Abstract
Background:
Dyspnea is a frequent and distressing symptom in people with cancer. High-flow nasal oxygen has been shown to relieve dyspnea and could offer several advantages over conventional oxygen delivery devices.
Aim:
To assess the effectiveness of high-flow nasal oxygen compared with other medical oxygen or air delivery devices on dyspnea in people with cancer.
Design:
This study was designed as a systematic review and meta-analysis and was registered prospectively with PROSPERO (CRD42021265395).
Data sources:
EMBASE, MEDLINE and Web of Science electronic databases were searched from inception to February 23, 2025. Randomized controlled trials and controlled clinical trials evaluating the effectiveness of high-flow nasal oxygen in adults with cancer were screened, and data were independently extracted by three authors. The primary outcome was the mean change in dyspnea scores.
Results:
Seven trials (374 patients) met the inclusion criteria. Six trials (272 patients) were included in the meta-analysis. Most trials were at a high risk of bias or had some concerns. The meta-analysis showed that high-flow nasal oxygen significantly improved dyspnea compared to other medical oxygen or air delivery devices (SMD: −0.60; 95% CI: −1.02 to −0.17; I2 = 65%, p < 0.014). Sub-group analysis showed that the improvement was only visible in hypoxemic patients (SMD: –0.87; 95% CI —1.33 to —0.40; I2 = 58.7%, p = 0.089).
Conclusion:
High-flow nasal oxygen could be an additional therapeutic option for alleviating dyspnea in hospitalized people with advanced cancer.
Persistent dyspnea is a distressing symptom in people with oncological diseases and therapeutic options are scarce.
High-flow nasal oxygen is mostly used for the treatment of acute respiratory failure.
The effectiveness of high-flow nasal oxygen on dyspnea is seldom evaluated.
High-flow nasal oxygen can be effective in managing persistent dyspnea among hospitalized people with cancer.
Sub-group analysis showed that the benefit was confined to hypoxemic patients with no significant improvement in non-hypoxemic individuals.
High-flow nasal oxygen could be considered as a supportive intervention to reduce dyspnea in hospitalized people with cancer.
Further research is needed to evaluate its influence on peoples’ overall quality of life and its role within the broader goals of care.
Introduction
Dyspnea, a common symptom in people suffering from cancer, has been shown to increase toward the end of life and is often difficult to alleviate.1,2 Dyspnea has been defined as a subjective experience of breathing discomfort, which consists of qualitatively distinct sensations that vary with intensity. 3 The prevalence of dyspnea greatly depends on the definition and scale used for its assessment, diagnosis, co-morbidities and time of assessment, with up to 75% of people with lung cancer suffering from this symptom at some point during their illness. 4
In contrast to other symptoms like pain, the mechanisms behind dyspnea are less understood. It is also a complex symptom to treat as objective signs often do not correlate with the person’s perception of dyspnea and cannot be predicted by pulmonary function testing. 3 Pharmacological management of this symptom relies mainly on the use of opioids. However, certain populations, such as people with heart failure or chronic obstructive pulmonary disease, do not seem to benefit from this therapeutic option.5,6
High-flow nasal oxygen (HFNO), also known as high-flow nasal cannula oxygen therapy or simply high-flow oxygen, is used primarily in patients with acute hypoxemic respiratory failure.2,7 Nevertheless, this device could offer several advantages over standard oxygen in people with cancer suffering from dyspnea. Indeed, high-flow nasal oxygen is an oxygen delivery device that provides up to 100 l/min of heated, humidified oxygen through a nasal cannula with a fraction of inhaled oxygen that can be increased from 21% to 100%. 8 Studies have shown that this device also induces nasopharyngeal washout and positive distending pressure and decreases airway resistance thus the metabolic cost of breathing. 9 It may also alleviate dyspnea by acting on the trigeminal nerve, as with the hand held fan. 10
However, the evidence of its effectiveness on alleviating dyspnea in people with cancer is inconsistent. 11 Therefore, we conducted this systematic review to examine the effectiveness of high-flow nasal oxygen compared to other respiratory therapies in the management of dyspnea in adults suffering from cancer.
Methods
We conducted a systematic review and meta-analysis, reported using the Preferred Reporting Items for Systematic Reviews and Meta-Analysis statement (PRISMA). 12 This study was registered prospectively with Prospero (CRD42021265395). Two deviations from the preregistered protocol occurred. First, although the protocol specified searches in five databases, we restricted the search to three due to substantial overlap in coverage. Second, although the protocol anticipated that a meta-analysis would not be feasible following a scoping review, the final number of included studies allowed for quantitative synthesis, and a meta-analysis was therefore conducted. All amendments were documented in PROSPERO.
Data sources and search strategy
In this systematic review and meta-analysis, MEDLINE (Ovid), EMBASE (Ovid) and Web of Science were searched from inception to November 30rd 2023. A search update was performed on February 23 2025 and found no other studies. These databases were searched using a combination of subject heading and keywords, reported separately (Supplemental Figure S1). The search strategy contained the terms related to the intervention, “oxygen therapy”, as well as terms related to “dyspnea” and “cancer.” Citation searching was performed for all included studies.
Study selection
Studies were included if they met the following criteria: (1) included more than 80% of adult patients with cancer, (2) compared high-flow nasal oxygen vs any other medical air or oxygen delivery device (i.e. excluding the handheld fan), (3) reported dyspnea with a validated scale, (4) took place in the hospital setting, (5) were written in the English language, (6) had a randomized controlled or a controlled clinical trial design. Reference selection process was performed using Rayyan QCRI, a specific tool designed to help authors performing systematic reviews. 13 Referencing in documents was managed using Zotero (version 4.0.29.15).
Three authors (LH, SC, TFS) independently screened titles of all citations identified by electronic and hand-search and classified them as included, unclear, excluded or duplicate of another citation. For all studies defined as unclear, the authors then screened abstracts and full articles when appropriate and classified them as included or excluded. Reasons for exclusion at this stage was specified. The selection process is presented using the PRISMA flow diagram. 12 For studies in which data on dyspnea was incomplete, study authors were contacted for further results.11,14
Data extraction
A data extraction sheet based on the Data Extraction Template for Included Studies of the Cochrane Consumers and Communication Group 15 was used to collect all data concerning: study design, sample size, patient characteristics such as main diagnosis, presence of hypoxemia, type of control intervention, outcome, and effect measure. The template was piloted with one study and tailored to contain all relevant information. The extraction template can be found in Supplemental Table S1. All data was extracted independently by two authors (SS, IS) and entered into an Excel spread sheet (Microsoft® Excel® 2011, version 14.7.7).
Data synthesis and analysis
We performed a quantitative synthesis using random-effects models to minimize the effect of between-study heterogeneity. The outcome of interest was self-administered dyspnea scores reported as the standardized mean difference (SMD) to account for the use of different dyspnea scales across studies. Analysis was conducted using Hedges’ g which corrects for the upward bias present in Cohen’s d in small samples.16,17 Secondary outcomes were presented as change in median with 25th–75th percentile and change mean with 95% CI or change mean and interquartile range (IQR) as mentioned in individual studies.
To enable a consistent approach to the meta-analysis and enhance interpretation of the findings, we converted estimates when appropriate. We used the DerSimonian-Laird (DL) method to combine summary measures using random-effects models to minimize the effect of between-study heterogeneity. We also reported the estimates using Inverse Variance (IV), which assumes a fixed effect models. We assessed the magnitude of the effect using Cohen’s guidelines for effect size benchmarks which distinguishes a SMD as low (SMD~0.2), moderate (SMD~0.5), or large (SMD~0.8). 18 In the meta-analysis, heterogeneity was assessed using the Cochrane χ2 statistic and the I2 statistic and was distinguished as low (I2 ⩽25%), moderate (25% < I2 <75%) or high (I2 ⩾75%). 19 Since we had fewer than 10 articles in our meta-analysis, we could not perform a meta-regression to evaluate sources of heterogeneity, in order to examine contributing factors. 20 Instead, we performed stratified analysis examining as potential source for the observed heterogeneity whether people had hypoxemia or not, acknowledging that this analysis was not pre-specified. All tests were two-tailed and p-values ⩽ 0.05 were considered significant. STATA release 17 (Stata Corp, College Station, Texas) was used for all statistical analyses.
Quality assessment
All studies were assessed for internal validity using the revised Cochrane risk of bias tool for randomized trials (RoB-2). 21 If one or more individual domains were assessed as having a high risk of bias, the trial was rated as having a high risk of bias. All domains had to be rated as having a low risk of bias for the overall risk of bias to be classified as low. In cases of unclear risk of bias or mixed assessment of low and unclear risk of bias, the overall score was classified as having an unclear risk of bias. Quality assessment followed the Critical Appraisal Skills Program (CASP) guidelines for the quality assessment of RCT and was used to support the risk of bias assessment. 22
Results
Electronic databases identified 469 eligible studies (MEDLINE:36, EMBASE: 272, Web of Science 161). Hand searching identified one more trial included in the analysis. 14 After removing duplicates, 428 citations were retained. Based on title and abstract screening, 19 articles were evaluated in detail. After full-text assessment, we excluded 12 papers. We included seven articles in the systematic review, six of which were also included in the meta-analysis for a total of 272 patients.11,23–27 The reasons for exclusion at the abstract and full text stage are specified in the study PRISMA flow-chart (Figure 1). 28
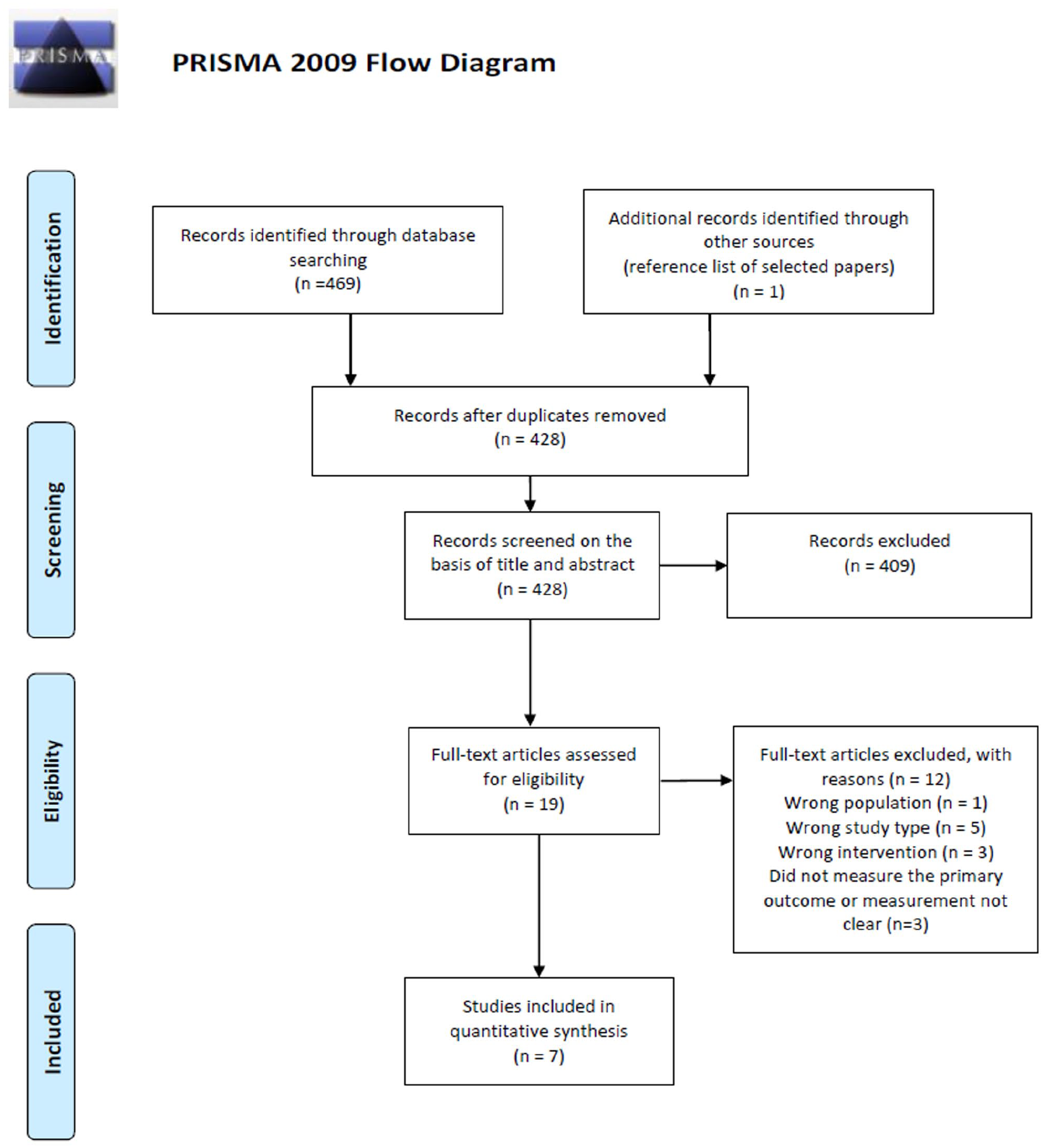
Study PRISMA flow chart of search results.
Description of included studies
Of the seven studies included in this systematic review, all were conducted in the hospital setting. Four studies were set in an acute care unit outside of intensive care unit (ICU) setting (N = 209),14,25–27 another in an ICU (N = 100), 11 and one in the emergency department (N = 44). 23 One study was conducted in an experimental setting (respiratory physiology laboratory; N = 21). 24 Six studies were conducted in a single center,14,23–27 and one was multi-centered. 11 Studies were conducted in the United States of America (N = 3),24–26 France (N = 1), 11 Thailand (N = 1), 23 China (N = 1) 27 and Turkey (N = 1). 14 Table 1 summarizes the study characteristics.
Study characteristics.
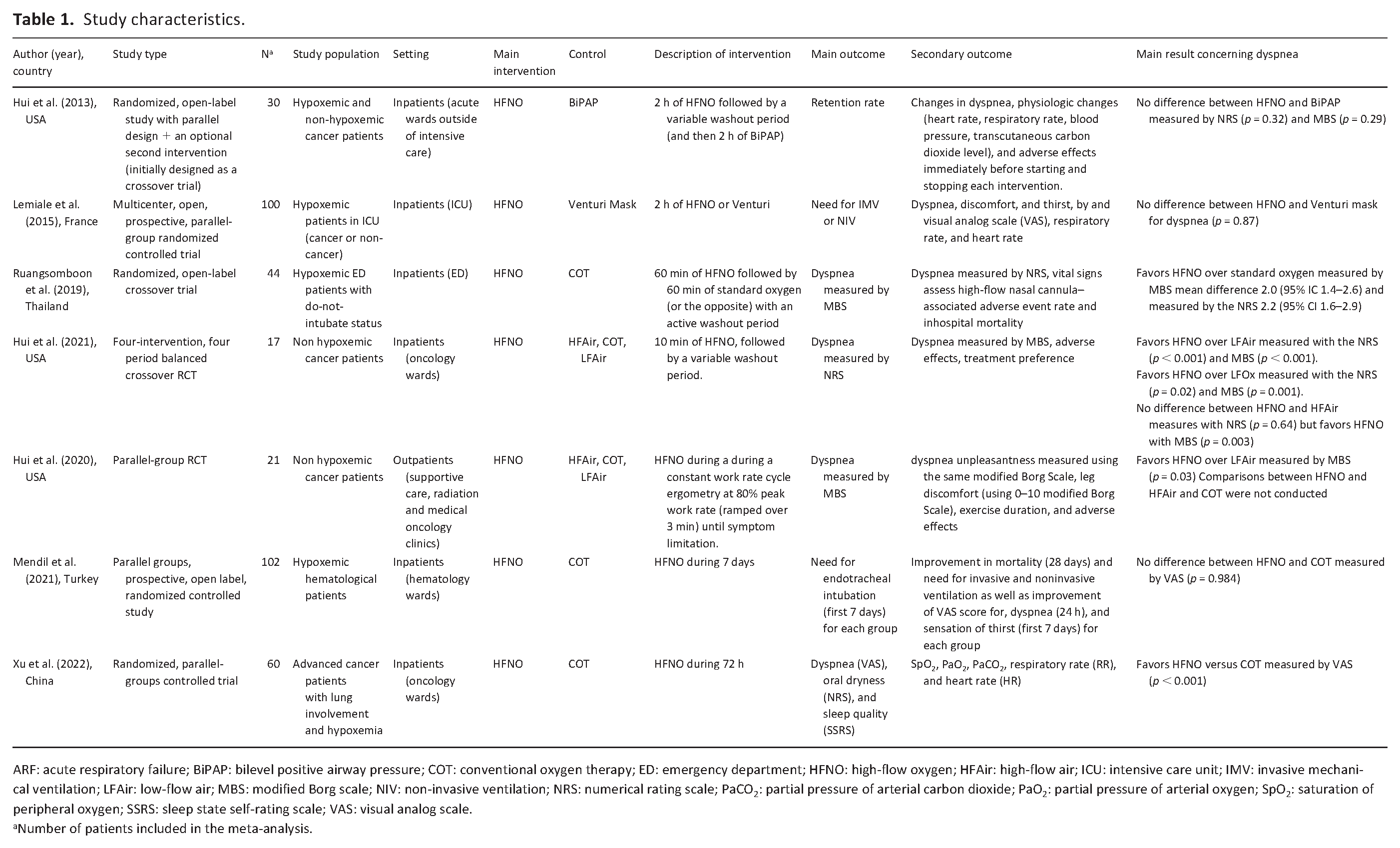
ARF: acute respiratory failure; BiPAP: bilevel positive airway pressure; COT: conventional oxygen therapy; ED: emergency department; HFNO: high-flow oxygen; HFAir: high-flow air; ICU: intensive care unit; IMV: invasive mechanical ventilation; LFAir: low-flow air; MBS: modified Borg scale; NIV: non-invasive ventilation; NRS: numerical rating scale; PaCO2: partial pressure of arterial carbon dioxide; PaO2: partial pressure of arterial oxygen; SpO2: saturation of peripheral oxygen; SSRS: sleep state self-rating scale; VAS: visual analog scale.
Number of patients included in the meta-analysis.
Sample size variated from 19 to 102 participants.14,25 Five studies only included oncology patients,14,24–27 and two studies included mostly oncology patients.11,23 We did not disaggregate data for participants with cancer but used the full data set available. Age groups were similar across studies with a range of mean age going from 51 to 66 years.
Four studies were conducted on hypoxemic patients.11,14,23,27 One study was conducted on a mixed hypoxemic and non-hypoxemic population. 26 Two studies were conducted on non-hypoxemic patients.24,25
One study compared high-flow nasal oxygen to bi-level positive airway pressure (BiPAP). 26 Another compared high-flow nasal oxygen to oxygen delivered via a Venturi mask. 11 Three studies compared high-flow nasal oxygen with conventional oxygen therapy.14,23,27 One study compared high-flow nasal oxygen to conventional oxygen therapy, high-flow air (HFAir) and low-flow air. 25 Finally, the physiological study compared low-flow air to high-flow nasal oxygen, conventional oxygen therapy and high-flow air. 24
In high-flow nasal oxygen groups, the oxygen flow was variable in all studies and generally titrated between 10 and —70 l/min.
Primary outcome
Dyspnea was assessed as a primary outcome in four studies,23–25,27 and a secondary outcome in three studies.11,14,26 Evaluation was conducted using the numerical rating scale (NRS) in two studies,23,25 the modified Borg scale (MBS) in three studies,23–25 and the visual analog scale (VAS) in four studies.11,14,26,27
One study was excluded from the meta-analysis as we were not able to acquire the necessary data. 14 In studies involving multiple comparisons, we chose to analyze only the intervention comparing high-flow nasal oxygen and conventional oxygen therapy.24,25 When NRS and MBS were both reported for the outcome of interest and did not provide the same answers, we chose to compare results using the scale that was pre-defined for the primary outcome of the study. Dyspnea was evaluated at varying time points across studies (5–1440 min),14,25 for consistency, only the longest time point was included in this review (Table 1).
High-flow nasal oxygen was significantly superior to conventional oxygen therapy in three studies and to Venturi mask in one study.11,23,25,27 Overall, the meta-analysis showed that high-flow nasal oxygen significantly improved dyspnea compared to the control group. The pooled standardized mean difference (SMD, Hedges’ g) using random-effect model was of −0.60 (95% CI: −1.02 to −0.17) with a moderate heterogeneity (I2 = 65%, p < 0.014; Figure 2). Among the six included studies in the meta-analysis, the effect sizes varied, with the largest improvement reported by Ruangsomboon et al. (−1.26, 95% CI: −1.89 to −0.62), while the data from two other studies showed a non-significant effect.24,26 The largest study contributed the most weight (36%) and found a moderate and significant improvement (−0.49, 95% CI: −0.89 to −0.10). 11
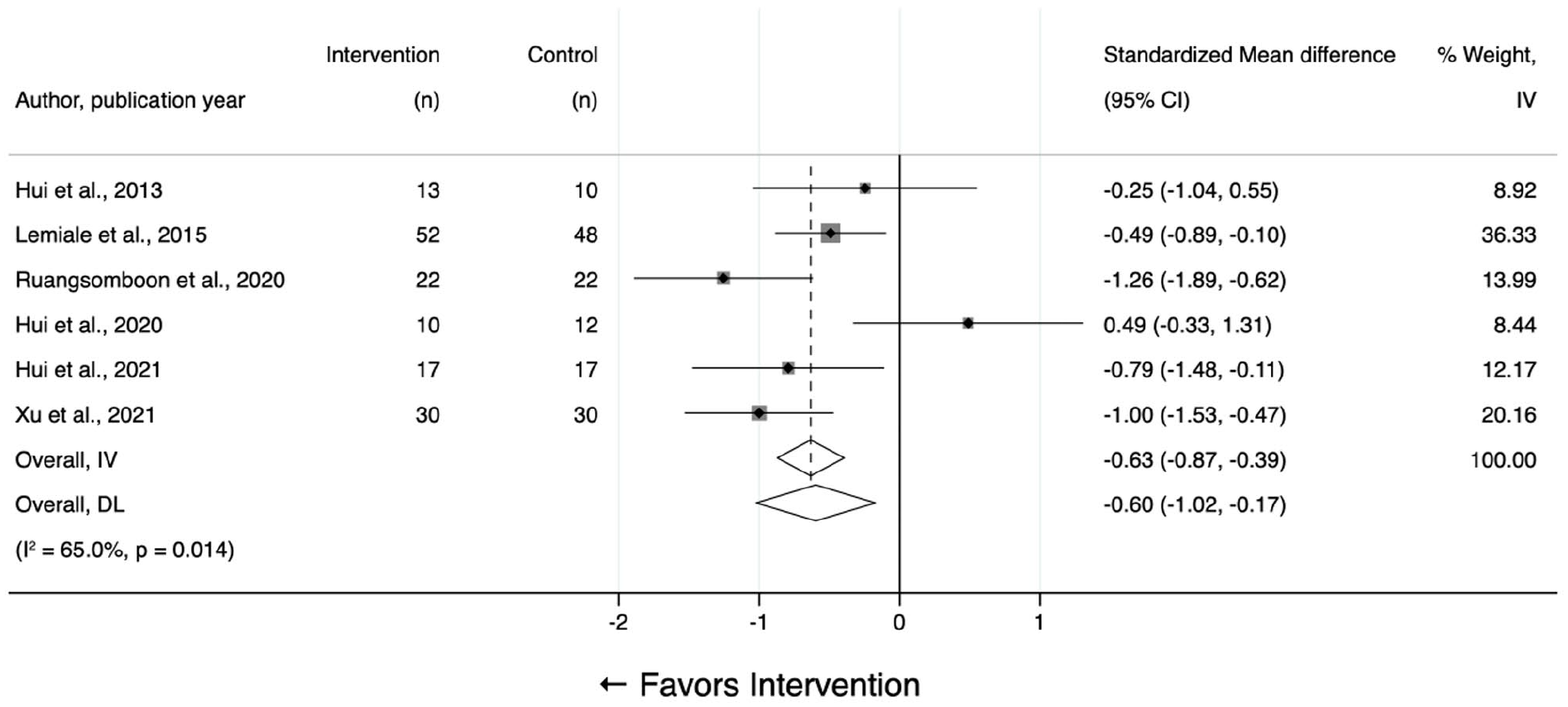
Overall meta-analysis.
Subgroup analyses stratified by hypoxemia status identified clinical differences (Figure 3). Among people without baseline hypoxemia, high-flow oxygen did not significantly reduce dyspnea (SMD: –0.21; 95% CI —0.95 to 0.52; I2 = 63.9%, p = 0.063), indicating only a small and clinically uncertain effect. In contrast, hypoxemic patients experienced a moderate-to-large improvement in dyspnea (SMD: –0.87; 95% CI —1.33 to —0.40; I2 = 58.7%, p = 0.089), 29 corresponding to a 87% probability that high-flow nasal oxygen- treated patients improved more than the average control. The test for subgroup differences was statistically significant (p = 0.048), suggesting baseline hypoxemia modifies the response to high-flow oxygen.
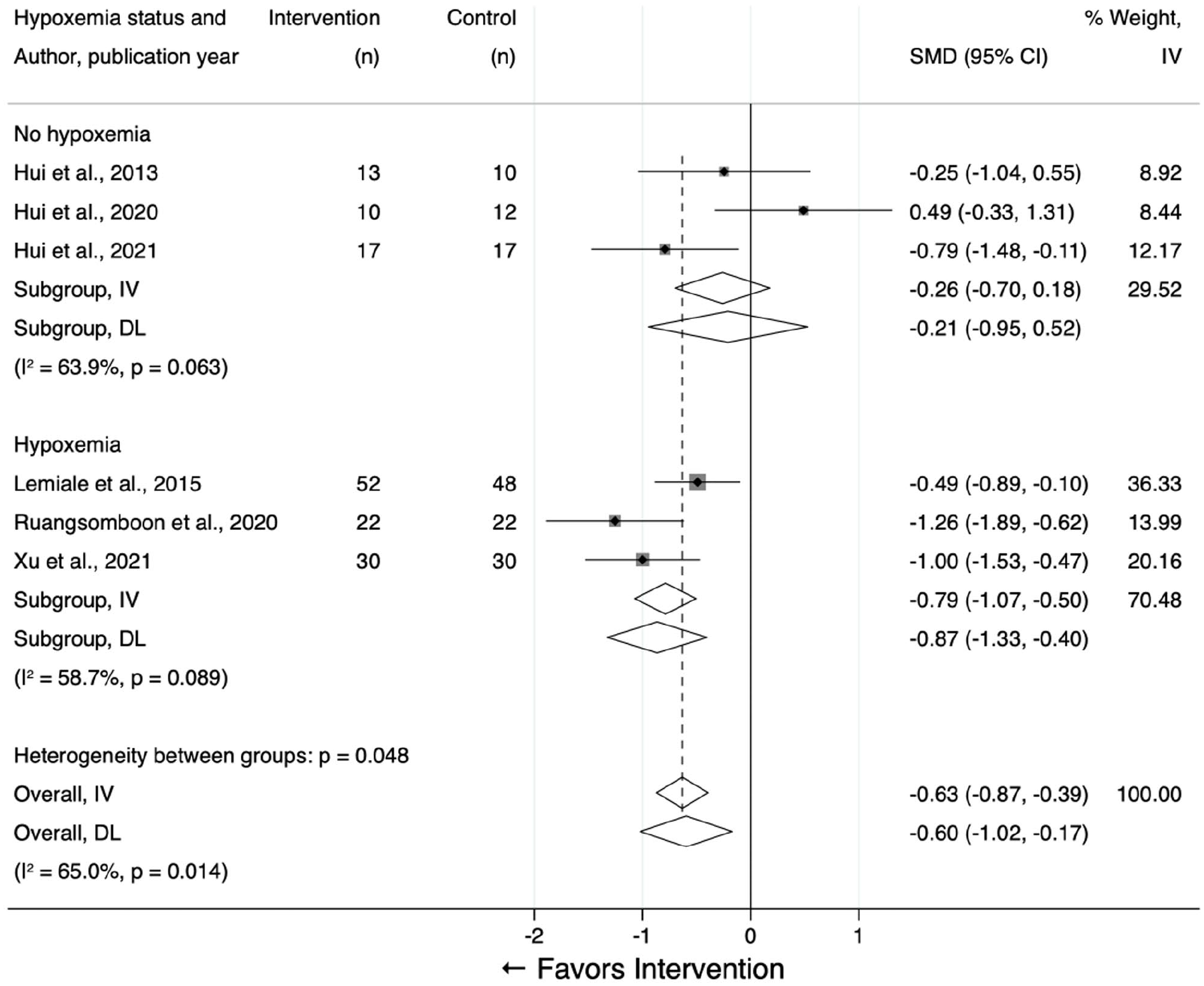
Meta-analysis stratified by hypoxemic status.
Secondary outcomes
None of the studies reported patient satisfaction. Two studies described mortality in the intervention and control groups at the end of follow up.14,23 Mendil et al. did not find any significant differences between the two groups (p = 0.395). Ruangsomboon et al. only gave a description of the number of deaths per group which was of 59.1% in the intervention group versus 72.7% in the control group without statistically comparing both groups. Lemiale et al. only gave a mortality rate for all participants, which was 24%.
Lemiale et al. assessed general comfort and thirst, with no significant difference between the high-flow nasal oxygen and conventional oxygen therapy (p = 0.88 and p = 0.44 respectively). In Hui et al., adverse effects including: dry eyes, eye irritation, feeling anxious, moist nose, mask painful, prong uncomfortable, stomach bloating, suffocation, trouble drinking, trouble eating and trouble talking were assessed by the NRS. Results were not statistically significant in the before-after analysis, and no difference was noted between the two groups. People had less trouble sleeping in the high-flow nasal oxygen group after the intervention, with a change of the median on the NRS of −6.0 (IQR: −8.0, 0.0, p = 0.04) compared to the BiPAP group which was of 0.0 (IQR: −1.0, 3.0, p > 0.99, p = 0.02). Xu et al., reported dryness of mouth which was increased in the control group (conventional oxygen therapy) versus the intervention group (p < 0.001). Sleepiness was also improved in the intervention group compared to conventional oxygen therapy (p < 0.001).
Quality assessment
Based on the Cochrane Handbook for Systematic Reviews of Intervention, we decided to assess the risk of bias by performing a domain-based evaluation (Supplemental Figures S2 and S3). 21 As predefined, CASP Checklist for Randomized Controlled Trials was performed in support of this quality assessment. 22 One of the included trials was assessed as having a low risk of bias. 25 Two had some concerns,11,27 and four were rated as having a high risk of bias.14,23,24,26
Discussion
Main findings
To our knowledge, this is the first systematic review and meta-analysis evaluating the effectiveness of high-flow nasal oxygen on dyspnea in people with cancer. We found that high-flow nasal oxygen was associated with a significant reduction in dyspnea compared to conventional oxygen therapy. Subgroup analysis by hypoxemic status confirmed that the benefit only concerned hypoxemic patients. There was no statistically significant effect in the non-hypoxemic subgroup, as has been found for people treated with conventional oxygen therapy. These findings underscore the potential of high-flow nasal oxygen as an effective intervention to alleviate dyspnea across a range of clinical scenarios, specifically in hypoxemic patients.
Other studies have previously shown a benefit of high-flow nasal oxygen in people with oncological diseases. In their meta-analysis, Heybati et al. found that high-flow nasal oxygen could be beneficial in terms of reduction of lengths of stay compared to conventional oxygen therapy and BiPAP in people receiving oncological therapies including surgery. Similarly to our results, people treated with high-flow nasal oxygen presented less nose and mouth dryness compared to conventional oxygen therapy and BiPAP. However, dyspnea was not evaluated in their study. 30 On the other hand, Takase et al. found a reduction of dyspnea on the MBS of 1.4 in their prospective single arm phase II study in hospitalized patients with advanced cancer treated by high-flow nasal oxygen. 31
Strengths and limitations
Our study has several limitations. Most included studies had small sample sizes (n = 19–102). In three studies, dyspnea was not the primary outcome,11,14,26 one lacked a pre-study power analysis, 14 two failed to meet target enrollment,25,26 and one underwent design modifications during the trial (from crossover to parallel) which may induce a bias in the interpretation of results. 26
There was marked heterogeneity in the patient populations, ranging from critically ill ICU patients 11 to highly functional outpatients. 24 It is therefore not possible at this point to state which population may benefit the most of high-flow nasal oxygen compared to other respiratory therapy. Selection bias is likely, as unstable critically ill patients were often excluded despite meeting eligibility criteria.
Dyspnea assessment was conducted using different scales which have been shown to yield different results. 25 In order to conduct this meta-analysis, we pooled the measures using the standardized mean difference (SMD) to account for these differences.
Study implications
This study highlights the benefits of high-flow nasal oxygen on dyspnea in people with cancer and therefore could be used in clinical practice to reduce this symptom, specifically in hypoxemic patients when other validated treatment, such as opioids, are no longer effective. High-flow nasal oxygen in palliative patients with persistent dyspnea present an ethical dilemma, as it can provide symptomatic relief but may inadvertently prolong dependence on hospital-based care. While high-flow nasal oxygen improves oxygenation and comfort, its continuous requirement may limit the feasibility of transitioning to a home setting, potentially conflicting with patient preferences for end-of-life care outside the hospital. Clinicians must weigh the benefits of symptom relief against the potential loss of autonomy and alignment with the person’s goals of care.
As one study has shown, non-hypoxemic patients seem to benefit from high-flow air as much as from high-flow nasal oxygen. 25 These findings suggest that other interventions requiring minimal organizational resources such as the use of a handheld fan may be sufficient to provide effective symptom relief in non-hypoxemic patients as has been shown in a previous study. 32 As dyspnea is a complex symptom, the effectiveness of high-flow nasal oxygen on dyspnea would benefit from a broader multidimensional assessment as recommended by international respiratory societies, such as the Dypnea-12 scale or the Multidimentional Dyspnea Profile.3,33,34 This would allow to determine which component of dyspnea benefits the most of high-flow nasal oxygen and select the people who are most likely to benefit.
Conclusion
High-flow nasal oxygen was associated with a significant reduction in dyspnea compared to conventional oxygen therapy, specifically in hypoxemic patients. High-flow nasal oxygen could be an additional option to reduce dyspnea in people with advanced cancer in the hospital setting if in alignment with the person’s goals of care.
Supplemental Material
sj-docx-1-pmj-10.1177_02692163261422559 – Supplemental material for High-flow nasal oxygen for the relief of persistent dyspnea in adult patients with cancer: A systematic review and meta-analysis
Supplemental material, sj-docx-1-pmj-10.1177_02692163261422559 for High-flow nasal oxygen for the relief of persistent dyspnea in adult patients with cancer: A systematic review and meta-analysis by Lisa Hentsch, Ivan Guerreiro, Valentina Gonzalez Jaramillo, Simon Singovski, Isabelle Santana, Sara Cocetta and Tanja Fusi-Schmidhauser in Palliative Medicine
Supplemental Material
sj-docx-2-pmj-10.1177_02692163261422559 – Supplemental material for High-flow nasal oxygen for the relief of persistent dyspnea in adult patients with cancer: A systematic review and meta-analysis
Supplemental material, sj-docx-2-pmj-10.1177_02692163261422559 for High-flow nasal oxygen for the relief of persistent dyspnea in adult patients with cancer: A systematic review and meta-analysis by Lisa Hentsch, Ivan Guerreiro, Valentina Gonzalez Jaramillo, Simon Singovski, Isabelle Santana, Sara Cocetta and Tanja Fusi-Schmidhauser in Palliative Medicine
Footnotes
Author contributions
LH drafted the article. LH, SC and TFS screened the databases. IS and SS extracted the trial-level data. VGJ conducted the meta-analysis. LH, IG and VGJ interpretated the data. SS, IS conducted the risk of bias analysis. All authors have revised the article critically for important intellectual content and approved the version to be published. All authors participated sufficiently in the work to take public responsibility for appropriate portions of the content.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: IG declare grant from the Ligue Pulmonaire Genevoise, honoraria for presentation and Advisory Board from AstraZeneca, GSK and Sanofi paid to his institution. The other authors declare that they have no conflict of interest.
Data availability statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.*
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
