofrontal cortex, suggesting distinct regional effects. rTMS not only of prefrontal, but also of cortical motor areas could also be shown to induce striatal dopamine release in healthy volunteers (Strafella et al., 2003).
Since dopamine is a neurotransmitter implied to be of great importance in the neurobiology of depression (Belujon and Grace, 2017), it seems plausible that blocking dopamine receptors by drugs for psychosis might attenuate positive effects of rTMS, the latter of which could at least in part be mediated by increased dopamine release.
Fittingly, it could be shown in the case of the motor cortex that excitatory and inhibitory effects of theta burst stimulation, a form of rTMS, can be suppressed in healthy volunteers by medicating them with 400 mg of sulpiride, a clinically relevant dose of a common drug for psychosis (Monte-Silva et al., 2011).
Seemingly contrary to most of these findings, a recent study by Schulze et al. seems to demonstrate a trend for patients on drugs for psychosis actually trending to benefitting more from theta burst TMS of the DMPFC (Schulze et al., 2017), which the authors admitted was surprising to them.
Our aim was thus to explore whether taking drugs for psychosis while being treated with rTMS might have an influence on the effectiveness of the latter in our naturalistic patient sample. As already clearly stated by Schulze et al. in their paper, in the light of the preclinical evidence pointing to detrimental effects of drugs for psychosis, prospective randomized trials on the subject would be difficult to justify, increasing the importance of retrospective naturalistic analysis, despite its obvious limitations.
Methods
Based on a retrospective cohort of patients with depression who were treated with rTMS at the Center for Neuromodulation at Regensburg University Hospital (Germany) between 2002 and 2017, a sample of 299 patients could be selected for this analysis. Patients gave written informed consent to treatment. The retrospective analysis of clinical data was approved by the local ethics committee of the University of Regensburg (16-104-0223).
The inclusion criteria were: naïve to rTMS (only the patient’s first treatment with rTMS was considered), diagnosis of depression according to ICD-10 of F31-F33, a completed Hamilton Depression Rating Scale (HDRS) at the beginning and at the end of the rTMS treatment, and absence of a serious somatic illness. Both in- and outpatients were included. For this analysis we included 299 patients (117 (39%) taking no drugs for psychosis). Both groups did not differ with respect to sex, resting motor threshold, stimulation intensity, and number of treatment sessions (Table 1), but differed with respect to age, baseline depressive symptoms (HDRS), number of pulses per session, type, and severity of depression on a significance level of 20% (Table 1). Both groups showed differences (all p-values < 0.2) with respect to the intake of medication, i.e. amitriptylinoxide, bromazepam, chlorprotixene, citalopram, desipramine, diazepam, imipramine, lamotrigine, lithium, lorazepam, nortriptyline, oxazepam, reboxetine, tranylcypromine, trazodone, trimipramine, venlafaxine, and zolpidem. Different study protocols were used – most were treated with high-frequency protocols over the left DLPFC (n = 255). Four patients were stimulated on the right DLPFC, 12 on the medial prefrontal cortex and 28 were stimulated on both the left and right DLPFC in consecutive order.
Characteristics of patients with depression taking vs. not taking drugs for psychosis.
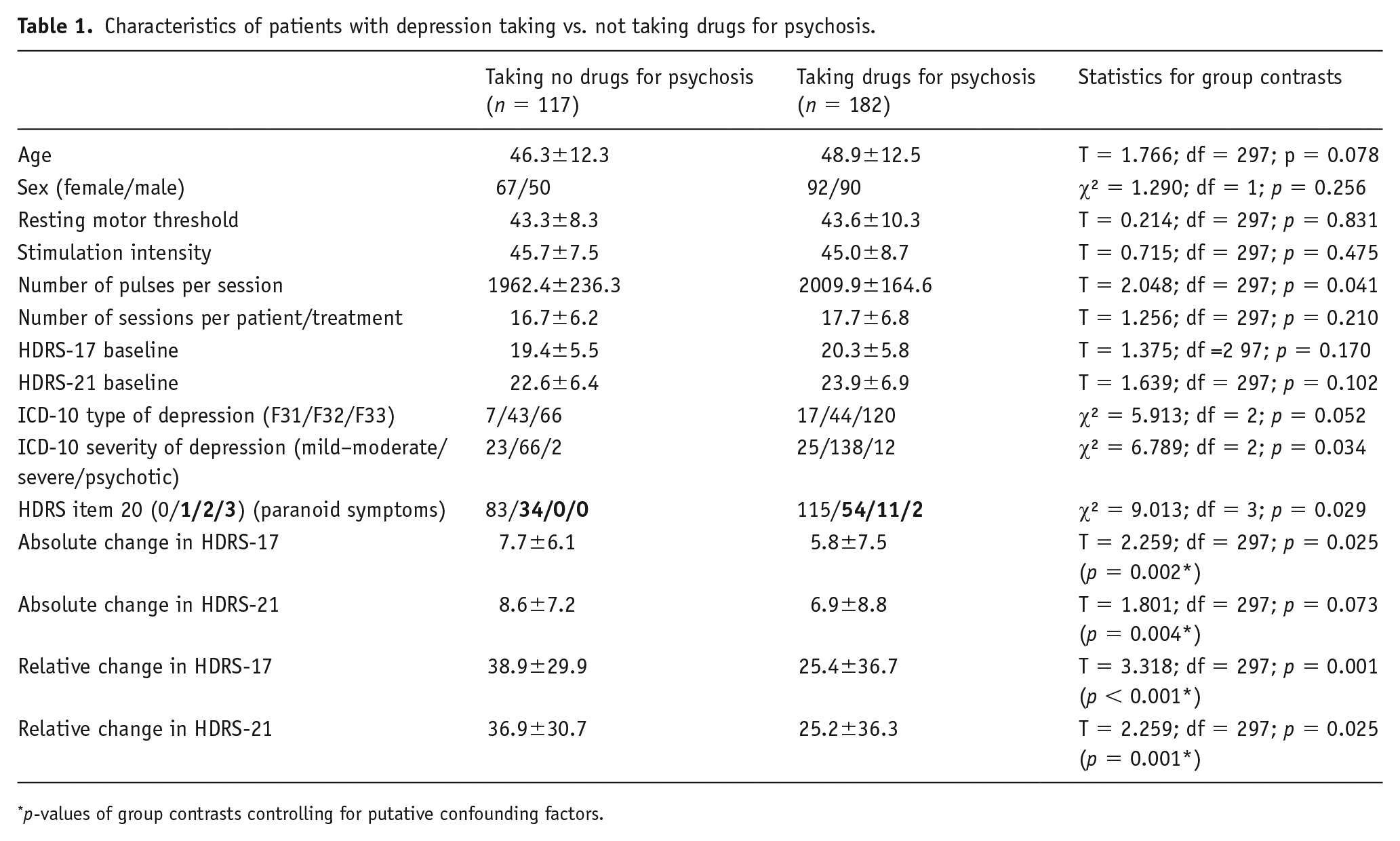
p-values of group contrasts controlling for putative confounding factors.
All data were analyzed using SPSS (IBM Corp., USA; Version 24.0.0.0). The significance level was set at p<0.05 for group contrasts testing for differences in HDRS measures without correcting for multiple comparisons. The significance level was set to p < 0.20 for group contrasts testing for no differences in descriptive variables with the aim to control type II error. For group contrasts, we used Student t-tests or chi-square-test of independence depending on the scales of measurement. Effect sizes for HDRS group contrasts were reported by Cohen’s d (Cohen, 1988) and number needed to treat (NNT) calculated from d (Cohen, 1988; Lenhard, 2016).
To control for group differences we repeated the Student t-tests for the change in the HDRS using analyses of covariance (ANCOVA) including confounding variables. That means that four student T-tests were repeated with between-subjects factor group (group taking and the group not taking drugs for psychosis) and the covariates age, number of applied TMS pulses, type or severity (according to ICD-10 and HDRS baseline values) of depression at baseline, frequency of paranoid/psychotic symptoms at baseline (according to item 20 of the HDRS), presence of comorbidities and intake of the medication lithium, lorazepam, reboxetine, venlafaxine, amitriptylinoxide, bromazepam, citalopram, chlorprotixene, desipramine, diazepam, imipramine, lamotrigine, nortriptyline, tranylcypromine, trazodone, trimipramine, zolpidem, and oxazepam. The dependent variable was absolute and relative HDRS change (for the 21 and the 17 items version) from pre to post rTMS.
To investigate the association of the drug dose with response to rTMS we calculated the haloperidol equivalents of the summarized total dose of drugs for psychosis over the course of the treatment. We used the defined daily dose (DDD) method, which was comprehensively discussed, for example, Leucht et al. (2016), with the advantages of being available for all the drugs present in our sample. For dose conversion, the calculator derived from this method presented by Leucht et al. online was used (http://www.cfdm.de/media/doc/Antipsychotic%20dose%20conversion%20website.xls). As three outliers were present in the scatter plot we used non-parametric Spearman correlations of these summarized equivalents with the four measures of HDRS change.
Results
Comorbidities are shown in Table 2.
Frequency of comorbidities.
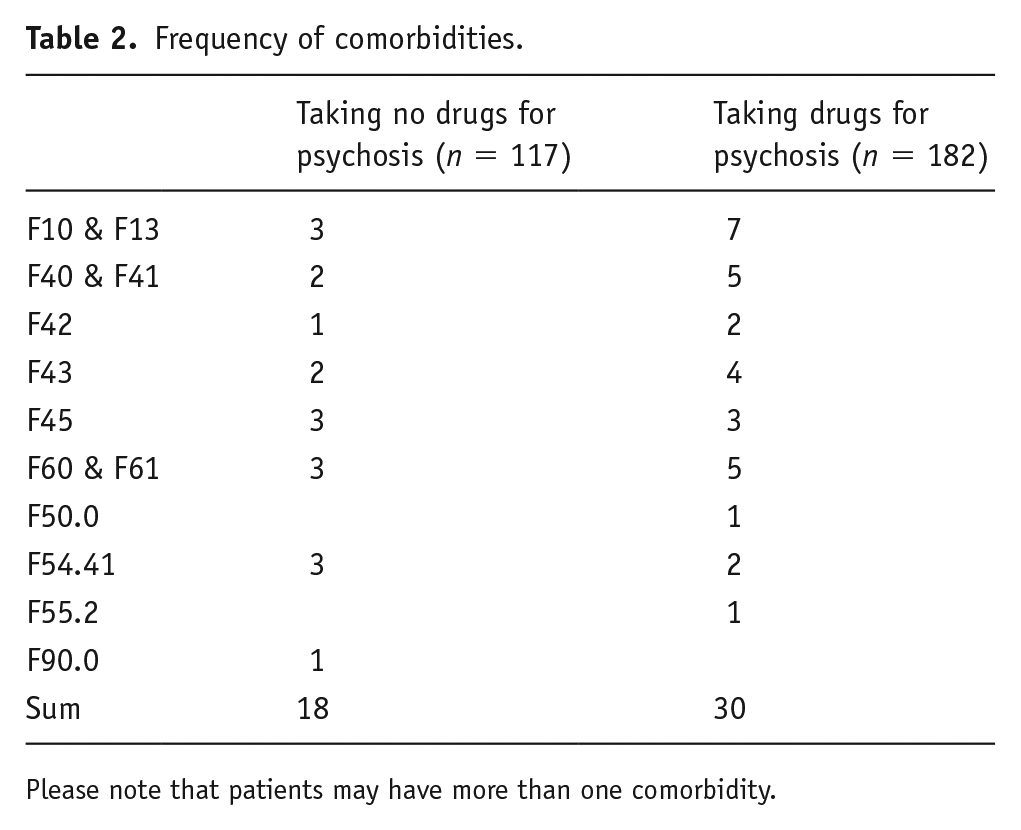
Please note that patients may have more than one comorbidity.
Overall, patients showed an amelioration of symptoms after rTMS treatment with reduced efficacy in the group of patients taking drugs for psychosis as indicated by significantly less amelioration of depressive symptoms as measured by absolute change in HDRS-17 and relative change in HDRS-17 and -21 and near significant less amelioration in absolute change of HDRS-21 (Table 1).
Accordingly, number of responders (decrease in HDRS sum score of at least 50%) were higher for group taking no (HDRS-17: 38.5%; HDRS-21: 35.9%) in contrast to the group taking drugs for psychosis (HDRS-17: 26.9%; HDRS-21: 25.8%) (HDRS-17: χ² = 4.399; df = 1; p = 0.036; HDRS-21: χ² = 3.457; df = 1; p = 0.063). In addition, number of remitters (HDRS-17 and HDRS-21 sum score below 11 or 9, respectively) were higher for group taking no (HDRS-17: 35.9%; HDRS-21: 37.6%) in contrast to the group taking drugs for psychosis (HDRS-17: 20.9%; HDRS-21: 21.4%) (HDRS-17: χ² = 8.197; df = 1; p = 0.004; HDRS-21: χ² = 9.296; df = 1; p = 0.002). That means that there are at least 10% more responders and remitters in the group of patients without in contrast to the patients with drugs for psychosis in their medication regimen.
Effect sizes were small for group contrasts (0.212 ⩽ d ⩽ 0.403; 8.4 ⩾ NNT ⩾ 4.5). Controlling significant group contrasts by repeating the group contrasts for the HDRS scores using ANCOVAs revealed again significant group effects highlighting that the effect of taking drugs for psychosis does not depend on age, number of applied TMS pulses, type or severity of depression or frequency of psychotic symptoms at baseline, presence of comorbidities and differences in the intake of additional medication. Table 3 shows an overview of the drugs for psychosis taken in this sample. For this sample correlations of the summarized haloperidol equivalents with treatment response show a significant association, i.e. the higher the dose the lower the treatment response (0.176 < r < 0.187; n = 182; 0.011 < p < 0.017). On a single item level, change in the items 1, 2, 10, and 20 turned out to be significantly different between both groups with superior amelioration in the group of patients without drugs for psychosis for items 1 (depressed mood), 2 (feelings of guilt), and 10 (anxiety – psychic) and with superior amelioration in the group of patients taking drugs for psychosis for item 20 (paranoid symptoms) (Figure 1).
Descriptive statistics with respect to intake of drugs for psychosis.
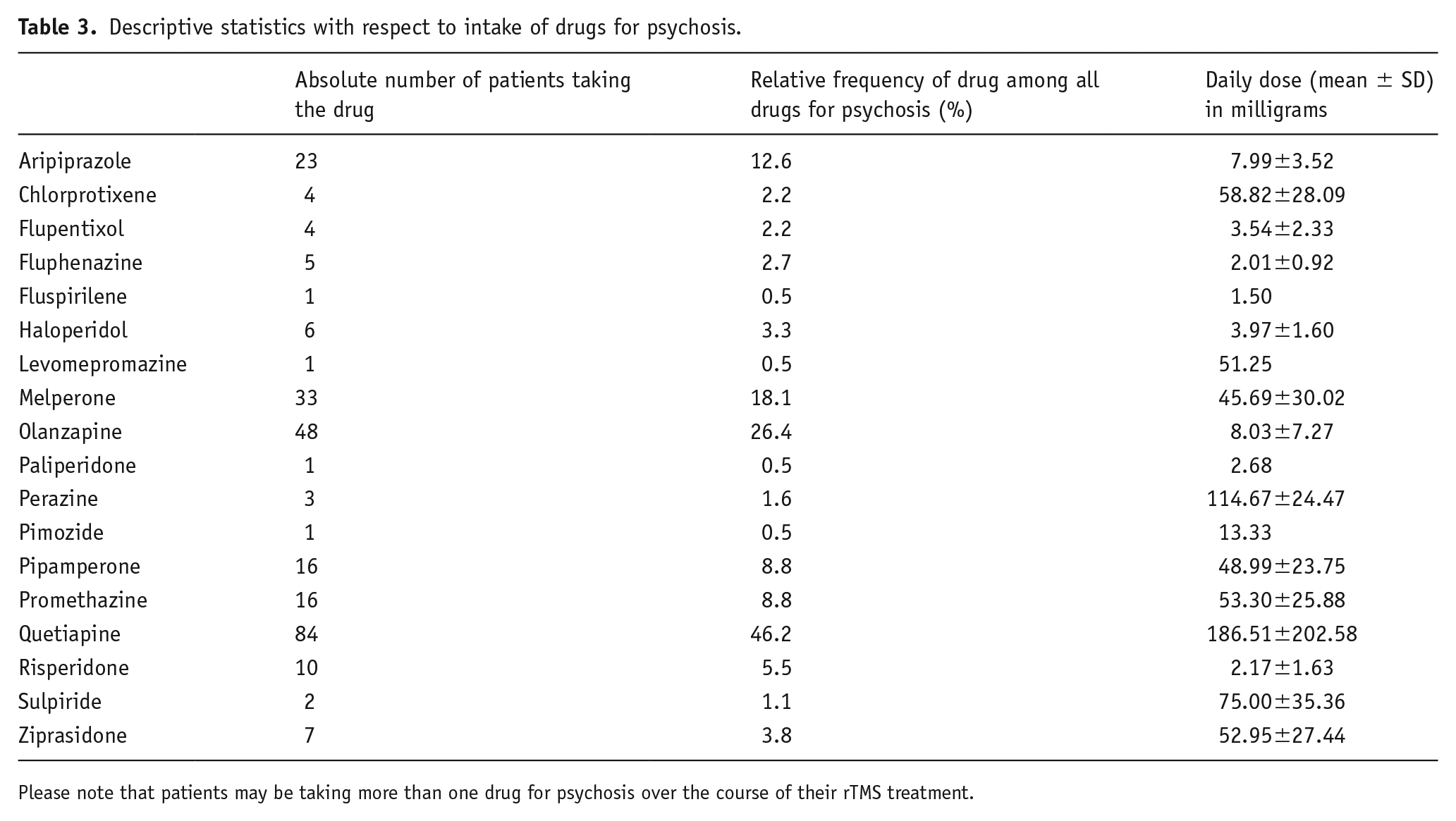
Please note that patients may be taking more than one drug for psychosis over the course of their rTMS treatment.
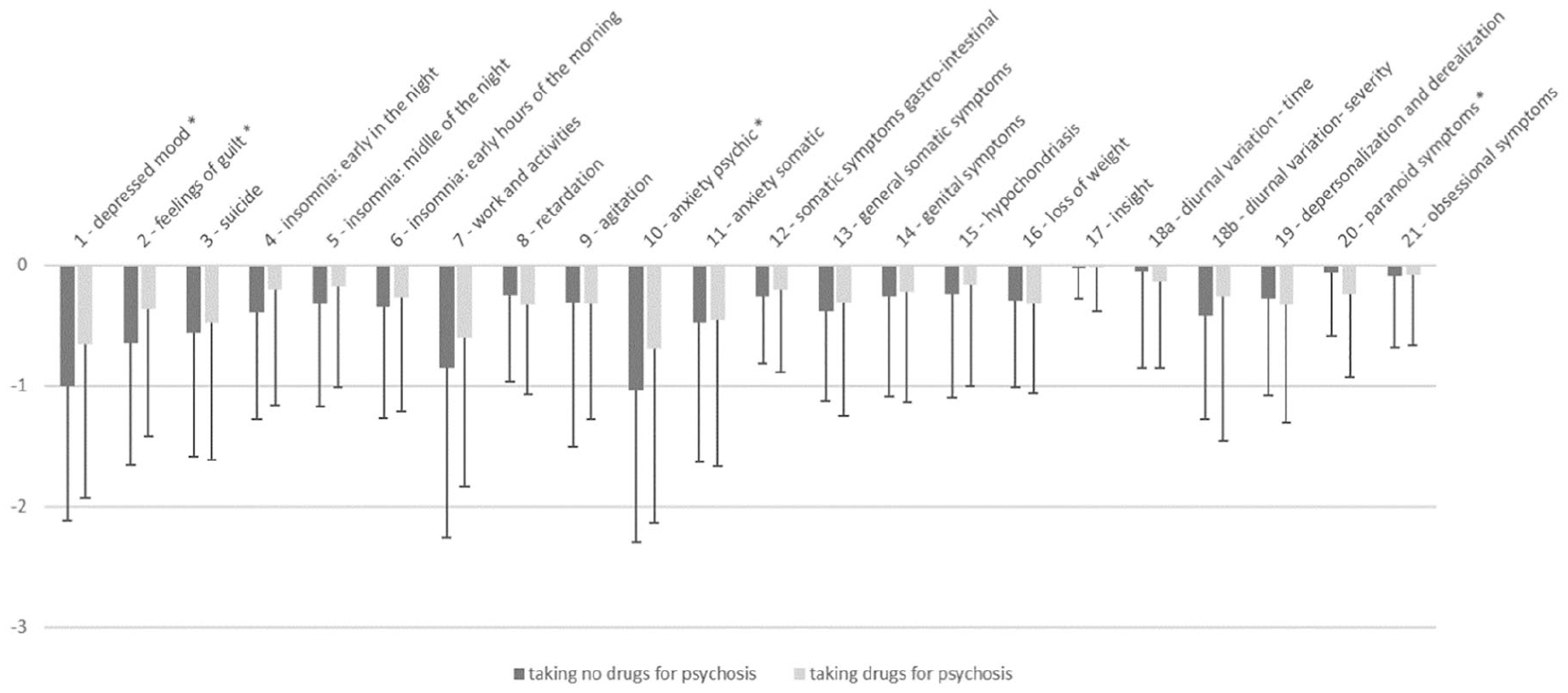
Change from pre- to post-intervention for the single items of the Hamilton Depression Rating Scale (mean ± SD).
Discussion
In our comparatively large patient sample, patients taking drugs for psychosis showed significantly less amelioration of depressive symptoms as measured by HDRS scores and response and remission rates. In our opinion, three main factors might explain the obtained results.
First, it is possible that there is a specific mechanism by which drugs for psychosis block the beneficial effects of rTMS.
Considering the preclinical work, it seems possible that at least a part of the antidepressive effects of rTMS might be traced back to increased dopaminergic activity in specific regions of the brain, and that these drugs hinder this mechanism, most likely by blocking D2-dopaminergic receptors (D2R).
In this case, a higher dose of a drug for psychosis or a more potent one should cause even more attenuation. When calculating haloperidol equivalents by the DDD method for total drug dose over the rTMS treatment course, we found a significant association, not proving but adding clues that such a mechanism might exist.
Further prospective work under better controlled conditions would be crucial to further investigate this possibility.
Secondly, drugs for psychosis may have unwanted side effects that show up on the Hamilton scale as indicators of depression, thereby exerting “pro-depressive” effects that show up in our sample and are independent of effects on rTMS treatment. Treatment with these substances may produce side effects such as anhedonia, tiredness and lack of energy, sexual dysfunction and weight gain. Some of these may actually lead to increased scores in depression ratings such as the Hamilton score, thereby masking putative positive effects of rTMS. On the other hand, some of these side effects may even be rated as “improved” depression scores (e.g. weight gain or no further weight loss, faster onset and longer duration of sleep). Also, it has to be considered that drugs for psychosis are actually used as an augmentation strategy for depressive illness, thus limiting the strength of the “pro-depressive” argument against these drugs, and that we clearly see better improvement of paranoia in the group treated with antipsychotics.
Third and last, it is possible that those patients who are preferably treated with drugs for psychosis by their clinicians suffer from a different disease entity other than “common” depression that has a different biological basis and responds less readily to rTMS. In this hypothesis, the medication has no relevant effect on the outcome by itself but merely serves as an indicator of another type of depression, which then again by its very nature responds less readily to the stimulation treatment (and perhaps other treatments, as well).
In the same way, these patients might not have another, but more treatment-resistant disease; as a limitation of our retrospective analysis, there was no systematic data on number of failed previous treatment courses.
However, the attenuating effect of drugs for psychosis appeared to be independent of baseline measures as shown by covariant analysis. On the other side, standard rating tools might not be sufficient to identify distinct biological phenotypes.
Our results suggest that patients with drugs for psychosis benefitting less from rTMS might not, or not only, be due to the nature and severity of their illness but perhaps may relate to the drugs these patients are commonly prescribed.
Regardless of the underlying mechanisms, our results are in contrast to the findings of Schulze et al. (2017). The authors of said paper point out several possible explanations for their results in the discussion. With only 29 patients on drugs for psychosis medication, their sample was considerably smaller than ours was. They also point out to the implementation of DMPFC stimulation and that the effects of DLPFC stimulation, with which the majority of our patients were treated, might yield different results. There is evidence that rTMS of the DLPFC modulates brain dopamine release in a topographically distinct manner, that is, left-sided stimulation has a different effect than right-sided stimulation (Cho and Strafella, 2009; Ko et al., 2008), and perhaps such effects might also be seen for DLPFC versus DMPFC stimulation, offering another explanation for our results distinct from those of Schulze et al. In addition, they point out that “classical” protocols such as stimulation with 1 Hz or 20 Hz might differ in their being affected from drugs for psychosis as compared to theta burst stimulation.
We must also consider the limitations of a clinical instrument like the Hamilton score for accurately depicting the severity and course of a complex biological process like “depression.” As can be seen from the data, different items respond differently to the co-therapy with drugs for psychosis.
Limitations of our study include the naturalistic setting with heterogeneity of exact psychiatric diagnoses (uni- and bipolar depression), type and variety of drugs used and concomitant use of other, mainly antidepressive drugs. However, this might also be seen as an advantage as it reflects the reality of everyday clinical practice.
Compliance with drug intake may have been an issue with both patient groups; however this is a general problem with psychiatric drug studies, especially if drugs for psychosis are involved. Due to the nature of a retrospective analysis, we have no systematic blood level testing data available.
As an additional limitation, there was no sham rTMS treatment, which could have provided further valuable clues as to whether the attenuating effect of antipsychotics is a dependent or independent of rTMS.
In conclusion, further studies are warranted to provide insight into this highly relevant area of interest. If further evidence accumulates that drugs for psychosis do indeed attenuate rTMS effects, then patients on that type of medication should perhaps be screened more carefully for the necessity of receiving it before undergoing rTMS treatment or even be excluded from receiving it for reasons of diminished hope of success.
Conclusion
Drugs for psychosis attenuate antidepressive effects of rTMS in depressed patients in this retrospective naturalistic analysis. The exact mechanisms remain to be elucidated. We suggest that either (a) rTMS exerts its antidepressive effects via increased dopaminergic activity which is blocked by drugs for psychosis, (b) that drugs for psychosis have side effects that interfere with or counteract antidepressive effects of rTMS, (c) that depressed patients that are treated with drugs for psychosis might suffer from a distinct disease entity less responsive to rTMS, a combination of all three explanations being possible and not unlikely. Further studies on this topic are warranted considering its clinical significance and possible practical consequences.
Footnotes
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Tobias Hebel, Mohamed Abdelnaim, Markus Deppe and Martin Schecklmann have no interests to disclose.
Berthold Langguth receives royalties from Springer for edited books, received research grants from the company Neuromod as well as from national (DFG, BMBF) and European institutions (EU), received speaker’s honoraria and advisory panel payments from Neuromod, Desyncra, Decibel Tx, and Servier.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
