Abstract
Compulsive and perseverative behaviour in binge-eating, female, Wistar rats was investigated in a novel food reward/punished responding conflict model. Rats were trained to perform the conditioned avoidance response task. When proficient, the paradigm was altered to a food-associated conflict test by placing a chocolate-filled jar (empty jar for controls) in one compartment of the shuttle box. Entry into the compartment with the jar triggered the conditioning stimulus after a variable interval, and foot-shock 10 seconds later if the rat did not leave. Residence in the ‘safe’ compartment with no jar did not initiate trials or foot-shocks. By frequently entering the chocolate-paired compartment, binge-eating rats completed their 10 trials more quickly than non-binge controls. Binge-eating rats spent a greater percentage of the session in the chocolate-paired compartment, received foot-shocks more frequently, and tolerated foot-shocks for longer periods; all consistent with compulsive and perseverative behaviour. The d-amphetamine prodrug, lisdexamfetamine, has recently received US approval for the treatment of moderate to severe binge-eating disorder in adults. Lisdexamfetamine (0.8 mg/kg po [d-amphetamine base]) decreased chocolate consumption by binge-eating rats by 55% and markedly reduced compulsive and perseverative responding in the model. These findings complement clinical results showing lisdexamfetamine reduced compulsiveness scores in subjects with binge-eating disorder.
Introduction
Binge-eating disorder (BED) was included in the American Psychiatric Association Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition in 2013 (American Psychiatric Association, 2013), and is defined as ‘eating, in a discrete period of time, an amount of food larger than most people would eat in a similar amount of time and under similar circumstances’. Patients often eat large amounts of food when they are not physically hungry. They may eat more rapidly than normal or on their own because they feel embarrassed, disgust, shame, guilt and loss of control following a binge-eating period. Characteristically, binge eating is not associated with inappropriate compensatory purging (de Zwaan et al., 1994; Masheb et al., 1999; Vannucci et al., 2013).
BED is characterised by the compulsive, repetitive and excessive consumption of highly palatable foods (‘binges’). Although BED can be associated with obesity (Fairburn et al., 2000; Goldschmidt et al., 2011; Hudson et al., 2007), a significant minority (17–30%) of subjects have normal body weights (body mass index (BMI) 18.0-25 kg/m2) (Fairburn et al., 2000; Goldschmidt et al., 2011), and according to Hudson et al. (2007), approximately 60% are in the normal weight/overweight categories (BMI 18.5–29.9 kg/m2). Research also shows that BED is often comorbid with anxiety and substance use disorders (Wonderlich et al., 2009). The prevalence rate of greater than 1% for BED in young women is higher than 0.3% for anorexia nervosa or approximately 1% for bulimia (Hoek and van Hoeken, 2003). In a recent meta-analysis, Qian et al. (2013) estimated that the lifetime prevalence of BED was 2.22% compared with 0.21% and 0.81% for anorexia nervosa and bulimia, respectively. Impulsivity is a core psychopathological symptom in attention deficit hyperactivity disorder (ADHD) (Bolea-Alamañac et al., 2014), and there is a growing body of preclinical and clinical evidence to indicate that a loss of impulse control and compulsivity may be causal in bingeing on palatable foods in BED (Colles et al., 2008; Galanti et al., 2007; Gearhardt et al., 2011; Schag et al., 2013; Svaldi et al., 2014; Wu et al., 2013). Evidence has also emerged to indicate that ADHD symptomatology is present in a significant proportion of individuals who have eating disorders including BED (Biederman et al., 2007; Cortese et al., 2007; Docet et al., 2012; Fernández-Aranda et al., 2013).
Using a variant of a limited intermittent, irregular access, to palatable food paradigm described by Corwin (2004), we developed a model of binge eating in rats using ground milk chocolate (Vickers et al., 2015). A range of compounds including the novel d-amphetamine prodrug, lisdexamfetamine dimesylate, and its active metabolite, d-amphetamine, preferentially and dose-dependently reduced chocolate binge eating, but not the consumption of normal chow (Vickers et al., 2015).
Lisdexamfetamine, which is approved to treat ADHD in North America, some South American countries and Europe, is also efficacious in treating BED (Citrome, 2015; McElroy et al., 2015, 2016a). Lisdexamfetamine has recently been approved in the USA for the treatment of moderate to severe BED in adults (US Food and Drug Administration, 2015). In common with most drugs used to treat ADHD, lisdexamfetamine enhances dopaminergic and noradrenergic neurotransmission in the brain (Heal et al., 2013a; Hutson et al., 2014; Rowley et al., 2012, 2014).
From the evidence above, it can be concluded that individuals with BED are fully aware of the adverse metabolic and psychological consequences of their bingeing episodes, but are nonetheless unable to prevent themselves from compulsively bingeing on highly calorific food and repeating this behaviour over and over again (perseveration). There is a link between ADHD and impulse control deficits in BED, and finally, lisdexamfetamine has been shown to be an effective treatment for both disorders and to reduce impulsivity in ADHD (Bolea-Alamañac et al., 2014; Mattingly et al., 2012; Najib, 2012; Weisler et al., 2014).
To examine if binge-eating rats develop compulsive behaviour, we first trained them and groups of controls in a conventional conditioned avoidance response task in a two-compartment shuttle box. The rat learned that a tone/light conditioning stimulus signalled it would receive a mild foot-shock if it did not relocate to the other compartment within 10 s. The paradigm was then modified to a novel food reward/punished responding conflict model by placing a chocolate-filled jar (empty jar for the non-binge-eating controls) into one of the chambers. Entry into this compartment triggered the presentation of the same light/tone conditioning stimulus used in the conditioned avoidance response test and a foot-shock 10 seconds later if the rat did not leave the compartment. As long as the rat remained in the compartment with no jar in it, no trial was initiated, no conditioning stimulus was presented and no foot-shock was administered. This compartment was always ‘safe’ because the rats were able remain in it for the 30-minute duration of the test without initiating a tone/light warning or receiving foot-shocks. Hence, the binge-eating rats were familiar with the adverse consequences of not heeding the warning given by the conditioning stimulus, if they entered the chamber with the jar in it to consume the chocolate. The model, therefore, mirrors the conflict experienced by subjects with BED; the compulsion to binge versus the adverse psychological and metabolic consequences of giving in to the compulsion to binge eat. We used the model to investigate whether chocolate bingeing rats developed compulsive behaviour, and because the test was run in a battery of 10 trials, we could also investigate the perseverative aspect of chocolate bingeing. Having discovered that binge-eating rats showed compulsive/perseverative behaviour in the model, we investigated whether lisdexamfetamine, which is clinically effective in treating BED, altered these abnormal behaviours. The findings are the first demonstration that lisdexamfetamine reduces compulsivity and perseverative responding in a model of binge eating in rats.
Methods and materials
Animals and housing
Thirty-four lean, female, Wistar rats (weight range 200–250 g) were obtained from Charles River, Margate, Kent, UK. They were singly housed in polypropylene cages (45 cm × 28 cm × 20 cm) with wire grid floors to enable the food intake of each rat to be recorded. Bedding material was present to provide a nest and an alternative surface for the rats to rest on. Single housing was adopted to enable accurate measurement of food intake. Rats were maintained at a temperature of 21±4°C and 55±20% humidity on a reverse phase light–dark cycle (lights off for 8 hours from 10:30 to 18:30 hours) and acclimatised to the facility for a two-week period and weighed once-weekly prior to the experimental study.
All experiments were performed in accordance with Home Office guidelines and were licensed under the Animals (Scientific Procedures) Act 1986.
Establishment of binge-eating behaviour in female rats using chocolate as a palatable food
After a two-week acclimatisation period, rats were allocated into treatment groups based on body weight. All rats had 24 hour free access to standard powdered diet (Harlan Teklad 2018; 13.0 kJ/g) and tap water throughout the study. The diet was contained in a glass feeding jar with an aluminium lid (Solmedia Laboratory Suppliers, Romford, Essex, UK). Each lid had a 3-4 cm hole cut in it to allow access to the food. On binge days, an additional jar containing ground chocolate (Cadbury’s Dairy Milk; 23.44 kJ/g) or an empty jar (control) was placed in each cage for a two-hour period. The diet and chocolate were provided as powdered diet to reduce spillage and make accurate recording of daily food intakes rapid and simple. Food was supplied dry so that grams consumed could be accurately converted to kJ to allow total food intake to be measured. Rats were allowed to binge eat on days 1, 2, 4, 6, 7, 9, 12, 14, 15, 18, 23, 28, 31, 33, 36, 39, 42, 46 and 48. Hence, opportunities to binge were provided throughout the entire study duration and the interval between the binge sessions was gradually increased as the training progressed. Once the rats showed robust, reproducible binge eating, the binge-eating behaviour could be maintained by giving the rats only one or two binge sessions per week. The jars were placed in the cages at approximately 10:30 hours, which was at the beginning of the dark phase for the rats. The weights of the jars were recorded before and after the two-hour test session ensuring that the binge-eating sessions took place in the dark phase when rats consume most of their food. The body weight of each rat and its food and water intake were measured on every morning of the study; readings were taken in the final stage of the light phase at about 09:45 hours. All diets were provided as a powder to control as far as possible for differences in physical form, which may have affected diet preference. One rat in the binge-eating cohort was removed from the study during this phase (day 23) as despite not undergoing procedures it exhibited weight loss coupled with poor condition.
Eating disorders are more prevalent in women than men and it is one of the reasons why female rats were selected for these experiments. Another is that female rats are relatively weight-stable when adult, whereas males continue to grow throughout adulthood, which is not physiologically relevant to man. It is possible the binge eating of the rats could be affected by the oestrous cycle. We have not noted any marked day-to-day variance in the responses of female rats in the model over a period of four weeks. With rats having a four-day oestrous cycle, we have relied on using large group sizes to provide a cross-section of oestrous cycling across the cohort on every day of testing.
Conditioned avoidance response training
Training in the conditioned avoidance response test was initiated from day 29 onwards after binge eating had been established in the rats using automated shuttle boxes (42 × 16 × 20 cm; Med Associates, St Albans, USA). The shuttle boxes consisted of two compartments each with a tone and light to present the conditioning stimuli that were separated by means of a partition. Boxes had infrared-sensitive photocells in each compartment to enable the location of the rats to be determined. Each box was placed in a sound-attenuated chamber. Rats were trained to move to the adjacent compartment during presentation of the conditioning stimuli (tone and light; presented for 10 seconds), in order to avoid administration of the subsequent unconditioned stimulus (foot-shock, 0.5 mA for a duration of 10 seconds) via the grid floor (Sotty et al., 2009). The shock was aversive, but generally regarded as mild (i.e. it was not associated with injury to the feet, and rats did not show signs of stress prior to or after repeated training).
Training was organised so that initially all the rats underwent 30 trials in a 30-minute test session with a variable inter-trial interval of 20-30 seconds. When a rat crossed to the other compartment to avoid the shock, this was recorded by the apparatus as an ‘avoidance’. When the animal crossed to the other compartment during presentation of the shock, it was recorded by the apparatus as an ‘escape’. When the animal did not cross to the other compartment during presentation of the shock, this was recorded by the apparatus as an ‘escape failure’. This protocol was repeated over 10 days (i.e. two weeks: days 29–40 inclusive). One rat (binge-eating cohort) showed less than 25% avoidance after 10 days and was removed from the study at this stage.
Subsequently, the 32 rats (15 binge-eating rats and 17 non-binge-eating controls) underwent further daily training consisting of 10 trials in a 10-minute session with a variable inter-trial interval of 20-30 seconds. This was repeated over 5 days. Rats were initially placed in alternate sides of the two-compartment box to avoid bias.
All conditioned avoidance response training sessions occurred during the afternoon. On those days when animals underwent a two-hour binge session (see earlier), this was timed to begin at 10:30 (lights out).
Evaluation of binge-eating and control groups of rats in the novel food reward/punished responding conflict model
After reaching the training criterion (⩾80% avoidance on the previous three sessions), the test was modified to introduce a potential conflict between chocolate reward and foot-shock punishment. Rats underwent four such training sessions (30 minutes duration) each week for two weeks (i.e. eight sessions). In half of those sessions (two each week), an empty jar was present in one of the chambers. The jar was identical to those used during the binge-eating development phase of the study and the location chamber was counter-balanced across groups. On these ‘control’ days, the apparatus was programmed so that entry into the chamber with the empty jar for a variable duration (e.g. 5-30 seconds) resulted in the presentation (10 in total) of the conditioning stimuli (light and tone). A variable duration was used so rats could not learn that they had a fixed period of time in the chamber with the jar, and therefore could repeatedly enter and exit the chamber thereby avoiding foot-shocks. The presentation of the conditioning stimuli (light and tone) was followed (10 seconds later) by administration of the unconditioned stimulus (foot-shock, 0.5 mA for 10 seconds) via the grid floor. Each animal underwent a maximum of 10 trials in a 30-minute period. Trials were only initiated by the rat entering the chamber with the jar in it. As long as the rat remained in the other compartment, no trial was initiated, no conditioning stimuli (tone/light) were presented and no foot-shock was administered. The day after each ‘control’ trial (i.e. twice each week), rats underwent the same procedure as above, but the jar was filled with chocolate in the case of the binge-eating rats (non-binge controls were presented with an empty glass jar).
Evaluation of lisdexamfetamine in the novel food reward/punished responding conflict model
Having discovered in the first part of the study that binge-eating rats showed compulsive/perseverative responding in the food reward/punished conflict model, we went on to investigate whether lisdexamfetamine influenced the consumption of chocolate or compulsive/perseverative behaviours in the model.
On completion of the training phase, the effects of lisdexamfetamine or vehicle were investigated. The study was a within-subjects design and each animal received either lisdexamfetamine or vehicle in a counterbalanced order at a one-week interval. The day prior to each drug or vehicle treatment, all rats underwent a baseline session identical to that previously described (i.e. in the presence of chocolate in the case of binge-eating rats) with the exception that they were dosed orally with vehicle 60 minutes prior to start of the test to familiarise them with oral dosing. Rats were dosed in their home cage and left in it with free access to chow until being placed in the shuttle boxes. On test days (days 65 and 72) all rats were dosed with lisdexamfetamine or vehicle in their home cages. One hour later each rat underwent a 30-minute test session identical to those run previously. A glass jar with either chocolate in it (binge-eating rats) or empty (non-binge-eating controls) was placed in one compartment of the apparatus. In the test session, when the rat entered the compartment with the jar the conditioned stimulus (light and tone) was presented after a variable interval (5–30 seconds). This stimulus was followed 10 seconds later by presentation of the unconditioned stimulus (foot-shock, 0.5 mA for 10 seconds) via the grid floor if the rat did not move to the other compartment with no jar in it.
As stated previously, all rats had 24 hour free access to standard powdered diet and they were kept in the home cages until they were placed in the two-chamber boxes for testing. Therefore, the rats were able to satisfy their hunger up to the point of testing. The test itself took an average of 10 minutes for the binge-eating rats. Thus, the responses of the binge-eating rats in the food reward/punished responding conflict model were motivated by a desire to consume chocolate and not hunger as a result of food restriction.
Drugs and reagents
Lisdexamfetamine was provided by Shire, UK. Lisdexamfetamine was dissolved in deionised water and administered orally by gavage in a dose volume of 3 ml/kg body weight. The dosing volume of 3 ml/kg was selected for consistency with previous studies, for example, Vickers et al. (2015). The dose of lisdexamfetamine is expressed in terms of d-amphetamine base (salt/base correction factor = 3.37). A dose of 0.8 mg/kg [d-amphetamine base] of lisdexamfetamine was selected for use in these experiments because Vickers et al. (2015) showed that it was approximately the ED50 to reduce chocolate bingeing by binge-eating rats and it preferentially decreased chocolate bingeing without decreasing the consumption of normal chow in the two-hour binge-eating test. Lisdexamfetamine or vehicle was administered 60 minutes before testing.
Data and statistical analysis
During the acquisition of binge eating by the rats, in each binge-eating session the following four measures were analysed by Student’s t-test:
Powdered chocolate intake (kJ) during the two-hour session Powdered chow intake (kJ) during the two-hour session Total food intake (kJ) during the two-hour session Total food intake (kJ) during the 24 hours including this two-hour session.
Body weight on each day of training was analysed by analysis of covariance (ANCOVA) with day 1 body weight as the covariate. In the novel food reward/punished responding conflict model, a square root transformation was used because the data were skewed. This transformation was applied to results for the number of occasions that the rats entered the chamber containing the jar, but left prior to a trial commencing, number of escapes, total time in chamber containing the jar without initiating a trial, total avoidance time, total escape time and chocolate intake. Because the results were forced between two values (e.g. a percentage may be forced to be between 0 and 100), an angular transformation arcsin(sqrt(×/100)) was used for the results on percentage avoidance, percentage escapes and percentage time spent in chamber containing the jar. Chocolate intake was analysed by mixed linear model with treatment and period as fixed factors, animal as a random factor. All other data were analysed by mixed linear model with treatment and period as fixed factors, animal as a random factor, baseline (the same variable on the day before treatment, using the same transformation) as a covariate. The primary endpoint of study 1 was to determine whether binge eating produced compulsive/perseverative behaviour in the food reward/punished responding conflict model. The primary endpoint in study 2 was to determine whether lisdexamfetamine altered the compulsive/perseverative responses of the binge-eating rats in the model. Multiple comparisons between the treatment groups were by the multiple t-test. The chance of both treatments (binge eating and lisdexamfetamine) being simultaneously significant at the 5% level if there was no treatment effect (type 1 error) was approximately 1 in 400, that is, 1/20 × 1/20. With such a low probability of a type 1 error, an adjustment for multiple comparisons was not deemed to be appropriate. The mixed linear model allows between-group comparisons (between binge-eating and non-binge-eating groups of rats) and within-group comparisons (e.g. between lisdexamfetamine and vehicle treatments within the group of binge-eating rats) to be made in the same analysis. Transformations were used, where appropriate, to ensure data were normally distributed. Where reported in the paper, adjusted means (least squares means) have been produced by ANCOVA.
A value of P<0.05 was regarded as being statistically significant. All statistical analysis was performed using SAS version 9.1.3 (SAS Institute Inc., Cary, NC, USA).
Results
Development of binge eating in the rats
When freely fed, lean, female, Wistar rats were given unpredictable intermittent two-hour access to chocolate during the 28-day acquisition schedule, they developed a characteristic pattern of hyperphagia on the binge session days followed by substantial reductions of food intake on the days immediately after (Figure 1(a)). This pattern of bingeing behaviour was maintained throughout the conditioned avoidance response training period, and the duration of the experimental phase of the project (Figure 1(a)).
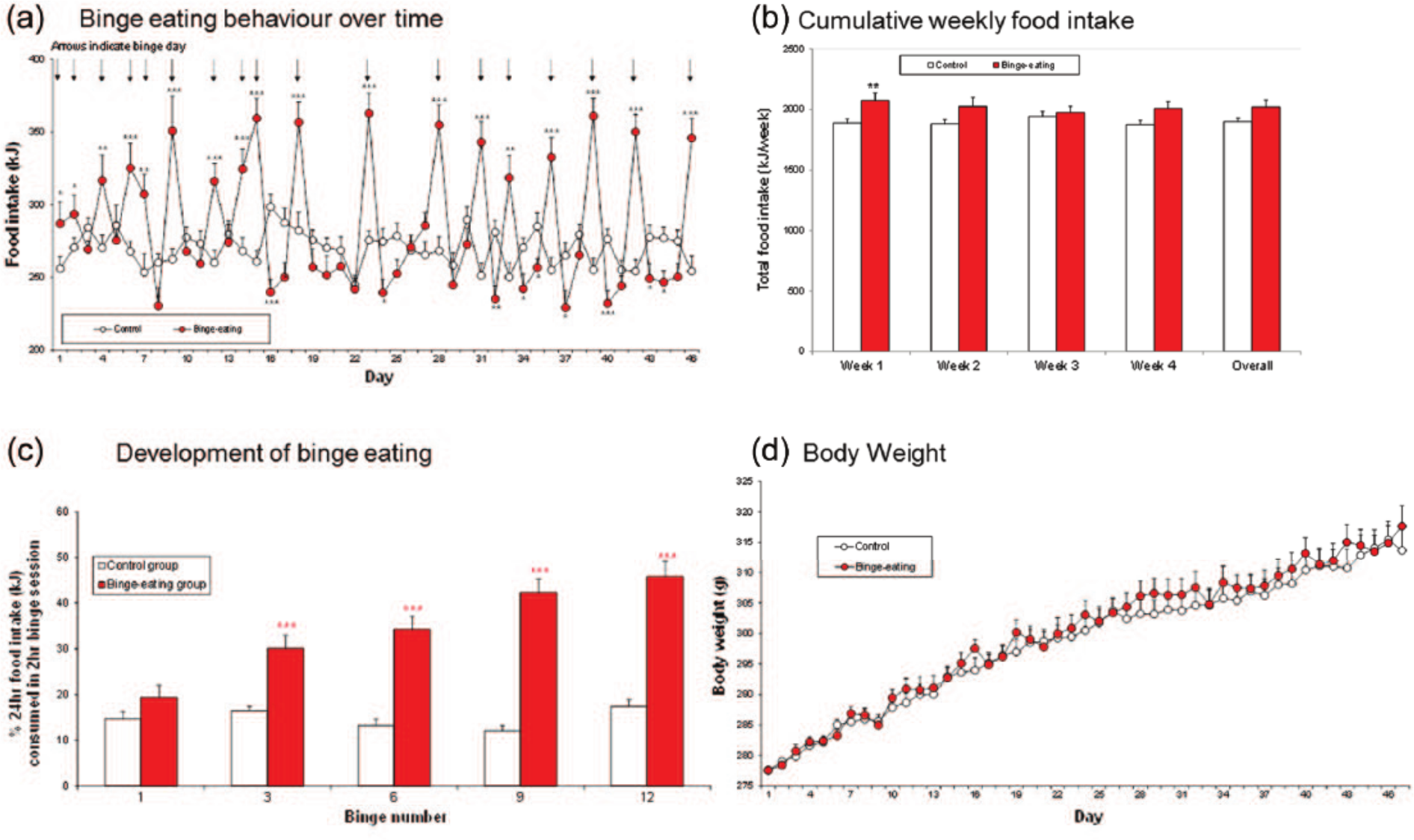
Development of binge eating in lean, female, Wistar rats. Binge-eating group, n=15. Non-binge controls, n=17. (a) Daily food intake of rats with and without unpredictable, irregular, limited access to ground milk chocolate. Results are arithmetic means ± SEMs. Comparisons are by Student’s t-test. Significantly different compared with the control group *P<0.05, **P<0.01, ***P<0.001. (b) Cumulative weekly food intake of binge-eating and control groups of rats. Results are arithmetic means ± SEMs. Comparisons are by Student’s t-test. Significantly different compared with the control group **P<0.01. (c) Development of binge eating showing the increasing percentage of the daily calorie intake consumed in the binge sessions over time. Results are arithmetic means ± SEM. Comparisons are by Student’s t-test, ***P<0.001. (d) Body weights of binge-eating and control groups of rats. Results are adjusted means. SEMs were calculated from the residuals of the statistical model.
The rats acquired a preference for chocolate as shown by the steady increase in the consumption of this palatable food in the two-hour binge sessions during the 28-day training schedule, and substantial consumption of chocolate was maintained during the study duration (Figure 1(a)). The consumption of chocolate was reflected by an overall increase in calorie intake on the days of the chocolate binge sessions. The pattern of robust hyperphagia on the binge session days was followed by reductions of food intake on the days immediately thereafter. This pattern of feeding behaviour produced a small increase in the average energy intake/week in weeks 1, 2 and 4, and overall, which was statistically significant during week 1 only (Figure 1(b)). There were some minor fluctuations in the water intake of the rats, but overall, drinking remained roughly constant throughout experimental duration (data not shown). As binge eating became established in the rats, they consumed increasing amounts of chocolate in the two-hour binge sessions (Figure 1(c)). By day 28, the binge-eating rats were ingesting over 40% of their daily food intake in these two-hour sessions. The body weights of the rats showed a gradual increase over the study duration. There was no significant difference between the body weights of the binge-eating and non-binge-eating groups of rats over the course of the experiment (Figure 1(d)).
Training performance of the binge-eating and non-binge-eating groups of rats in the conditioned avoidance response procedure
Groups of binge-eating lean, female, Wistar rats and non-binge-eating controls both learned to perform the conditioned avoidance response task within a very short period of time, with both groups of rats achieving over 80% foot-shock avoidances after three training sessions (Figure 2). There was no difference between the rate at which the groups of binge-eating rats and non-binge-eating controls learned the task or their level of proficiency in performing in the conditioned avoidance response test (Figure 2).
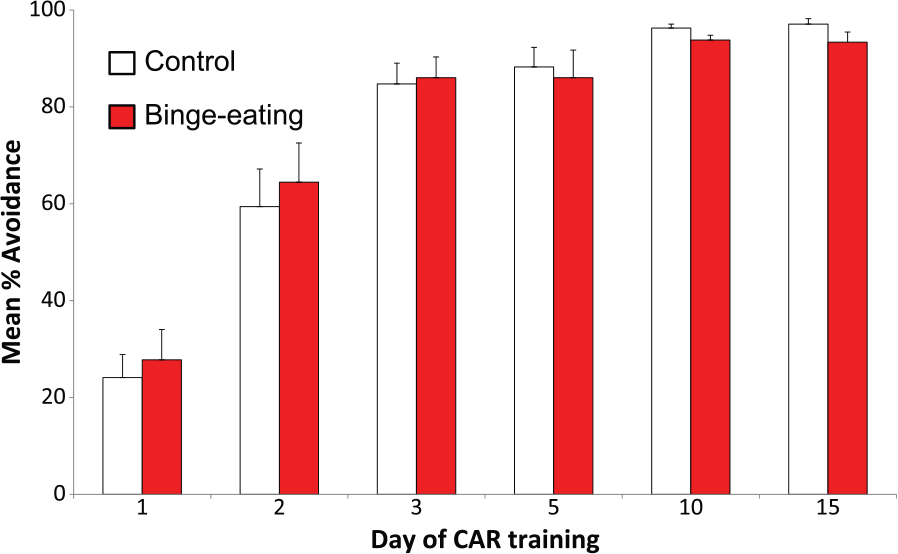
Acquisition and proficiency of binge-eating rats (n=15) and non-binge controls (n=17) in performing a standard conditioned avoidance response (CAR) task. Results are mean ± SEM percentage avoidance of foot-shocks in the CAR test. Days 1-10: training 30 trials/rat, days 11–15: 10 trials/rat.
Effects of binge-eating on behaviour in the novel food reward/punishment conflict model
The binge-eating rats consumed an average of 35.2 ± 5.2 kJ (1.50 ± 0.22 g) of chocolate during their completed sessions in the food reward/punishment conflict model (Table 1).
Effects of lisdexamfetamine on chocolate consumption by binge-eating rats in the food reward/foot-shock punished conflict test.

Results are expressed as adjusted means (least squares means produced by analysis of covariance) ± SEM. SEMs were calculated from the residuals of the statistical model. Data were analysed by analysis of covariance with baseline as covariate. Significantly different from vehicle-treated, binge-eating rats. F=9.19 on 1,13 df ††P<0.01.
There was no difference between the number of completed trials in the food reward/punishment conflict model made by the binge-eating rats compared with non-binge-eating controls (Figure 3(a)). However, the time that the binge-eating rats took to complete the 10 trials was less than 50% of that of the non-binge-eating controls (main effect of treatment: F=8.70 on 3,28 df P<0.001; P<0.001 by multiple t-test) (Figure 3(b)).
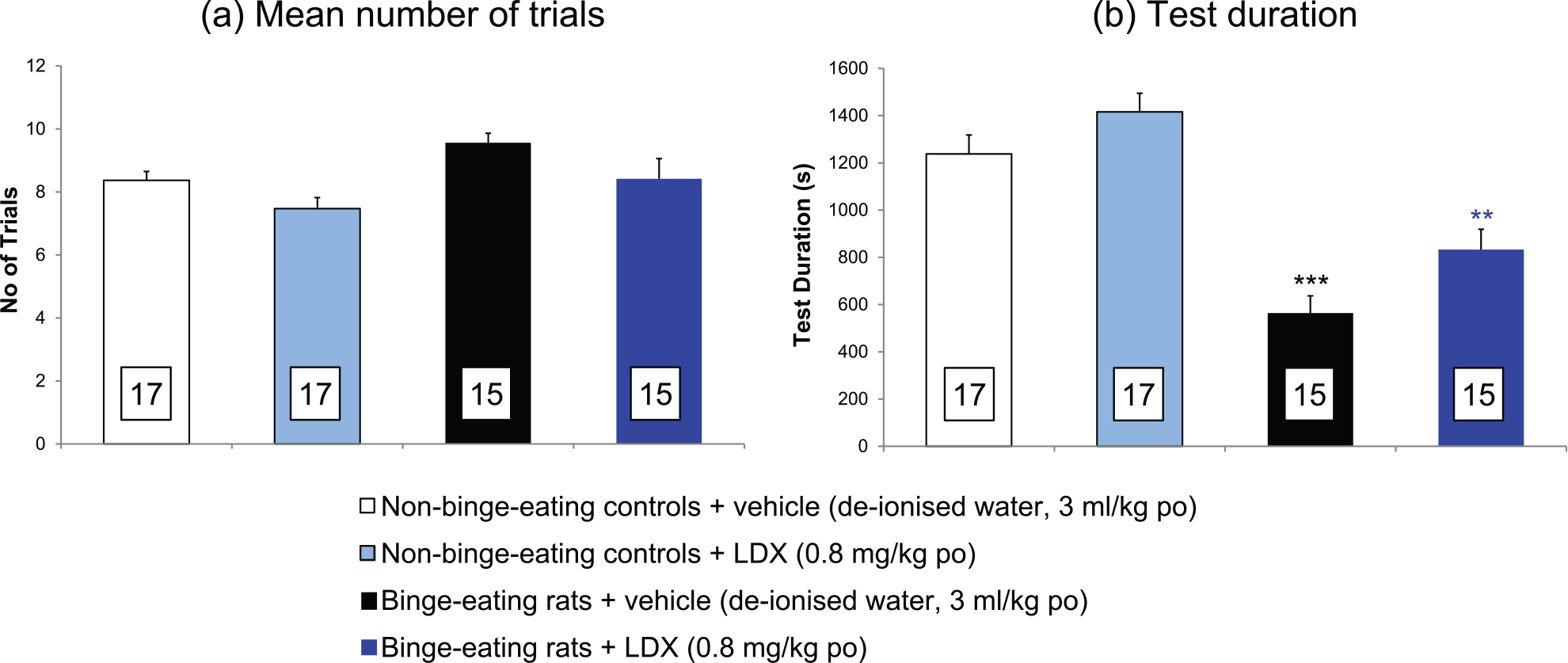
A comparison of the responses of binge-eating rats and non-bingeing controls in a novel food reward/punished responding conflict model and the effects of lisdexamfetamine on these responses. (a) Number of trials completed. (b) Test duration. Results are expressed as adjusted means ± SEM (n values in boxes in the histobars). SEMs were calculated from the residuals of the statistical model. Data analysed by analysis of covariance with baseline as covariate. Comparisons versus non-binge-eating control group or vehicle-treated, binge-eating rats are by the multiple t-test.
The binge-eating group of rats spent 73.5% of the duration test sessions in the compartment paired with the chocolate jar, which was significantly greater (main effect of treatment: F=14.23 on 3,28 df P<0.001; P<0.001 by multiple t-test) than the 22.4% of the test that the non-binge-eating controls spent in the compartment with the empty jar (Figure 4(a)).
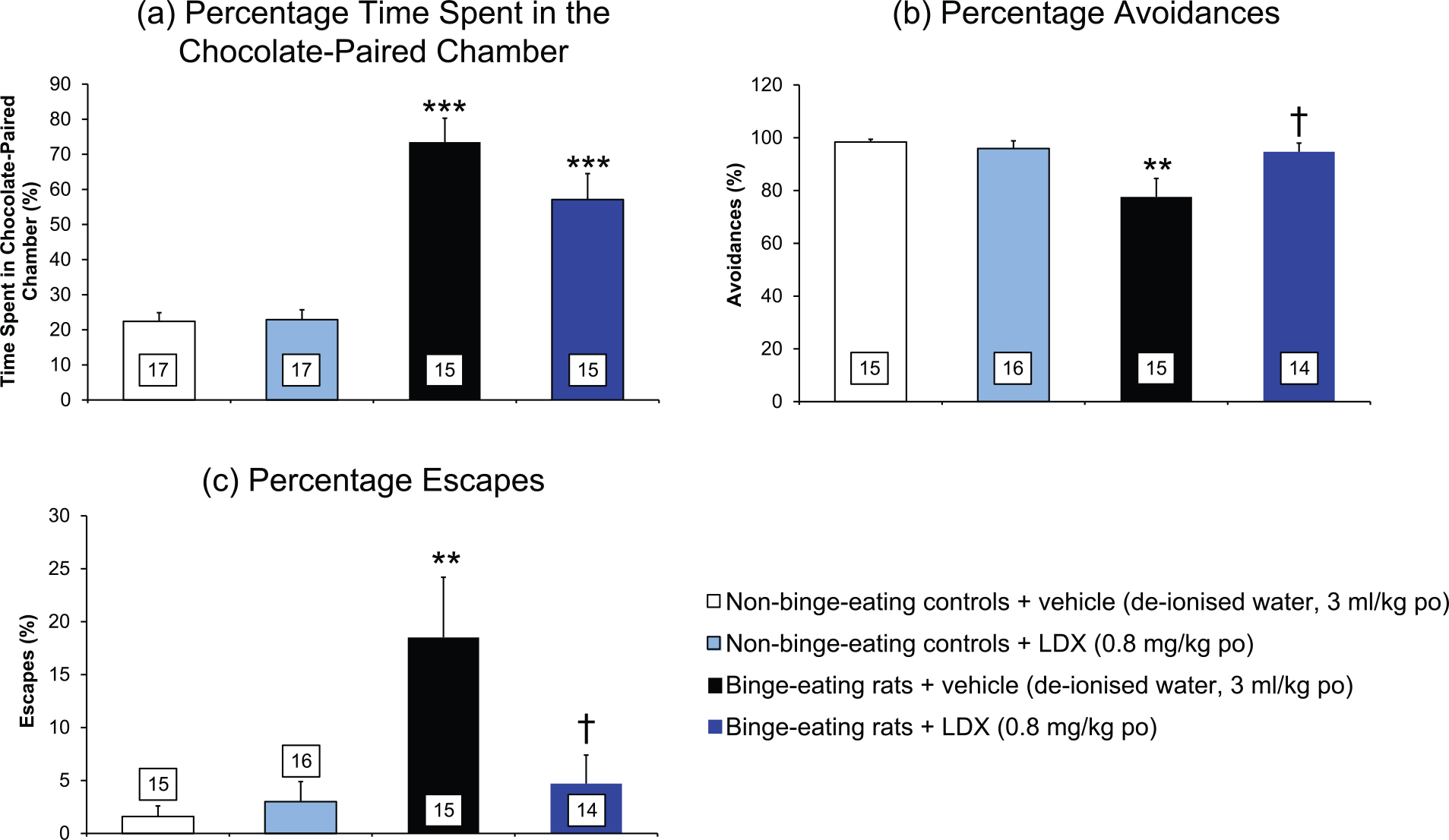
A comparison of the responses of binge-eating rats and non-bingeing controls in a novel food reward/punished responding conflict model and the effects of lisdexamfetamine on these responses. (a) Percentage time spent in the chocolate-paired chamber. (b) Percentage avoidances (an avoidance is when the rat leaves the compartment on presentation of the conditioned tone/light stimulus prior to receiving a foot-shock). (c) Percentage escapes (an escape is when the rat leaves the compartment after receiving a foot-shock). Results are expressed as adjusted means ± SEM (n values in boxes in the histobars). SEMs were calculated from the residuals of the statistical model. Data analysed by analysis of covariance with baseline as covariate. Comparisons versus non-binge-eating control group or vehicle-treated, binge-eating rats are by the multiple t-test.
The percentage of avoidance responses (trials when the rats heeded the tone/light conditioning stimulus and left before receiving a foot-shock) made by the binge-eating rats was 77.6 ± 7.0%, which was significantly lower (main effect of treatment: F=3.65 on 3,25 df P<0.05; P<0.01 by multiple t-test) than the 98.4 ± 1.0% avoidances made by the non-binge-eating controls in the food reward/punished responding conflict model (Figure 4(b)).
There was no difference (main effect of treatment: F=1.51 on 3,28 df P=0.23) between the mean number of avoidances made by binge-eating rats (6.60 ± 0.60, n=15) and non-binge-eating controls (8.1 ± 0.28, n=17) in the model.
The average time taken to make an avoidance was 25.1% longer for the binge-eating rats (3.88 ± 0.29 seconds, n=15) than the non-binge-eating controls (3.10 ± 0.26 seconds, n=15) with a trend for statistical significance (main effect of treatment: F=1.85 on 3,25 df P=0.164; P=0.074 by multiple t-test).
The percentage of escapes (sessions when rats did not heed the conditioning tone/light stimulus and received a foot-shock before leaving the compartment) made by the binge-eating rats was 18.5 ± 5.7% compared with 1.6 ± 1.0% for the non-binge-eating group; a significant increase of 1190% (main effect of treatment: F=3.53 on 3,25 df P<0.05; P<0.01 by multiple t-test) (Figure 4(c)).
The mean number of escape failures (occasions when the rats receiving foot-shocks failed to leave the shock-paired compartment in the model) was extremely low and did not differ between the binge-eating rats (0.04 ± 0.05, n=15) and the non-binge-eating controls (0.00 ± 0.00, n=17); main effect of treatment: F=1.06 on 3,28 df P=0.38.
The mean number of occasions when the rats received a foot-shock before leaving the compartment containing the jar (empty for the non-binge-eating controls and chocolate filled for the binge-eating rats) was significantly increased by 1290% (main effect of treatment: F=4.78 on 3,28 df P<0.01; P<0.01 by multiple t-test) for the binge-eating rats compared with the non-binge-eating controls (Figure 5(a)).
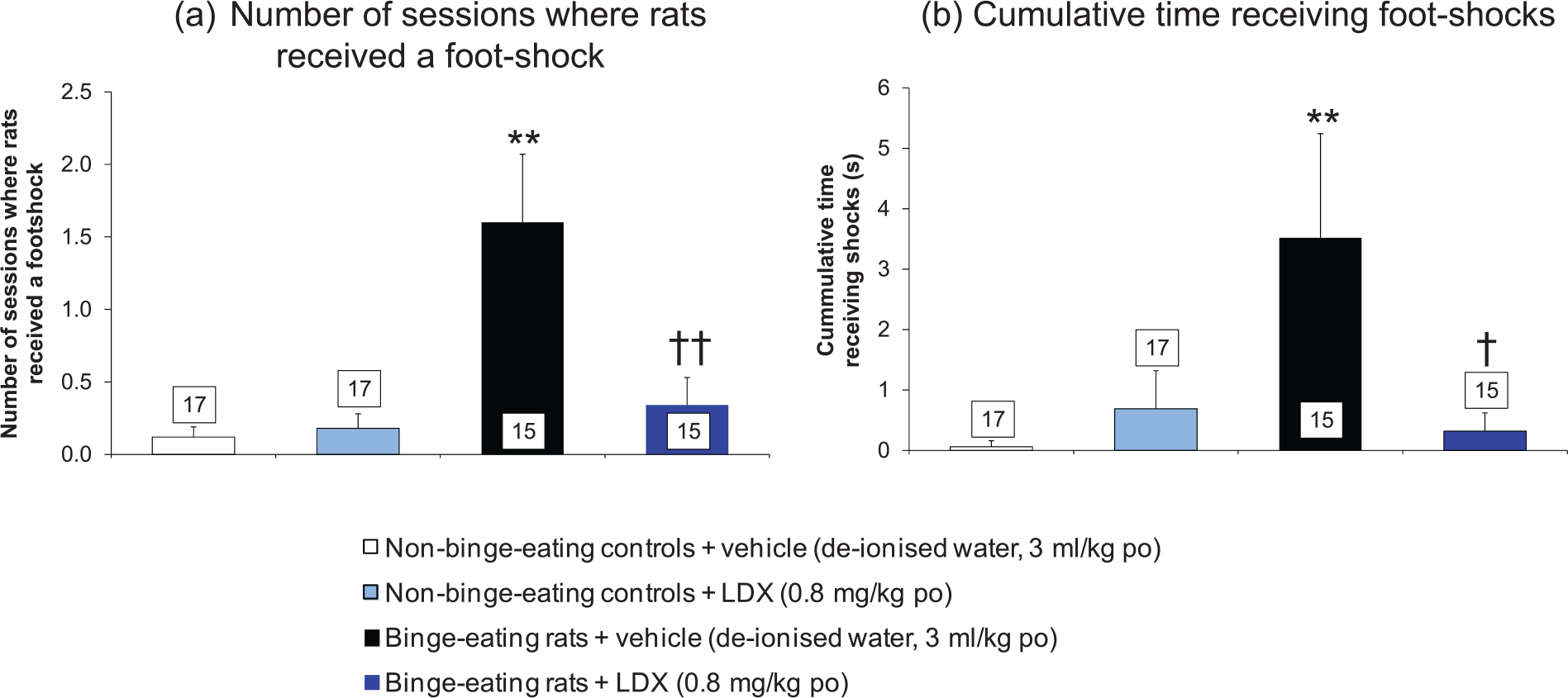
A comparison of the responses of binge-eating rats and non-bingeing controls in a novel food reward/punished responding conflict model and the effects of lisdexamfetamine on these responses. (a) Number of sessions when rats received a foot-shock. (b) Cumulative time receiving foot-shocks. Results are expressed as adjusted means ± SEM (n values in boxes in the histobars). SEMs were calculated from the residuals of the statistical model. Data analysed by analysis of covariance with baseline as covariate. Comparisons versus non-binge-eating control group or vehicle-treated binge-eating rats are by the multiple t-test. Significantly different from non-binge-eating control group: **P<0.01.
The cumulative foot-shock time (seconds) tolerated by the binge-eating group was significantly longer than for the non-binge-eating controls (main effect of treatment: F=4.14 on 3,28 df P<0.05; P<0.01 by multiple t-test) (Figure 5(b)).
Effect of lisdexamfetamine on chocolate consumption by binge-eating rats in the food reward/punished responding conflict model
Administration of lisdexamfetamine (0.8 mg/kg po [d-amphetamine base]) 60 minutes before testing in the food reward/punished responding conflict model significantly reduced the consumption of chocolate by the binge-eating group of rats to 15.8 ± 1.9 kJ (0.67 ± 0.08 g), which was 55% lower (P<0.01) than the vehicle-treated, binge-eating rats (Table 1).
Lisdexamfetamine administration did not alter the number of trials completed by either the binge-eating or non-binge-eating control groups of rats in the test (Figure 3(a)).
The time taken by the binge-eating rats to complete the 10 trials in this model was less than 50% of the time taken by the non-binge-eating controls (P<0.001). There was no effect of lisdexamfetamine administration on the time taken for the non-binge-eating controls to complete the 10 trials (Figure 3(b)); however, lisdexamfetamine increased the time taken by the binge-eating rats by 47.8% (Figure 3(b)) with a strong trend towards statistical significance (P=0.063).
The binge-eating group of rats spent 73.5% of the test period in the compartment paired with the chocolate jar. Lisdexamfetamine reduced the percentage of time that the binge-eating rats spent in the chocolate-paired compartment by 22.3%, with a strong trend towards statistical significance (P=0.060) (Figure 4(a)).
The percentage of time the non-binge-eating controls spent in the compartment paired with the empty food jar was 22.4% and this parameter was not influenced by the administration of lisdexamfetamine (Figure 4(a)).
Administration of lisdexamfetamine (0.8 mg/kg po [d-amphetamine base]) 60 minutes before testing significantly (P<0.05) increased the percentage avoidances made by the binge-eating rats to 94.7 ± 3.3% when compared with the vehicle-treated, binge-eating group (Figure 4(b)). The percentage avoidances made by the lisdexamfetamine (0.8 mg/kg po)-treated binge-eating group of rats was not significantly different from the response of the non-binge-eating controls. Lisdexamfetamine (0.8 mg/kg po) had no effect on the percentage avoidances made by the non-binge-eating control group of rats.
There was no difference between the mean number of avoidances made by binge-eating rats and non-binge-eating controls in the food reward/punishment conflict test, and lisdexamfetamine (0.8 mg/kg po) did not significantly alter the number of avoidances made by either group of rats (main effect of treatment: F=1.51 on 3,28 df P=0.23; data not shown).
Lisdexamfetamine significantly (P<0.05) reduced the mean latency to avoid the shock stimulus by the binge-eating rats (lisdexamfetamine group: 3.01 ± 0.24 seconds, n=14; vehicle group: 3.88 ± 0.29 seconds, n=15), but it had no effect on this parameter in the non-binge-eating controls (lisdexamfetamine group: 3.24 ± 0.28 seconds, n=15; vehicle group: 3.10 ± 0.26 seconds, n=15). The mean avoidance time of the binge-eating rats receiving lisdexamfetamine was not significantly different from that of the vehicle-treated, non-binge-eating control group.
Lisdexamfetamine significantly (P<0.05) reduced the percentage of escapes by the binge-eating group from 18.5 ± 5.7% to 4.7 ± 2.7% versus the vehicle-treated, binge-eating group (Figure 4(c)). The percentage of escapes made by the non-binge-eating control group of rats was not influenced by lisdexamfetamine (0.8 mg/kg po) pretreatment (Figure 4(c)). The percentage of escapes made by the lisdexamfetamine-treated binge-eating group was not significantly different from the non-binge-eating controls.
Lisdexamfetamine did not alter the number of escape failures in the binge-eating rats or the non-binge-eating control groups.
Lisdexamfetamine significantly reduced the number of sessions in which the binge-eating rats received foot-shocks by 79% (P<0.01) compared with the vehicle-treated, binge-eating group (Figure 5(a)). Lisdexamfetamine had no effect on this parameter in the non-binge-eating controls (Figure 5(a)). The mean number of sessions when the lisdexamfetamine-treated, binge-eating rats received foot-shocks was not significantly different from the vehicle-treated, non-binge-eating control group (Figure 5(a)).
The cumulative time the binge-eating rats tolerated foot-shocks was markedly higher than the non-binge-eating controls. Pretreatment with lisdexamfetamine produced an approximately 90% reduction in the cumulative time that binge-eating rats received foot-shocks (Figure 5(b)). Lisdexamfetamine did not influence this parameter in the non-binge-eating controls (Figure 5(b)). The cumulative foot-shock time experienced by the lisdexamfetamine-treated binge-eating group was not significantly different from the non-binge-eating controls (Figure 5(b)).
Effects of lisdexamfetamine on body weight and the intake of food and water over 24 hours
In the 24-hour period, which included testing in the food reward/punished responding conflict model, the total food intake (chocolate + normal chow) of the binge-eating rats was significantly (P<0.05) greater than that of the non-binge-eating control group (Table 2). Administration of lisdexamfetamine (0.8 mg/kg po [d-amphetamine base]) significantly decreased the total food consumed by the binge-eating rats by approximately 20% (Table 2). Lisdexamfetamine also significantly reduced the amount of normal chow consumed by the non-binge-eating controls by approximately 10% (Table 2). Lisdexamfetamine treatment did not significantly affect 24-hour water intake or body weight of either the binge-eating or non-binge-eating animals compared to appropriate controls (Table 1).
Effects of binge eating and lisdexamfetamine on daily food and water intake and bodyweight.
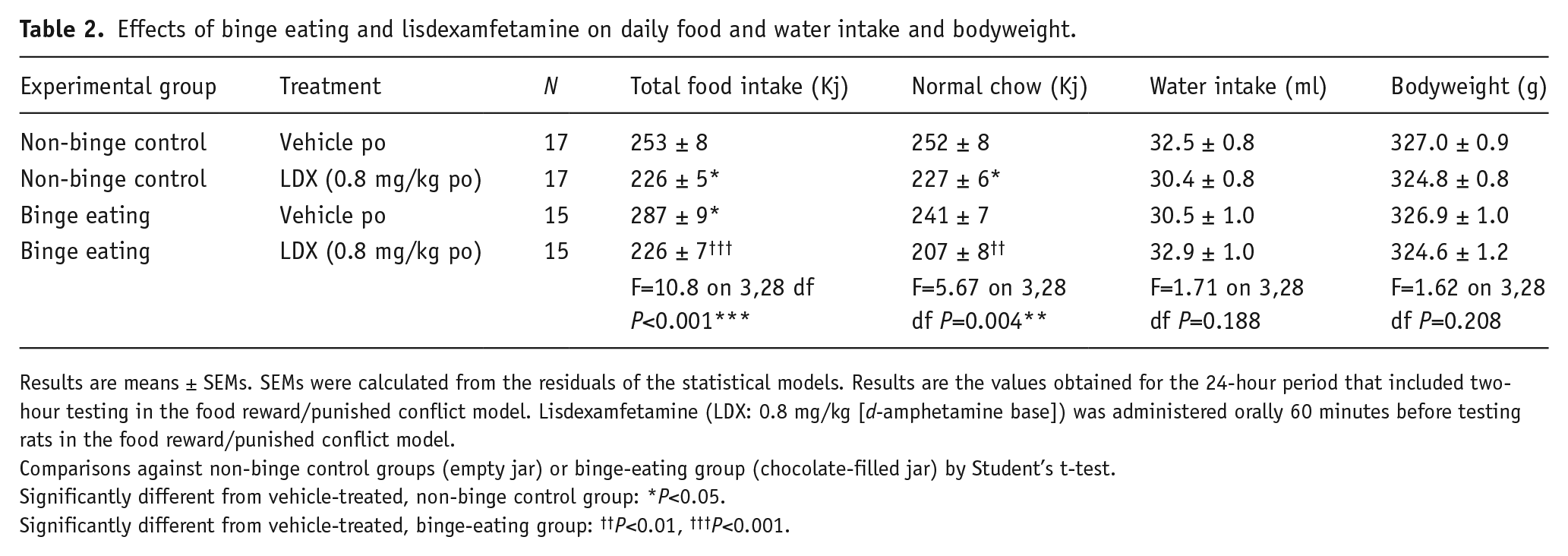
Results are means ± SEMs. SEMs were calculated from the residuals of the statistical models. Results are the values obtained for the 24-hour period that included two-hour testing in the food reward/punished conflict model. Lisdexamfetamine (LDX: 0.8 mg/kg [d-amphetamine base]) was administered po 60 minutes before testing rats in the food reward/punished conflict model.
Comparisons against non-binge control groups (empty jar) or binge-eating group (chocolate-filled jar) by Student’s t-test.
Significantly different from vehicle-treated, non-binge control group: *P<0.05.
Significantly different from vehicle-treated, binge-eating group: ††P<0.01, †††P<0.001.
Discussion
We have developed a model in which rats are given unpredictable, irregular, limited access to chocolate, which leads to the establishment of robust binge eating (hyperphagia) with concomitant reductions in the consumption of normal chow (Vickers et al., 2015). It is based on the work of Corwin (2004) except ground milk chocolate rather than vegetable shortening is used as the palatable food. Chocolate is high in fat and sugar with a highly palatable texture and taste, and it was chosen because it is a powerful driver of over-consumption in humans (Dingemans et al., 2002; Jääskeläinen et al., 2014; Rigaud et al., 2014; Yanovski, 2003) and rodents (de Jong et al., 2013; Giuliano et al., 2012).
When modelling binge eating, it is important to differentiate this aberrant behaviour from the behaviourally normal consumption of palatable food. Although we have not included a control group of rats given free access to chocolate, data from our dietary-induced obesity model in female Wistar rats showed that they consumed 56.3 kJ of chocolate in an equivalent two-hour period at the start of the dark phase (unpublished, in-house data), which is three times less than the 167-176 kJ levels seen over the final three baseline sessions in the present study. In our opinion, a multiple of over two-fold between these two food intakes is the minimum required for the acceptance of a model of binge eating. The food consumption of the binge eating in this period was also four to five-fold greater than the control group of rats (33-41 kJ).
Acute administration of lisdexamfetamine (0.1-1.5 mg/kg, po [d-amphetamine base]) to binge-eating rats dose-dependently reduced chocolate consumption by 15-71% (Vickers et al., 2015). A lisdexamfetamine dose of 0.8 mg/kg, po [d-amphetamine base] decreased the amount of chocolate consumed by the rats in the binge session by 49.6%, without significantly reducing normal chow intake (Vickers et al., 2015), and it was therefore selected as an appropriate dose for testing in the food reward/punished responding conflict model. In the model described by Vickers et al. (2015), the rats consumed 140-200 kJ (5.96–8.51 g) of chocolate in a two-hour binge-eating test, whereas in the 10-minute period that the binge-eating rats spent performing the 10 trials in the food reward/punishment conflict test the rats consumed approximately one fifth of that amount. Nonetheless, the reduction produced by lisdexamfetamine administration of 55% is remarkably consistent with its previously reported effect (Vickers et al., 2015). The congruence between the results, therefore, provides support for the selection of the 0.8 mg/kg, oral dose of lisdexamfetamine, which had previously been reported to reduce the consumption of chocolate but not chow, in the current investigation. That being said, the authors acknowledge that these findings would be more robust if the effects of lisdexamfetamine had been demonstrated using more than one dose of the prodrug.
We have not investigated any other compounds, which have been clinically evaluated as treatments for BED, for example, ALKS-33 and GSK1521498 (μ-opioid antagonists) and baclofen (GABA-B agonist). Although the outcomes from these trials have been mixed (McElroy et al., 2013; Wong et al., 2009; Ziauddeen et al., 2013), it will be interesting to investigate some of these compounds in the food reward/punishment conflict model to elucidate whether their poor showing in clinical trials could be explained by a failure to treat the compulsive and perseverative pathology underlying binge eating.
Binge-eating rats learned the conventional conditioned avoidance response task as quickly and performed it as competently as the non-binge-eating controls, which showed no evidence of cognitive impairment in the former group. The paradigm was then modified to a food reward/punished responding conflict model by placing chocolate (or an empty jar) into one of the compartments and pairing it with the conditioned avoidance response test tone/light stimulus and the administration of a foot-shock 10 seconds later if the rats did not leave the chamber. Our hypothesis was if the binge-eating rats had less inhibitory control, they would enter the chocolate-paired chamber and consume chocolate even though it would result in the presentation of the warning tone/light stimulus and a foot-shock if they did not leave. The binge-eating rats were also predicted to exhibit other compulsive and perseverative behaviours in the model, for example, an increase in the frequency of trials when the rats received foot-shocks (escapes). Both predictions were confirmed by the results that we obtained. Binge-eating rats entered the ‘unsafe’ food jar-paired chamber and consumed substantial amounts of chocolate while in this compartment. As summarised in Table 3, the binge-eating rats exhibited a wide range of responses in the model demonstrating compulsive and perseverative behaviours that were not displayed by the non-binge-eating controls. As summarised in Table 1, administration of lisdexamfetamine decreased chocolate consumption in the test. Lisdexamfetamine also produced a strong trend for a reduction in the percentage of time that binge-eating rats spent in the chocolate-paired compartment and the average time it took them to respond to the warning tone/light and avoid a shock. In many instances, lisdexamfetamine decreased compulsive and perseverative responding to the level of the behaviourally normal, non-binge-eating control group, that is, the total time spent in the chocolate-paired compartment, the percentage of ‘avoidances’, the number of escapes (trials when the rats received foot-shocks) and the percentage of escapes (Table 4).
Summary of the evidence for compulsive and perseverative behaviour in lean, female, binge-eating rats.
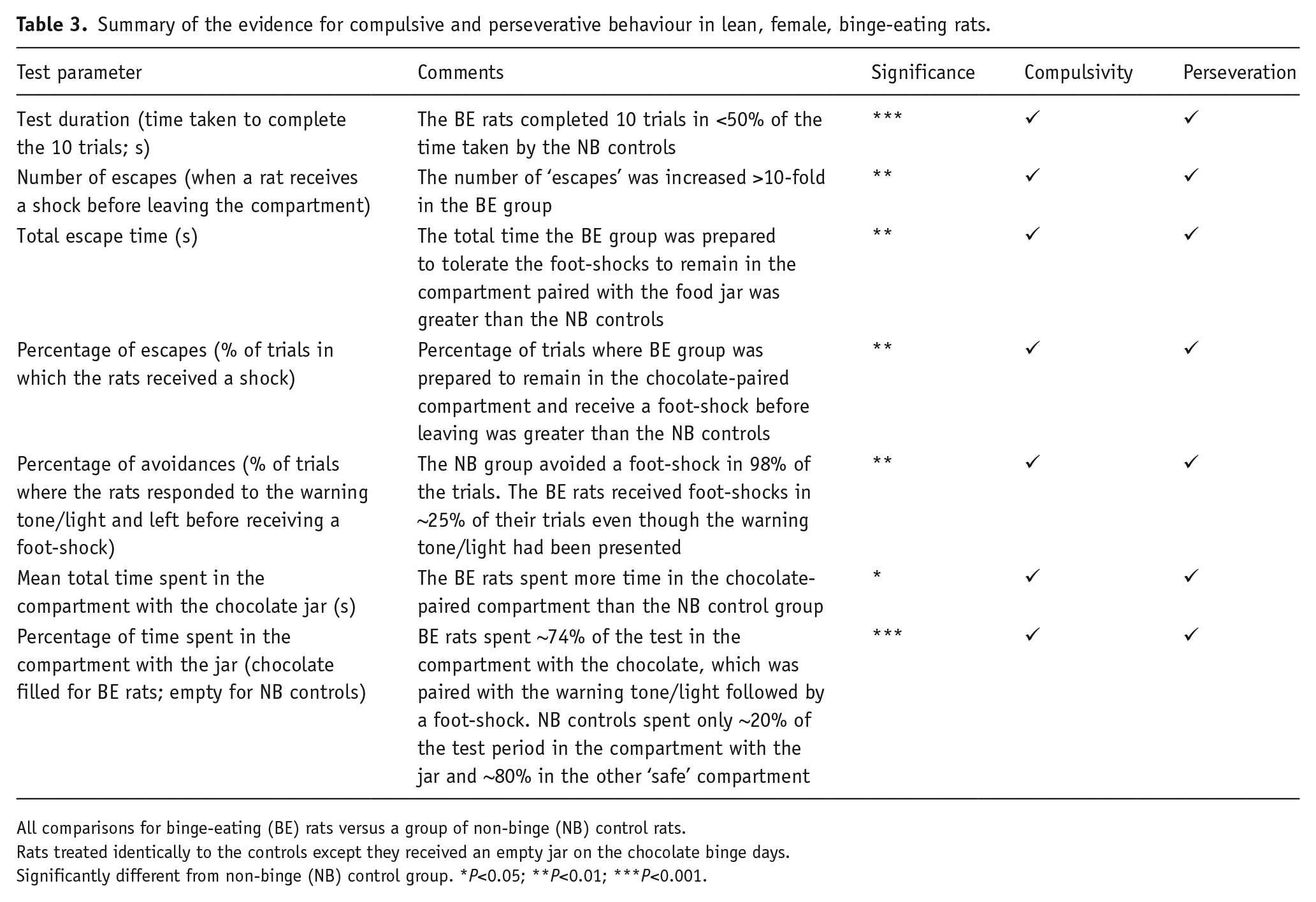
All comparisons for binge-eating (BE) rats versus a group of non-binge (NB) control rats.
Rats treated identically to the controls except they received an empty jar on the chocolate binge days.
Significantly different from non-binge (NB) control group. *P<0.05; **P<0.01; ***P<0.001.
Summary of effects of lisdexamfetamine on compulsive and perseverative behaviours in lean, female, binge-eating rats.
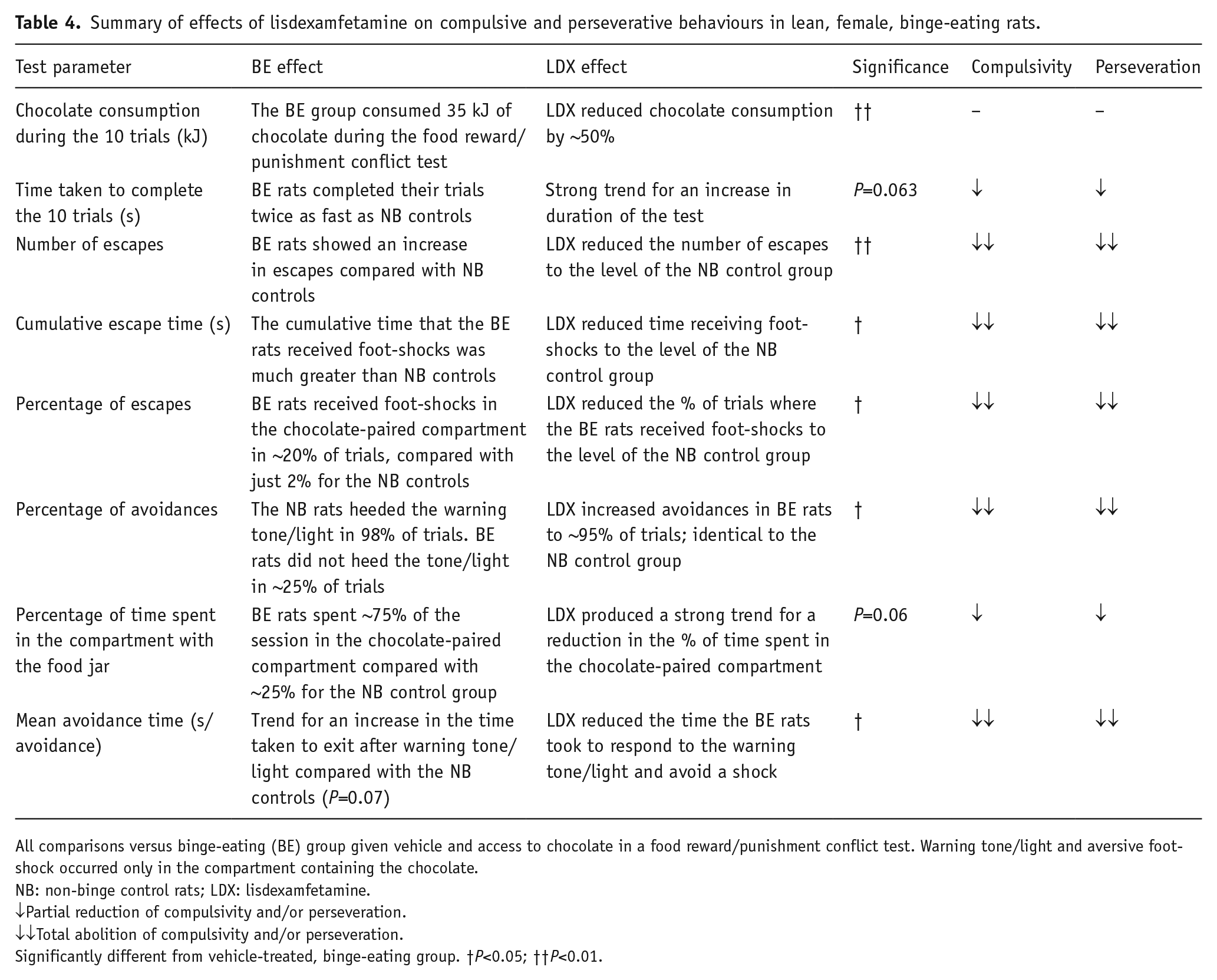
All comparisons versus binge-eating (BE) group given vehicle and access to chocolate in a food reward/punishment conflict test. Warning tone/light and aversive foot-shock occurred only in the compartment containing the chocolate.
NB: non-binge control rats; LDX: lisdexamfetamine.
↓Partial reduction of compulsivity and/or perseveration.
↓↓Total abolition of compulsivity and/or perseveration.
Significantly different from vehicle-treated, binge-eating group. †P<0.05; ††P<0.01.
There have been several previous attempts to explore compulsivity in binge-eating rats using punished conflict paradigms with varying degrees of success (de Jong et al., 2013; Oswald et al., 2011; Rossetti et al., 2013). De Jong et al. (2013) used restricted access to a palatable food in freely fed, male rats to induce binge eating. Groups of binge-eating and control rats were then trained to lever-press for chocolate rewards on a fixed ratio 5 schedule of food reinforcement. The first lever-press resulted in the presentation of a tone conditioning stimulus and a mild foot-shock was administered to the rats on the third and fourth prior to obtaining the chocolate reward on the fifth lever-press. Although this model had previously been employed to study motivation for cocaine-seeking, the authors reported that even mild foot-shocks suppressed all food-seeking by the rats. De Jong et al. (2013) then switched to quinine adulteration of the palatable food as an alternative to evaluate punished responding in the rats. They observed that quinine adulteration suppressed operant responding for food to the same extent in the binge-eating and control groups of rats. Furthermore, there were no differences in the numbers of active lever-presses made during the time-out periods on fixed ratio testing, or lever-presses made for food rewards on a progressive ratio schedule of reinforcement. When the cohort of binge-eating rats was subdivided, those with the most severe binge eating showed greater operant responding for palatable food when paired with punishment, a result broadly consistent with the present findings.
Rossetti et al. (2013) induced binge eating in adolescent, female rats given free access to normal chow by allowing them restricted access to a palatable diet for a period of seven weeks. The rats were trained to lever-press for food rewards on a fixed ratio 1 schedule of reinforcement. Compulsive and perseverative food-seeking was evaluated on the first and fifth day after withdrawal of the preferred palatable food by pairing operant responding for sweet pellets or the preferred chocolate pellets with moderate foot-shocks in a 30-minute test session. The authors claimed that operant responding for the chocolate pellets in the second punished conflict test was more resilient in the binge-eating rats than the non-binge control group. On the other hand, punished operant responding for sweet pellets was equally suppressed in both groups of rats. Scrutiny of the results revealed a complex picture because in the first test session the rats, which had been trained to binge eat by giving them restricted access to the palatable diet, took approximately 20% fewer chocolate rewards than the non-binge controls, suggesting they were less motivated to consume palatable food under punished conditions than normal rats. Furthermore, in the second punished conflict session, operant responding by the binge-eating group for chocolate pellets was decreased by approximately 35% compared with the first test. Although this reduction was not statistically significant compared with the binge-eating rats’ response in test 1, their decreased responding for chocolate rewards in the first test is a potential confounding factor.
Oswald et al. (2011) investigated food-seeking under punishment in binge-prone and binge-resistant groups of female rats. These populations represent susceptibility rather than established binge-eating behaviour. A two-arm maze was used baited in one arm with normal chow pellets and the other with chocolates (M&Ms). In the acclimatisation sessions, the rats were allowed to retrieve chow pellets or M&Ms from the hoppers without punishment. Under test conditions, access to the palatable food was paired with a foot-shock that increased in intensity with each succeeding day of testing. The binge-prone rats retrieved M&Ms paired with more intense shocks than the binge-resistant group, and a substantial proportion of the group was prepared to retrieve at least one M&M under shock intensities at which the binge-resistant rats had given up. However, the high variability in the responses of both groups restricted significant differences to a small number of observation points.
When viewed overall, the findings from these studies support the hypothesis that binge-eating rats exhibit compulsive and perseverative responding in punished conflict tests, and therefore, are in general agreement with our results from the food reward/punishment conflict test. One key difference in our model was not pairing consumption of the palatable food with punishment, and only administering foot-shocks to the rats that were resident in the chocolate-paired compartment for longer than 10 seconds after the tone/light stimuli had been presented. Also, by testing the rats in blocks of 10 trials, the effect of binge eating and lisdexamfetamine on perseverative responding could be evaluated.
When we created the model, we wanted to draw parallels with what happens to subjects with BED. Normal and BED subjects have free access to palatable foods; however, it is only the latter who lose control and regularly binge on these foods. In our model, the binge-eating rats were initially trained to respond to a tone/light signal by transferring from one chamber of the shuttle box to the other. In the food reward/punishment conflict test, the consequence of not leaving the chocolate-paired chamber after the tone/light signal was to receive a foot-shock (an attempt to simulate adverse sequelae experienced by BED subjects after each binge-eating episode). However, the rats were allowed to consume chocolate for periods of 30 seconds or less with no adverse consequences. Moreover, because the tone/light signal was not paired with immediate punishment, the rat could continue to consume chocolate for a short while after it had been presented. All the rat had to do to avoid punishment was leave before the 10 seconds has elapsed. What differentiates the model from others is moderate chocolate consumption is not punished; only prolonged compulsive feeding on chocolate when the rat ignores a previously learned warning of future adverse consequences. Linking the reward directly to punishment lacks translational validity because eating palatable foods is not bad for subjects with BED, it is not being able to control binges on them which is. Linking the reward directly to the punishment does not differentiate between normal and abnormal feeding behaviour, and in our opinion, it is important to employ a model that differentiates between the two when modelling binge eating. Linking the reward directly to the punishment also brings potential confounders when exploring the actions of novel drug treatments for BED. The Geller Seifter test uses food and the Vogel test uses water in reward/punished conflict tests, which are established models to detect the anxiolytic properties of drugs. Thus, the actions of a drug on food consumption in a model where eating the palatable food is punished could be due to its anxiolytic effects or a reduction in pain threshold. These alternative mechanisms would require an additional raft of control experiments to provide reassurance that they are not confounders for the outcome of the experiment.
Of interest both to the present study and other tests exploring compulsivity, impulsivity and perseveration that involve effects on bingeing on palatable foods by rats or humans is to what extent drug-induced effects are secondary to changes in appetite or satiety?
The binge-eating rats consumed 287 kJ of food during the 24-hour period that included the food reward/punishment conflict test (Table 1), compared with an average of 346 kJ on a day with a two-hour chocolate binge session. Furthermore, the chocolate intake by the binge-eating rats in the brief food reward/punishment conflict test was only 35 kJ (12% of their daily intake) compared with 95 kJ (27.4% of their daily intake) on a chocolate binge day. Therefore, the 24-hour consumption of chocolate and chow on the food reward/punishment conflict test days is not representative of the rats’ feeding pattern on a typical binge-eating day because of their very restricted (average 10 minutes) access to chocolate. In the situation in which rats were not allowed a prolonged chocolate binge, it is not surprising that lisdexamfetamine’s action to reduce food intake (Heal et al., 2013b) produced a small reduction in the 24-hour consumption of normal chow as well as chocolate (Table 1).
Lisdexamfetamine and sibutramine, which is an anti-obesity drug, both decreased food intake of cafeteria-fed, obese, female rats by increasing satiety and reduced their body weights (Heal et al., 2013b). The free-access cafeteria diet that was employed included separate pots of high-fat chow, ground salted peanuts and ground milk chocolate (as used to induce binge eating). However, in the behaviourally abnormal, binge-eating rats, only lisdexamfetamine selectively reduced chocolate bingeing without affecting normal chow intake; sibutramine dose-dependently decreased the consumption of palatable and normal chow in the model (Vickers et al., 2015). These results demonstrate that the ability to decrease palatable food consumption in normal rats does not confer an ability to reduce binge eating. Further evidence for this hypothesis comes from a study that we have very recently reported showing that when groups of binge-eating and control rats were trained to lever-press for chocolate pellets in a delay-discounting task (one lever delivered a single pellet immediately and the other lever three pellets, but after increasing delay intervals), the binge-eating rats were intolerant of delayed gratification and responded impulsively for the immediate smaller reward and consumed fewer pellets than the controls as a result (Hutson et al., 2015). Lisdexamfetamine (0.8 mg/kg po), which is the dose we used in the food reward/punishment conflict test, abolished the intolerance of delayed reward in binge-eating rats. Rather than decreasing the consumption of chocolate pellets by binge-eating rats, lisdexamfetamine increased the consumption of chocolate pellets by the binge-eating rats to level of the non-binge-eating control group (Hutson et al., 2015). Evidence from clinical sources suggests that effects of drugs on the psychopathology of binge eating are independent of effects on food intake and body weight. In the dose-ranging trial of lisdexamfetamine in BED, subjects receiving the 30 mg/day dose experienced substantial weight loss relative to placebo, but no reduction in the number of binge episodes or binge frequency (McElroy et al., 2015). It was only the higher doses of 50 and 70 mg/day that reduced BED severity (McElroy et al., 2015). Similar separations between the actions of drugs to reduce body weight in overweight or obese subjects, but not clinically improve their BED status have also been reported for bupropion (White and Grilo, 2013), lamotrigine (Guerdjikova et al., 2009) and topiramate (Claudino et al., 2007). As lisdexamfetamine reduces food intake in normal rats as well as having an additional effect to selectively decrease bingeing on palatable foods, the picture is a complex one. However, the effects of lisdexamfetamine directly on compulsive and perseverative behaviours in the food reward/punishment conflict test in addition to chocolate consumption suggest an action of the prodrug on the pathology underlying binge eating rather than a simple secondary consequence of reduced appetite or increased satiety.
McElroy et al. (2016b) have recently published on the effects of lisdexamfetamine on various measures of compulsivity in subjects with BED using data taken from their phase 2, dose-ranging trial. In subjects with BED, lisdexamfetamine significantly reduced the composite score of the Yale–Brown obsessive compulsive scale modified for binge eating (Y-BOCS-BE), and also, the obsessional and compulsive subscales (McElroy et al., 2016b). Together, these findings provide evidence to support the view that increased compulsivity is a core feature of BED and lisdexamfetamine is effective in treating the underlying psychopathology of this disorder. Our findings that binge-eating rats are compulsive in a novel food reward/punishment conflict model and this behaviour is attenuated by lisdexamfetamine mirror the clinical results.
This study has revealed that lisdexamfetamine has the ability to prevent the compulsive and perseverative behaviours underpinning the psychopathology of binge eating, which provide clues to its effectiveness in treating BED. It also suggests that to achieve efficacy in BED in overweight/obese subjects, a drug needs to combine an ability to treat the psychopathology of binge eating with an effect on hunger and/or satiety to produce weight loss and weight loss maintenance.
In summary, a novel food reward/punishment conflict model has been developed. The results obtained using this model provide good evidence to demonstrate that binge-eating rats exhibit compulsive and perseverative responding when given access to their preferred binge food. Even though the binge-eating rats had been trained to recognise that the tone/light stimuli preceded an aversive foot-shock, they were nonetheless prepared to consume chocolate in the compartment where they would receive foot-shocks. The binge-eating rats spent more time in the chocolate-paired compartment even though it initiated the warning tone/light and a possible aversive foot-shock. Finally, these rats received foot-shocks in the chocolate-paired compartment significantly more often than the behaviourally normal, non-binge-eating controls. A single administration of lisdexamfetamine decreased chocolate bingeing in the food reward/punished responding conflict model, and in addition, it normalised some of the compulsive and perverative behaviours of the binge-eating rats suggesting that its actions were not simply due to reduced hunger or appetite.
Footnotes
Acknowledgements
The authors wish to acknowledge that this programme of research was funded by Shire Pharmaceuticals.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: DJ Heal is a full time employee of and a major shareholder in RenaSci Ltd. SP Vickers, S Goddard and RJ Brammer are full time employees of RenaSci Ltd. PH Hutson is a full time employee of Shire and holds stock/stock options in Shire.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by Shire Pharmaceutical Development Ltd, UK.
