Abstract
Background:
Terminally ill patients often have difficulty taking medications. Suitable, minimally invasive, fast-acting administration routes are particularly important. Trials on alternative drug administration routes in palliative care are essential to base therapy decisions on evidence rather than experience.
Aim:
To evaluate the effectiveness of intranasal compared with subcutaneous midazolam for the initial management of terminal agitation in palliative care patients.
Design:
Randomized, investigator-initiated open-label phase-II trial, August 2022–July 2024.
Setting/Participants:
Monocentric trial at the palliative care ward of a German University Hospital. 180 patients were assessed for suitability, 60 patients (median age, 68 years (SD 12.3), 26 female) were randomized 1:1 and analyzed. Patients with terminal agitation fulfilling all eligibility criteria were randomized to midazolam 5 mg administered either intranasal (n = 30) or subcutaneous (n = 30). Primary outcome: Improvement of terminal agitation by midazolam administration assessed by RASS-PAL-score. Secondary outcome: Midazolam plasma concentrations after administration.
Results:
Median RASS-PAL-scores decreased significantly in both groups. Intranasal group: 2 at baseline, to –1 at 5 min and –2 at 20 min. Subcutaneous group: 1,–0 and –1. Within-group reductions exceeded the clinically relevant threshold of ⩾ 1 point at 5 and 20 min (all p < 0.0001). Median midazolam plasma concentrations were significantly higher intranasal (5 min: 90 ng/ml, 20 min: 83 ng/ml) than subcutaneous (5 min: 15 ng/ml, 20 min: 24 ng/ml). No patients withdrawn due to adverse effects.
Conclusion:
Midazolam intranasal administration was effective in reducing terminal agitation and achieves higher plasma concentrations compared to subcutaneous administration. It is an alternative route of application to improve end-of-life care for terminally ill patients.
Trial Registration:
MinTU Study 173-01, Eudra CT No.: 2021-004789-36, DRKS ID: 00026775 (07.07.2022, https://drks.de/search/de/trial/DRKS00026775).
Midazolam is used intravenously or subcutaneously for terminal agitation, but intranasally in other indications.
Alternative routes of administration are essential for optimal end-of-life care when standard routes are no longer feasible. Therapies need to be easy to use, fast-acting and minimally invasive.
Currently, there is no clinical evidence or research demonstrating the efficacy of intranasal midazolam in the management of terminal agitation.
This study provides evidence that intranasal midazolam
is effective in treatment for terminal agitation,
shows comparable clinical effectiveness to subcutaneous administration,
rapidly achieves therapeutic plasma concentrations.
Midazolam intranasal represents an alternative route of administration that can support high-quality end-of-life care, particularly in outpatient settings, by caregivers and in hospices.
Introduction
Terminal agitation is a distressing and frequently observed symptom during the dying process. 1 It is often characterized by psychomotor and/or inner restlessness and anxiety, which may be intensified by existential distress as patients confronted with death. 2 This constellation of symptoms represents a substantial clinical challenge in palliative care, particularly at the end-of-life. Ensuring effective symptom management is essential to uphold patient comfort, reduce suffering and maintain dignity. 3
Medication administration in palliative care is particularly challenging at the end-of-life as conventional routes, such as oral or intravenous administration, may no longer be feasible due to poor vein access or patient discomfort, especially in hospice and home care.4,5 In such scenarios, alternative routes of drug administration become essential to ensure prompt, effective and minimally burdensome symptom control. Despite their clinical importance, these alternatives are frequently based on empirical experience rather than on evident data from clinical trials. 6 The scarcity of clinical trials at the end-of-life, due to ethical, regulatory and financial constraints may be responsible for this data gap.7,8 As a result, palliative care trials are generally reliant on investigator-initiated trials. In a placebo-controlled trial evaluating intravenous lorazepam in combination with haloperidol for agitated delirium in patients with advanced cancer, 9 as well as in a randomized clinical trial investigating proportional sedation for persistent agitated delirium in palliative care, 10 the feasibility and clinical utility of the Richmond Agitation Sedation Scale-Palliative Version (RASS-PAL-score) 11 were demonstrated, along with the successful conduct of placebo-controlled trials within palliative care populations. These trials highlight that rigorous clinical research, including randomized and placebo-controlled designs, is possible even in this vulnerable patient group. Despite these important advances, there remains a clear need to explore less invasive treatment alternatives, particularly for patients in the terminal phase who may benefit from simplified or non-intravenous approaches. The present study aims to contribute to this expanding evidence base by investigating an alternative route of administration.
The subcutaneous (SC) route is commonly used in clinical practice. However, it can be invasive and distressing, particularly when repeated administrations are required. 12 This has led to growing interest in less invasive alternatives such as intranasal (IN) administration.3,13 Midazolam, a short-acting benzodiazepine, is widely used in palliative care for multiple indications due to its rapid onset of action and established efficacy14,15 as well as ease of use and less invasive character. The safety and efficacy of midazolam IN have been demonstrated in various medical contexts, including the treatment of epileptic seizures and procedural sedation.16,17 Nevertheless, its administration in the management of terminal agitation in the palliative care setting has not been investigated within prospective studies. Therefore, the present trial aimed to evaluate that midazolam as ready-to-use nasal spray can effectively reduce terminal agitation.
Methods
Trial design
This was a randomized, controlled, open-label, monocenter phase-II clinical trial. Details of the protocol and the statistical analysis plan have been published earlier. 18 Approval was obtained from the Federal Institute for Drugs and Medical Devices (4045382, 29.06.2022) and the Ethics Committee II of the Medical Faculty Mannheim, University of Heidelberg (2022-4 monocentric, 15.06.2022, Amendment 31.01.2023). The trial was registered in the German Clinical Trials Register (DRKS ID: 00026775).
Ethical considerations
The trial was conducted in accordance with the Declaration of Helsinki and Good Clinical Practice guidelines. Written informed consent was obtained from all participants.
Setting
The trial was conducted on the palliative care ward of the University Hospital Mannheim, Germany.
Population
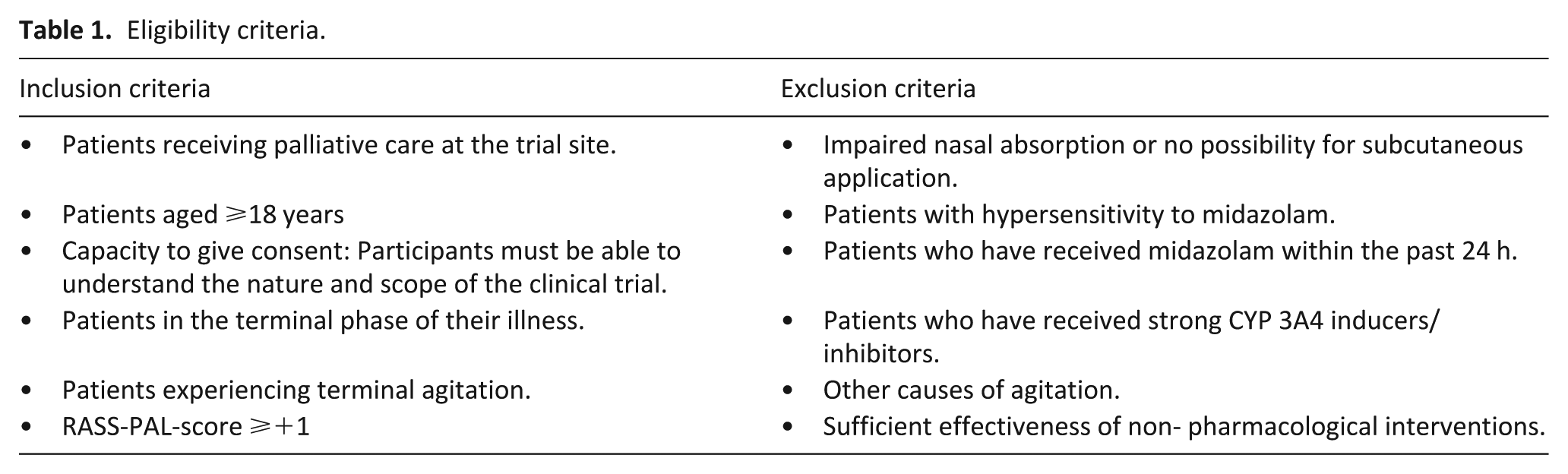
According to requirements of the federal competent authority, only patients capable of giving informed consent, assessed by trained physicians according to routine procedures, were permitted to be enrolled. Inclusion and exclusion criteria are listed in Table 1.
Eligibility criteria.
Recruitment
No active recruitment was performed. After admission to the palliative care ward, all patients were asked to give informed consent. Only those who developed terminal agitation during their terminal illness were randomized and treated.
Sampling
As the development of terminal agitation cannot be predicted at the time of admission, all patients assessed as being in the terminal phase were informed about the study and asked to provide written informed consent. After consent, general eligibility criteria were assessed (screening 1), and eligible patients were observed prospectively. If terminal agitation subsequently developed, agitation-specific eligibility criteria were confirmed in a second screening step (screening 2), followed by randomization to midazolam IN or SC. This two-step screening procedure explains the high number of patients who were screened but did not receive treatment, as many did not develop terminal agitation. Only patients randomized after screening 2 contributed to the trial data and analyses.
Intervention
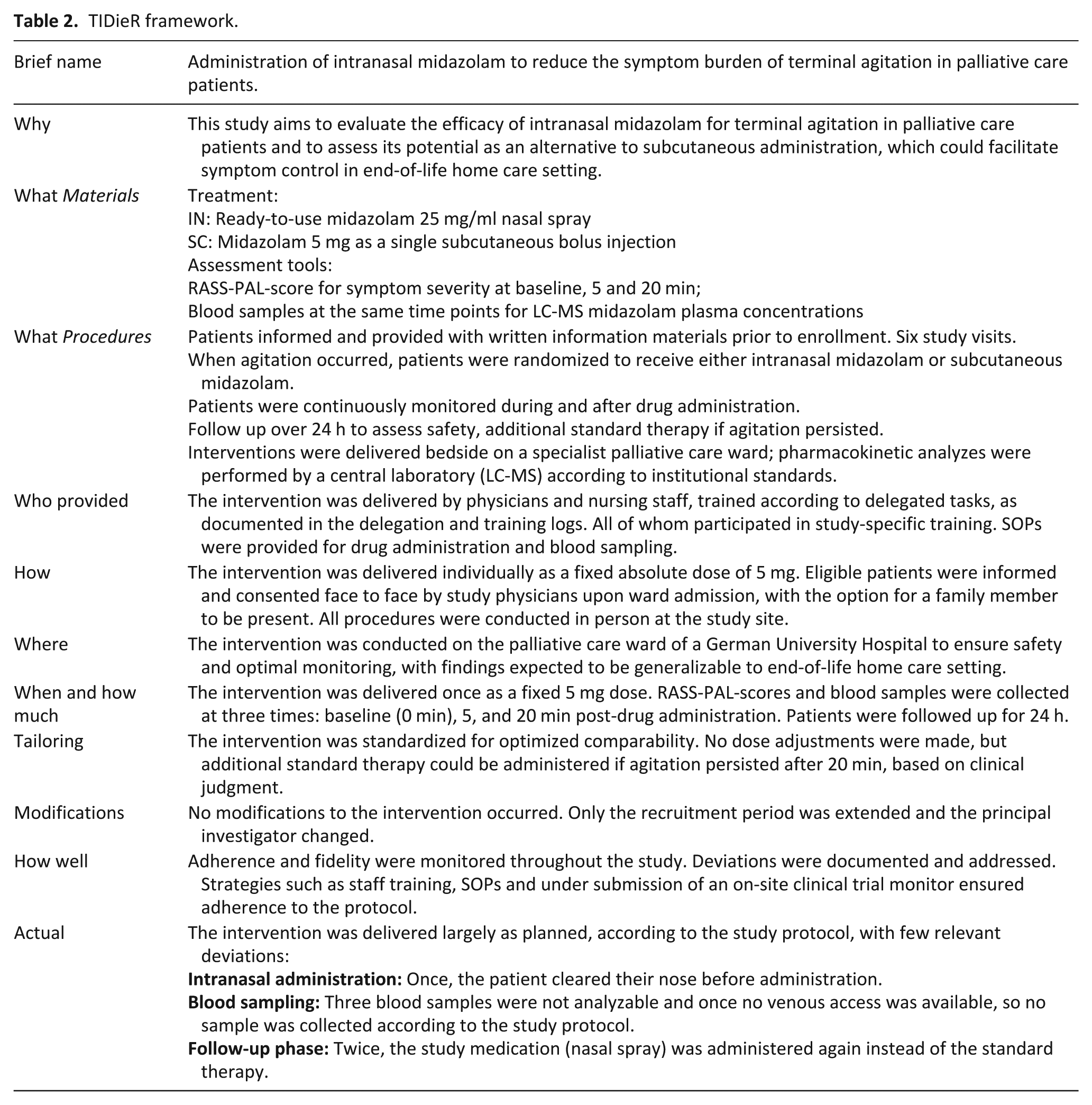
See the TIDieR framework in Table 2 as detailed description of the study intervention.
TIDieR framework.
The trial was conducted open-label. Due to differing routes of administration, a double-dummy design was avoided to minimize patient burden. To prevent withholding effective end-of-life symptom relief, no placebo control was used.
Once patients began to develop terminal agitation, screening 2 was initiated. Agitation was defined as RASS-PAL-score ⩾1, after exclusion of other causes and unsuccessful non-pharmacological interventions, resulted in patients being randomized into the two treatment arms (IN vs SC). After randomization, study treatment per protocol was immediately initiated with a single dose of 5 mg midazolam either IN (treatment group; 2 sprays per each nostril) or SC (control group; bolus injection). The ready-to-use nasal spray (25 mg/ml) was prepared by the pharmacy of the University Hospital Erlangen. Reduction of symptom burden was assessed using the RASS-PAL-score at baseline (prior to drug administration), 5 and 20 min after midazolam administration to assess symptom severity. Blood samples were taken at these time points for pharmacokinetic analysis using Liquid Chromatography-Mass Spectrometry (LC-MS). After 20 min, subsequent treatment was provided according to standard clinical practice, if required. Due to the limited life expectancy of the patient population, the follow-up period was restricted to 24 h (see Figure 1). Patients who did not develop agitation during inpatient treatment were classified as screening failures.
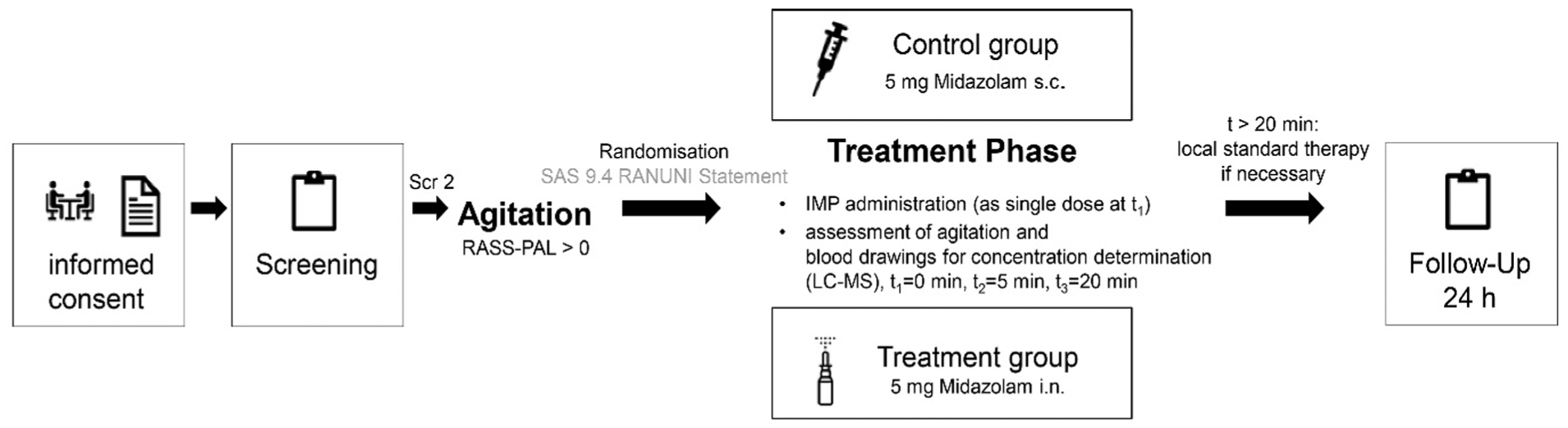
Trial flow chart.
Data collection
Data collection was carried out using study-specific paper case report forms. The data collected during the course includes demographic patient information, details of treatment and symptom progression, data from the observation sheet, laboratory parameters and the quantitative analytical evaluation of the blood samples to determine the level of midazolam plasma concentration. The planned blood samples were taken exclusively via existing intravenous indwelling catheters according to the standard operating procedures of the trial site. If no catheter was available, no blood samples were taken and this data was missing.
Data verification was performed by an on-site monitor in accordance with the trial specific monitoring plan. Data collection plan is depicted in Table 3.
SPIRIT table: data collection plan.
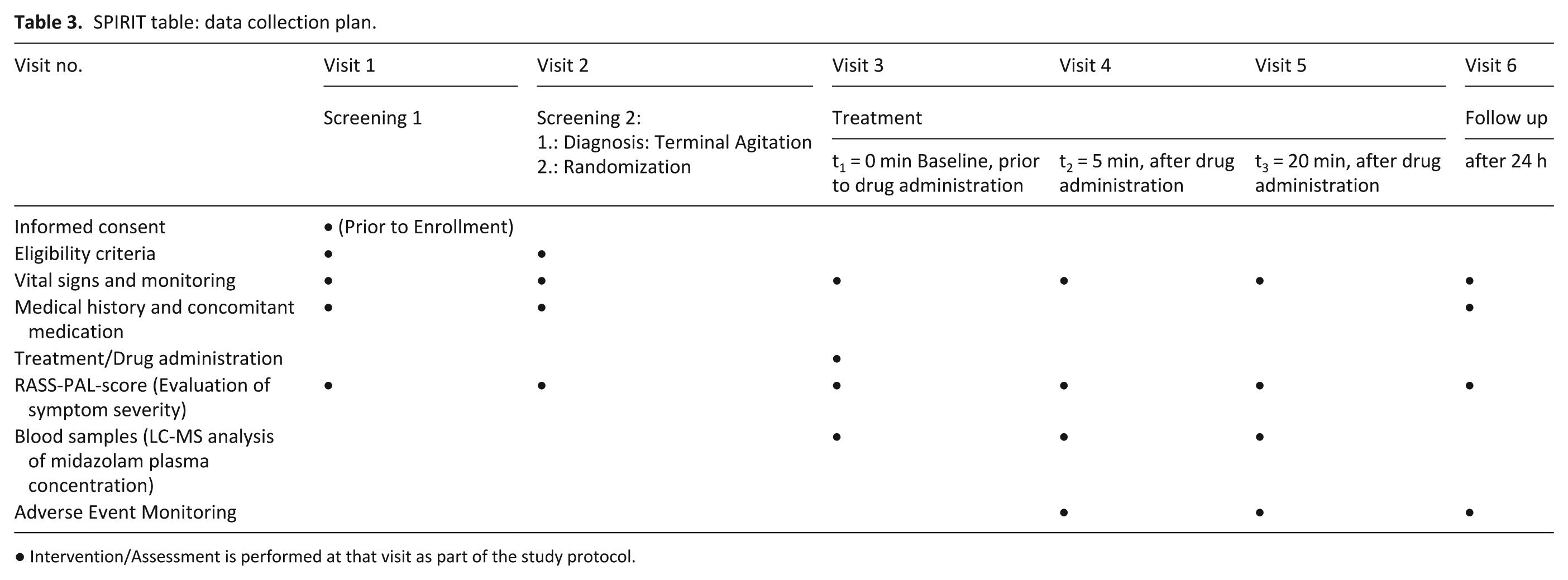
● Intervention/Assessment is performed at that visit as part of the study protocol.
Randomization
Patients were randomly assigned in a 1:1 ratio using a computer-generated randomization list (RANUNI function, SAS 9.4), created by the Department of Medical Statistics, Biomathematics and Information Processing of the Medical Faculty Mannheim, University of Heidelberg.
Outcomes
Primary outcome was the severity of agitation symptoms before and after drug administration (5 and 20 min) using the RASS-PAL-score. Improvement by at least one point between two observation times is defined as treatment efficacy. One-point reduction in RASS-PAL-score was defined as sufficient, as the aim of the trial was to demonstrate effectiveness through the reduction of symptom burden at its onset. Further treatment needs beyond this initial effect were not included in the investigation.
Secondary outcome was the detection of a midazolam plasma concentration ⩾30 ng/ml at t3 = 20 min after the first midazolam administration, regardless of the administration route (IN vs SC) to determine therapeutic plasma concentrations. Quantitative analysis of blood samples after t2 = 5 min, to determine whether midazolam is absorbed at least as quickly after IN administration as after SC administration.
All adverse events (AEs) and reactions were documented and monitored throughout the follow-up period as they occurred. For safety parameters heart rate and respiratory rate were monitored.
Sample size
Due to the lack of previous trials on this topic, it was not possible to estimate the exact distribution of study outcomes. Therefore it was decided to enroll a total of 60 patients (25 patients in each study arm with a drop-out rate of 20%), analogous to a pilot study. This goes in line with the central limit theorem which states that if the sample size n is sufficiently large (approximately n ⩾25), these mean values will be normally distributed (even if the population is not normally distributed). The two-step screening process allowed for a high number of screening failures without impacting the final analysis.
Statistical analysis
Statistical calculations were performed using SAS 9.4 (SAS Institute Inc), by the statistical institute, following the intention-to-treat principle. Descriptive statistics were used to summarize baseline characteristics and outcome parameters. Depending on the data distribution, results are reported as mean ± standard deviation (SD) or median with interquartile range (IQR). To assess for normal distribution, the Shapiro–Wilk test was applied. Within-group comparisons of repeated measurements (e.g. RASS-PAL-score) were conducted using the Generalized Estimating Equations model. For these analyses the SAS procedure PROC GENMOD has been used. Comparisons between the two groups were performed using either independent-samples t-test (for normally distributed variables) or Wilcoxon-Mann-Whitney test (for non-normally distributed data or ordinal variables, e.g. plasma concentration or RASS-PAL-score). These tests were performed on two sides. The one-sided Wilcoxon test was used to compare plasma concentrations at 30 ng/ml. A p-value < 0.05 was considered statistically significant.
Results
Trial flow and baseline characteristics of participants
From August 2022 to July 2024 180 patients were identified as eligible for participation. 46 patients rejected trial participation, resulting in 134 patients included in the trial (Figure 2) of whom 60 were treated within the trial with either midazolam IN (n = 30) or SC (n = 30). The remaining 74 patients did not receive treatment mainly because they did not meet the inclusion criterion of terminal agitation at screening period 2 (n = 72). Two patients were excluded at screening period 1 because the study team determined that their capacity to provide informed consent was not adequately established. The median time between signing the informed consent and the administration of midazolam in response to agitation within the trial was 72 h (range: 24–624 h).
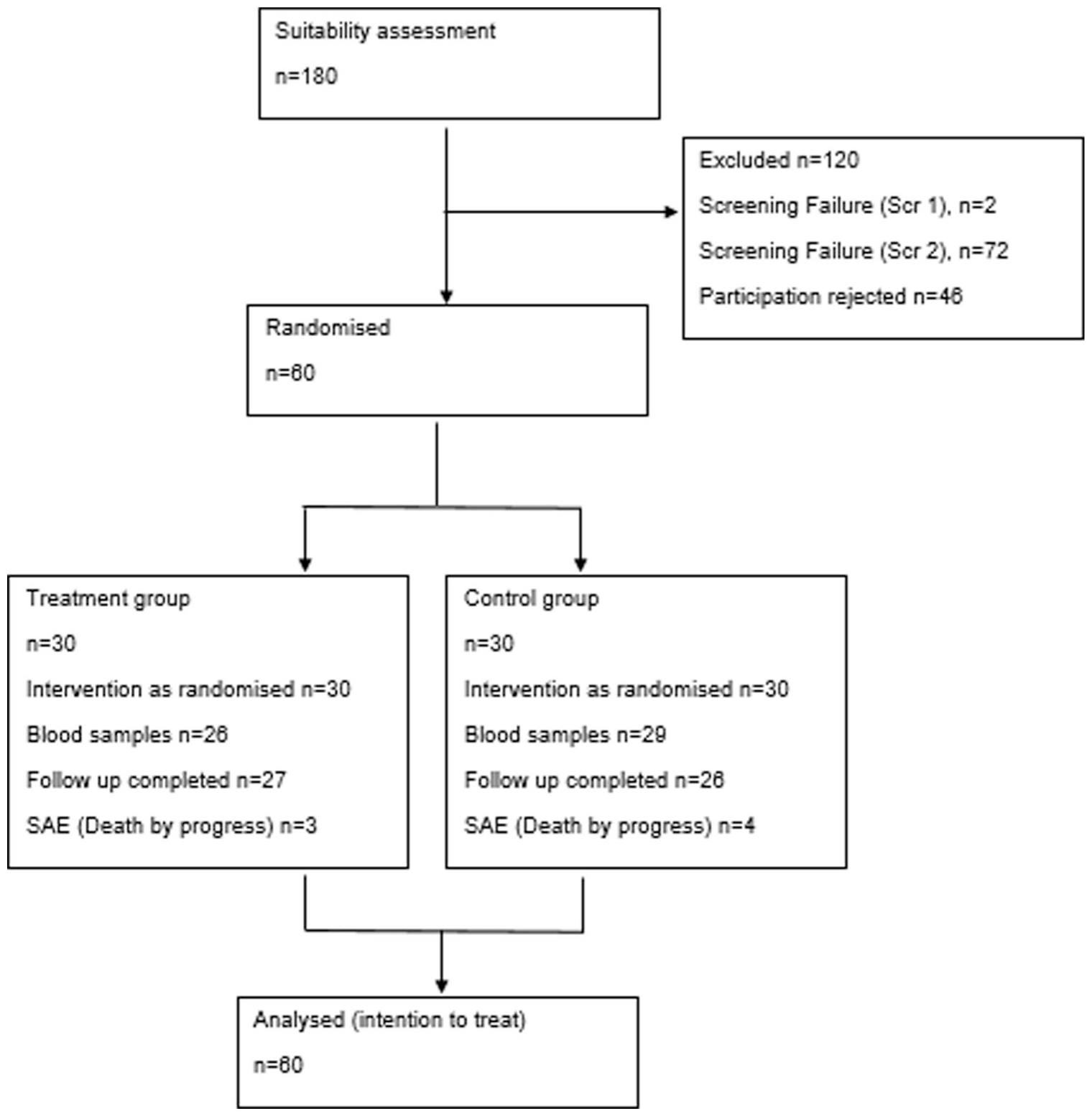
Consort chart of trial flow.
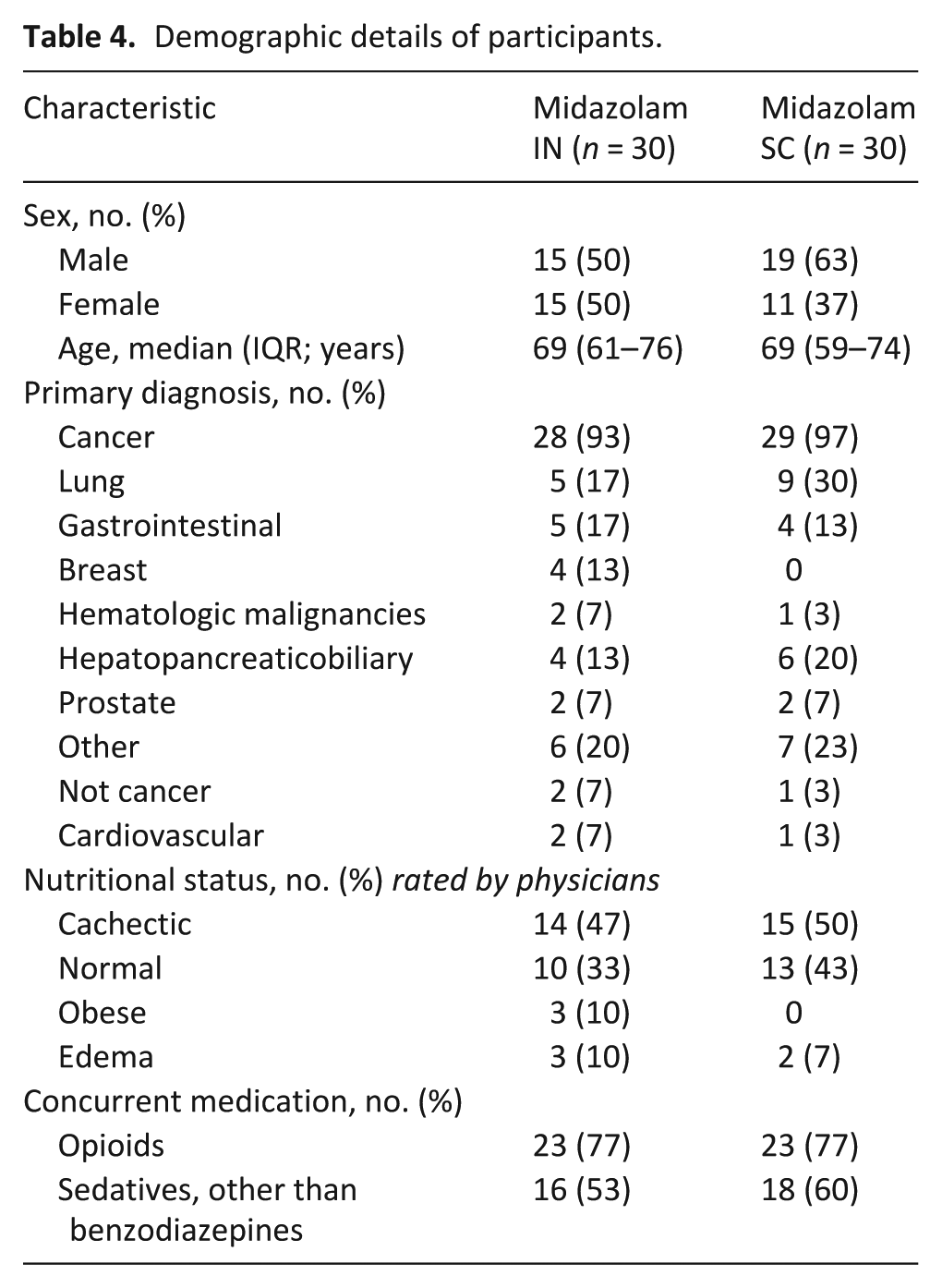
Among all 60 patients, 57 (95%) had cancer as primary diagnosis, with lung cancer being the most common tumor entity (14/57 (25%)). Baseline characteristics for the two treatment groups are presented in Table 4.
Demographic details of participants.
Primary outcome
At baseline, a RASS-PAL-score ⩾2 was observed in 28 patients, including 1 patient with a score of 4. In the intervention group (midazolam IN), the mean RASS-PAL-score decreased by 2.2 points (SD 1.58, 95% CI = 1.61–2.79) after 5 min and by 3.94 points (SD 1.57, 95% CI = 3.35–4.52) after 20 min. Both reductions were statistically significant compared to the clinically relevant threshold of 1 point, as demonstrated by one-sided t-tests (p < 0.0001). In terms of distribution, the median RASS-PAL-score in the IN group decreased from 2 (IQR: 1.5–2.5) at baseline to –1 (IQR: –1.5—0.5) at 5 min and –2 (IQR: –3—1) at 20 min. In the control group (midazolam SC), the mean score decreased by 1.72 points (SD 1.43, 95% CI = 1.23–2.3) after 5 min (p = 0.003) and by 3.2 points (SD 1.45, 95% CI = 2.66–3.74) after 20 min (p < 0.0001), both significantly greater than the threshold of 1 point. Median RASS-PAL-scores in the SC group were 1 (IQR: 0.5–1.5) at baseline, 0 (IQR: –1–1) at 5 min, and –1 (IQR: –2–0) at 20 min. Within-group analysis confirmed a statistically significant reduction in agitation over time for both groups (p < 0.001). A clinically relevant reduction in RASS-PAL-score of ⩾ 1 point was achieved in all patients, in both groups. Notably, there was a trend toward a more pronounced reduction in the IN group after 20 min (p = 0.07). RASS-PAL-scores over time are shown in Figure 3.
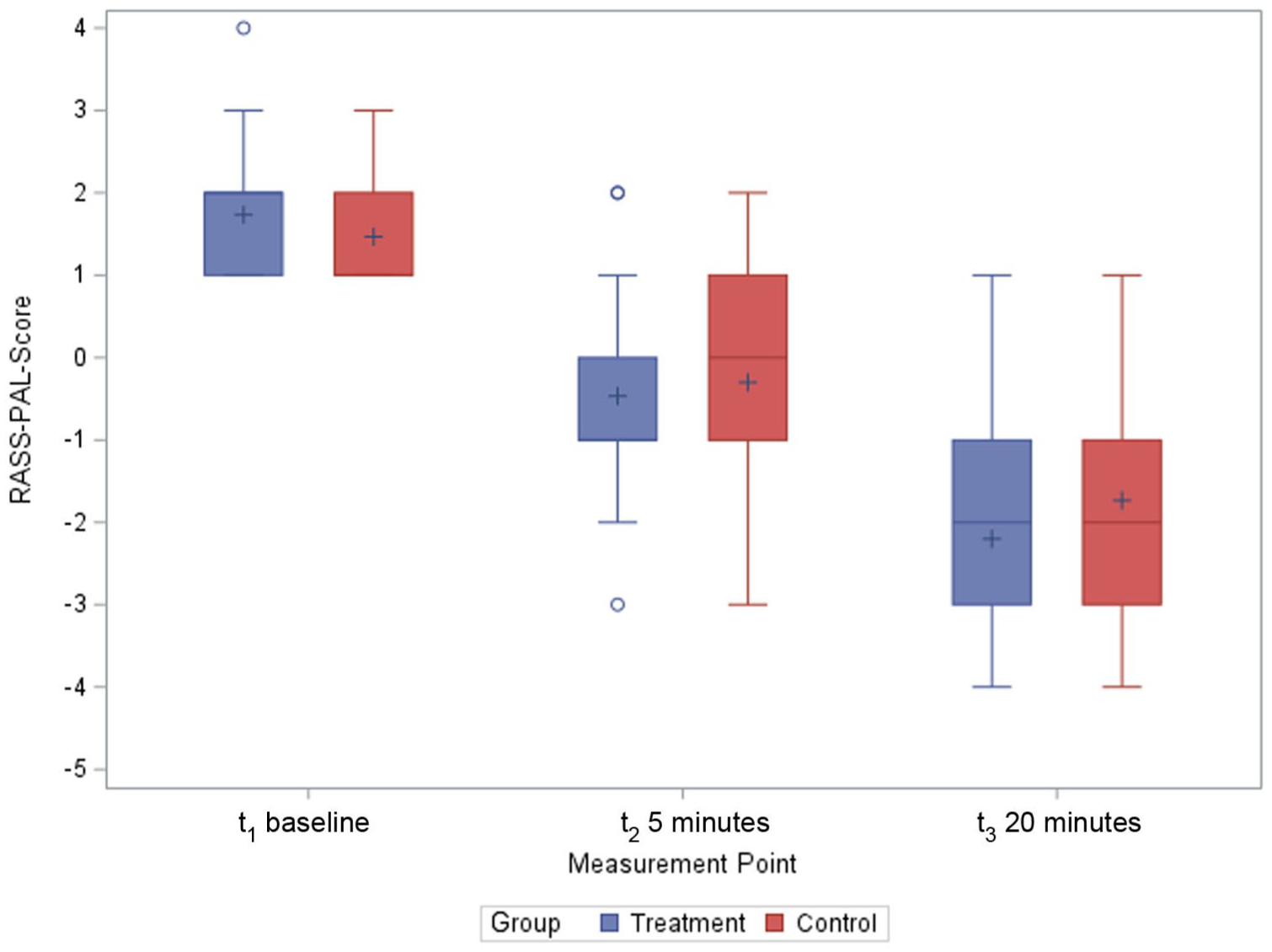
Primary outcome (RASS-PAL-score).
Secondary outcome
As expected, baseline midazolam plasma concentrations were 0 ng/ml in both groups. In the IN group, median plasma concentrations increased to 90 ng/ml (IQR: 43–137) at 5 min and remained elevated at 83 ng/ml (IQR: 44–123) at 20 min, which was significantly higher than the clinically relevant threshold of 30 ng/ml (p < 0.0001). In contrast, patients receiving midazolam SC had substantially lower concentrations: 15 ng/ml (IQR: 3.8–27) at 5 min and 24 ng/ml (IQR: 5.9–41) at 20 min, which was not significantly higher than 30 ng/ml (p = 0.620). Between-group comparisons confirmed significantly higher plasma concentrations in the IN group at both time points (p < 0.0001). Midazolam plasma concentrations over time are shown in Figure 4.
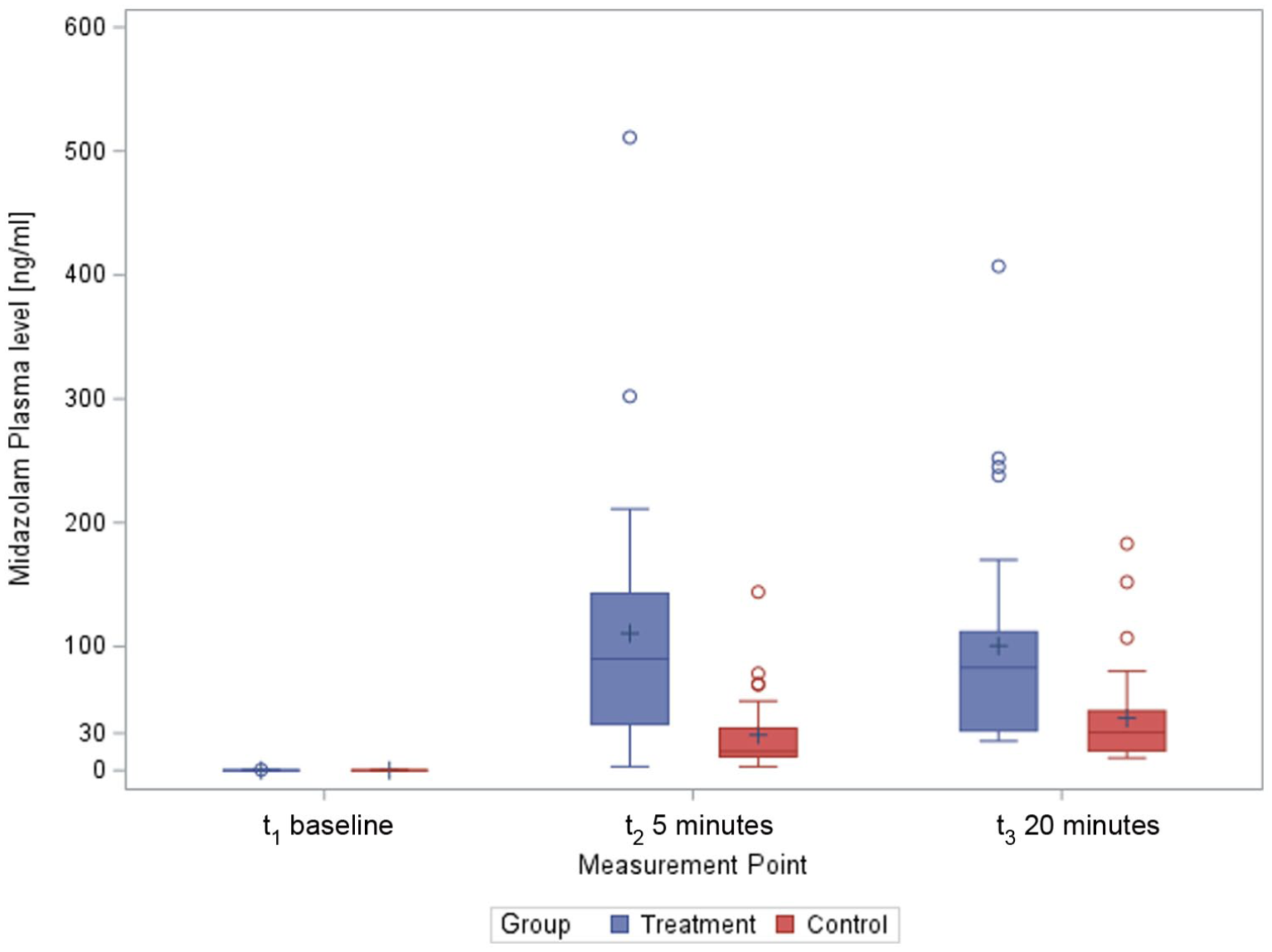
Secondary outcome (Midazolam plasma level).
Reasons for blood sample data missing (5/60, 4 IN, 1 SC) included avoidance of additional burden in accordance with the protocol (in one case (IN) venous access was not available), or deviations in trial conduct (in four cases (3 IN, 1 SC) samples were not evaluable in the lab as they were either improperly stored or could not be retrieved).
Adverse events
Seven patients (3 IN, 4 SC) died during the follow-up period due to progression of the underlying disease. These events were classified as serious AEs but were clearly unrelated to the clinical trial, as determined during the medical review process in accordance with the protocol. No serious treatment-related AEs occurred in either group. The most frequently patient or caregiver reported treatment-related AE was transient irritation of the nasal mucosa (n = 11), which occurred immediately after administration in the midazolam IN group. All cases were of mild severity and resolved spontaneously. In the midazolam SC group, one patient reported mild pain at the injection site and another one experienced a brief respiratory pause. Both events were self-limiting and did not require medical intervention.
Discussion
Main findings
Primary outcome of the trial was the efficacy of midazolam IN compared to SC administration to reduce terminal agitation in palliative care patients. The baseline RASS-PALL-scores were similar between cohorts (median 2 (IN) vs 1 (SC); p = 0.22, not significant). Importantly, the controlled environment at the palliative care ward allowed for early recognition and intervention, which may have contributed to the overall low incidence of severe agitation. The results suggest comparable clinical effectiveness between administration routes, with comparable levels of reducing agitation in terminally ill patients in both treatment arms.
Pharmacokinetic results confirm previous studies on the rapid absorption and bioavailability of midazolam IN.19–21 Blood samples taken at multiple time points revealed a significantly faster and higher increase in midazolam plasma concentrations 5 min after IN administration (with individual outliers showing notably high values) compared to SC administration. After 20 min, plasma concentrations began to decrease or remained stable following IN administration. Whereas SC administration resulted in a slower increase in drug concentrations, which remained lower, possibly due to slower absorption from the subcutaneous tissue into the systemic circulation. This pharmacokinetic profile suggests that midazolam IN may be particularly favorable for the rapid treatment of acute arousal states.
Midazolam plasma concentrations show considerable inter-individual variability as previous studies have indicated.19–21 The pharmacokinetics of midazolam IN have been described in healthy volunteers and in patients with epilepsy.22,23 Our results demonstrate the expected efficacy of midazolam IN and SC administration during the first 20 min, with significantly higher plasma concentrations for the IN route in the initial 5 min. Moreover, there is no universally defined target plasma concentration for sedation. A threshold between 40 and 100 ng/ml is commonly cited.24,25 However, in this trial, achieving sedation was not the primary aim. Rather, symptom relief, such as calming the patient, was considered clinically sufficient. Thus, lower plasma concentrations may have been adequate in this context. The primary outcome was to assess whether midazolam IN and SC is effective in the management of terminal agitation immediately after drug administration with the aim of reducing symptoms and alleviating patients discomfort sufficiently. A single dose of 5 mg midazolam was chosen, which was effective to control terminal agitation in the study population. On this basis, a reduction of one point in the RASS-PAL score was considered sufficient, leaving the option to administer additional doses after the study treatment phase, if needed. Given this context, clinical benefit was reached in all treated patients within the study population. Future studies could investigate the relationship between dose and symptom reduction and establish optimal dosing strategies. Furthermore, therapeutic target values should be defined to guide clinical management.
The less invasive IN approach eliminates the need for painful injections, which could be burdensome for some patients, particularly in cachectic nutritional state. In our trial 29/60 patients were classified as cachectic and in at least one case, SC administration was problematic. In such cases, IN administration may offer a valuable alternative.
Limitations
Limitations must be acknowledged, particularly the methodological decision to administer a fixed 5 mg dose of midazolam to all patients across both treatment arms. This standardized approach was chosen to ensure comparability between groups and reflects common clinical practice, where initial doses typically do not exceed 5 mg. While this enhances the internal validity of the study, it does not allow individualized dose adjustments that may be necessary in cases of severe or persistent agitation. Future studies should consider flexible dosing protocols depending symptom severity to better reflect clinical realities. After the initial 20 min trial window, further treatment was administered at the discretion of the attending physicians and documented accordingly, although it was not included in the statistical analysis. A longer follow-up period with repeated dosing as clinically indicated could provide additional insights into the long-term efficacy and safety profile of midazolam IN. Moreover, the relatively short observation period may have limited the ability to assess the full therapeutic potential of midazolam, particularly in terms of sustained symptom control. However, it is important to emphasize that the primary outcome of this trial was to evaluate the reduction of terminal agitation at its onset, not long-term management. Within this timeframe, meaningful and statistically significant reductions in agitation were observed in both treatment groups, supporting the rapid effectiveness of midazolam, particularly via the IN route. Importantly, no significant differences were observed in depth of sedation or recovery of vigilance between the two administration routes, indicating that midazolam IN, despite higher plasma concentrations, does not result in clinically relevant prolonged sedation or undesirable hangover effects during the trial. This suggests that clinically meaningful effects were achieved regardless of pharmacokinetic variations. Additionally, it indicates that the trend toward deeper sedation at 20 min did not persist over time.
The trial was conducted in a controlled inpatient setting, which may not fully reflect the conditions in home-based palliative care. To ensure patient safety and data integrity, this trial was conducted within the setting of a specialized palliative care unit. While this allowed for rigorous monitoring and precise data collection, it also introduced the limitation of reduced relevance of a simplified administration route in a hospital setting, where intravenous access is commonly available. Previous studies have shown that caregiving self-efficacy, defined as a caregiver’s confidence in managing care-related tasks, is closely linked to reduced caregiver burden and improved psychological well-being. 26 By providing an easily applicable alternative to midazolam SC, IN administration may alleviate caregiver distress and enhance their ability to manage terminal agitation effectively.13,27
Future research may incorporate caregiver-reported outcomes to better understand the impact of midazolam IN on family distress and feasibility of administration in home care settings.
Because no prior investigation had evaluated the use of midazolam IN in palliative care patients with terminal agitation, no sufficiently robust data were available to support a reliable sample-size calculation. Consequently, the trial was not powered to function as a formal non-inferiority trial. Nevertheless, the results provide the empirical rationale to justify subsequent trials to definitively test and statistically confirm, the non-inferiority of midazolam IN versus SC.
Finally, the trial was conducted not placebo-controlled. Given the ethical implications, withholding effective treatment from patients experiencing acute terminal agitation was not deemed acceptable. Instead the study compared two active treatment strategies both aligned with current palliative care standards.13,28,29
Implications for practice
In this trial, we demonstrated that midazolam IN is a valid therapeutic option for terminally agitated palliative care patients to reduce terminal agitation. Furthermore, the data suggest, that midazolam IN is at least equally effective as midazolam SC. Given its less invasive nature and ease of use, midazolam IN has the potential to facilitate end-of-life care, particularly in hospice or home care settings.
Availability of any formulation of midazolam should not pose a problem. Although midazolam IN is not commercially authorized (except in the US for another indication 30 ) and is supplied as an individually prescribed, pharmacy-prepared formulation. 31 Costs are expected to remain moderate if compounded by public pharmacies.
Conclusion
In conclusion, the trial implicates that midazolam IN is an effective treatment option for terminal agitation in palliative care patients. Its ease of use, rapid onset of action and less invasiveness makes it suitable for home-based care, where it can empower caregivers and improve the quality of care.
Supplemental Material
sj-xlsx-1-pmj-10.1177_02692163261428947 – Supplemental material for A randomized controlled clinical trial of intranasal versus subcutaneous midazolam for agitation in terminal illness (MinTU study)
Supplemental material, sj-xlsx-1-pmj-10.1177_02692163261428947 for A randomized controlled clinical trial of intranasal versus subcutaneous midazolam for agitation in terminal illness (MinTU study) by Hanna Hirschinger, Constanze Rémi, Evelyn Jäger, Stefanie Nittka, Svetlana Hetjens, Susanne Saussele, Kirsten Merx, Jan Koch, Mohammed Abba, Mahmoud Ghazal, Ralf-D Hofheinz, Thomas Michaeli, Elena Sperk, Katharina Kohlbrenner, Wolf-K Hofmann, Deniz Gencer and Tobias Boch in Palliative Medicine
Footnotes
Acknowledgements
We thank all the patients and their relatives who were willing to participate in this challenging study. We also thank Sandra Porter, Gabriele Bartsch, Janna Kirchhof, Birgit Gryger, the physicians and the palliative care team of the 3rd department of medicine, University Hospital Mannheim, especially Claudius Voss, Marcel Lorch, Christina Reyser, Eric Thanbichler and the pharmacy team of the University Hospital Mannheim.
Ethical considerations
Ethics approval (2022-4, Ethics committee II, Medical Faculty Mannheim, University Heidelberg).
Consent to participate
Written, informed consent to participate was obtained from all participants.
Author contributions
HH and DG designed the trial. TB is principal investigator. EJ and SN performed the LC-MS analysis and analyzed the blood samples. SH performed the statistical analysis. SS, KM, JK, MA, MG, RDH, TM contributed to the operational realization of the trial. HH drafted the manuscript, which was critically revised by the steering committee TB, DG, WKH, RDH, ES, KK, CR and SS. All authors read and approved the final manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors. Investigator initiated trail, without external funding: 3rd Department of Medicine, Medical Faculty Mannheim, University Medical Center Mannheim, Heidelberg, Theodor-Kutzer-Ufer 1-3, D-68167 Mannheim, Germany. For the publication fee we acknowledge financial support by Heidelberg University.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
