Abstract
Background:
Polypharmacy and inappropriate medication are common amongst people with life-limiting conditions. Whilst deprescribing may help reduce these medication-related issues, supporting evidence in this population group is limited.
Aim:
To synthesise evidence on the outcomes of deprescribing in people with life-limiting conditions.
Design:
Systematic review.
Data source:
MEDLINE, Embase, Scopus, PsycINFO and CINAHL were searched. Original studies published between Jan 2000 and Dec 2024 in English were included.
Result:
A total of 17,457 hits were screened, of which 46 original studies met the inclusion criteria. Most eligible studies were pre-post interventional (n = 14) or cohort studies (n = 14), conducted primarily in nursing homes or long-term care facilities (n = 20) and hospitals (n = 16). The majority originated from North America (n = 20) and Australia (n = 7). A wide range of outcome variables were examined, with a primary focus on clinical outcomes. All studies assessing the impact on the number of medications used reported either a reduction in overall medication burden or inappropriate medications (n = 15), or no significant change (n = 3). Regarding mortality, most studies (10 studies) reported no impact, while 3 studies each reported increased and decreased mortality. For other outcomes, the majority of studies reported that deprescribing had no effect.
Conclusion:
This systematic review suggests that deprescribing offers some benefits, including reduced medication burden and costs in people with life-limiting conditions. While there is no strong evidence for harm, a small proportion of patients reported increased risks, so careful monitoring is essential. Further research should explore how deprescribing outcomes vary by disease condition and medication type.
Polypharmacy and inappropriate medication use is common in people with life-limiting conditions, and is exacerbated by the addition of symptom management medication to a legacy of preventative treatments.
While deprescribing can reduce the medication burden, it remains a complex and challenging intervention in people with life-limiting conditions.
A broad range of deprescribing outcomes are reported across clinical-, medication- and system-related impacts.
Existing studies are mostly from the US and Australia with little representation from low-and middle-income countries.
Deprescribing is generally safe, with most studies demonstrating no negative effects. However, careful monitoring is recommended to mitigate potential risk in minority of cases.
Careful monitoring is essential while implementing deprescribing interventions in individuals with life-limiting conditions.
Further research is needed to strengthen the evidence base for deprescribing, particularly in relation to specific diseases and medication classes.
Introduction
Life-limiting conditions are progressive, incurable illnesses that are expected to shorten a person’s life, such as cancer, organ failure, and neurodegenerative conditions.1,2 People with life-limiting conditions frequently experience polypharmacy and the use of inappropriate medications.3–7 Previous studies have reported polypharmacy and potentially inappropriate medication use in people with advanced cancer8,9 and dementia,10–12 as well as people receiving palliative3,13,14 or specifically, hospice care.15,16 In people with life limiting conditions, the presence of multiple long-term conditions and other age-related factors (e.g. frailty), can further increase the risk of polypharmacy and the use of potentially inappropriate medications.5,17 It has also been shown that polypharmacy and the use of inappropriate medication can adversely affect patients’ health outcomes, including adverse drug events, reduced medication adherence, hospitalisation, and increased risk of morbidity and mortality – although studies have not specifically focussed on people with life-limiting conditions.18–21
For people with life-limiting conditions, medication optimisation can be challenging for a number of reasons, including; the complex and dynamic medical needs of such patients, the potential psychological impact of changing medication – something which is relevant for both patients and caregivers, as well as the risk of symptom exacerbation upon reducing or stopping medication.22–25 In this context, it is important that medication use aligns with the evolving health status and goals of care of the patient. 26
One potential solution to the challenge of polypharmacy and potential inappropriate medication use is deprescribing. Deprescribing aims to reduce medication burden, specifically ensuring discontinuation of inappropriate medications.27–29 It is a systematic process of identifying and discontinuing medications that are no longer beneficial, or where the risks outweigh the intended benefits. Previous studies have reported that deprescribing can improve survival or reduce mortality risk in specific contexts, such as among early older adults (aged 65–79 years) 30 or older patients in end-of-life care.27–31 The impact of deprescribing on other health-related outcomes (e.g. falls or hospitalisation), or among vulnerable patient groups (e.g. people with frailty or dementia) remain limited.27,29
In the context of people with life-limiting conditions, evidence for deprescribing to improve patient outcomes is lacking.27,31–33 Previous systematic reviews, published by Shrestha et al. in 2020 33 and 2021, 31 reviewed deprescribing outcomes amongst older people with limited life expectancy and concluded there was evidence that deprescribing improved medication appropriateness; the review also concluded that evidence needs to be better established for other outcomes. Another systematic review, published in 2021, examined the effect of deprescribing interventions in older adults close to end-of-life, using the Criteria for Screening and Triaging to Appropriate aLternative care (CriSTAL) risk prediction tool,32,34 concluded it was difficult to ascertain if deprescribing improved patient outcomes. Since the publication of these systematic reviews, there has been an exponential increase in deprescribing research; for examples Hurley et al.,35,36 (2024), Etherton-Beer et al. 37 (2023), Tapper et al. 38 (2022), Niznik et al.39,40 (2020). As such, a comprehensive updated systematic review exploring evidence on deprescribing outcomes among older people with life-limiting conditions is warranted. This study aimed to address this gap and examine the evidence for outcomes of deprescribing for people with life limiting conditions.
Methods
The protocol for this systematic review was developed and registered in the Prospective Register of Systematic Reviews (PROSPERO; CRD42024622342). This review is reported to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guideline (Supplemental Table 1).
Inclusion criteria
The population, intervention, comparison, outcome, study design (PICOS) framework was used to conceptualise the review inclusion criteria (Table 1). To be eligible for inclusion, studies had to be published in English, from January 2000 to December 2024. The date restrictions were implemented to ensure the inclusion of recent and relevant research that accurately reflects current (de)prescribing practices.
The PICOS framework used for the systematic review.

Search strategy
Following the PICOS refinement, a search strategy was developed. Relevant keywords and controlled vocabulary with appropriate synonyms and Boolean logic were used. MEDLINE, Embase, Scopus, PsycINFO and CINAHL were systematically searched to identify relevant literature. The literature search was supplemented by forward citation searching of relevant studies. Detailed search strategies employed for each database are provided (Supplemental Table 2 to 6).
Study selection and data extraction
Two reviewers (RS and LM) independently screened titles and abstracts for eligibility. Any disagreement during this stage was resolved through discussion between the two reviewers. No studies were excluded at this stage without mutual agreement. Following this, the same reviewers (RS and LM) independently assessed full text articles for eligibility. Any uncertainties that arose were discussed with the clinical members of the research team (AT, a pharmacist and FD, a palliative medicine doctor) who had final consensus.
A standardised data extraction form was developed to capture information on study characteristics, population characteristics, intervention descriptions including medication details (method of deprescribing, medications deprescribed, duration, follow-up, pattern of medication use) and outcomes (information on clinical, medication, and system-related outcomes). Corresponding authors of eligible papers were contacted for further clarification, if required. The data were extracted in full by one author (RS) and checked by a second author (ES). Any disagreement was discussed between RS and ES and, if agreement could not be reached AT had consensus as the senior author.
Quality appraisal
Joanna Briggs Institute critical appraisal tools were used to assess the quality of included studies. This tool provides a comprehensive suite of tools tailored to different study designs aligning with established evidence synthesis methodologies, with recent revisions focussing on evaluating the risk of bias.41–43 RS assessed the risk of bias of the included studies which was then checked, in full, by ES. Any conflicts were resolved through discussion with the senior author (AT) who had consensus. No studies were excluded based on the results of the critical appraisal. All studies meeting the eligibility criteria were included to provide a comprehensive overview of the available evidence.
Data synthesis
Due to the considerable heterogeneity in studies of interest (e.g. different study designs, patient populations, interventions and reported outcomes), a descriptive synthesis approach was undertaken to analyse the data, grouped by theme. Outcomes of deprescribing were organised into common groups under three broad categories: clinical-, medication- and system- related outcomes. Extracted data were summarised in tabular format to facilitate comparison across studies.
When a study reported multiple measures using a validated or stated instrument or scale for a single outcome domain, each measure was listed separately in the summary table. For example, if a study assessed cognitive function using (i) the Mini-Mental State Examination (MMSE) scale and (ii) the Cognitive Performance Scale (CPS) in across the intervention and control groups, all four measurements (i.e. two outcomes from each group) were reported as separate data points. Similarly, when a study reported outcomes separately for distinct patient subgroups over for total patients, each subgroup was treated as a separate data point in the synthesis. For example, in studies that presented deprescribing outcomes separately for patients with dementia and without dementia, the data from each group was extracted and reported as separate entries. The effects of deprescribing on each outcome, as reported by the included studies, were categorised into three groups:
Positive effect: Studies reporting improvement or beneficial effects for patients with statistical analysis for significance were categorised as positive effects (e.g. reduced medication burden, improved clinical parameters, reduced adverse drug events).
No effect: Studies showing no statistically significant changes were categorised as no effect.
Negative effect: Studies reporting potential harm or worsening of outcomes for patients with statistical analysis for significance were categorised as negative effects (e.g. increased adverse drug events, symptom deterioration, increased hospitalisations).
For example, if a study reported a statistically significant lower MMSE for the deprescribing group compared to control group, it was categorised that deprescribing had a ‘Negative effect’ on cognitive function.
For findings reported without statistical analysis for significance, the categorisation into positive, negative, or no effect was based on the direction of the reported trend or the authors’ interpretation. For example, if a study reported a reduction in the number of patients with falls after deprescribing intervention without performing statistical analysis for significance, it was also categorised as a ‘Positive effect’ in the summary table.
To distinguish findings with and without statistical analysis for significance, an indication mark was given in the summary table. The term ‘significant’ in the result section refers to the outcome with statistical analysis for significance.
Results
Study selection
Database and reference searching identified 17,457 records. After removal of duplicates, 10,284 records remained for title and abstract screening. Of these, 537 progressed to full-text review, resulting in 46 original studies meeting the inclusion criteria. The study selection process is detailed in Figure 1.
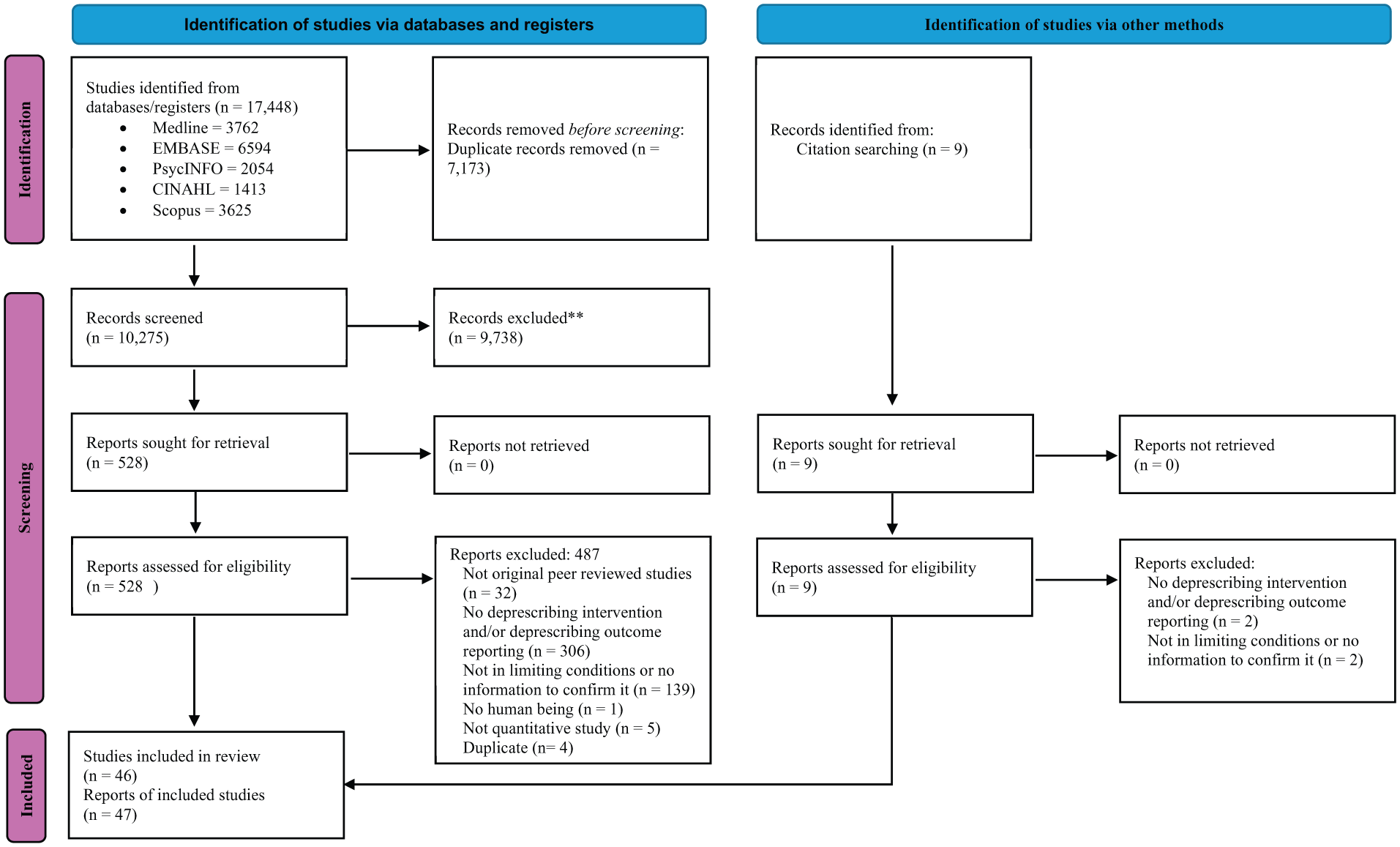
PRISMA flowchart of included studies.
Characteristics of included studies
Among the included studies, the most common study designs were cohort (n = 14),39,40,44–55 pre-post interventional (n = 14)35,56–68 and randomised control studies (n = 11).37,69–78 The majority of the included studies were conducted in nursing homes/long-term care facilities (n = 20)35–37,39,40,44,46,48,52,60,61,63,67,71,72,73,74–77,84 hospitals (n = 16)45,47,51,53–55,57,59,64,65,68,69,78,79,80,81 and hospice/palliative care units (n = 5).49,56,62,68,70 Studies were also conducted in specialist care clinics (n = 2),58,82 a telemedicine palliative care clinic (n = 1), 66 and home-based palliative care settings (n = 1). 50 Most studies were conducted in the US (n = 13),38,40,44,46–48,58,59,62,66,67,70,71 and Australia (n = 7)37,54,60,65,74,77,79 (Table 2 and Figure 2).
Characteristics of included studies.
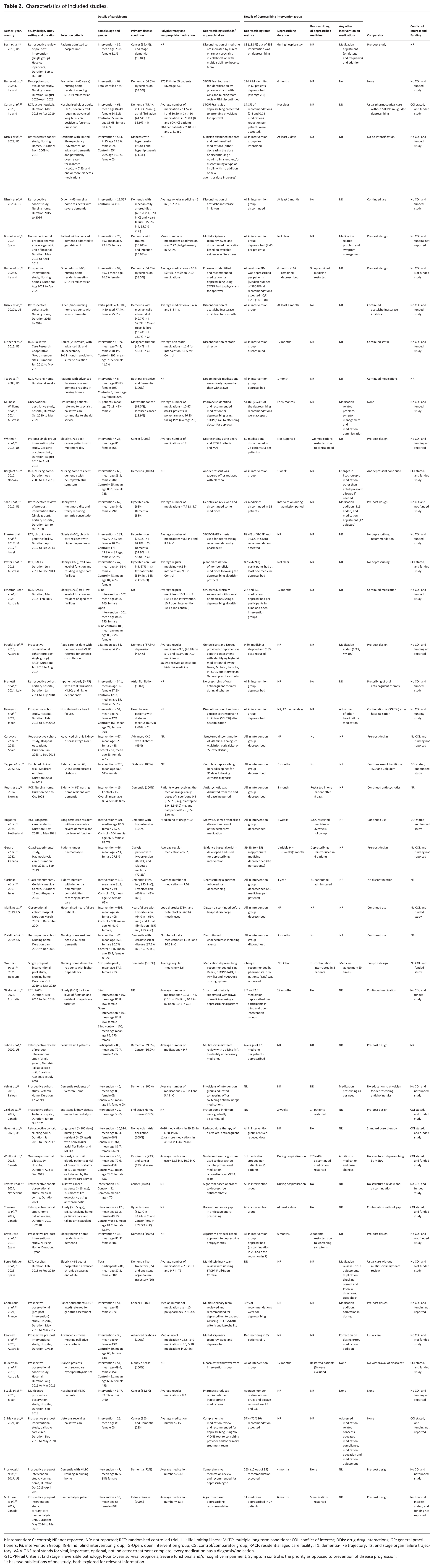
I: intervention: C: control; NR: not reported; NR: not reported; RCT: randomised controlled trial; LLI: life limiting illness; MLTC: multiple long term conditions; COI: conflict of interest; DDIs: drug-drug interactions; GP: general practitioners; IG: intervention Group; IG-Blind: blind intervention group; IG-Open: open intervention group; CG: control/comparator group; RACF: residential aged care facility; T1: dementia-like trajectory; T2: end stage organ failure trajectory; VA VIONE tool stands for vital, important, optional, not indicated/treatment complete, every medication has a diagnosis/indication.
STOPPFrail Criteria: End stage irreversible pathology, Poor 1-year survival prognosis, Severe functional and/or cognitive impairment, Symptom control is the priority as opposed to prevention of disease progression.
It has two publications of one study, both explored for relevant information.
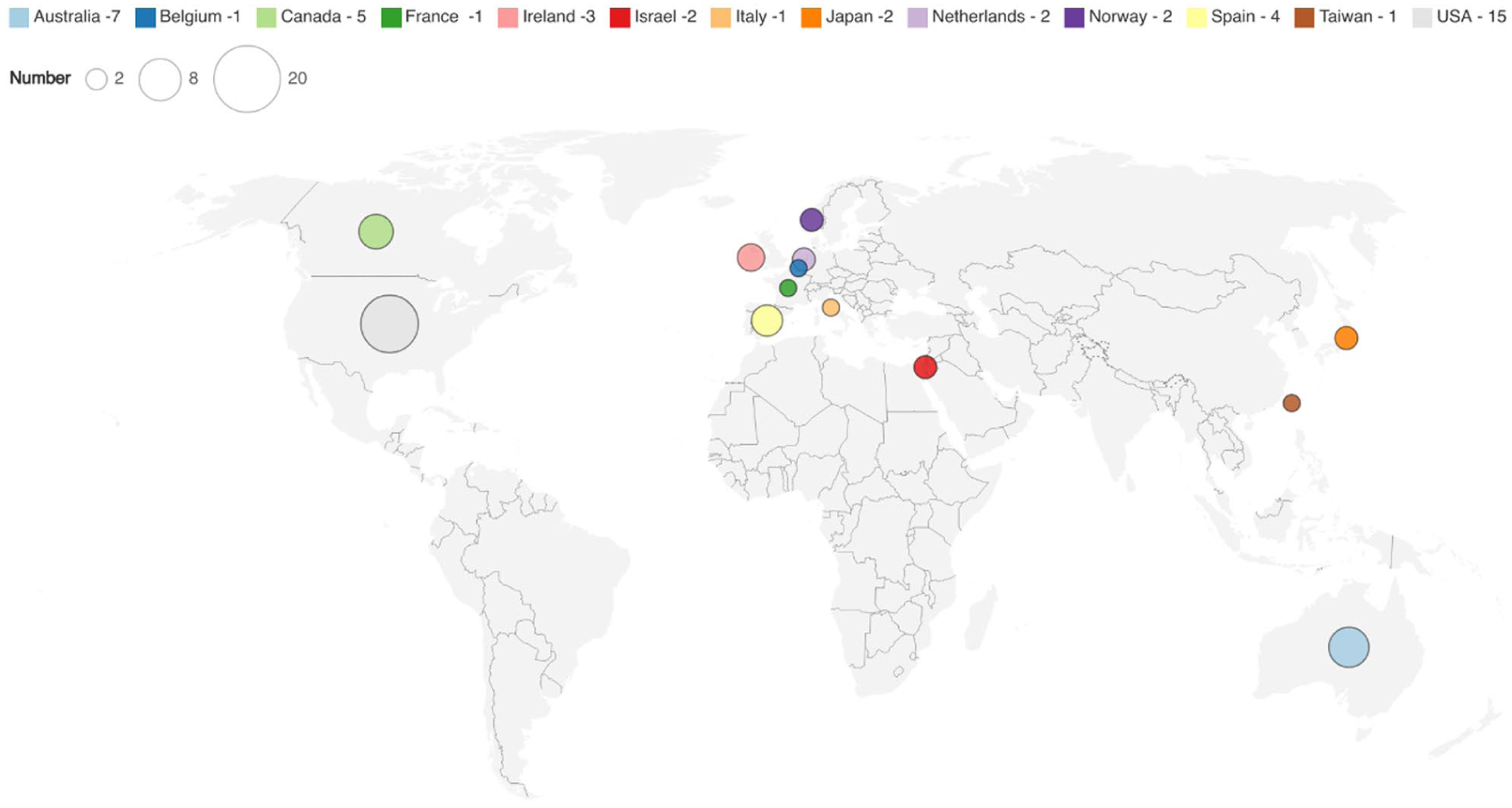
Map of geographical locations of included studies.
In terms of the population from the included studies, the majority were older adults who reported multiple disease conditions, taking a mean of 4.6 52 to 13.3 80 medications. The most reported disease conditions were dementia (18 studies)35,36,39,40,46,52,57,60–63,67,69,72,75,76,78,83 and cancer (8 studies)49,56,58,64,66,70,79,81 (Table 2 and Figure 3).
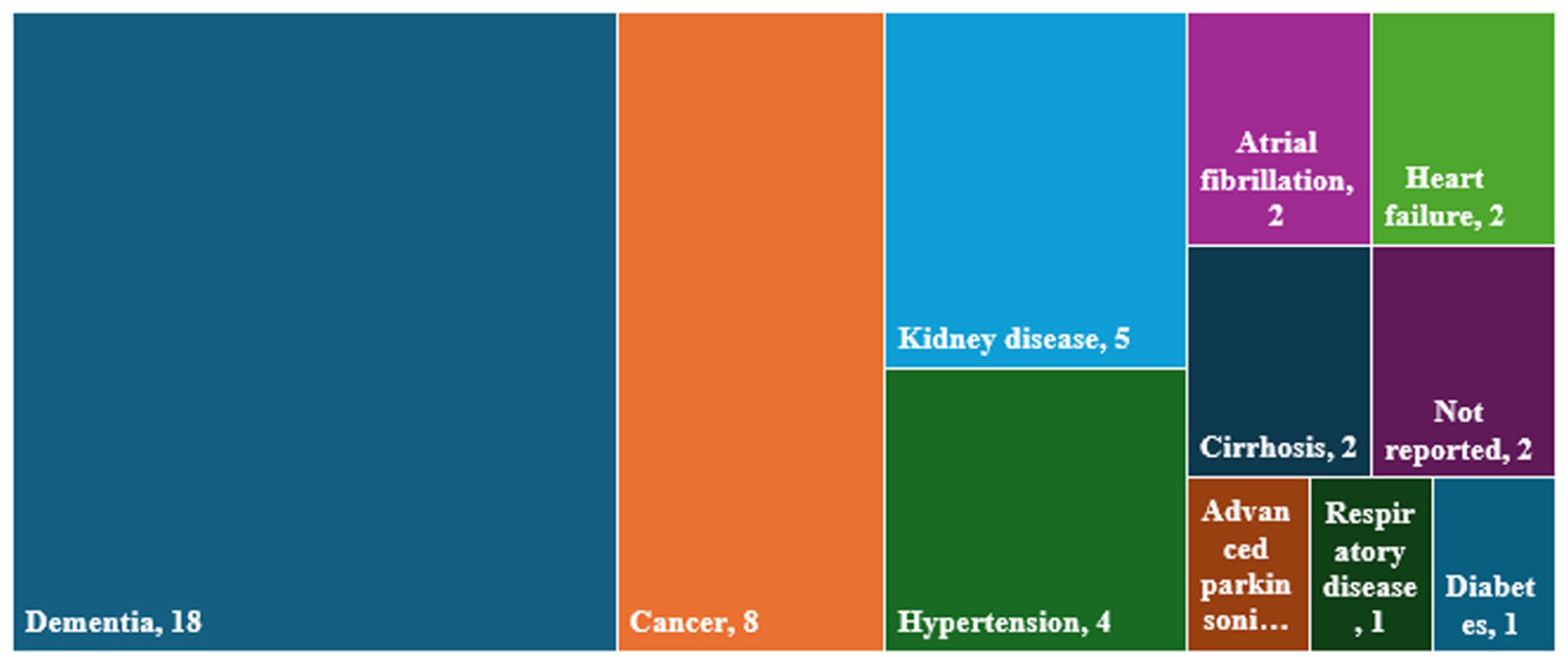
Primary disease conditions of included studies.
Deprescribing characteristics
The deprescribing approaches undertaken among the included studies were heterogeneous. Most studies directly deprescribed targeted medicines38–40,44–55,63,70–72,75,76 and utilised a validated tool or criteria-based approach to identify inappropriate medication.35,58,60–62,66,69,73,78,79 Some studies used an algorithm-based approach, meaning they followed a structured, step-by-step decision-making process or flowchart, to identify medication for deprescribing,37,68,74,77,80,82,83 while other studies employed medication reviews without utilising any tools56,57,59,65,67,81 (Table 2).
Commonly reported deprescribed medications included acid suppressants, anti-thrombotics, oral hypoglycaemics, anti-hypertensives, lipid lowering agents, analgesics, psychotropics, dementia medications and supplements. Other reported deprescribed medications included anticholinergics, diuretics and antidepressants. A detailed description of the reported deprescribed medications is included as a Supplemental file (Supplemental Table 7).
Deprescribing outcomes
Deprescribing outcomes were organised into three categories: clinical-, medication- and system-related outcomes (Table 3). The detailed outcomes reported by each study is available as a Supplemental file (Supplemental Tables 8–10).
Summary of clinical, medication, and system-related outcomes reported by included studies.
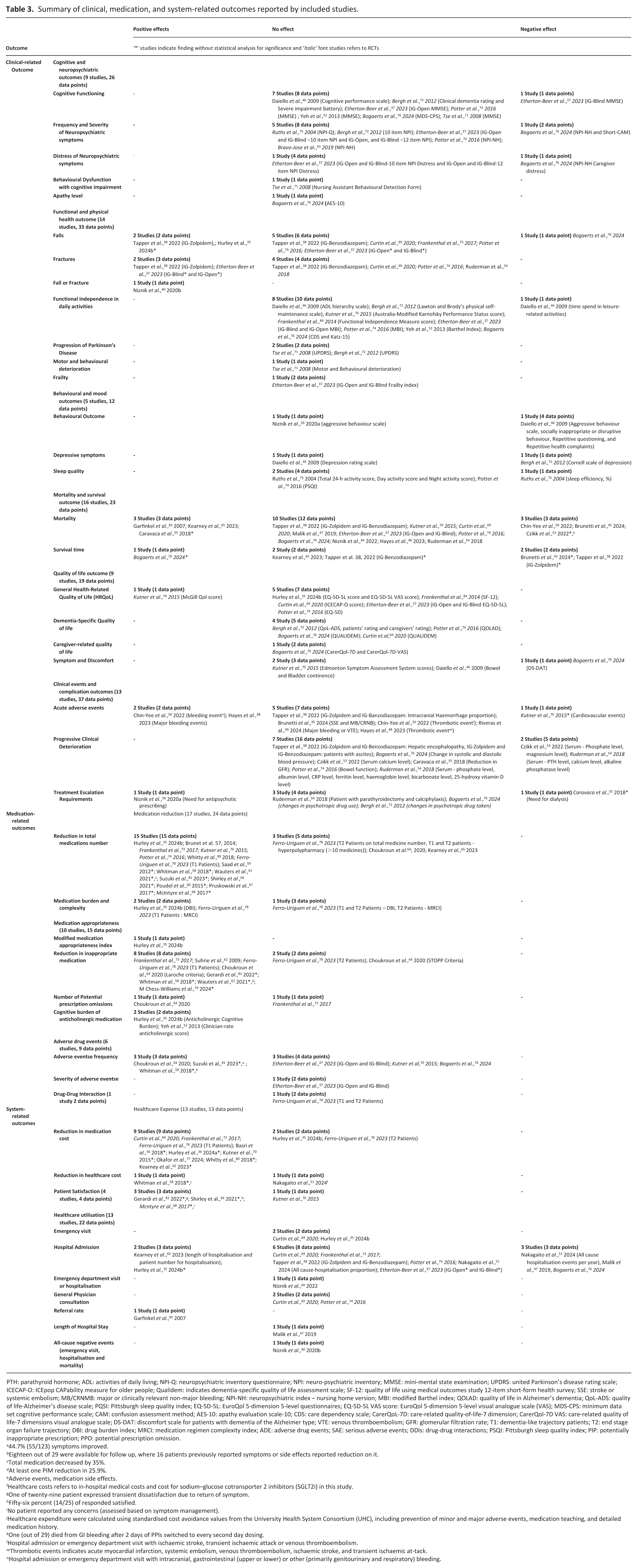
PTH: parathyroid hormone; ADL: activities of daily living; NPI-Q: neuropsychiatric inventory questionnaire; NPI: neuro-psychiatric inventory; MMSE: mini-mental state examination; UPDRS: united Parkinson’s disease rating scale; ICECAP-O: ICEpop CAPability measure for older people; Qualidem: indicates dementia-specific quality of life assessment scale; SF-12: quality of life using medical outcomes study 12-item short-form health survey; SSE: stroke or systemic embolism; MB/CRNMB: major or clinically relevant non-major bleeding; NPI-NH: neuropsychiatric index – nursing home version; MBI: modified Barthel index; QOLAD: quality of life in Alzheimer’s dementia; QoL-ADS: quality of life-Alzheimer’s disease scale; PQSI: Pittsburgh sleep quality index; EQ-5D-5L: EuroQol 5-dimension 5-level questionnaires; EQ-5D-5L VAS score: EuroQol 5-dimension 5-level visual analogue scale (VAS); MDS-CPS: minimum data set cognitive performance scale; CAM: confusion assessment method; AES-10: apathy evaluation scale-10; CDS: care dependency scale; CarerQoL-7D: care-related quality-of-life-7 dimension; CarerQol-7D VAS: care-related quality of life-7 dimensions visual analogue scale; DS-DAT: discomfort scale for patients with dementia of the Alzheimer type; VTE: venous thromboembolism; GFR: glomerular filtration rate; T1: dementia-like trajectory patients; T2: end stage organ failure trajectory; DBI: drug burden index; MRCI: medication regimen complexity index; ADE: adverse drug events; SAE: serious adverse events; DDIs: drug-drug interactions; PSQI: Pittsburgh sleep quality index; PIP: potentially inappropriate prescription; PPO: potential prescription omission.
44.7% (55/123) symptoms improved.
Eighteen out of 29 were available for follow up, where 16 patients previously reported symptoms or side effects reported reduction on it.
Total medication decreased by 35%.
At least one PIM reduction in 25.9%.
Adverse events, medication side effects.
Healthcare costs refers to in-hospital medical costs and cost for sodium–glucose cotransporter 2 inhibitors (SGLT2i) in this study.
One of twenty-nine patient expressed transient dissatisfaction due to return of symptom.
Fifty-six percent (14/25) of responded satisfied.
No patient reported any concerns (assessed based on symptom management).
Healthcare expenditure were calculated using standardised cost avoidance values from the University Health System Consortium (UHC), including prevention of minor and major adverse events, medication teaching, and detailed medication history.
One (out of 29) died from GI bleeding after 2 days of PPIs switched to every second day dosing.
Hospital admission or emergency department visit with ischaemic stroke, transient ischaemic attack or venous thromboembolism.
Thrombotic events indicates acute myocardial infarction, systemic embolism, venous thromboembolism, ischaemic stroke, and transient ischaemic at-tack.
Hospital admission or emergency department visit with intracranial, gastrointestinal (upper or lower) or other (primarily genitourinary and respiratory) bleeding.
Clinical-related outcomes
Medication-related outcomes
Two studies reported medication burden and medication complexity outcomes. Hurley et al. 35 assessed the Drug Burden Index (DBI) and found deprescribing significantly reduced DBI scores (meaning the medication burden was reduced for patients), while another study, by Ferro-Uriguen et al., 78 found deprescribing did not significantly change DBI scores (Table 3).
System-related outcomes
Critical appraisal of included studies
For randomised controlled studies, blindness to participants and those delivering the intervention were common concerns. Non-similar study groups, identification and addressing compounding factors, and follow-up related information were the major non-complying criteria for cohort studies. A common factor identified in most quasi-experimental studies was having a single arm without comparison groups. The detailed scoring and overall rating of each study are available in the Supplemental file (Supplemental Tables 11–15).
Discussion
In this systematic review, we have comprehensively summarised deprescribing outcomes for people with life-limiting conditions and organised these outcomes into three broad groups representing clinical, medication and system-level domains.
Our review highlights several key findings with implications for patients, healthcare professionals and policymakers. The evidence shows that, for people with life-limiting conditions, deprescribing can improve medication-related outcomes: for example, deprescribing can reduce the total medication number taken by patients, as well as improving medication appropriateness. There was strong evidence to support this, and this finding corresponds to previous reviews: Shrestha et al. 33 reported that deprescribing reduces the number of medications and improves appropriateness of medication in people with life limiting illness and limited life expectancy. A recent 2025 systematic review and network meta-analysis among older adults with chronic diseases also confirmed that deprescribing is an appropriate intervention for reducing inappropriate prescribing. 85 Similar findings of medication reduction have also been reported by two umbrella reviews on deprescribing interventions in older people.27,29 While it is perhaps not surprising that deprescribing approaches reduce the total number of medication used by patients, the reduction of inappropriate medication is less predictable, but importantly, this is essential to reduce potential adverse effects from medication. In view of these findings, healthcare professionals whose responsibilities include prescription and review of medication for people with life limiting illnesses should initiate deprescribing to reduce the medication count and improve prescribing appropriateness, whilst carefully monitoring the effects.
The impact of deprescribing on the other outcomes was found to be more complex. For clinical outcomes, evidence suggests that deprescribing does not necessarily lead to improvement, but in most cases, it does not result in harm. For example, for studies reporting on mortality (16 studies), the majority of studies (10 studies) reported that deprescribing had no impact; 3 studies reported deprescribing increased mortality, while 3 reported deprescribing reduced mortality. 31 In line with this, a recent 2024 umbrella review on deprescribing interventions in older adults by Chua et al., 29 which reported that 17 out of 19 systematic reviews found no significant impact on mortality outcomes with none of the reviews reporting increased mortality. This suggests that while there is a higher likelihood of no effects on mortality and survival time, there is a possibility that deprescribing can increase mortality in people with life-limiting illness in certain contexts. Understanding the factors influencing the risk of mortality in life-limiting conditions is complex due to the involvement of multifaceted aspects, including physiological changes associated with the progression of life-limiting conditions.26,86,87 Mostly neutral findings were also found for the behavioural and mood outcomes and the functional and physical health outcomes. Therefore, when considering deprescribing in relation to health-related outcomes, it is essential to carefully assess the patient’s underlying conditions, the specific medications targeted for deprescribing, and to ensure ongoing monitoring to maintain safety.
Literatures generally reported beneficial effects like medication-related domains in most system-related outcomes. The evidence from the included studies indicated that deprescribing consistently reduced medication cost, which is an expected outcome. However, the evidence on overall healthcare cost reduction was limited, with one reported beneficial and another no significant difference. This might be linked with the differences in their methodology applied for healthcare cost estimation: Whitman et al. 58 modelled cost assumptions associated with adverse events prevention and pharmacist interventions, and did not assess with statistical analysis of significance, whereas Nakagaito et al. 51 calculated actual hospitalisation and medication costs and assessed difference with statistical analysis of significance. Notably, none of them accounted for the cost associated with long-term health consequences of deprescribing. Furthermore, external evidence (Wojtowycz et al. 88 ) suggests that the overall healthcare cost could be increased in some cases, such as when new medications are added after deprescribing, highlighting a critical and unexplored areas. Therefore, further studies are essential to explore these nuances. Followingly, deprescribing approach did not appear to adversely affect patient satisfaction. However, healthcare utilisation outcomes were largely unchanged, indicating for further research to clarify whether specific patients population and deprescribed medication are more associated with adverse events for re-admission. To summarise the system-related outcomes, the majority studies showed that deprescribing generally had no negative effects.
The neutrality reported across most outcomes underscore the complexity of measuring the impact of deprescribing. While the absence of harmful effects in the majority of studies is encouraging, the predominantly neutral effects raise a critical question on whether deprescribing retains modest-benefit only or current outcome measures are insufficient to capture meaningful changes or make appropriate conclusions. Further research is needed to elucidate the specific factors that drive variations in deprescribing outcomes. Additionally, some reported negative effects on some outcome variables highlight the need for a careful monitoring and individualised patient-centred approaches to deprescribing. A recent qualitative study highlighted that patient wanted to have a voice in deprescribing decision-making, but at present this does not always occur. 89
Strength and limitations
A key strength of this review is its comprehensive synthesis of deprescribing outcome across a broad spectrum of life-limiting conditions, which identified a larger body of evidence (46 studies) compared to previous similar review by Shrestha et al. 33 in 2020 (9 studies). While Shrestha et al. 33 focussed on older adults (>65) with a short life expectancy (up to 2 years), our pragmatic approach draws the evidence from a wider range of serious illnesses, reflecting the clinical reality of life-limiting conditions where age and life expectancy are often uncertain. However, this fundamental difference precludes a direct quantitative comparison of findings but demonstrates the relevance of deprescribing to a wider palliative care context.
The study does, however, have few limitations that should be acknowledged. First, the included studies were heterogenous in terms of study design, settings, outcome assessment and reporting, selection and deprescribing of medication that limited to conduct meta-analysis. Similarly, this study did not include those studies which were not published or available in English language, not peer reviewed, published before 2000, and did not provide sufficient information to conclude that their study population had life-limiting conditions.
Furthermore, while this systematic review included 46 original studies there are a number of important evidence gaps. First, the included studies were mostly conducted in North America, Europe and Australia. There were few representations from Asia and no representation from other continents. It is not clear if – or how – the healthcare system in which the deprescribing takes place impacts on the deprescribing outcome. Second, there were variations in how the deprescribing process was undertaken. Some studies directly targeted specific medications, while other studies have reported deprescribing a range of medications with the help of various tools and clinicians’ recommendations. The deprescribing duration, success or failure rate of deprescribing, and incorporation of other medication-related interventions (e.g. medication review, medication reconciliation, adjustment in medicine and their dosage) were not consistently reported across the studies. Finally, patients in the included studies had a variety of life-limiting conditions, with dementia and cancer being the most common reflecting the diverse disease trajectories being considered.
Future directions
To strengthen the literature to further support evidence-based deprescribing, future studies could explore the impact of the deprescribing of specific medication classes in specific patient and disease contexts. Similarly, future studies should clearly define the success rate of deprescribing, including reporting data on whether the deprescribed medication remained stopped or reduced or restarted. Improving the consistency of deprescribing reporting across studies should further strengthen the evidence on deprescribing outcomes. Future studies could further investigate the factors influencing successful deprescribing to inform the development of future deprescribing interventions in people with life-limiting conditions.
Moreover, the number of studies contributing to each outcome variable was not uniform. While medication reduction, healthcare utilisation and mortality were well reported, patient-centred outcomes (such as quality of life, patient satisfaction), long-term safety endpoints (such as specific clinical complications, adverse drug events), and overall healthcare cost were reported by small number of studies. Thus, these gaps should be prioritised in future studies.
Conclusion
This systematic review suggests that deprescribing approach offers several benefits, including reduced medication burden and costs in people with LLCs. While there is no strong evidence for harm, a small proportion of patients may face risks, so careful monitoring is essential. Further studies exploring deprescribing interventions specific to patients’ disease conditions are warranted to strengthen the evidence on deprescribing outcomes.
Supplemental Material
sj-docx-1-pmj-10.1177_02692163261416281 – Supplemental material for Outcomes of deprescribing for people with life-limiting conditions: A systematic review
Supplemental material, sj-docx-1-pmj-10.1177_02692163261416281 for Outcomes of deprescribing for people with life-limiting conditions: A systematic review by Rajeev Shrestha, Emily Shaw, Liam Mullen, David Sinclair, Felicity Dewhurst and Adam Todd in Palliative Medicine
Footnotes
Author contributions
RS, DS, FD and AT designed the study. RS and LM performed the screening independently. RS performed data extract and quality assessment with the help of ES. RS, DS, FD and AT performed data synthesis. RS wrote the initial draft. DS, FD and AT critically reviewed and supervised the study. All authors approved the final version of the manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The work described in this is funded by the NIHR Newcastle Patient Safety Research Collaboration (PSRC) [grant number: NIHR204291]. The views expressed are those of the present and not necessarily those of the NIHR or the Department of Health and Social Care. The NIHR NPSRC is a partnership between the Newcastle Upon Tyne Hospitals NHS Foundation Trust, the University of Newcastle Upon Tyne, Cumbia, Northumbria, Tyne and Wear NHS Foundation Trust, Queen Mary University of London, University of Northumbria at Newcastle and Northumbria Healthcare NHS Foundation Trust.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
